NEURO MUSCULAR MONITORING
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive information from the textbooks. Let me compile a thorough response.
Neuromuscular Monitoring
Why It Matters
Neuromuscular blocking drugs (NMBDs) do not alter consciousness or cause amnesia — a paralyzed patient can be awake and aware. Residual neuromuscular blockade after emergence occurs in approximately 30–40% of patients reversed with neostigmine and up to 5% with sugammadex. This renders monitoring critical to patient safety.
Key risks of residual block include:
- Impaired pharyngeal function and airway obstruction
- Increased risk of aspiration
- Impaired hypoxic ventilatory control
- Unpleasant symptoms of muscle weakness (diplopia, facial weakness, dysphagia)
- Prolonged PACU length of stay
"Quantitative neuromuscular monitoring is the only method of determining whether full recovery of muscular function has occurred and whether pharmacologic reversal can be safely avoided." — Miller's Anesthesia, 10e
Methods of Monitoring
1. Clinical / Time-Based Assessment (Unreliable)
Historically used but now discouraged:
- Time since last NMBD dose
- Physical tests: head lift ×5 seconds, handgrip, tidal volume, vital capacity, tongue protrusion, eye opening
Limitation: The 5-second head lift was accomplished by 11/12 volunteers despite a TOF ratio ≤0.5. These assessments are too insensitive to detect clinically significant residual paralysis.
2. Peripheral Nerve Stimulators (Qualitative Monitoring)
Battery-operated devices delivering a square-wave current (10–80 mA, 100–300 µs duration). The response is assessed visually or by palpation.
Common stimulation patterns:
| Pattern | Description | Clinical Use |
|---|---|---|
| Single twitch | Single supramaximal stimulus at 0.1–1 Hz | Baseline comparison |
| Train-of-Four (TOF) | 4 stimuli at 2 Hz, every 0.5 s | Most common; assess fade & count |
| Tetanic stimulation | 50–100 Hz for 5 s | Detect partial block; painful in awake patients |
| Post-tetanic count (PTC) | Count of twitches after tetanus | Deep/profound block (no TOF response) |
| Double-burst stimulation (DBS) | 2 short tetanic bursts separated by 750 ms | More sensitive than TOF for tactile fade detection |
Preferred site: Ulnar nerve at the wrist → adductor pollicis (thumb adduction). Negative (black) electrode placed distally, positive proximally.
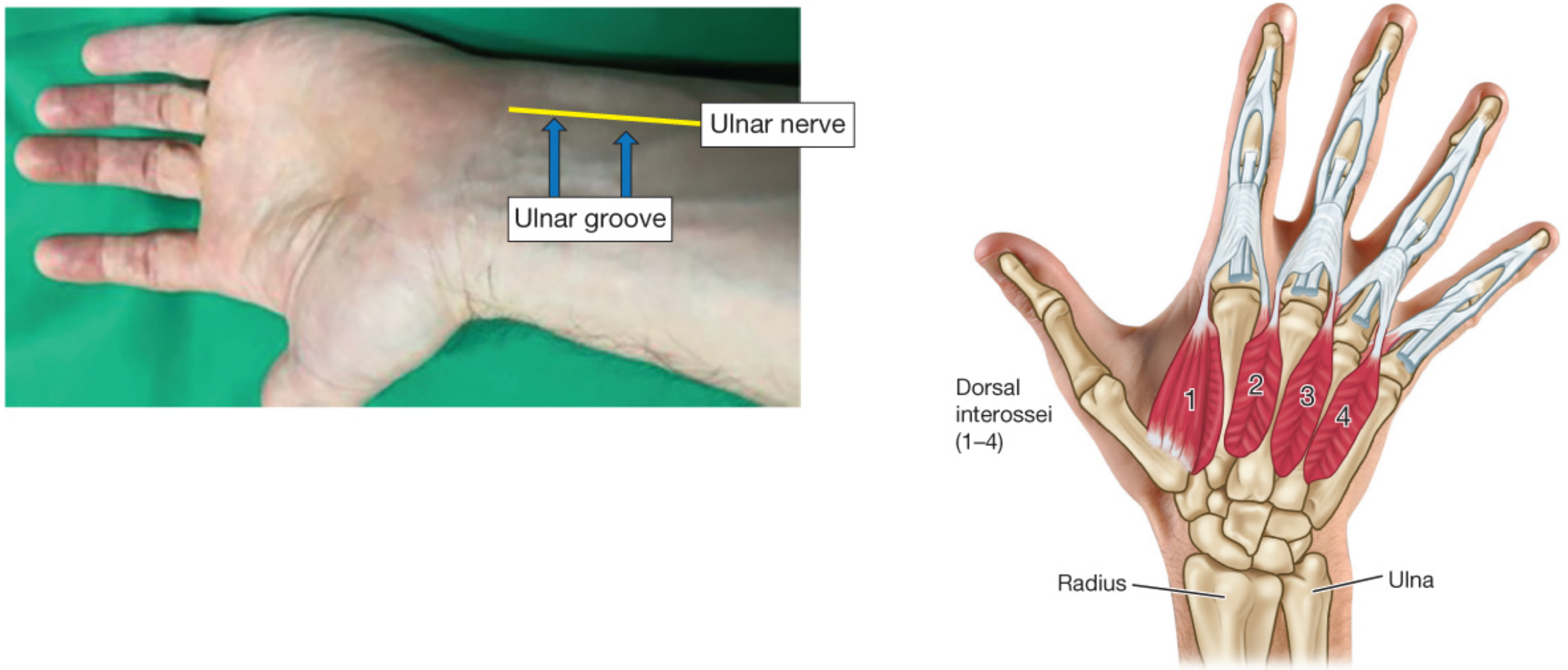
Critical limitation: Clinicians are unable to subjectively detect fade when TOF ratios exceed 0.30–0.40. Qualitative monitoring cannot exclude residual paralysis.
3. Quantitative Monitoring (Objective) — Gold Standard
Measures the evoked muscle response with precision rather than subjective feel or vision.
Technologies:
| Technology | Mechanism | Notes |
|---|---|---|
| Mechanomyography (MMG) | Measures force of muscle contraction | Reference standard; impractical clinically |
| Electromyography (EMG) | Measures compound muscle action potential | Close to MMG; EMG values may be slightly higher than MMG |
| Acceleromyography (AMG) | Measures acceleration of thumb movement (piezoelectric) | Most widely used clinically; AMG TOFR may be 5–10% higher than MMG |
| Kinemography (KMG) | Measures movement using a bending sensor | Slightly higher values than MMG |
| TOF-Cuff | Pneumatic cuff measures pressure from muscle contraction | Higher values vs. MMG (~4.7%) |
AMG bias note: Because AMG tends to overestimate the TOF ratio, a threshold of TOF ratio ≥ 0.9 (with normalized AMG) or higher may be needed before extubation.
