Essay on pulmonary TB
Reading File
Finding Sources
Finding Sources
Searching PubMed
"tuberculosis"[MeSH Terms] AND "treatment"[MeSH Terms]
Reading File
Reading File
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Reading File
Reading File
I now have comprehensive material from multiple authoritative textbooks. Let me compile the full essay.
Pulmonary Tuberculosis
1. Introduction and Epidemiology
Tuberculosis (TB) is one of humanity's oldest and most devastating infectious diseases, caused by Mycobacterium tuberculosis — discovered by Robert Koch in 1882. Despite more than a century of scientific understanding, TB remains a leading cause of death from a single infectious agent worldwide. The lungs are the primary site of infection in over 80% of cases, making pulmonary TB the dominant clinical form of the disease. Globally, TB disproportionately affects populations with poverty, malnutrition, overcrowding, HIV co-infection, and limited healthcare access. The WHO estimates approximately 10 million new cases and 1.5 million deaths annually, with the highest burden in Southeast Asia, Africa, and the Western Pacific.
2. The Pathogen
M. tuberculosis belongs to the M. tuberculosis complex (MTBC), which also includes M. africanum, M. bovis, and several animal-adapted species. Humans are the only significant reservoir; infected humans are the nearly exclusive source of transmission. The organism has a number of biologically distinctive features:
- Acid-fastness: The mycobacterial cell wall is composed of mycolic acids with acyl chains up to 90 carbons in length, containing cyclopropane rings, methyl branches, ketones, and methoxy groups. This lipid-rich envelope renders the bacterium resistant to decolorization by acid-alcohol, forming the basis of Ziehl-Neelsen and auramine staining.
- Slow growth: The generation time of M. tuberculosis is approximately 15–20 hours, compared with minutes for most common bacteria. This profoundly influences treatment duration.
- Genome: The MTBC genome (~4.4 × 10⁶ nucleotides) encodes 4006 proteins. It is guanine-cytosine rich (65.6%) and includes unique multigene families — the proline-glutamate (pe) and proline-proline-glutamate (ppe) genes — markedly expanded in pathogenic mycobacteria.
- Secretion systems: At least three distinct protein secretion systems exist. The type VII secretion system — particularly the ESX-1 locus — secretes ESAT-6 and CFP-10, which are essential virulence factors and immune recognition targets. ESX-3 secretes EsxH, which impedes phagosome maturation and MHC class II antigen presentation.
— Murray & Nadel's Textbook of Respiratory Medicine
3. Transmission
M. tuberculosis is transmitted almost exclusively by the airborne route. An infectious person with pulmonary TB generates droplet nuclei (1–5 µm) by coughing, sneezing, speaking, or singing. These particles remain suspended in the air for hours and, when inhaled by a susceptible host, reach the alveoli. Close, prolonged contact in enclosed, poorly ventilated spaces greatly increases risk. Transmission is strongly correlated with smear-positivity of the index case; cavitary disease carries the highest infectiousness.
