Lupus nephritis
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Loading Image
I now have comprehensive, well-sourced content to write a thorough answer on lupus nephritis. Here is the full synthesis:
Lupus Nephritis
Overview
Over 50% of patients with systemic lupus erythematosus (SLE) develop some form of kidney involvement, ranging from microscopic hematuria and subnephrotic proteinuria to nephrotic syndrome, nephritic syndrome, or rapidly progressive glomerulonephritis (RPGN). Because urinalysis findings often do not correlate with histologic severity, kidney biopsy is the most important diagnostic tool in this condition. — NKF Primer on Kidney Diseases, 8e
Pathogenesis
Lupus nephritis is an immune complex–mediated glomerulonephritis. Circulating autoantibodies (especially anti-dsDNA) deposit in the glomeruli, activating complement and triggering inflammation. Key features:
- Immunofluorescence: The hallmark "full-house" pattern — simultaneous glomerular deposition of IgG, IgA, IgM, C3, and C1q.
- Electron microscopy: Deposits in mesangial, subepithelial, and subendothelial locations, plus tubuloreticular inclusions in endothelial cells (a marker of interferon-α activity).
- A recently identified antigen, Exostosin 1/2, has been associated with Class V (membranous) lupus nephritis.
- Lupus can also manifest as tubulointerstitial nephritis, thrombotic microangiopathy, or vasculitis in the kidney.
Active disease is typically associated with elevated anti-dsDNA antibodies and low complement levels (C3, C4), though these are not specific for nephritis.
ISN/RPS Classification
The International Society of Nephrology/Renal Pathology Society (ISN/RPS) classification describes six classes based primarily on light microscopy:
| Class | Morphologic Type | Key Clinical Features | Deposit Location | Management |
|---|---|---|---|---|
