Esophegial atresia in pedia everything about it and some little related terms to tomorrow i have exam of that topic ospe exam so i need detailed information about everything
esophageal atresia tracheoesophageal fistula types classification
Esophageal Atresia (EA) & Tracheoesophageal Fistula (TEF) — Complete OSCE Review
1. DEFINITION
2. INCIDENCE & EPIDEMIOLOGY
| Fact | Value |
|---|---|
| Incidence | ~1 in 3,000–4,000 live births |
| Sex predilection | Mild male predominance |
| Low birth weight | ~33% of affected neonates |
| Associated anomalies | 60–70% of cases |
| Polyhydramnios (prenatal) | Present in ~2/3 of EA cases |
3. EMBRYOLOGY & PATHOGENESIS
- At ~4 weeks gestation, a diverticulum forms off the anterior aspect of the primitive foregut (proximal pharyngeal foregut).
- This diverticulum elongates caudally forming the laryngotracheal groove, creating a separate trachea and esophagus — separated by the tracheoesophageal septum.
- EA = failure of the primitive foregut to recanalize (esophageal tube continuity is interrupted).
- TEF = failure of the lung bud to separate completely from the foregut.
- The process involves Sonic Hedgehog (SHH) signaling, N-myc, Sox2, and CHD7 mutations in syndromic cases.
- It is a multifactorial sporadic condition — genetic predisposition + environmental exposures (alcohol, smoking, medications, infections, agricultural chemicals).
4. CLASSIFICATION (Gross Classification — Most Used)
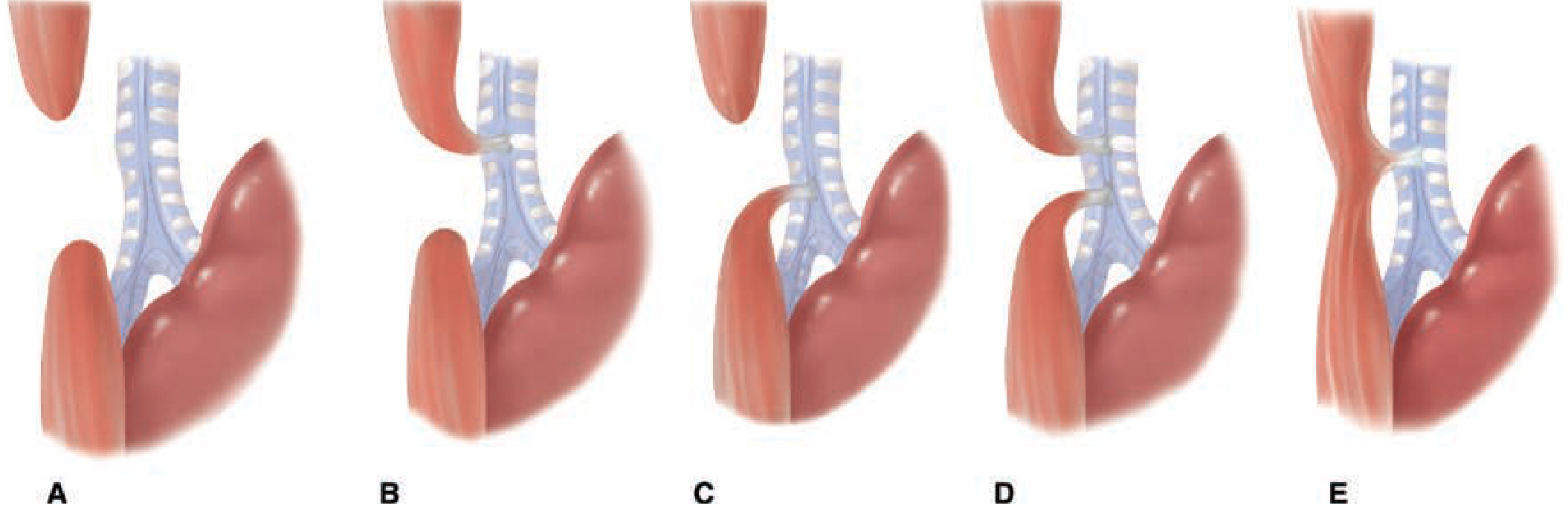
| Type | Description | Frequency |
|---|---|---|
| Type A | Pure/isolated EA — blind proximal pouch + blind distal segment (no fistula), NO gas in abdomen | 8–10% |
| Type B | EA + proximal TEF (fistula between proximal esophageal pouch and trachea) | ~1% |
| Type C ⭐ | EA + distal TEF — most common — blind proximal pouch + distal esophagus connects to trachea, GAS in abdomen | ~85% |
| Type D | EA + double TEF (both proximal and distal fistulas) | ~2% |
| Type E | TEF without EA = "H-type fistula" — looks like letter H, no atresia | ~4–8% |
Key OSCE point: Type C (EA + distal TEF) = most common. The gas in the abdomen on X-ray is the hallmark — air travels via TEF → trachea → distal esophagus → stomach.