Bias summary (versus MMG reference at TOFR 0.9):
| Technology | Bias | Adjusted Goal TOFR |
|---|---|---|
| MMG | +2% | 0.92 |
| EMG | −4.7% | 0.85 |
| AMG (various studies) | −8 to +10% | 0.82–0.99 |
| TOF-Cuff | +4.7% | 0.95 |
TOF Ratio Interpretation
| TOF Ratio | Clinical Significance |
|---|---|
| < 0.40 | Profound-to-deep block; tactile fade detectable |
| 0.40–0.70 | Partial block; insensitive clinical signs begin to disappear |
| 0.70–0.90 | Residual paralysis; pharyngeal dysfunction, aspiration risk |
| ≥ 0.90 | Threshold for extubation (with quantitative monitor) |
| ≥ 1.0 | Full recovery by quantitative measure |
At TOF ratios < 0.90, awake volunteers exhibit impaired pharyngeal function, airway obstruction, and increased aspiration risk even though they may appear clinically recovered. — Miller's Anesthesia, 10e
Depth of Block Classification
| Depth | PTC | TOF Count | TOF Ratio |
|---|---|---|---|
| Profound block | 0 | 0 | — |
| Deep block | 1–10 | 0 | — |
| Moderate block | — | 1–3 | — |
| Shallow block | — | 4 | < 0.90 |
| Minimal block | — | 4 | > 0.90 |
| Full recovery | — | 4 | ≥ 0.90–1.0 |
Clinical Practice Recommendations
- Quantitative monitoring should be used whenever NMBDs are administered — subjective assessment alone is insufficient.
- Monitoring guides dosing of NMBDs intraoperatively and reversal drug selection and dosing at end of surgery.
- Neostigmine is most effective for reversal of minimal block (TOF count 4, minimal fade); it is unreliable for deep block.
- Sugammadex can reverse moderate-to-deep block but quantitative monitoring is still needed to confirm recovery.
- Time elapsed since last NMBD dose is not a reliable substitute for monitoring — up to 37% of patients have TOF ratio < 0.90 two or more hours after a single intubating dose of intermediate-duration NMBD.
- International guidelines from Canada, France, Spain, Australia, New Zealand, Czech Republic, Portugal, Japan, and the USA all recommend quantitative monitoring.
Summary
| Method | Detects fade at TOFR | Objective? | Clinical value |
|---|---|---|---|
| Clinical assessment | ≤ 0.50 | No | Low (unreliable) |
| Peripheral nerve stimulator (qualitative) | ≤ 0.30–0.40 | No | Moderate (better than clinical alone) |
| Quantitative (AMG/EMG) | Any level | Yes | High — required for safe extubation |
— Barash Clinical Anesthesia 9e, Miller's Anesthesia 10e
Definition and physiology
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Excellent — I now have all the physiological detail needed for a comprehensive answer.
Neuromuscular Monitoring — Definition & Physiology
Definition
Neuromuscular monitoring is the objective or subjective assessment of the evoked muscle response to peripheral nerve stimulation, used to quantify the degree of neuromuscular blockade produced by neuromuscular blocking drugs (NMBDs) at the neuromuscular junction (NMJ). Its purpose is to:
- Guide intraoperative dosing of NMBDs
- Determine the depth of block for optimal surgical conditions
- Confirm adequate recovery of neuromuscular function before tracheal extubation
- Guide timing and dosing of reversal agents
"Good evidence-based practice dictates that clinicians always quantitate the extent of neuromuscular block by objective monitoring." — Miller's Anesthesia, 10e
Physiology of the Neuromuscular Junction
1. Anatomy of the NMJ
The neuromuscular junction is the specialized synapse between a motor nerve terminal and the skeletal muscle fiber. Key structural components:
