Lupas nephritis
Reading File
Finding Sources
Finding Sources
Searching Images
lupus nephritis histology WHO class
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
Searching Images
lupus nephritis wire loop lesion diffuse proliferative glomerulonephritis light microscopy
Loading Image
Reading File
Reading File
I now have comprehensive information from multiple authoritative textbooks. Let me compile the full response.
Lupus Nephritis
Overview
Lupus nephritis (LN) is renal involvement in systemic lupus erythematosus (SLE) and represents the greatest source of morbidity and mortality in lupus patients. Clinically significant kidney disease occurs in approximately 30% of SLE patients. LN is most common in Black females between 15 and 45 years of age. In Asia, LN accounts for >50% of secondary glomerular diseases in countries such as China, Korea, and Japan. — Henry's Clinical Diagnosis and Management by Laboratory Methods
Pathogenesis
LN involves all parenchymal compartments — glomeruli, vessels, tubules, and interstitium. The pathogenesis involves:
- Genetic and epigenetic predisposition: >50 polymorphisms associated with LN susceptibility, including variants affecting programmed cell death, immune clearance of apoptotic debris, and innate/adaptive immunity
- Immune complex deposition: Circulating anti-dsDNA antibodies form immune complexes that deposit in the mesangium, subendothelial, and subepithelial zones
- Complement activation: C1q deposits prominently in glomerular mesangium and peripheral capillary loops (the "full house" pattern on immunofluorescence: IgG, IgA, IgM, C3, C1q)
- Th17 effector pathways: CCR6 variants (a surface marker for Th17 cells) are associated with susceptibility
ISN/RPS Classification (Revised 2004)
The International Society of Nephrology / Renal Pathology Society (ISN/RPS) classification is the standard framework:
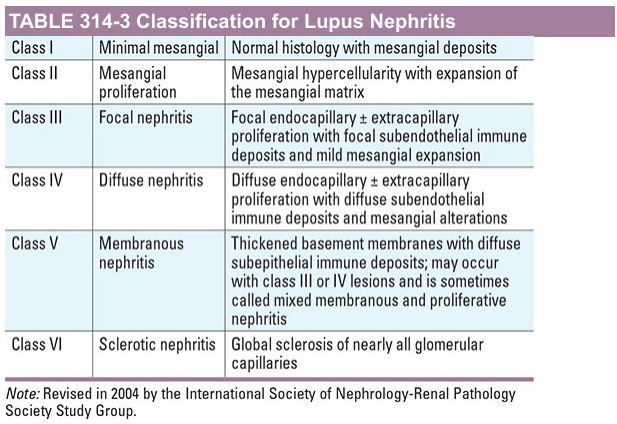
| Class | Name | Key Features |
|---|---|---|
| I | Minimal mesangial | Normal LM; mesangial immune deposits on IF |
| II | Mesangial proliferative | Mesangial hypercellularity/matrix expansion; mesangial deposits |
| III | Focal nephritis | Endo/extracapillary proliferation in ≤50% glomeruli; focal subendothelial deposits. III(A) = active; III(A/C) = active + chronic; III(C) = chronic |
| IV | Diffuse nephritis | Endo/extracapillary proliferation in >50% glomeruli; diffuse subendothelial deposits. IV-S (segmental) or IV-G (global). Most severe and common |
| V | Membranous nephritis | Diffuse subepithelial deposits ± thickened BM; may coexist with class III/IV |
| VI | Sclerotic nephritis | Global sclerosis of >90% glomeruli; represents end-stage |
— Harrison's Principles of Internal Medicine, 21st ed.
Hallmark histologic features of Class IV LN:
- "Wire-loop" lesions (massive subendothelial immune deposits thickening capillary walls)
- Hyaline thrombi (intraluminal eosinophilic deposits)
- Endocapillary hypercellularity, monocyte infiltration
- "Full house" immunofluorescence: IgG, IgA, IgM, C3, C1q
Lupus podocytopathy — a distinct entity not included in the ISN/RPS classification. SLE patients present with nephrotic syndrome and biopsies show minimal change–like lesions or FSGS with foot process effacement but no subendothelial/subepithelial deposits. High relapse rate. — Henry's Clinical Diagnosis
Clinical Manifestations
| Finding | Notes |
|---|---|
| Microscopic hematuria | Most common urinary finding |
| Proteinuria | Ranges from mild to nephrotic range (>3.5 g/day) |
| Red blood cell casts | Indicate active glomerulonephritis |
| Nephrotic syndrome | Especially in class V |
| Renal insufficiency | Elevated creatinine, reduced GFR |
| Tubular defects | Proximal/distal RTA |
| Hypertension | Common |
Indications for kidney biopsy: All SLE patients with suspected renal involvement should undergo biopsy before treatment, as class dictates therapy. Biopsy provides class, activity index, and chronicity index.
Diagnosis
Serological markers:
- ANA (screening, sensitive but non-specific)
- Anti-dsDNA antibodies (specific; levels correlate with disease activity)
- Anti-Sm antibodies (highly specific; associated with worse renal prognosis in some studies)
- Low complement (C3, C4) — hypocomplementemia correlates with active nephritis
- Antiphospholipid antibodies (associated with thrombotic microangiopathy, worse outcomes)
Urinalysis: Active urinary sediment (hematuria, proteinuria, RBC casts), urine protein:creatinine ratio for monitoring.
