Raas mechanism
Reading File
Finding Sources
Finding Sources
Searching Images
RAAS renin angiotensin aldosterone system mechanism diagram
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of the Renin-Angiotensin-Aldosterone System (RAAS):
The RAAS Mechanism
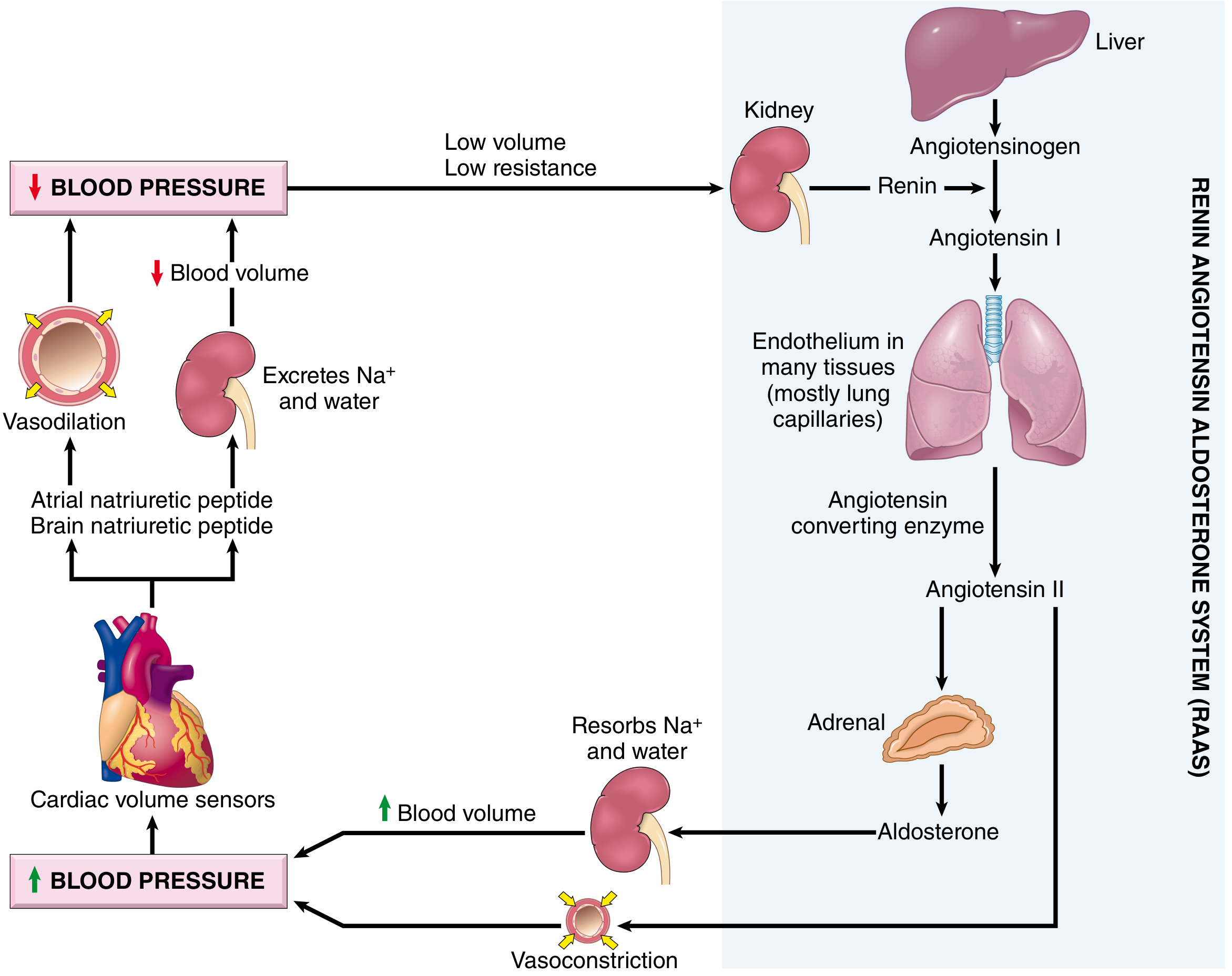
Overview
The RAAS is a hormonal cascade that regulates blood pressure, sodium/water balance, and vascular tone. It operates through a chain of enzymatic steps linking the kidney, liver, lung, and adrenal gland.