| I | Minimal mesangial | Bland urinalysis, normal function | Mesangial (no proliferation) | Supportive only |
| II | Mesangial proliferative | Microscopic hematuria ± non-nephrotic proteinuria | Mesangial (with proliferation) | Non-nephrotic: treat extrarenal disease; Nephrotic: steroids or CNI |
| III | Focal proliferative (<50% glomeruli) | Active urinalysis, nephritic syndrome | Subendothelial | Corticosteroids + MMF or cyclophosphamide |
| IV | Diffuse proliferative (≥50% glomeruli) | Nephritic/nephrotic syndrome, RPGN | Subendothelial (diffuse) | Corticosteroids + MMF or cyclophosphamide |
| V | Membranous | Nephrotic syndrome | Subepithelial + intramembranous | Conservative initially; steroids + MMF/CNI |
| VI | Advanced sclerosing | Advanced kidney failure | >90% global glomerulosclerosis | Prepare for kidney replacement therapy |
Class IV — diffuse proliferative lupus nephritis — is the most severe form and requires the most vigorous treatment. Class III with active necrotizing lesions and large subendothelial deposits also warrants aggressive therapy. Patients can transition between classes over the course of their disease. — NKF Primer, 8e; Goldman-Cecil Medicine
Histology: Class IV
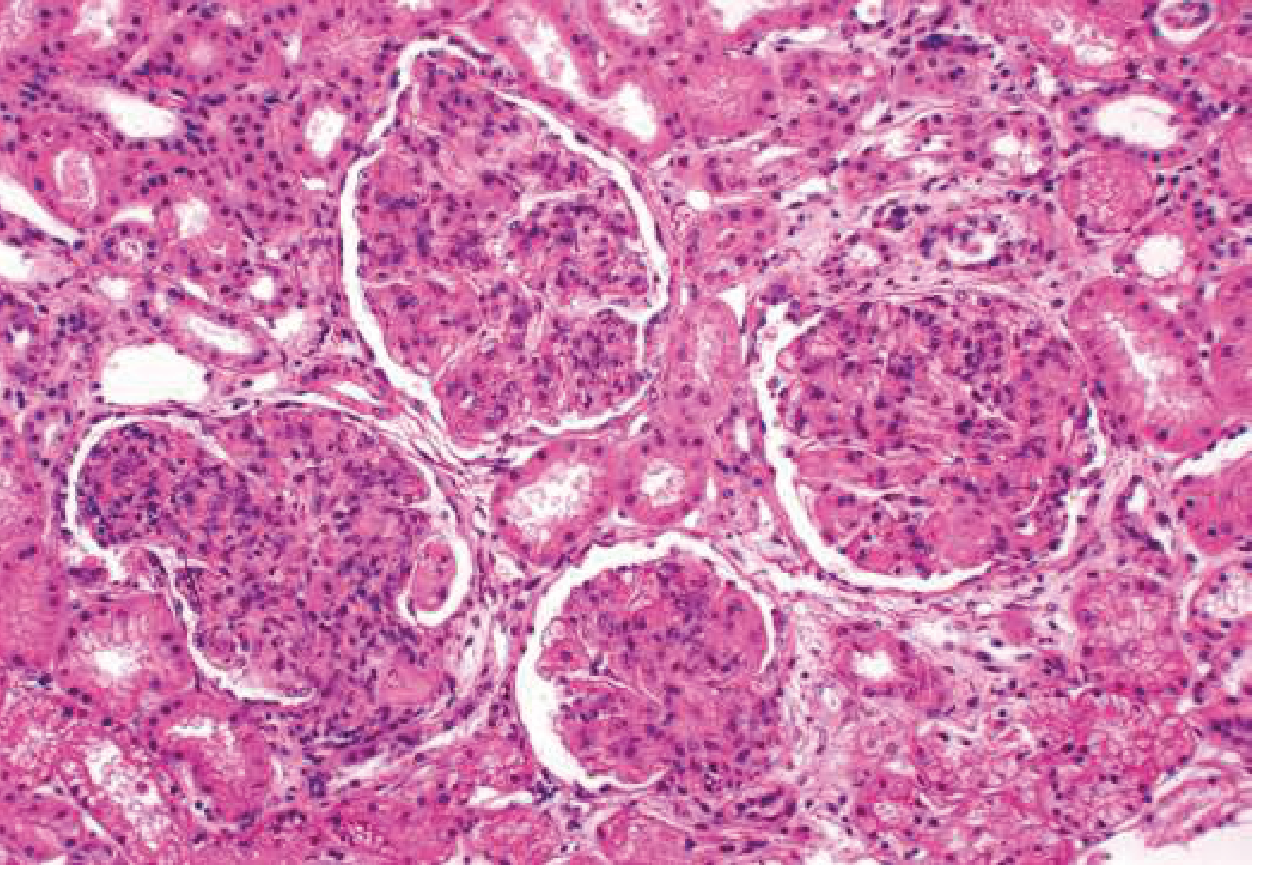
Diffuse proliferative lupus nephritis (Class IV) — H&E showing endocapillary proliferation involving all glomeruli. — Goldman-Cecil Medicine
Treatment
Universal: Hydroxychloroquine
All patients with lupus nephritis should receive hydroxychloroquine (6.5 mg/kg ideal body weight/day, max 400 mg/day) or an equivalent antimalarial unless contraindicated. It reduces flare risk, including during pregnancy. — Goldman-Cecil Medicine; Comprehensive Clinical Nephrology, 7e
Class I & II
No specific renal-directed immunosuppression required beyond what is given for extrarenal SLE. Patients with nephrotic-range proteinuria in Class II may have lupus podocytopathy — treat as minimal change disease (steroids ± CNI).