SLICC criteria allow biopsy-proven LN in the presence of ANA or anti-dsDNA as a standalone criterion for SLE diagnosis. — Henry's Clinical Diagnosis
Management
Induction Therapy (Classes III & IV — Proliferative LN)
Goal: Rapid suppression of active inflammation
| Regimen | Details |
|---|---|
| High-dose corticosteroids | IV methylprednisolone pulses (250–500 mg/day × 3 days) followed by oral prednisolone 0.5–1 mg/kg/day |
| + Mycophenolate mofetil (MMF) | 2–3 g/day — preferred first-line agent; equivalent efficacy to IV cyclophosphamide with better tolerability |
| + IV cyclophosphamide (IVC) | Alternative; low-dose Euro-Lupus (500 mg × 6 pulses) or high-dose NIH protocol |
Treatment has evolved since the 1980s with a shift from corticosteroids + cyclophosphamide toward corticosteroids + mycophenolate therapy, with improved outcomes and reduced adverse effects. — Brenner & Rector's The Kidney
Newer biologic agents:
- Belimumab (anti-BAFF): FDA-approved as add-on therapy for active LN
- Voclosporin (calcineurin inhibitor): FDA-approved in combination with MMF
- Rituximab (anti-CD20): Used in refractory cases; a large RCT showed no additional benefit over conventional therapy in non-refractory disease
Therapeutic plasma exchange (TPE): Does NOT improve outcomes in proliferative LN. A large RCT (Lupus Nephritis Collaborative Study Group) showed TPE added to standard therapy did not reduce kidney failure or mortality. TPE is considered Category III (uncertain benefit) by the American Society for Apheresis. Exception: may be considered in LN with severe thrombotic microangiopathy. — Brenner & Rector's The Kidney
Maintenance Therapy
Goal: Prevention of flares while minimizing cumulative immunosuppression
- MMF 1–2 g/day or azathioprine 2 mg/kg/day — preferred over long-term cyclophosphamide
- Hydroxychloroquine: All SLE/LN patients should receive this unless contraindicated — reduces flares, mortality, and renal damage accrual
- Low-dose corticosteroids: Tapered to lowest effective dose
- RAAS blockade (ACE inhibitor or ARB): Antiproteinuric and reno-protective in all patients with proteinuria
Class V (Membranous LN)
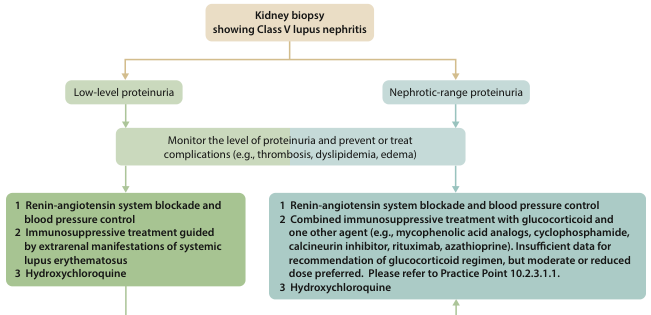
- Low-level proteinuria: RAAS blockade + hydroxychloroquine; immunosuppression guided by extrarenal SLE manifestations
- Nephrotic-range proteinuria: RAAS blockade + hydroxychloroquine + combined immunosuppression (glucocorticoid + MMF, cyclophosphamide, calcineurin inhibitor, or rituximab)
- Monitor for complications: thrombosis (anticoagulate if albumin <20 g/L), dyslipidemia, edema
Class VI
- Supportive care; prepare for renal replacement therapy (dialysis/transplant)
- Minimize immunosuppression to lowest dose needed for extrarenal disease control
Monitoring Response
- Urine protein:creatinine ratio (target <0.5 g/g)
- Serum creatinine/GFR
- Complement C3/C4 and anti-dsDNA titers (correlate with disease activity)
- CBC (monitor for cytopenias, drug toxicity)
- Complete renal response: protein <0.5 g/g + stable/improved GFR
Prognosis and Outcomes
| Factor | Impact |
|---|---|
| Class IV | Most severe; highest risk of ESKD without treatment |
| High baseline creatinine | Unfavorable long-term prognosis |
| Significant chronic damage on biopsy | Unfavorable |
| Vascular/endothelial lesions, crescents | Aggressive disease |
| Anti-Sm positivity (Korea data) | Associated with worse renal prognosis |
| Age >50 at onset | Higher chronicity, inferior outcomes |
| Non-White ethnicity | Higher disease burden |
| Antiphospholipid syndrome | Worse outcomes |
With modern immunosuppressive regimens, 10-year renal survival in class IV LN is approximately 70–80%. LN remains an important cause of ESKD in Asia. — Brenner & Rector's The Kidney, Comprehensive Clinical Nephrology 7th ed.
Pregnancy in Lupus Nephritis
- Ideally, patients should be in remission ≥6 months before conception
- MMF and cyclophosphamide are contraindicated (teratogenic); switch to azathioprine
- Hydroxychloroquine should be continued — reduces flares and fetal growth restriction
- Low-dose aspirin to reduce preeclampsia risk
- Lupus nephritis flares occur in 30% during pregnancy, 15% postpartum
- Distinguishing LN flare from preeclampsia is challenging (both cause hypertension, proteinuria, renal injury, thrombocytopenia)
- Poor pregnancy outcome predictors: Cr >0.9 mg/dL, proteinuria >0.5 g/24h, antiphospholipid syndrome, hypertension, non-White ethnicity
— Comprehensive Clinical Nephrology 7th ed., NKF Primer on Kidney Diseases 8th ed.
This is a shared conversation. Sign in to Orris to start your own chat.