| Component | Description |
|---|---|
| Motor nerve terminal (presynaptic) | Unmyelinated terminal of the lower motor neuron; contains synaptic vesicles loaded with acetylcholine (ACh) |
| Synaptic cleft | ~20–50 nm gap between nerve terminal and motor endplate |
| Motor endplate (postsynaptic) | Thickened region of the muscle membrane with deep junctional folds concentrating nicotinic ACh receptors (nAChRs) at the crests |
| Acetylcholinesterase (AChE) | Enzyme concentrated in the synaptic cleft and junctional folds; rapidly hydrolyzes ACh |
Each terminal fiber of a lower motor neuron innervates a single muscle fiber. The motor cortex → upper motor neurons → lower motor neurons (ventral horn of spinal cord) → ventral roots → muscle endplate.
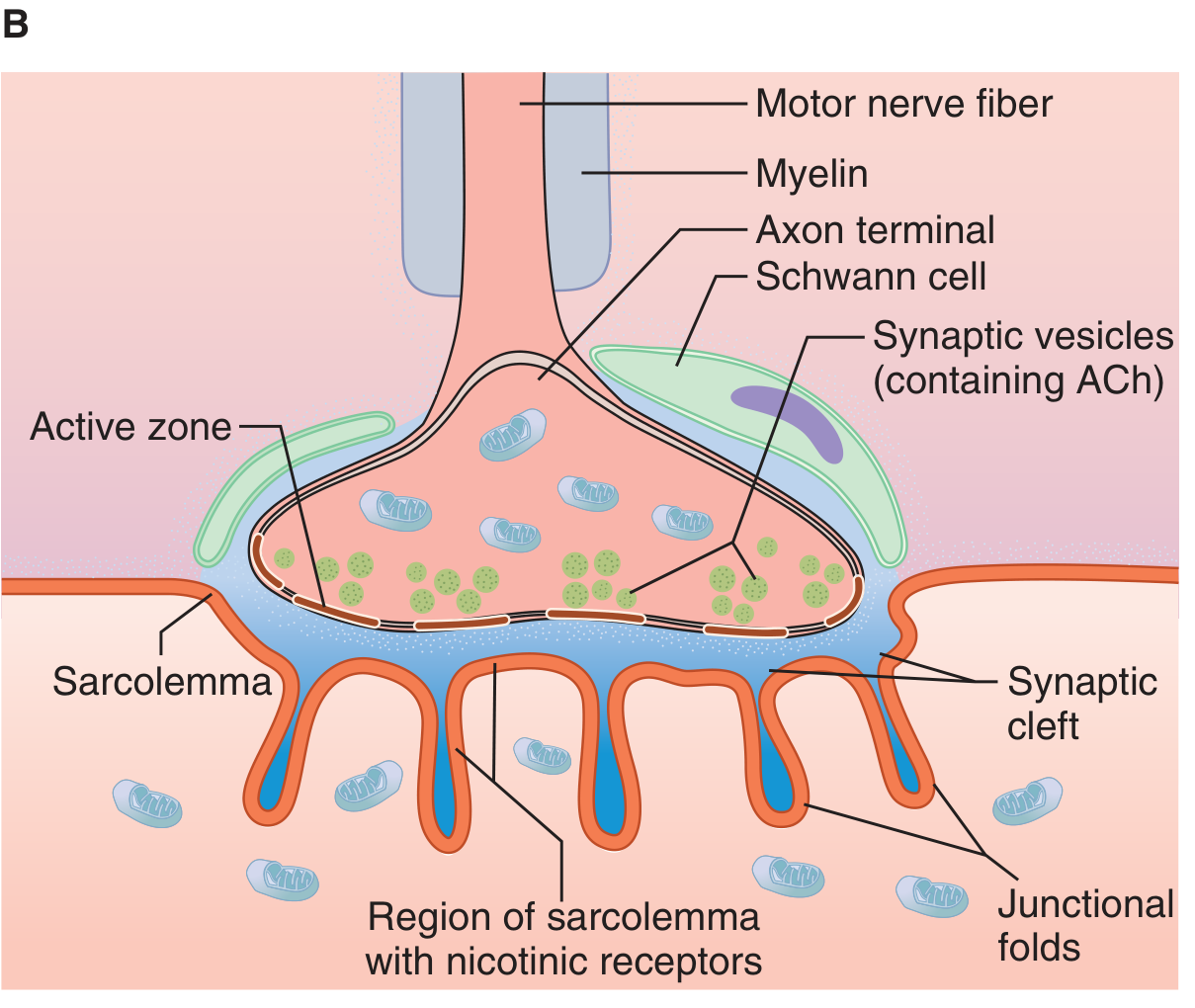
2. Sequence of Events: Nerve Impulse → Muscle Contraction
- Action potential propagates down the myelinated lower motor neuron; becomes unmyelinated at the terminal
- Depolarization of the nerve terminal opens voltage-gated Ca²⁺ channels
- Ca²⁺ influx triggers exocytosis of ACh-containing synaptic vesicles into the synaptic cleft
- ACh diffuses across the cleft and binds to nicotinic cholinergic receptors (N_M type) concentrated at the tops of the junctional folds
- Receptor activation → increased Na⁺ and K⁺ conductance → Na⁺ influx → endplate potential (EPP)
- The EPP depolarizes the adjacent muscle membrane to its firing threshold
- Action potentials propagate in both directions along the muscle fiber → excitation-contraction coupling → muscle contraction
- ACh hydrolysis: AChE rapidly breaks down ACh into acetate and choline, terminating the signal; choline is re-taken up for ACh re-synthesis
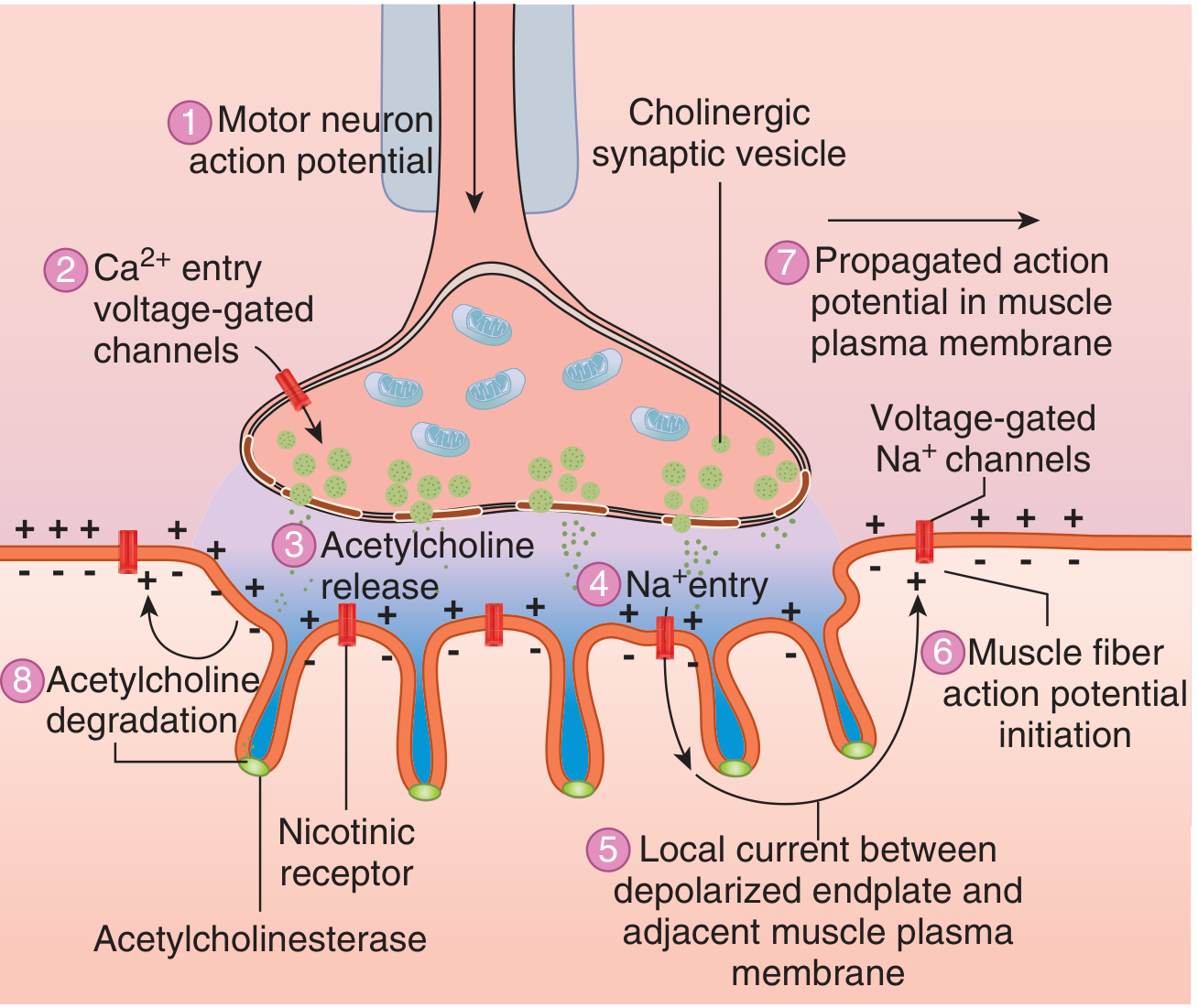
3. The Nicotinic Acetylcholine Receptor (nAChR)
The postsynaptic nAChR is a ligand-gated ion channel with a pentameric structure:
- Mature (junctional) receptor: α₁α₁βδε subunits — located at the motor endplate; requires binding of 2 ACh molecules (one at each α subunit) for activation
- Immature (extrajunctional) receptor: α₁α₁βδγ subunits — expressed throughout the entire muscle membrane in fetal tissue or pathological states (denervation, burns, immobilization)
Clinical relevance of immature receptors:
- Extrajunctional receptors have prolonged channel opening times and increased sensitivity to depolarizing NMBDs
- If succinylcholine is administered ≥24 hours after denervation injury (spinal cord injury, severe burns, stroke, Guillain-Barré, prolonged ICU immobilization), massive K⁺ efflux through these receptors → life-threatening hyperkalemia
Presynaptic receptors (α₃β₂ nAChR):
- Located on the nerve terminal
- Nondepolarizing NMBDs bind here → reduce mobilization of ACh vesicles → contribute to the fade phenomenon (progressive decrease in twitch height during sustained or repetitive stimulation)
4. Receptor Occupancy and Stimulation Response
| % Receptor Occupancy (by NMBD) | Clinical Effect |
|---|---|
| < 70–75% | No visible twitch depression |
| ~80% | Single twitch begins to decrease |
| ~90% | Significant twitch suppression |
| ~95% | Twitch abolished (clinical paralysis) |
| ~70–75% | TOF fade begins to appear |
Because of the large margin of safety at the NMJ (only ~25% of receptors need to be free for normal twitch), significant receptor occupancy can exist with no clinical signs of weakness — the basis for residual neuromuscular block.
