Now I have comprehensive information to compose a detailed, well-illustrated answer. Let me build the complete response.
ICU Management of Mucus Overload with Respiratory Failure in a COPD Patient
Clinical Snapshot
- SpO₂: 74–80% → Severe hypoxemia (normal ≥ 95%)
- Findings: Bilateral rhonchi, alveolar mucus impaction
- Comorbidity: Hypertension
- Already given: Mucomelt (acetylcysteine) nebulisation + IV hydrocortisone
This is a severe acute exacerbation of COPD (AECOPD) with hypoxemic respiratory failure. The priority is: secure oxygenation → clear airways → relieve bronchospasm → treat underlying trigger.
STEP 1 — Airway Clearance: The Immediate Priority
🔬 Mechanism of Mucus Impaction in COPD
In COPD, excess mucus accumulates because:
- Goblet cell hyperplasia → overproduction of viscid mucus
- Disrupted mucociliary escalator → impaired clearance
- Inflammatory exudate floods alveoli → consolidation + rhonchi
- Disulfide bonds crosslink mucin glycoproteins → gel-like consistency
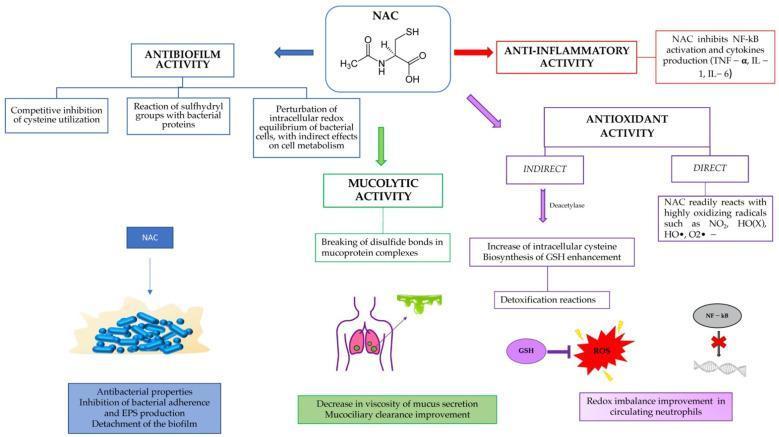
DRUG 1: N-Acetylcysteine (NAC / Mucomelt) — ✅ Already Given
Mechanism:
| Action | How it works |
|---|
| Mucolytic | Free sulfhydryl (–SH) groups break disulfide bonds in mucin glycoprotein complexes → reduces viscosity → improves mucociliary clearance |
| Antioxidant (Direct) | Directly scavenges free radicals: NO₂, HO•, O₂•⁻ |
| Antioxidant (Indirect) | Deacetylase → cysteine → glutathione (GSH) synthesis → protects alveolar epithelium |
| Anti-inflammatory | Inhibits NF-κB → suppresses TNF-α, IL-1, IL-6 |
| Antibiofilm | Disrupts bacterial biofilms; inhibits EPS production |
ICU Dosing:
- Nebulisation: 300 mg in 3 mL NS via jet nebuliser every 6–8 hours
- IV form (for paracetamol toxicity): 150 mg/kg loading dose
- Oral: 600 mg twice daily (maintenance)
Outcome: Decreases mucus viscosity, restores mucociliary escalator function, and clears alveolar impaction.
DRUG 2: Hydrocortisone (IV Corticosteroid) — ✅ Already Given
Mechanism:
Hydrocortisone binds to intracellular glucocorticoid receptors (GR-α) → GR-α/ligand complex translocates to nucleus → binds glucocorticoid response elements (GRE) on DNA:
Hydrocortisone → GR-α complex → Nucleus
↓
Trans-activation: Trans-repression:
Annexin-1 (anti-inflammatory) NF-κB ↓ → IL-1, IL-6, IL-8, TNF-α ↓
MAPK phosphatase-1 AP-1 ↓ → MMP-9 ↓ (less tissue damage)
β₂-adrenoceptor upregulation Mucin gene MUC5AC expression ↓
Clinical effects in AECOPD:
- Reduces airway oedema → less obstruction
- Decreases eosinophilic inflammation → faster FEV₁ recovery
- Reduces treatment failure rate and length of hospital stay
- Upregulates β₂-receptor sensitivity (synergy with bronchodilators)
ICU Dosing:
- IV Hydrocortisone: 100–200 mg every 6–8 hours (or methylprednisolone 1–2 mg/kg/day for 5 days)
- Do NOT use >7–14 days → myopathy, hyperglycaemia, hypertension risk
Important for your patient: Since he has hypertension, monitor BP carefully — mineralocorticoid effects of hydrocortisone can cause sodium retention and BP rise. Consider switching to methylprednisolone (no mineralocorticoid effect) after stabilisation.
