Latest guidelines for management of hypertension in pregnancy
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive summary of current guidelines for the management of hypertension in pregnancy, drawn from multiple authoritative textbooks.
Management of Hypertension in Pregnancy
Classification
Hypertension in pregnancy is defined as BP ≥140/90 mm Hg on at least two separate occasions and complicates up to 10% of pregnancies. There are four categories:
| Category | Definition |
|---|---|
| Chronic hypertension | Pre-existing or diagnosed before 20 weeks; does not resolve postpartum |
| Gestational hypertension | New-onset after 20 weeks, no proteinuria, resolves within 3 months of delivery |
| Preeclampsia | New-onset hypertension after 20 weeks + proteinuria or end-organ damage |
| Superimposed preeclampsia | Preeclampsia developing on a background of chronic hypertension |
Blood Pressure Targets
- Severe hypertension (SBP ≥160 or DBP ≥105–110 mm Hg): drug therapy is clearly and urgently indicated to prevent stroke and cardiovascular complications.
- Mild-to-moderate hypertension: The CHIPS trial (multicenter RCT) showed that treating to a tight target (DBP 85 mm Hg) versus less-tight (DBP 100 mm Hg) had no difference in adverse fetal outcomes, but the tight-control group had fewer episodes of severe hypertension. Treating to <140/90 mm Hg reduces risk of severe preeclampsia features, indicated preterm birth, and placental abruption without increasing SGA risk. — Creasy & Resnik's Maternal-Fetal Medicine
- Non-pharmacologic measures (exercise, dietary sodium restriction, weight loss) have not been adequately evaluated in pregnant women and are not recommended as primary interventions.
Antihypertensive Drug Therapy
Chronic Hypertension: First-Line Oral Agents
| Drug | Notes |
|---|---|
| Labetalol | α/β-blocker; preferred over pure β-blockers due to beneficial α-blockade effect on uteroplacental flow; multiple daily dosing |
| Long-acting Nifedipine | Once-daily slow-release preparation; mild edema as side effect |
| α-Methyldopa | Centrally acting; most extensive safety data, including 7-year neurological follow-up in exposed children; sedation and short duration of action |
Second-Line Oral Agents
- Metoprolol (long-acting formulation), Verapamil, Diltiazem — limited data but no proven fetal harm
Drugs to Avoid
- Atenolol — associated with fetal growth restriction
- Diuretics — theoretically impair the pregnancy-associated expansion of plasma volume; may be used cautiously for frank volume overload but contraindicated in preeclampsia
Contraindicated