5. Mechanism of Neuromuscular Blockade
Depolarizing Block (Phase I) — Succinylcholine
- Structurally resembles ACh (two ACh molecules linked by methyl groups)
- Binds and activates both postsynaptic and extrajunctional nAChRs → sustained depolarization (fasciculations followed by flaccid paralysis)
- NOT hydrolyzed by AChE; metabolized by pseudocholinesterase (plasma cholinesterase) — slower degradation than ACh → prolonged depolarization
- The continuously depolarized membrane cannot respond to subsequent ACh release → paralysis
- At TOF stimulation: no fade (all 4 twitches equally reduced)
Nondepolarizing Block — Rocuronium, Vecuronium, Cisatracurium, etc.
- Bind to one or both α subunits of the nAChR without activating the ion channel — competitive antagonism
- Also block presynaptic α₃β₂ receptors → impair ACh mobilization → fade on repetitive stimulation
- At TOF stimulation: fade is present (T4 < T3 < T2 < T1)
- Reversed by: anticholinesterases (neostigmine) or selective binding agents (sugammadex)
6. Why Interpatient Variability Demands Monitoring
There is extraordinary variability between patients in response to all NMBDs. In a classic study of 100 patients given 0.1 mg/kg curare: 6 patients showed no twitch depression; 7 had complete abolishment; the rest showed variable depression — unpredictable by age, sex, weight, or ASA class.
In a cohort of 526 patients receiving an equipotent dose of rocuronium, atracurium, or vecuronium (no reversal, no additional doses): the degree of spontaneous recovery at arrival to PACU was entirely unpredictable. Time alone cannot substitute for monitoring.
Summary: The Physiological Basis of NMJ Monitoring
Motor neuron action potential
↓
Voltage-gated Ca²⁺ channel opens at nerve terminal
↓
ACh vesicle exocytosis → ACh into synaptic cleft
↓
ACh binds 2 α subunits of postsynaptic nAChR
↓
Na⁺/K⁺ channel opens → Na⁺ influx → Endplate Potential
↓
Muscle membrane depolarizes → Action potential → Contraction
↓
AChE hydrolyzes ACh → channel closes → repolarization
NMBDs interrupt this pathway — monitoring the evoked muscle response to a supramaximal peripheral nerve stimulus tells us how deeply this pathway is blocked.
— Barash Clinical Anesthesia 9e, Ganong's Review of Medical Physiology 26e, Miller's Anesthesia 10e
PHYSIOLOGICAL CHANGES THAT OCCUR DURING POSITIVE PRESSURE VENTILATION, EFFECTS OF PPV ON FLUID BALANCE
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive, authoritative content from multiple textbooks to construct a thorough answer.
Physiological Changes During Positive Pressure Ventilation (PPV) & Effects on Fluid Balance
Comparison: Spontaneous vs. Positive Pressure Breathing
| Parameter | Spontaneous Breathing | Positive Pressure Ventilation |
|---|---|---|
| Inspiratory mechanism | Active (diaphragm/intercostals contract) → negative intrathoracic pressure | Passive — gas driven into lungs under positive pressure |
| Intrathoracic pressure during inspiration | Falls (negative) | Rises (positive) |
| Venous return | Augmented | Diminished |
| Cardiac preload | Increased | Decreased |
| Cardiac output | Increased | Decreased |
| Pressure gradient LV → Aorta | Increased | Decreased |
| Diaphragmatic movement | Greatest in dependent zones (optimising V/Q) | Uniform, non-dependent distribution |
| Exhalation | Passive (recoil) | Passive (recoil) |
"With the initiation of positive-pressure ventilation (PPV), the opposite occurs: venous return is diminished, cardiac output falls, and there is a decreased pressure gradient between the left ventricle and aorta." — Rosen's Emergency Medicine, 9e
I. Cardiovascular Effects
A. Reduced Venous Return and Preload
- PPV increases mean intrathoracic pressure, which diminishes the venous pressure gradient from peripheral capillaries to the right atrium
- Normally this gradient is relatively small (~5–8 mmHg), so even modest increases in intrathoracic pressure significantly impair right heart filling
- PEEP further amplifies mean intrathoracic pressure → greater ventricular preload reduction
- Conditions with low lung compliance (asthma, obesity, ascites, ARDS, light anesthesia) exaggerate this effect
B. Reduced Cardiac Output
- Decreased RV preload → decreased RV stroke volume → decreased LV filling → decreased cardiac output
- Hypotension is common after initiation of PPV, especially in:
- Hypovolemic patients
- Vasodilatory states (sepsis, anaphylaxis)
- Patients taking diuretics or ACE inhibitors
- Cyanotic heart disease (preload-dependent)
C. Effect on Left Ventricular Afterload
- Increased intrathoracic pressure reduces LV transmural pressure (the effective afterload on the LV)
- This is actually beneficial in LV failure — PPV can improve LV ejection in cardiogenic pulmonary oedema by reducing afterload
II. Pulmonary Effects
A. Lung Volumes and V/Q Matching
- Under general anaesthesia with spontaneous breathing: reduced tidal volume, reduced FRC, increased closing volumes → V/Q mismatch and atelectasis
- PPV with PEEP counters atelectasis, improves minute ventilation, and reduces intrapulmonary shunt
- However, PPV distributes ventilation to non-dependent lung zones, while perfusion remains gravity-dependent → potential V/Q mismatch
B. Alveolar Pressures
- Peak inspiratory pressure (PIP) and plateau pressure are monitored as surrogates of lung stretch
- Excessive pressures → barotrauma (pneumothorax, pneumomediastinum) and volutrauma
- Exhalation is passive via chest wall recoil when ventilator pressure is removed
III. Effects on Fluid Balance
PPV impairs fluid balance via three interacting mechanisms: haemodynamic, neurohumoral, and direct renal effects.