4. Pathogenesis and the Granuloma
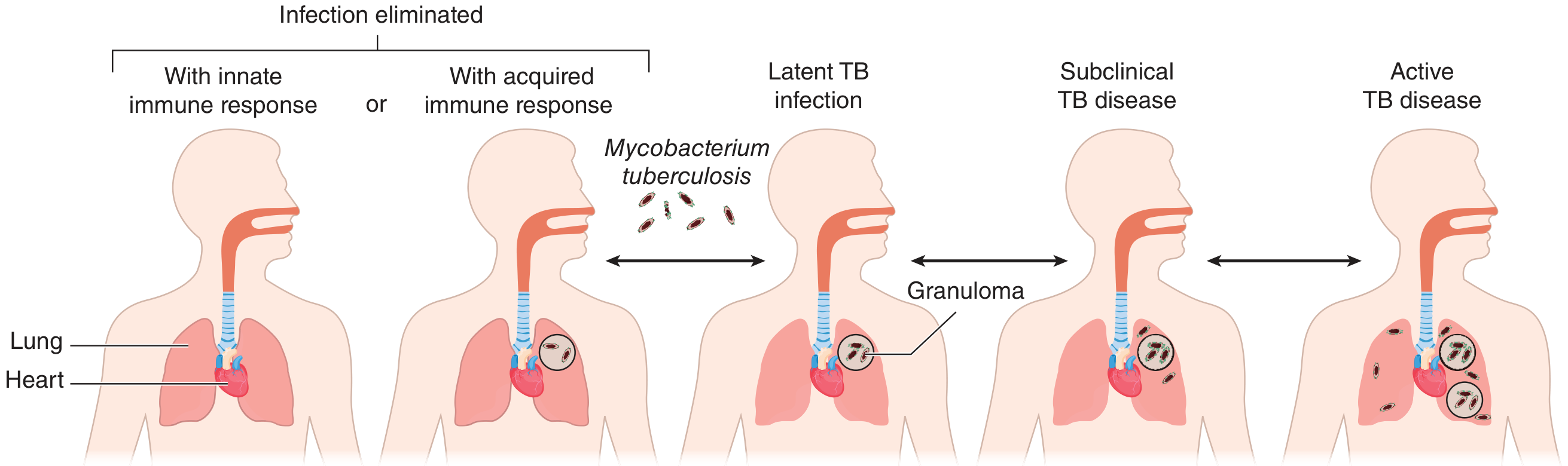
Figure: Spectrum of outcomes after M. tuberculosis infection (Goldman-Cecil Medicine)
4.1 Initial Infection
Inhaled bacilli are phagocytosed by alveolar macrophages. Pathogenic mycobacteria perturb phagosome maturation: instead of proceeding to an acidified phagolysosome armed with hydrolases and antimicrobial peptides, the mycobacterial vacuole retains early endosomal markers. The NADPH oxidase, LC3, and late lysosomal markers are excluded, allowing M. tuberculosis to survive and replicate intracellularly. The bacilli also exploit the cytosol and can rupture the phagosomal membrane, detected by cytosolic innate immune sensors that activate type I interferon responses.
4.2 The Granuloma
As infection progresses, an organized collection of cells — the granuloma — forms around the bacilli, consisting of:
- Macrophages (some undergoing epithelioid transformation; others fusing into multinucleated giant cells)
- CD4⁺ T lymphocytes (dominant) and CD8⁺ T cells
- B cells in and adjacent to granulomas
- Dendritic cells and neutrophils
Caseating necrosis is the hallmark of tuberculous granulomas: central necrosis creates a cheese-like material (from Latin caesum, cheese) in which M. tuberculosis can replicate extracellularly at high numbers. The granuloma was historically viewed as purely host-protective, walling off bacteria and limiting dissemination. This is supported by the fact that HIV infection and TNF-α inhibitors (which disrupt granuloma architecture) greatly increase the risk of dissemination. However, current understanding is more nuanced: granulomas also facilitate cell-to-cell bacterial spread within macrophage aggregates, stimulate the bacilli to enter a non-replicating drug-tolerant state, and block access of adaptive immune cells and antibiotics to the bacteria. Progressive disease is paradoxically characterised by increasing granuloma size and number, with escalating tissue damage rather than effective microbial killing. — Murray & Nadel's
4.3 Outcomes of Primary Infection
After initial infection, outcomes lie on a spectrum:
- Elimination by innate or acquired immune response (~50%)
- Latent TB infection (LTBI): Immunologic containment without disease (~45%); the Ghon complex may leave a calcified Ghon lesion in the mid-lung fields; Ghon lesion + calcified hilar nodes = Ranke complex
- Subclinical TB disease: Positive cultures, minimal or no symptoms
- Progressive primary disease: Active TB within the first year — more common in children, the elderly, and the immunocompromised
- Reactivation TB ("post-primary disease"): Reactivation of LTBI, typically years to decades later
A lifetime risk of reactivation of approximately 5–10% exists in immunocompetent individuals. HIV co-infection raises this to ~10% per year.