STEP 2 — Oxygen Therapy (SpO₂ 74–80% → Life-threatening)
Target: SpO₂ 88–92% in COPD (NOT 100% — hypercapnic drive risk)
| Device | FiO₂ | Use |
|---|
| Venturi mask (24–28%) | 0.24–0.28 | First-line controlled O₂ in COPD |
| Non-rebreather mask | Up to 0.85 | If SpO₂ < 80% — temporary |
| High-Flow Nasal Cannula (HFNC) | Up to 1.0 | Preferred in hypoxemic respiratory failure |
| NIV (BiPAP) | Titrated | Best choice in AECOPD with hypercapnia |
In this patient with SpO₂ 74–80%: Start with NIV (BiPAP) — IPAP 12–16 cmH₂O, EPAP 4–6 cmH₂O. NIV reduces work of breathing, recruits atelectatic alveoli, and improves mucus clearance through positive pressure.
STEP 3 — Bronchodilators (Critical — Must Add)
DRUG 3: Salbutamol (Albuterol) — Short-Acting β₂ Agonist (SABA)
Mechanism:
Salbutamol → β₂ receptor (Gs protein)
↓
Adenylyl cyclase activated
↓
cAMP ↑ → Protein Kinase A (PKA) activated
↓
Myosin Light Chain Kinase (MLCK) phosphorylated → INACTIVE
↓
Airway smooth muscle RELAXATION → Bronchodilation
(also: ↑ mucociliary beat frequency → mucus clearance)
Additional benefit: β₂ stimulation activates chloride channels → fluid secretion into airway lumen → lubricates mucus plug → eases expectoration.
ICU Dosing: 2.5 mg via nebuliser every 20 minutes × 3 doses, then every 1–4 hours
DRUG 4: Ipratropium Bromide — Short-Acting Muscarinic Antagonist (SAMA)
Mechanism:
Parasympathetic overactivity in COPD:
ACh → M₃ receptor on smooth muscle → bronchospasm + mucus secretion ↑
Ipratropium blocks M₃ receptors →
• Bronchodilation (smooth muscle relaxation)
• Reduced mucus secretion from submucosal glands
• Reduced cholinergic bronchospasm
ICU Dosing: 0.5 mg nebulised every 4–6 hours (combine with salbutamol in same nebuliser = Combivent/Duolin)
⚡ Salbutamol + Ipratropium combination is superior to either agent alone in acute COPD exacerbation and is the standard of care in ICU.
STEP 4 — Antibiotics (Treat Infectious Trigger)
Most AECOPD requiring ICU admission are triggered by bacterial infection (Haemophilus influenzae, Streptococcus pneumoniae, Moraxella catarrhalis, Gram-negatives in severe cases).
DRUG 5: Antibiotics
| Severity | Drug | Dose |
|---|
| Moderate AECOPD | Amoxicillin-clavulanate | 625 mg PO/IV TDS |
| Severe/ICU | Co-amoxiclav + Azithromycin OR | IV |
| Pseudomonas risk | Piperacillin-tazobactam + Ciprofloxacin | IV |
| Penicillin allergy | Levofloxacin 500 mg OD IV | 5–7 days |
STEP 5 — Additional ICU Drugs for COPD
DRUG 6: Aminophylline (IV Methylxanthine) — Second-line
Mechanism:
- Inhibits phosphodiesterase → cAMP ↑ → bronchodilation
- Adenosine receptor antagonism → reduced bronchoconstriction
- Improves diaphragmatic contractility (important in fatigued respiratory muscles)
- CNS respiratory centre stimulation → helps in hypercapnia
ICU Dosing: Loading dose 5 mg/kg IV over 30 min → Maintenance 0.5 mg/kg/hr infusion
Caution: Narrow therapeutic window (10–20 µg/mL). Monitor levels. Arrhythmia risk.
DRUG 7: Magnesium Sulfate (IV)
Mechanism:
- Natural calcium antagonist → smooth muscle relaxation → bronchodilation
- Inhibits mast cell degranulation → reduces histamine release
- Inhibits ACh release at neuromuscular junction → relaxes bronchial smooth muscle
ICU Dosing: 2 g IV over 20 minutes (single dose)
Use in: Severe bronchospasm not responding to β₂ agonists
DRUG 8: Carbocysteine / Erdosteine (Mucoregulators)
Mechanism:
- Carbocysteine: Stimulates serous cell secretion, thins mucus, restores the IgA content of bronchial secretions
- Erdosteine: Pro-drug → active metabolites with free thiol groups → breaks disulfide bonds (similar to NAC) + antioxidant + inhibits bacterial adhesion
Dosing: Carbocysteine 750 mg TDS orally; Erdosteine 300 mg BD
DRUG 9: Furosemide (If Fluid Overload Component)
In your patient with hypertension, there may be a component of pulmonary oedema compounding the mucus overload.