A. Haemodynamic Mechanism
- ↑ Intrathoracic pressure → ↑ Inferior vena cava (IVC) pressure → ↑ Renal venous pressure
- Elevated renal venous pressure → ↑ peritubular capillary pressure → increased tubular sodium reabsorption → antinatriuresis
- ↓ Cardiac output → ↓ Systemic arterial pressure → baroreceptor-mediated ↑ sympathetic tone to the kidney → renal vasoconstriction, ↓ renal blood flow (RBF), ↓ GFR, ↓ urine output
B. Neurohumoral Mechanism (the dominant pathway)
1. Atrial Natriuretic Peptide (ANP) Suppression
- Decreased cardiac filling → decreased atrial stretch → decreased ANP secretion
- ANP normally promotes natriuresis and diuresis; its suppression → salt and water retention
- Decreased ANP also leads to ↑ sympathetic tone and ↑ renin activation
2. Renin–Angiotensin–Aldosterone System (RAAS) Activation
- ↓ Renal perfusion pressure + ↑ sympathetic tone → ↑ Renin release → ↑ Angiotensin II → ↑ Aldosterone
- Aldosterone → ↑ sodium reabsorption and ↑ potassium excretion in distal tubule
- With PEEP, renin and aldosterone levels are further elevated proportional to the degree of haemodynamic compromise
3. ADH (Vasopressin / AVP) Release
- Decreased atrial stretch → impulses project from atrial stretch receptors → hypothalamus → ↑ ADH synthesis and release
- PPV is a recognised nonosmotic stimulus for ADH secretion
- ADH → ↑ water reabsorption in collecting tubules → oliguria and water retention
- This results in a syndrome resembling SIADH:
"Positive-pressure ventilation of the lungs... can promote the release of ADH." — Barash Clinical Anesthesia, 9e "Several chronic, nonmalignant pulmonary conditions, including positive-pressure ventilation, impede venous return... decreased atrial stretch increases AVP release... resulting in a syndrome of inappropriate AVP release — SIADH." — Medical Physiology (Boron & Boulpaep)
C. Summary: Net Renal Effects of PPV
| Parameter | Effect of PPV |
|---|---|
| Renal blood flow (RBF) | Decreased |
| GFR | Decreased |
| Urine output | Decreased (oliguria) |
| Sodium excretion (FENa) | Decreased (antinatriuresis) |
| ADH | Increased (water retention) |
| RAAS | Activated (Na⁺ and water retention) |
| ANP | Decreased (less natriuresis) |
"The renin–angiotensin–aldosterone system undoubtedly augments the renal responses to positive pressure ventilation. An increase in PEEP can depress cardiac output, RBF, GFR, and urine volume, and increase renin and aldosterone." — Miller's Anesthesia, 10e
D. Effect of PEEP on Fluid Balance
- PEEP further increases mean airway and intrathoracic pressure beyond PPV alone
- Each additional cmH₂O of PEEP worsens haemodynamic compromise → greater RAAS activation and ADH release
- The magnitude of renal depression correlates with mean airway pressure, not the specific ventilator mode (no difference between volume-controlled and pressure support in creatinine clearance and FENa)
E. Clinical Consequences
- Intraoperative oliguria — may occur regardless of IV fluid administered, due to ADH secretion and PPV effects combined with surgical stress; not automatically a sign of hypovolaemia
- Positive fluid balance — water and sodium retention during PPV contributes to perioperative fluid accumulation
- Fluid overload risk — aggressive fluid resuscitation during PPV can cause dangerous positive fluid balance; in AKI, positive fluid balance is independently associated with increased mortality
- Post-extubation diuresis — return to negative intrathoracic pressure with spontaneous breathing restores venous return, increases ANP, suppresses ADH → brisk diuresis often follows extubation
- Fluid responsiveness assessment — PPV enables the use of dynamic indices: respiratory variation in pulse pressure (PPV), stroke volume variation (SVV), and IVC collapsibility to predict preload responsiveness
"The suppression of renal urine production is related to perioperative ADH secretion and may also be influenced by the effects of positive-pressure ventilation. Increased intrathoracic pressure reduces venous return and cardiac output, which combine with a variety of neurohumoral responses such as sympathetic activation and suppression of ANP release to decrease GFR and urine output. As a result, intraoperative urine output may be low regardless of the volume of IV fluid administered." — Miller's Anesthesia, 10e
Summary Diagram
PPV / PEEP
↓
↑ Mean Intrathoracic Pressure
├──→ ↓ Venous Return → ↓ RV preload → ↓ CO → ↓ BP
│ └──→ Baroreceptors → ↑ Sympathetic → Renal vasoconstriction
│ ↓ RBF, ↓ GFR, ↓ UO
│
├──→ ↑ IVC pressure → ↑ Renal venous pressure → ↑ Na⁺ reabsorption
│
└──→ ↓ Atrial stretch
├──→ ↓ ANP → Na⁺ / water retention
├──→ ↑ Renin → ↑ Angiotensin II → ↑ Aldosterone → Na⁺ retention
└──→ ↑ ADH (AVP) → Water retention → Oliguria / SIADH-like state
— Miller's Anesthesia 10e, Rosen's Emergency Medicine 9e, Barash Clinical Anesthesia 9e, Boron & Boulpaep Medical Physiology, Comprehensive Clinical Nephrology 7e
DESCRIBE THERMOREGULATION UNDER ANESTHESIA. DISCUSS IMPACT OF PERIOPERATIVE HYPOTHERMIA