Step-by-Step Cascade
| Step | Organ/Tissue | Event |
|---|---|---|
| 1 | Liver | Constitutively produces angiotensinogen (the substrate) |
| 2 | Kidney (JGA) | Secretes renin in response to triggers (see below) |
| 3 | Plasma | Renin cleaves angiotensinogen → Angiotensin I (inactive decapeptide) |
| 4 | Lung endothelium (also other tissues) | ACE (Angiotensin Converting Enzyme) cleaves Ang I → Angiotensin II (active octapeptide) |
| 5 | Adrenal cortex (zona glomerulosa) | Ang II stimulates aldosterone release |
| 6 | Kidney tubule (DCT/CD) | Aldosterone → Na⁺ and water reabsorption → ↑ blood volume → ↑ BP |
| 6 | Vasculature | Ang II also directly causes vasoconstriction → ↑ BP |
Triggers for Renin Release
The juxtaglomerular (JG) cells release renin in response to:
- ↓ Afferent arteriole pressure (detected by baroreceptors within JG cells)
- ↓ NaCl delivery to the macula densa (tubuloglomerular feedback)
- Sympathetic nervous system activation (β₁-adrenergic stimulation of JG cells)
- Hypovolemia / hypotension
Actions of Angiotensin II
Ang II acts primarily via AT1 receptors:
- Vasoconstriction — raises systemic vascular resistance
- Aldosterone release — from adrenal zona glomerulosa → Na⁺/water retention
- Direct tubular effect — stimulates proximal tubular Na⁺ reabsorption
- Efferent arteriole constriction — maintains GFR when perfusion pressure is low
- AVP stimulation — acts on circumventricular subfornical organ (SFO) in the brain to stimulate ADH release and thirst
- Cardiac/vascular remodeling — hypertrophy, fibrosis (via TGF-β), upregulated by pressure overload
Aldosterone Actions
- Acts on mineralocorticoid receptors in the DCT/collecting duct
- Increases apical ENaC and basolateral Na⁺/K⁺-ATPase expression
- Net effect: Na⁺ retention, K⁺ and H⁺ excretion, water retention
- Locally produced in the heart → contributes to interstitial fibrosis in heart failure
Counter-Regulatory: ACE2 / Ang 1–7 / MAS Axis
A protective counter-regulatory arm exists:
- ACE2 converts Ang II → Angiotensin 1–7
- Ang 1–7 acts on MAS receptors → vasodilation, anti-inflammatory, anti-fibrotic, anti-oxidative effects
- This axis is downregulated in conditions like heart failure, hypertension, and COVID-19 (SARS-CoV-2 uses ACE2 as its entry receptor)
RAAS in Disease
| Condition | RAAS Role |
|---|---|
| Renovascular hypertension | Renal artery stenosis → ↓ afferent pressure → ↑ renin → ↑ Ang II → ↑ BP |
| Heart failure | Systemic RAAS activated acutely; local renal RAAS dominates in chronic HF; Ang II drives Na⁺ retention and myocardial remodeling |
| Primary hyperaldosteronism | Autonomous aldosterone excess → Na⁺ retention, hypertension, hypokalemia |
| Liddle syndrome | Gain-of-function ENaC mutation → mimics hyperaldosteronism |
| CKD | RAAS blockade slows progression by reducing proteinuria and hemodynamic injury |
Pharmacological Targets
| Drug Class | Target | Example |
|---|---|---|
| ACE inhibitors | Block ACE → ↓ Ang II | Enalapril, Lisinopril |
| ARBs | Block AT1 receptor | Losartan, Valsartan |
| Direct renin inhibitors | Block renin | Aliskiren |
| Aldosterone antagonists (MRAs) | Block mineralocorticoid receptor | Spironolactone, Eplerenone |
| ARNIs | ARB + neprilysin inhibitor (↑ natriuretic peptides) | Sacubitril/Valsartan |
Sources: Brenner and Rector's The Kidney, 2-Volume Set | Robbins, Cotran & Kumar Pathologic Basis of Disease | National Kidney Foundation Primer on Kidney Diseases, 8e | Histology: A Text and Atlas (Wheater)
This is a shared conversation. Sign in to Orris to start your own chat.