5. Clinical Features
5.1 Symptoms
The cardinal symptom is a persistent productive cough, often lasting weeks or months before presentation. Importantly, 10–25% of bacteriologically confirmed TB cases do not report cough, and cough of any duration combined with systemic symptoms may be a better screening criterion in high-risk groups. Systemic features include:
- Fever (in ~35–80% of patients)
- Night sweats
- Weight loss / anorexia ("consumption")
- Malaise and fatigue
- Hemoptysis: Seen with more extensive disease. May also result from inactive TB — from bronchiectasis, rupture of a vessel in a cavity wall (Rasmussen aneurysm), an aspergilloma in an old cavity, or broncholithiasis from eroded calcified lesions. — Murray & Nadel's
Dyspnea suggests extensive pulmonary involvement, large pleural effusion, or pneumothorax.
5.2 Physical Examination
Physical findings are often non-specific. Post-tussive rales in the upper lung zones may be heard. Amphoric (cavernous) breath sounds indicate a cavity communicating with a large airway. Lymphadenopathy is uncommon in immunocompetent adults with pulmonary TB but may be prominent in HIV-infected individuals and children.
6. Chest Radiographic Features
Radiography is central to both diagnosis and classification:
- Progressive primary TB: Dense consolidation, predominantly in the middle and lower lobes
- Reactivation TB (the classic pattern): Cavitary disease in the apical and posterior segments of the right upper lobe and the apical-posterior segment of the left upper lobe — remote from the site of primary infection. Cavitation reflects liquefactive necrosis and is a hallmark of high bacterial burden and infectivity.
- Additional findings: Fibrotic scars, loss of lung volume, calcification, and endobronchial spread to lower lobes producing tree-in-bud opacities
- Erosion into blood vessels or lymphatics → miliary pattern (1–2 mm nodules throughout both lungs), representing haematogenous dissemination
- Pleural effusions: Usually unilateral and exudative; seen in primary TB pleuritis
- HIV co-infection: Disease may be atypical — less cavitation, lower smear-positivity; a normal chest radiograph is possible despite active disease. CT scan is more sensitive in these cases. — Goldman-Cecil Medicine
7. Diagnosis
7.1 Microbiologic Testing
- Sputum smear microscopy (Ziehl-Neelsen or fluorochrome): Rapid and inexpensive but lacks sensitivity (~45–80%) and cannot distinguish species. Three sputum samples collected on separate days optimise yield.
- Mycobacterial culture: The gold standard. Solid media (Löwenstein-Jensen) takes 3–8 weeks; liquid broth systems (BACTEC MGIT) yield results in 7–14 days. Culture allows drug susceptibility testing.
- Molecular tests — Xpert MTB/RIF: WHO-recommended as the initial diagnostic test where available. Simultaneously detects M. tuberculosis DNA and rifampin-resistance mutations within 2 hours. Sensitivity ~88% vs. culture; ~98% in smear-positive specimens. Rifampin resistance on Xpert strongly correlates with MDR-TB. — Goldman-Cecil Medicine
- Nucleic acid amplification tests (NAATs): More broadly, include various platforms beyond Xpert.
7.2 Immunologic Tests
- Tuberculin skin test (TST / Mantoux): Intradermal injection of purified protein derivative (PPD). A positive result (induration ≥5–15 mm depending on risk group) indicates prior sensitisation but cannot distinguish LTBI from active disease, or TB from BCG vaccination. False-negatives occur in severe immunosuppression.
- Interferon-gamma release assays (IGRAs): Whole-blood assays (QuantiFERON-TB Gold Plus, T-SPOT.TB) measuring IFN-γ production by T cells in response to TB-specific antigens (ESAT-6, CFP-10). More specific than TST (not affected by BCG). Cannot distinguish LTBI from active disease.