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive material. Let me compile a thorough, well-structured answer.
Thermoregulation Under Anaesthesia & Impact of Perioperative Hypothermia
Part 1: Normal Thermoregulation
The hypothalamus maintains core body temperature within a very narrow interthreshold range — normally between 36°C and 38°C. This range has two extremes:
| Trigger | Autonomic Response |
|---|---|
| Core temp rises slightly above upper threshold | Sweating + cutaneous vasodilation (heat loss) |
| Core temp falls slightly below lower threshold | Peripheral vasoconstriction + shivering (heat conservation/generation) |
The interthreshold range in a healthy, unanesthetized adult is approximately 0.2–0.4°C — remarkably tight. Any temperature within this range requires no autonomic thermoregulatory response.
Part 2: Thermoregulation Under Anaesthesia
A. How Anaesthetics Impair Thermoregulation
Both general and regional anaesthetics widen the interthreshold range to approximately 2–4°C, effectively impairing the hypothalamic thermostat:
General anaesthetics:
- Impair hypothalamic thermoregulatory reflex integration
- Volatile agents: Produce a concentration-dependent reduction in the vasoconstriction threshold — e.g., isoflurane decreases the vasoconstriction trigger temperature by ~3°C per 1% inhaled concentration
- Shift both the sweating threshold upward and the shivering/vasoconstriction threshold downward → the body tolerates a much wider temperature range without responding
- Neuromuscular blocking agents (paralytics) abolish shivering entirely, removing the only significant heat-generating response
Regional/neuraxial anaesthesia (spinal/epidural):
- Blocks efferent sympathetic fibres → prevents compensatory vasoconstriction in the anaesthetized dermatomes
- Blocks afferent thermal sensory input from the lower body → the hypothalamus receives falsely normal temperature information (altered perception of temperature from anaesthetised dermatomes)
- This miscommunication allows continued heat loss without triggering corrective responses
- Lowers the shivering threshold and vasoconstrictive response to hypothermia
Combined general + regional anaesthesia:
- Effects are roughly additive — the greatest degree of thermoregulatory impairment
- Particularly relevant given increasing use of regional anaesthesia in modern ERAS pathways
"General anesthesia lowers the cold-response threshold of the body. Volatile and intravenous anesthetics impair thermoregulation, paralytics prevent the body's shivering response to hypothermia, and compensatory vasoconstriction is downregulated." — Sabiston Textbook of Surgery, 7e
B. The Three Phases of Heat Loss Under Anaesthesia
When no active warming is applied, core temperature follows a characteristic three-phase pattern:
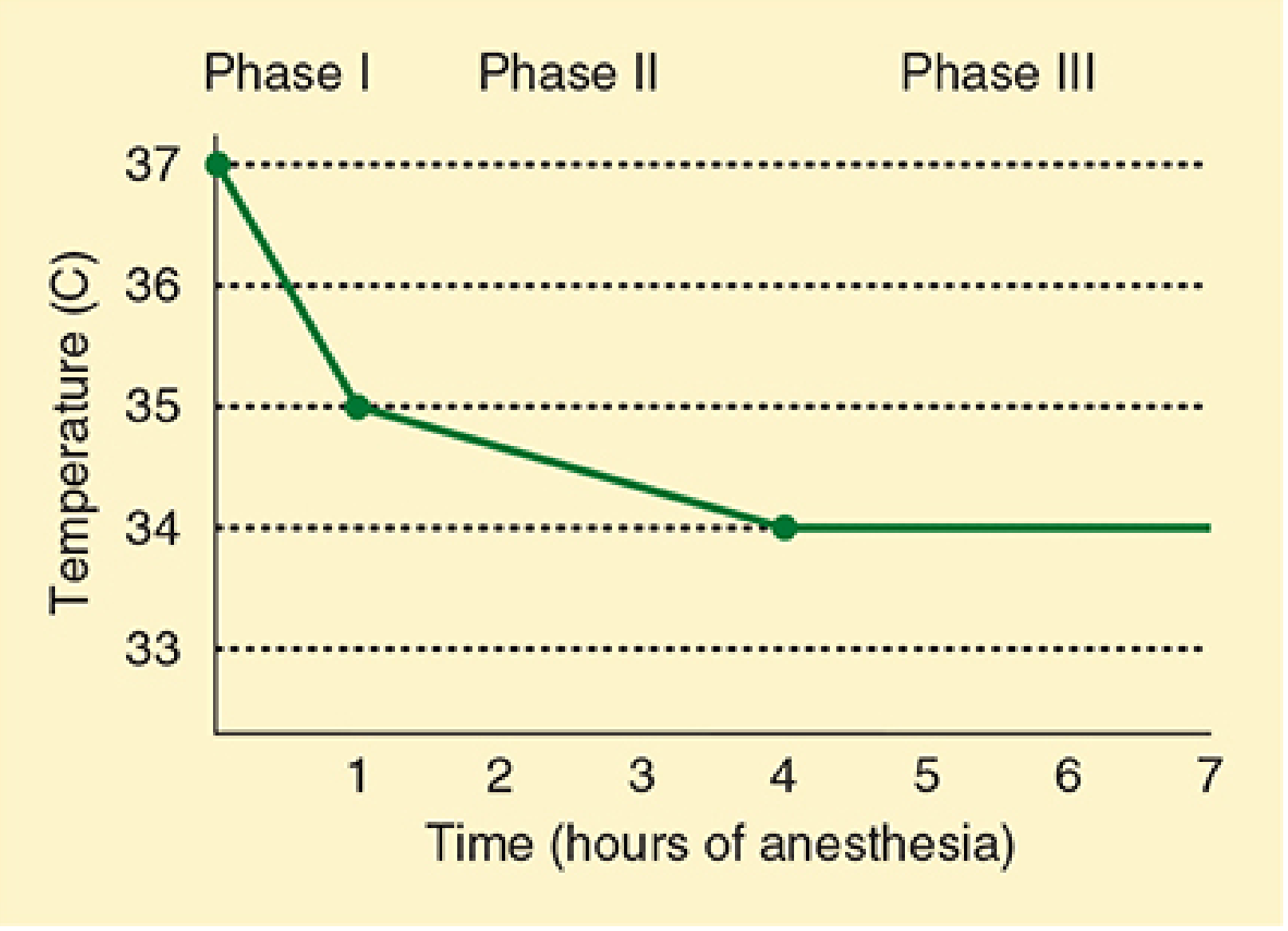
| Phase | Time | Mechanism | Magnitude |
|---|---|---|---|
| Phase 1: Redistribution | First 60 minutes | Anaesthesia-induced vasodilation → rapid transfer of heat from warm central compartments (abdomen, thorax) to cooler peripheral tissues (arms, legs) | 1–2°C drop |
| Phase 2: Continuous heat loss | Next 3–4 hours | Ongoing heat dissipation to the environment exceeds metabolic heat production | Slower, gradual decline |
| Phase 3: Steady state | After 3–5 hours | Heat loss equals metabolic heat production; vasoconstriction (if still functional) limits further loss | Temperature plateau |
Key point on Phase 1: The initial fall is primarily from internal redistribution, not environmental loss. Prewarming for 30 minutes with forced-air blankets before induction reduces this by diminishing the central–peripheral temperature gradient.
C. Environmental and Surgical Factors Contributing to Heat Loss
| Factor | Mechanism |
|---|---|
| Cold operating room temperature | Radiation and convection losses |
| Large wound exposure (laparotomy, thoracotomy) | Evaporative heat loss from serosal surfaces |
| Cold IV fluids (>1 L/h at room temperature) | Direct heat conduction away from patient |
| Unhumidified breathing gases | Respiratory heat and moisture loss |
| Prolonged procedure duration | Cumulative losses |
| Body habitus extremes | ↑ risk in elderly, thin, female, malnourished patients |
D. Temperature Monitoring Sites
| Site | Accuracy | Notes |
|---|---|---|
| Distal oesophagus | Excellent — best surrogate for core | Gold standard for intubated patients |
| Pulmonary artery | Excellent (true core) | Invasive; used when PA catheter in situ |
| Nasopharynx | Good | Reflects brain temperature |
| Tympanic membrane | Good | Close to hypothalamic temperature |
| Bladder | Good | Correlates well; useful in long cases |
| Sublingual/Axillary | Moderate | Acceptable if consistently measured |
| Skin / Forehead / Rectal | Poor | Do not reliably reflect core temperature |
Part 3: Impact of Perioperative Hypothermia
Perioperative hypothermia is defined as core temperature < 36°C. Mild–moderate hypothermia is 33°C–35°C. Its consequences span every organ system.