Mechanism: Loop diuretic → inhibits Na⁺/K⁺/2Cl⁻ co-transporter in Loop of Henle → diuresis → reduces pulmonary venous congestion → clears alveolar fluid
ICU Dosing: 40–80 mg IV stat, then reassess
DRUG 10: Doxapram (Respiratory Stimulant) — If NIV Fails
Mechanism: Stimulates peripheral chemoreceptors (carotid body) → increases respiratory drive → increases tidal volume and respiratory rate
Use: When NIV is unavailable or not tolerated, as bridge to prevent intubation
Dose: 1.5–4 mg/kg/hr IV infusion
INTERVENTIONAL / NON-PHARMACOLOGICAL ICU MANAGEMENT
| Intervention | Mechanism/Purpose |
|---|
| Chest physiotherapy (percussion + postural drainage) | Mechanically mobilises mucus from peripheral airways to central airways for suction/expectoration |
| Endotracheal suctioning | Direct removal of mucus via ETT if intubated |
| Bronchoscopy with BAL | In refractory mucus plugging — direct washout of alveolar secretions |
| Mucolytic nebulisation | Hypertonic saline (3–7%) increases osmotic gradient → draws fluid into mucus → reduces viscosity |
| Hydration | IV fluids maintain mucociliary clearance; dehydration thickens mucus |
| Positioning | Semi-recumbent 30–45° → reduces aspiration, improves diaphragmatic excursion |
Complete ICU Drug Regimen Summary
| Priority | Drug | Route | Mechanism |
|---|
| 🔴 Immediate | Controlled O₂ + NIV (BiPAP) | Mask | Oxygenation + alveolar recruitment |
| 🔴 Immediate | Salbutamol 2.5 mg + Ipratropium 0.5 mg | Nebuliser | Bronchodilation (β₂ + M₃ block) |
| 🔴 Immediate | NAC (Mucomelt) 300 mg | Nebuliser | Mucolytic — breaks disulfide bonds |
| 🟠 Urgent | Hydrocortisone/Methylprednisolone IV | IV | Anti-inflammatory, oedema reduction |
| 🟠 Urgent | Antibiotics (co-amoxiclav ± azithromycin) | IV | Treat bacterial trigger |
| 🟡 Adjunct | Aminophylline | IV infusion | Bronchodilation + diaphragm support |
| 🟡 Adjunct | Magnesium sulfate 2 g | IV | Bronchodilation (refractory spasm) |
| 🟡 Adjunct | Carbocysteine/Erdosteine | Oral | Mucoregulatory, maintenance |
| 🟡 Adjunct | Furosemide (if fluid overload) | IV | Decongest pulmonary circulation |
| ⚪ If needed | Doxapram | IV infusion | Respiratory stimulant (NIV failure) |
COPD Exacerbation-Reducing Interventions (Evidence-Based)
As shown above from the GOLD 2025 Report, long-term prevention of exacerbations includes:
- LABAs (salmeterol, formoterol) + LAMAs (tiotropium, glycopyrronium)
- ICS + LABA combinations (budesonide/formoterol)
- Triple therapy (LABA + LAMA + ICS) for high-risk patients
- Roflumilast (PDE4 inhibitor) — for chronic bronchitis phenotype
- Mucoregulators: NAC, carbocysteine, erdosteine (reduce exacerbation frequency)
- Macrolides (azithromycin 250 mg OD) — long-term anti-inflammatory
Hypertension Consideration
Since the patient has hypertension:
- Avoid high-dose salbutamol without monitoring → tachycardia, BP fluctuation
- Prefer methylprednisolone over hydrocortisone (no mineralocorticoid effect → less sodium retention)
- Magnesium sulfate is actually beneficial — mild antihypertensive effect
- Avoid aminophylline toxicity → arrhythmias worsen hypertensive cardiac risk
Summary: The cornerstone of ICU management in this patient is the combination of NIV for oxygenation + salbutamol/ipratropium for bronchospasm + NAC nebulisation for mucolysis + systemic corticosteroids for inflammation + antibiotics for infection. Chest physiotherapy and bronchial hygiene should run in parallel. Mucomelt (NAC) was correctly initiated and works by cleaving the disulfide crosslinks in mucin — this reduces gel viscosity and restores mucociliary clearance, directly targeting the alveolar mucus overload described.