7.3 Other Investigations
- Bronchoscopy with BAL: For patients unable to produce sputum; useful for endobronchial disease
- CT chest: Superior to plain radiography, particularly in HIV-infected patients
- Pleural fluid: Exudate; lymphocyte-predominant; ADA (adenosine deaminase) elevation supports the diagnosis; culture positivity is low (~25%)
- Baseline blood tests: LFTs (especially if aged >35, HIV-positive, or alcohol use); CBC; HIV testing should be offered to all TB patients
8. Treatment
8.1 Treatment of Active Drug-Susceptible Pulmonary TB
Treatment requires multidrug therapy to prevent emergence of resistance. The standard 6-month regimen has two phases:
Intensive phase (2 months): Rifampin (R) + Isoniazid (H) + Pyrazinamide (Z) + Ethambutol (E) — the RIPE regimen
Continuation phase (4 months): Rifampin (R) + Isoniazid (H)
This achieves relapse-free cure in ~95% of drug-susceptible cases. — Goldman-Cecil Medicine; Red Book 2021
| Drug | Mechanism | Key Adverse Effects |
|---|---|---|
| Isoniazid (H) | Prodrug activated by KatG catalase-peroxidase; inhibits InhA (ketoenoyl-reductase) → blocks mycolic acid synthesis | Hepatotoxicity, peripheral neuropathy (prevented by pyridoxine 25–50 mg/day), drug-induced lupus |
| Rifampin (R) | Inhibits bacterial RNA polymerase (β-subunit) | Hepatotoxicity, drug-drug interactions (potent CYP inducer), orange discolouration of secretions |
| Pyrazinamide (Z) | Converted to pyrazinoic acid; disrupts membrane potential in acidic pH (active in macrophages) | Hepatotoxicity, hyperuricaemia, arthralgia |
| Ethambutol (E) | Inhibits arabinosyl transferase → inhibits arabinogalactan synthesis | Optic neuritis (dose-dependent; monitor visual acuity and colour vision) |
Isoniazid is always given with pyridoxine (vitamin B₆) 25–50 mg/day to prevent peripheral neuropathy.
8.2 Monitoring and Follow-Up
- Monthly clinical assessment and sputum examination until sputum is negative on two consecutive cultures
- ~75–80% of patients are sputum culture-negative by 2 months; ~95% by 3 months
- Persistent positive culture at 4 months = treatment failure — requires expert input and never addition of a single drug (which promotes further resistance)
- Patients are considered infectious until at least 2 weeks of effective therapy + clinical response + three negative AFB smears — Goldman-Cecil Medicine
8.3 Directly Observed Therapy (DOT)
DOT — where a healthcare provider observes each dose — is the global standard to ensure adherence and detect adverse events. When administered by DOT, short-course therapy is equivalent in efficacy to 9 months of isoniazid and achieves superior completion rates.
8.4 Drug-Resistant TB
- MDR-TB: Resistance to at minimum rifampin and isoniazid (the two most potent first-line drugs)
- XDR-TB: MDR-TB plus resistance to fluoroquinolones and at least one injectable second-line drug
- Treatment of MDR-TB requires second-line agents including bedaquiline (inhibits ATP synthase), linezolid, fluoroquinolones (levofloxacin/moxifloxacin), cycloserine, and pretomanid. The BPaLM regimen (bedaquiline + pretomanid + linezolid + moxifloxacin) represents current best practice per WHO 2022 consolidated guidelines.
- A single drug should never be added to a failing regimen.