A. Cardiovascular Effects
- Cardiac arrhythmias — increased risk at temperatures below 35°C; ventricular fibrillation below 28°C
- Myocardial ischaemia and infarction — postoperative shivering dramatically increases myocardial oxygen demand; vasoconstriction raises afterload; catecholamine surge increases heart rate and BP
- Increased peripheral vascular resistance — tissue hypoperfusion and ↑ cardiac workload
- Hypertension on emergence — sympathetic activation from cold stress
- In the PACU, hypothermic patients have a significantly increased incidence of postoperative myocardial ischaemia
B. Haematological Effects — Coagulopathy
- Hypothermia impairs platelet aggregation and activation — platelets sequester in the spleen and peripheral vasculature
- Reduces activity of coagulation factor enzymes (the coagulation cascade is highly temperature-dependent; standard laboratory coagulation tests are performed at 37°C and will appear falsely normal)
- Associated with nearly a 20% increase in operative blood loss
- Increased transfusion requirements
- This coagulopathy is reversible with rewarming
C. Surgical Site Infection (SSI)
- Hypothermia causes peripheral vasoconstriction → decreased oxygen delivery to the wound
- Tissue hypoxia impairs:
- Neutrophil oxidative killing of bacteria
- Collagen synthesis and wound healing
- Associated with significantly increased rate of SSI — this is one of the strongest evidence-based drivers for maintaining normothermia intraoperatively
D. Drug Metabolism
- Most anaesthetic agents are hepatically metabolised by cytochrome P450 enzymes — all enzyme activity is temperature-dependent
- Hypothermia prolongs the duration of action of:
- Inhalational agents
- Opioids
- Neuromuscular blocking drugs (NMBDs) — prolonged neuromuscular blockade
- Benzodiazepines, propofol
- May cause delayed awakening from anaesthesia
- Impairs correction of metabolic acidosis and electrolyte derangements (e.g., hyperkalaemia)
E. Postoperative Shivering
- The body's attempt to generate heat once anaesthetic-induced thermoregulatory inhibition wanes
- Shivering can increase oxygen consumption by up to 5-fold
- Increases CO₂ production, cardiac output, heart rate, and systemic BP
- Increases intraocular pressure
- Poorly tolerated in patients with cardiac or pulmonary disease
- May cause metabolic acidosis if intense enough
- Treatment: meperidine (pethidine) 12.5–25 mg IV in adults; active rewarming is the better strategy
F. Renal Effects
- Impaired renal function
- Hypothermia reduces GFR and may contribute to intraoperative oliguria
G. Neurological / CNS Effects
- Altered mental status and confusion
- Cognitive impairment in PACU
- Delayed emergence from anaesthesia
H. Summary of Consequences
| System | Consequence |
|---|---|
| Cardiovascular | Arrhythmias, ischaemia, MI, hypertension, ↑ afterload |
| Haematological | Coagulopathy, ↑ blood loss, ↑ transfusion |
| Immune/Wound | ↑ Surgical site infection, impaired healing |
| Pharmacological | Prolonged NMBD, delayed awakening, ↑ drug duration |
| Metabolic | ↑ Protein catabolism, impaired acid-base correction |
| Renal | ↓ GFR, oliguria |
| Neurological | Confusion, delayed emergence |
| Systemic | Shivering, ↑ O₂ consumption, prolonged PACU stay, ↑ mortality |
"Patients who are hypothermic on arrival in the PACU should be actively warmed to avoid these immediate complications as well as delayed consequences of hypothermia... long-term deleterious effects include an increased incidence of myocardial ischemia and myocardial infarction, delayed wound healing, and increased perioperative mortality." — Miller's Anesthesia, 10e
Part 4: Prevention and Management
Preoperative
- Prewarming for 30 minutes with convective forced-air blankets before induction → reduces Phase 1 redistribution significantly by narrowing the central–peripheral temperature gradient
Intraoperative
| Method | Notes |
|---|---|
| Forced-air warming blanket | Most effective; easiest to use; low cost — first choice |
| Warm-water mattress blanket | Effective, especially in conjunction with forced air |
| IV fluid warming | Prevents cooling from cold fluids; pre-warm all infusions >1 L/h (ambient-temperature fluids can worsen hypothermia) |
| Heated, humidified inspired gases | Reduces respiratory heat loss |
| Increase ambient OR temperature | Reduces radiation/convection losses; impractical for large core temperature recovery |
| Passive insulation (blankets, drapes) | Reduces cutaneous loss by ~30%; limited utility alone unless full-body coverage |
Postoperative
- Forced-air warming device is first choice in PACU
- Alternatively: warming lights, heating blankets
- Continue until core temperature ≥ 36°C
- IV meperidine 12.5–25 mg for symptomatic shivering while rewarming
"Hypothermia is most drastic within the first hour after induction because of the rapid effect of anesthesia-induced vasodilation. Therefore, it is during this period that rewarming techniques are most critical." — Sabiston Textbook of Surgery, 7e
— Morgan & Mikhail's Clinical Anaesthesiology 7e, Miller's Anesthesia 10e, Sabiston Textbook of Surgery 7e, Barash Clinical Anaesthesia 9e
PREOPERATIVE FASTING GUIDELINES. ROLE OF CARBOHYDRATE DRINKS
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive, authoritative content from multiple textbooks. Here is the complete structured answer.
Preoperative Fasting Guidelines & Role of Carbohydrate Drinks
Part 1: Rationale for Preoperative Fasting
The primary purpose of preoperative fasting is to minimise gastric volume and acidity at the time of anaesthetic induction, thereby reducing the risk of pulmonary aspiration of gastric contents — a potentially fatal complication.
Historical Context: Mendelson Syndrome
In 1946, Mendelson first described pneumonitis from pulmonary aspiration of acidic gastric secretions in obstetric patients under anaesthesia. The classical threshold for aspiration risk was: gastric volume > 25 mL with pH < 2.5 — though this threshold is derived from primate studies and remains debated. Clinically significant aspiration occurs in approximately 1 in 2,000–3,000 elective anaesthetics but rises to 1 in 600 for emergency cases.
Of patients in whom aspiration is suspected:
- Fewer than half exhibit pulmonary injury
- ~⅓ require postoperative intubation and ventilation
- Most are extubated within 6 hours
- ~10% require ventilation >24 hours
- ~50% of those requiring ventilation >24 hours die of pulmonary complications
The Traditional "NPO after Midnight" Rule
The old dictum of fasting from all oral intake after midnight was a blanket rule that failed to differentiate between clear liquids (gastric emptying time 1–2 hours) and solid food (gastric emptying time ~6 hours). Evidence showed that prolonged fasting from clear liquids does not further reduce gastric volume or raise pH — in fact, adults fasting for 2–4 hours had smaller gastric volumes and higher gastric pH than those fasting for >4 hours.
Part 2: Current ASA Preoperative Fasting Guidelines
The American Society of Anesthesiologists (ASA) first published guidelines in 1998/1999, updated in 2011, and most recently updated in 2017 (with additional updates in 2023). They apply to healthy patients of all ages undergoing elective procedures under general, regional, or monitored sedation anaesthesia.
Summary Table — ASA Fasting Guidelines
| Ingested Material | Minimum Fasting Period |
|---|---|
| Clear liquids (water, fruit juice without pulp, carbonated drinks, clear tea, black coffee) | 2 hours |
| Breast milk | 4 hours |
| Infant formula | 6 hours |
| Non-human milk | 6 hours |
| Light meal (toast, non-fatty foods) | 6 hours |
| Fried or fatty food / heavy meal | 8 hours (or more) |
"The purpose of the guidelines is not only to minimize the risk of pulmonary aspiration but also to avoid case delays as well as prolonged fasting leading to dehydration, hypoglycemia, and patient dissatisfaction." — Barash Clinical Anaesthesia, 9e
Clear liquids include: water, fruit juices without pulp, carbonated beverages, clear tea, black coffee
Clear liquids do NOT include: alcohol, milk, orange juice (non-clear), or fluids with particulates
Notes on Specific Items
- Chewing gum, hard candies, tobacco/smoking: European Society of Anaesthesiology does not recommend delaying anaesthesia if consumed immediately before induction
- 2023 ASA update: Explicitly includes carbohydrate-containing clear liquids as permissible up to 2 hours preoperatively
Part 3: Patients Who Require More Conservative Fasting
Standard guidelines apply to healthy patients undergoing elective procedures. More conservative intervals are warranted for:
| Risk Factor | Concern |
|---|---|
| Pregnancy | Delayed gastric emptying, ↑ aspiration risk |
| Obesity / Morbid obesity | Increased aspiration risk, difficult airway |
| Gastro-oesophageal reflux disease (GERD) | ↑ risk of regurgitation |
| Hiatal hernia | Incompetent lower oesophageal sphincter |
| Diabetic gastroparesis | Markedly delayed gastric emptying |
| Ileus / bowel obstruction | Gastric stasis |
| Opioid medications | Delayed gastric emptying |
| Difficult or anticipated difficult airway | Need for modified technique |
| Emergency surgery | Cannot guarantee fasting status |
For emergency procedures where fasting guidelines cannot be met, rapid sequence induction (RSI) with cricoid pressure is the primary strategy to reduce aspiration risk.