- ACE inhibitors and ARBs — second-trimester exposure causes fetal renal dysplasia, oligohydramnios, and pulmonary hypoplasia
Acute/Urgent BP Control (Severe Hypertension)
For SBP ≥160 or DBP ≥105–110 mm Hg, IV or rapid-acting agents are used:
| Agent | Notes |
|---|---|
| IV Labetalol | Good safety data; first-line IV agent |
| IV Nicardipine | Extensive safety data as a tocolytic; effective |
| Hydralazine (IV/IM) | Extensive clinical experience but increased risk of maternal hypotension and possibly placental abruption; now considered second-line |
| Oral nifedipine (immediate release) | Effective for acute lowering |
A meta-analysis of 21 trials showed hydralazine was associated with a higher risk of maternal hypotension compared to labetalol or nifedipine for acute management. — Brenner & Rector's The Kidney
Avoid: Sodium nitroprusside (risk of fetal cyanide toxicity if used >4 hours)
Preeclampsia Management
Severe Features Criteria
- SBP ≥160 mm Hg or DBP ≥110 mm Hg
- Headache or visual changes
- Epigastric/right upper quadrant pain
- Pulmonary edema
- Platelets <100,000/µL
- Creatinine >1.1 mg/dL
- AST >2× the upper limit of normal
Management Flowchart
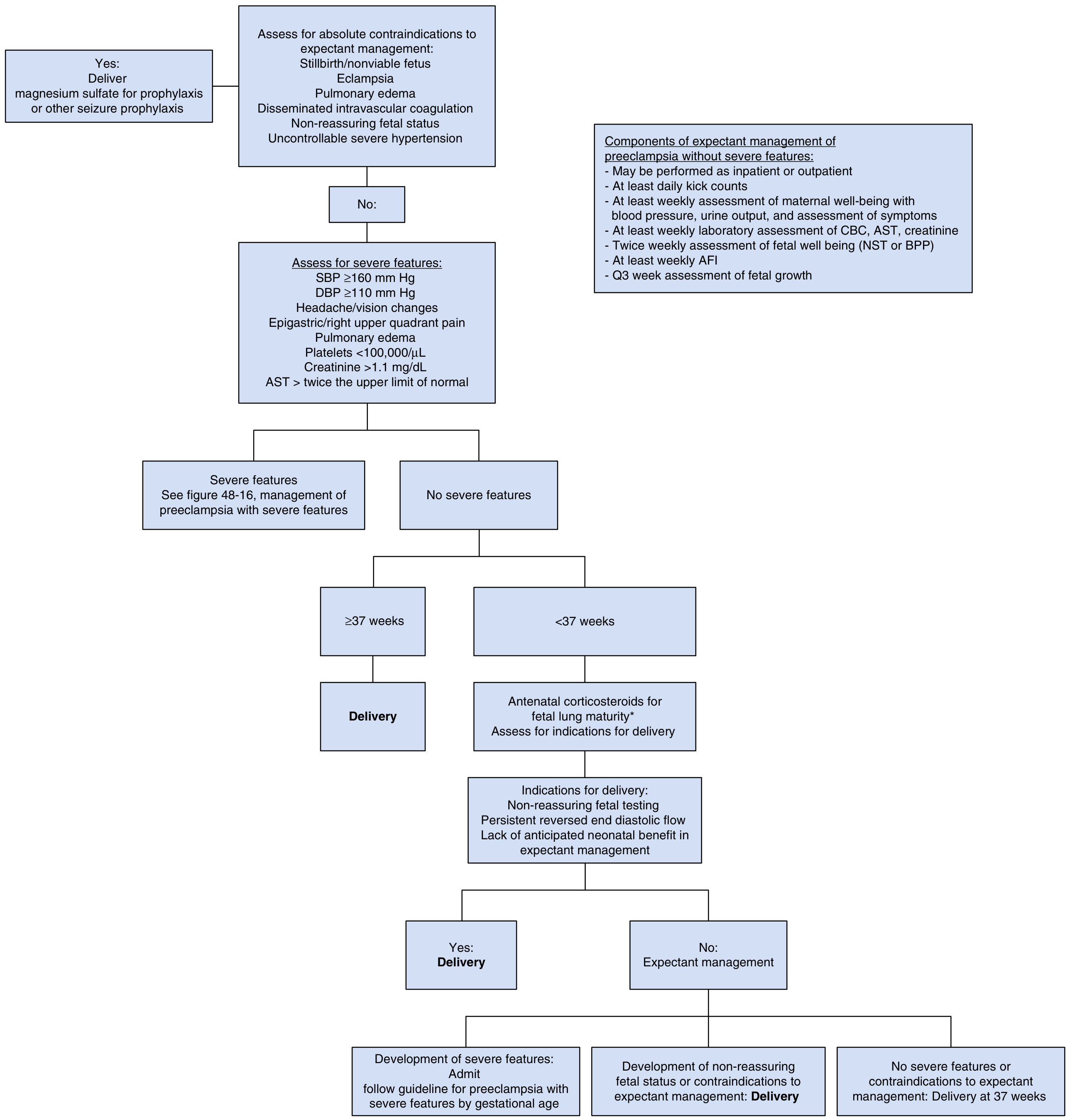
Modified from ACOG Task Force on Hypertension in Pregnancy — Creasy & Resnik's Maternal-Fetal Medicine
Key Decision Points
- Absolute indications for immediate delivery: Eclampsia, stillbirth/nonviable fetus, pulmonary edema, DIC, non-reassuring fetal status, uncontrollable severe hypertension
- ≥37 weeks + no severe features: Deliver
- <37 weeks + no severe features: Expectant management with antenatal corticosteroids if appropriate; monitor with at least weekly BP, labs (CBC, AST, creatinine), NST/BPP twice weekly, AFI weekly, growth scans every 3 weeks
- Delivery at 37 weeks if no complications arise
Seizure Prophylaxis
Magnesium sulfate is the agent of choice for eclampsia prophylaxis and treatment in severe preeclampsia.
Monitoring for Superimposed Preeclampsia
All women with chronic hypertension should be closely followed throughout pregnancy with:
- Serial BP measurements
- Urine protein monitoring
- Serum creatinine baseline and repeat testing
- Fetal growth scans (IUGR risk ~17%)
Postpartum Considerations
Women with gestational hypertension and preeclampsia are at increased long-term risk for hypertension and cardiovascular disease and should receive cardiovascular risk counselling postpartum.
Sources: Creasy & Resnik's Maternal-Fetal Medicine, 9e | Brenner & Rector's The Kidney, 11e | NKF Primer on Kidney Diseases, 8e | Comprehensive Clinical Nephrology, 7e
This is a shared conversation. Sign in to Orris to start your own chat.