Class III & IV (Induction)
Initial therapy for active Class III or IV (with or without a membranous component):

Initial therapy algorithm for active Class III/IV LN. — Goldman-Cecil Medicine (adapted from KDIGO 2021)
Corticosteroids (backbone of induction):
- IV methylprednisolone 0.25–0.5 g/day × 1–3 days (pulse), then
- Oral prednisone 0.6–1.0 mg/kg/day (max 80 mg/day), tapering
Plus one of:
- Mycophenolate mofetil (MMF) 1.0–1.5 g orally twice daily (preferred in most guidelines), or mycophenolate sodium 0.72–1.08 g twice daily
- Cyclophosphamide: IV 500 mg every 2 weeks × 6 doses (Euro-Lupus low-dose regimen), or 0.5–1.0 g/m² IV monthly × 6 months
Plus (optional augmentation):
- Belimumab (anti-BAFF monoclonal antibody): 10 mg/kg IV every 2 weeks × 3 doses, then every 4 weeks — added to standard therapy to increase response rates
- Voclosporin (novel CNI): 23.7 mg orally twice daily — used with MMF + steroids in patients with eGFR ≥45 mL/min/1.73 m², with improved response rates
Class III & IV (Maintenance)
After induction, taper prednisone to <5–7.5 mg/day. Preferred maintenance:
- MMF 1000 mg twice daily (preferred over azathioprine)
- Azathioprine 2 mg/kg/day (alternative)
- If neither tolerated: tacrolimus (trough ~4–6 ng/mL), cyclosporine A (trough ~60–100 ng/mL), or mizoribine 3 mg/kg/day
Class V (Membranous)
Initial conservative management; add steroids + MMF or CNI if nephrotic syndrome is severe or if there is serological activity.
Antiphospholipid Syndrome
40–50% of lupus nephritis patients produce anticardiolipin antibodies. Those with glomerular/arteriolar thrombosis require anticoagulation (warfarin or antiplatelet therapy) in addition to immunosuppression. — Goldman-Cecil Medicine
Monitoring & Disease Activity
Key markers of active nephritis:
- Rising serum creatinine
- Active urine sediment (RBC casts, dysmorphic red cells)
- Increasing proteinuria
- Elevated anti-dsDNA antibody titers
- Depressed serum complement (C3, C4)
Repeat kidney biopsy may be indicated to distinguish an active flare from chronic scarring, particularly if the clinical response is unexpected.
Prognosis
Despite aggressive therapy, 10–30% of patients with lupus nephritis develop kidney failure. Patients with lupus nephritis carry a higher risk of death compared to SLE patients without renal involvement. Poor prognostic markers include:
- High serum creatinine at presentation
- Proteinuria >0.5 g/24 h
- Hypertension
- Non-White ethnicity
- Vascular/endothelial abnormalities and crescents on biopsy
- Significant chronic damage (chronicity index) on biopsy
The risk of lupus nephritis recurrence in a kidney allograft is low. — Goldman-Cecil Medicine; Brenner & Rector's The Kidney
Pregnancy and Lupus Nephritis
- Women should ideally be in remission for ≥6 months before conception.
- All women with lupus nephritis in pregnancy should receive hydroxychloroquine (associated with reduced flare risk and fetal growth restriction).
- Lupus nephritis flares occur in ~30% during pregnancy and ~15% postpartum.
- Safe in pregnancy: corticosteroids, azathioprine, tacrolimus, hydroxychloroquine.
- Contraindicated: MMF (microtia, cleft palate, micrognathia — teratogenic), cyclophosphamide.
- Distinguishing a nephritic flare from preeclampsia can be challenging; complement levels and anti-dsDNA titers may help.
- Adverse outcome predictors: baseline creatinine >0.9 mg/dL, proteinuria >0.5 g/24 h, antiphospholipid syndrome, hypertension, non-White ethnicity, disease flare. — Comprehensive Clinical Nephrology, 7e
Sources: NKF Primer on Kidney Diseases, 8e | Goldman-Cecil Medicine, International Edition | Comprehensive Clinical Nephrology, 7e | Brenner and Rector's The Kidney, 2-Volume Set
This is a shared conversation. Sign in to Orris to start your own chat.