Top 10 Risk Factors for Aspiration (AIMS Study)
- Emergency surgery
- Inadequate anaesthesia
- Abdominal pathology
- Obesity
- Opioid medication
- Neurological deficit
- Lithotomy position
- Difficult intubation/airway
- Gastro-oesophageal reflux
- Hiatal hernia
Part 4: Pharmacological Prophylaxis Against Aspiration
The ASA does not recommend routine use of aspiration prophylaxis in low-risk patients but considers it beneficial in high-risk individuals.
| Drug Class | Agents | Mechanism | Use |
|---|---|---|---|
| H₂-receptor antagonists | Ranitidine 150 mg PO / 50 mg IV; Cimetidine 150–300 mg | Block histamine-stimulated gastric acid secretion → ↑ gastric pH | High-risk patients; ranitidine preferred (fewer side effects, longer acting — up to 9 hours) |
| Proton pump inhibitors (PPIs) | Omeprazole, pantoprazole | Block the proton pump → ↓ acid production | Alternative to H₂ antagonists |
| Non-particulate antacids | Sodium citrate 0.3 M 30 mL | Rapidly neutralises existing gastric acid | Immediate onset; particularly useful in obstetrics and emergency settings |
| Prokinetics | Metoclopramide 10 mg IV | ↑ lower oesophageal sphincter tone; promotes gastric emptying | Useful in full stomach, diabetic gastroparesis, hiatal hernia |
| Antiemetics | Ondansetron | Reduces PONV risk | Adjunct |
Multiple doses of H₂ antagonists are more effective than a single dose alone.
Part 5: Role of Preoperative Carbohydrate Drinks
Background — The Problem with Prolonged Fasting
Traditional overnight fasting creates a catabolic, insulin-resistant state even before the patient enters the operating room. Surgery then adds further metabolic stress:
- ↑ Cortisol and catecholamines → gluconeogenesis and protein catabolism
- ↑ Insulin resistance → hyperglycaemia, impaired wound healing, muscle wasting
- ↑ Immunosuppression
- Dehydration, hypoglycaemia, patient anxiety and discomfort
The surgical stress response, measured by the development of postoperative insulin resistance, is the key metabolic marker of perioperative harm. Insulin resistance correlates with:
- Increased length of hospital stay
- Increased complications (infection, delayed healing)
- Impaired nitrogen balance
The Carbohydrate Loading Concept
Preoperative carbohydrate loading is a cornerstone of Enhanced Recovery After Surgery (ERAS) protocols. The rationale is to:
- Transition the patient from a fasted (catabolic) state to a fed (anabolic) state before anaesthesia
- Maximise preoperative glycogen stores
- Blunt the surgical stress response
- Reduce postoperative insulin resistance
The Drink
- Typically a 12.5% maltodextrin solution (complex carbohydrate — rapidly absorbed, low osmolality)
- Commercially available examples: Nutricia preOp, Gatorade (used practically in many ERAS programmes)
- Dose: typically 400 mL the evening before surgery + 200–400 mL 2–3 hours before induction
- As a clear liquid with rapid gastric emptying (~90 minutes), it is safe to administer up to 2 hours before anaesthesia
- Does not increase gastric residual volume or aspiration risk compared to water in healthy patients
Physiological Benefits
| Benefit | Mechanism |
|---|---|
| ↓ Postoperative insulin resistance | Carbohydrates stimulate insulin secretion preoperatively, improving insulin receptor sensitivity before the stress of surgery hits |
| ↓ Protein catabolism | Anabolic state suppresses muscle breakdown; nitrogen balance preserved |
| ↓ Surgical stress response | Blunts cortisol/catecholamine surge; reduces immunosuppression |
| ↓ Nausea and vomiting | Reduced preoperative hunger and anxiety; less hypoglycaemia |
| ↑ Patient comfort and wellbeing | Reduces thirst, hunger, anxiety on the day of surgery |
| ↓ Length of hospital stay | Multiple RCTs and meta-analyses demonstrate reduced LOS |
| Improved muscle function | Glycogen repletion; better early mobilisation |
"Drinks containing 12.5% maltodextrin given 2 to 3 hours preoperatively have been shown to improve insulin resistance and decrease hospital stay." — Berek & Novak's Gynaecology
"One study reported almost no insulin resistance developing in patients undergoing major open colorectal surgery when employing a combination of epidural anesthesia that minimizes catecholamine and cortisol release, preoperative carbohydrate loading boosting insulin sensitivity before surgery, and early enteral feeding." — Sabiston Textbook of Surgery, 7e
Practical ERAS Carbohydrate Loading Protocol
| Time | Action |
|---|---|
| Evening before surgery (≥ 8 hours pre-op) | Last solid meal |
| Midnight to 2–3 hours before induction | Carbohydrate drink 400 mL (12.5% maltodextrin) |
| 2–3 hours before induction | Final carbohydrate drink 200 mL |
| After this point | Clear water only until 2 hours before; then NBM |
Contraindications to Carbohydrate Drinks
- Diabetes mellitus with gastroparesis (delayed gastric emptying — use with caution; some protocols allow it with monitoring)
- Bowel obstruction or ileus
- Emergency surgery (insufficient time)
- Known aspiration risk (GERD, hiatal hernia, pregnancy)
- Type 1 diabetes and poorly controlled Type 2 diabetes (risk of perioperative hyperglycaemia)
Part 6: Paediatric Considerations
European and Canadian paediatric anaesthesia societies have moved to a 1-hour clear fluid fasting policy for healthy paediatric patients, citing evidence of no increased aspiration risk and equivalent or better gastric emptying compared with 2-hour fasting. The ASA maintains a 2-hour minimum for clear liquids in children as the standard, with breast milk at 4 hours and formula/solids at 6 hours.
Summary
INGESTED MATERIAL FASTING INTERVAL
─────────────────────────────────────────────
Clear liquids (incl. CHO) → 2 hours
Breast milk → 4 hours
Infant formula / milk → 6 hours
Light meal (toast) → 6 hours
Fatty / fried / heavy meal → 8 hours
─────────────────────────────────────────────
Prolonged fasting = dehydration + insulin
resistance + catabolism + patient dissatisfaction
─────────────────────────────────────────────
CHO loading (12.5% maltodextrin 2–3 h pre-op):
• ↓ insulin resistance
• ↓ catabolism
• ↓ hospital stay
• ↑ patient comfort
→ Safe, evidence-based, central to ERAS
— ASA Practice Guidelines 2017/2023, Barash Clinical Anaesthesia 9e, Miller's Anesthesia 10e, Sabiston Textbook of Surgery 7e, Berek & Novak's Gynaecology, Mulholland & Greenfield's Surgery 7e
This is a shared conversation. Sign in to Orris to start your own chat.