8.5 Treatment of Latent TB Infection (LTBI)
Preferred regimens:
- 3HP: Isoniazid + rifapentine weekly × 12 doses (most preferred — superior completion rates)
- 4R: Rifampin daily × 4 months
- 3HR: Rifampin + isoniazid daily × 3 months
- 6H or 9H: Isoniazid daily × 6 or 9 months (alternatives)
9. Special Populations
HIV Co-infection
- TB is the leading cause of death among people living with HIV
- Clinical manifestations depend on CD4 count: at relatively preserved CD4 counts, disease resembles immunocompetent TB; at low CD4 counts, atypical presentation, lower smear-positivity, absent cavitation, more extrapulmonary and miliary disease
- Antiretroviral therapy (ART) should be initiated within 2–8 weeks of starting TB treatment (unless TB meningitis, where delay may be beneficial)
- Rifampin's potent CYP3A4 induction significantly reduces protease inhibitor and integrase inhibitor levels; rifabutin (a less potent inducer) is often substituted
Pregnancy
- TB in pregnancy is associated with premature birth, low birth weight, and vertical transmission
- Rifampin, isoniazid, and ethambutol are considered safe; pyrazinamide is generally used despite limited formal data. Streptomycin is absolutely contraindicated (ototoxicity to the fetus)
Paediatric TB
- Children more commonly develop progressive primary disease and extrapulmonary TB (including miliary and CNS TB)
- Smear microscopy is less reliable (paucibacillary disease)
- Gastric lavage or induced sputum may be required for specimen collection
10. Extrapulmonary TB
Because primary infection involves haematogenous dissemination before immunologic control, virtually any organ can be affected:
- Pleural TB: Most common extrapulmonary form; lymphocytic exudative pleural effusion; elevated ADA
- Miliary TB: 1–2 mm granulomatous nodules in multiple organs (lungs, liver, bone marrow, kidneys, adrenals, spleen); choroidal tubercles on fundoscopy
- TB meningitis: Most severe form; associated with high mortality and neurological sequelae; in patients with advanced AIDS and miliary TB, blood cultures may be positive in 20–40%
- Spinal TB (Pott's disease): Vertebral destruction, gibbus deformity, paraplegia
- Lymph node TB (scrofula): Most common extrapulmonary form in children
- Renal TB: "Sterile pyuria"; calcification; "putty kidney"
- Pericardial TB: Constrictive pericarditis
11. Prevention and Public Health
BCG Vaccination
Bacille Calmette-Guérin (BCG), a live attenuated strain of M. bovis, is the only licensed TB vaccine. It provides ~80% protection against severe TB in children (miliary, meningeal) but highly variable (0–80%) protection against adult pulmonary TB. BCG is given at birth in high-burden countries per national immunisation programmes.
Infection Control
- Respiratory isolation of suspected/confirmed cases until non-infectious
- Negative-pressure rooms with ≥12 air changes per hour
- N95 respirators for healthcare workers
- UV germicidal irradiation and HEPA filtration
Contact Tracing and LTBI Screening
All close contacts of an index case should be screened with TST or IGRA. Those with positive immunologic tests and no evidence of active disease should be offered LTBI treatment. Systematic screening is prioritised in high-risk groups: immunocompromised individuals, children under 5 years, and healthcare workers.
12. Complications
| Complication | Mechanism |
|---|---|
| Haemoptysis | Rasmussen aneurysm, aspergilloma in old cavity, bronchiectasis |
| Spontaneous pneumothorax | Rupture of subpleural bulla or cavity |
| Bronchopleural fistula | Erosion of cavity into pleural space |
| Respiratory failure | Extensive bilateral disease |
| Aspergilloma | Fungal colonisation of residual cavity |
| ARDS | Severe active TB or immune reconstitution inflammatory syndrome (IRIS) |
| Amyloidosis | Chronic granulomatous inflammation → AA amyloidosis |
13. Conclusion
Pulmonary TB is a complex infectious disease with deep evolutionary ties to human history. Its pathogenesis hinges on a delicate and often fatal interplay between M. tuberculosis's sophisticated immune evasion strategies and the host granulomatous response. The clinical picture — chronic productive cough, constitutional symptoms, upper-lobe cavitary disease — is classic but can be substantially modified by HIV infection, immunosuppression, and age. Diagnosis has been transformed by molecular tools, and the RIPE regimen remains the global backbone of treatment for drug-susceptible disease. The growing challenge of drug-resistant TB, the need for shorter regimens, and the search for a more effective vaccine remain the defining priorities of the field.
Sources: Goldman-Cecil Medicine, 26th Ed. | Murray & Nadel's Textbook of Respiratory Medicine | Harrison's Principles of Internal Medicine, 22nd Ed. | Katzung's Basic and Clinical Pharmacology, 16th Ed. | Red Book 2021 (AAP) | Fishman's Pulmonary Diseases and Disorders
This is a shared conversation. Sign in to Orris to start your own chat.