Key OSCE point: Type A (pure EA) = absent abdominal gas = NO fistula.
5. CLINICAL FEATURES (Presentation in the Neonate)
Classic "3 Cs"
- Coughing with first feed
- Choking with first feed
- Cyanosis with first feed
Full symptom list:
- Excessive salivation / frothy mucus from mouth and nose (pooling in blind pouch) — often the first sign noticed
- Inability to swallow / regurgitation on first feeding attempt
- Coughing, choking, cyanosis during feeding
- Respiratory distress — aspiration of saliva → aspiration pneumonia (especially right upper lobe)
- Abdominal distension — in Type C, air from trachea enters stomach via TEF
- Maternal polyhydramnios — fetus cannot swallow amniotic fluid (most prominent in isolated EA/Type A, ~86%)
- H-type fistula (Type E) presents subtly — feeding-induced coughing/choking, often diagnosed late (postneonatally), recurrent chest infections
6. DIAGNOSIS
Bedside/Clinical Test — MOST IMPORTANT
- Failure to pass an orogastric (NG) tube — tube coils back at the thoracic inlet (~10 cm from lips). This is the hallmark of EA.
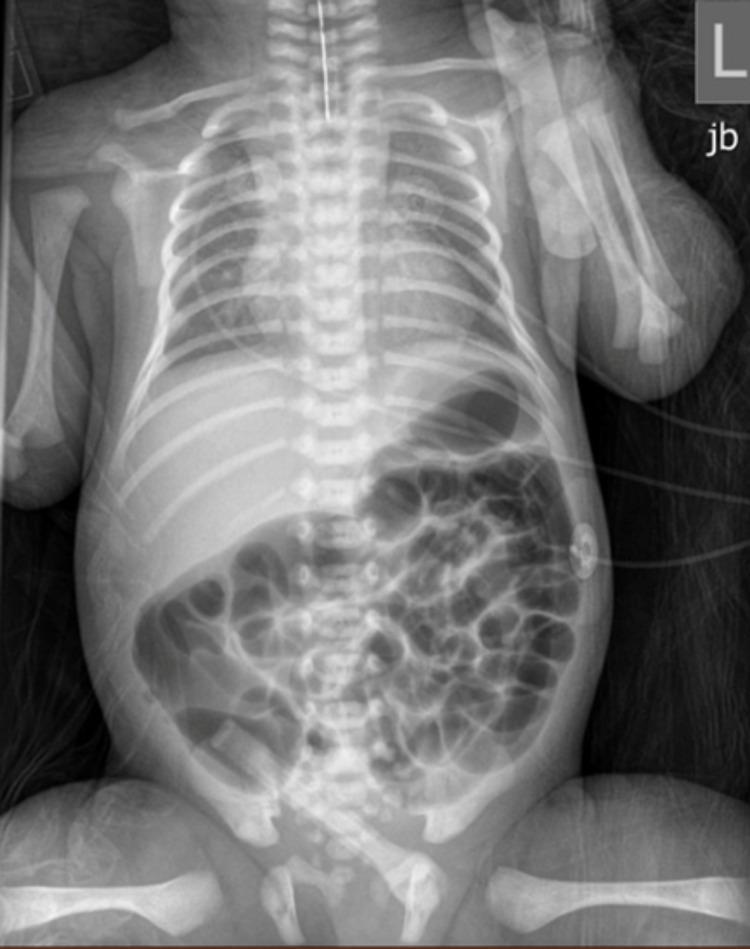
Plain Chest X-Ray (CXR)
- Coiled/recoiled orogastric tube in the proximal esophagus (upper mediastinum) — confirms EA
- Gas in the abdomen → confirms distal TEF (air travels trachea → distal esophagus → stomach)
- No abdominal gas → isolated EA (Type A), no fistula
Prenatal Ultrasound
- Absent/small stomach bubble (fetus cannot swallow amniotic fluid)
- Polyhydramnios — develops in 3rd trimester (86.7% at ≥27 weeks)
- "Pouch sign" — dilated upper esophagus posterior to the fluid-filled trachea = diagnostic
| Ultrasound Finding | Detection Rate at ≥27 weeks |
|---|---|
| Polyhydramnios | 86.7% |
| Small/absent stomach | 29.3% |
| Diagnosed EA | 33.9% |
Other Investigations
- Echocardiography — mandatory to detect associated cardiac defects
- Renal ultrasound — to detect genitourinary anomalies
- Bronchoscopy — evaluates tracheomalacia, identifies fistula location (especially for recurrent/H-type), used preoperatively
- Barium swallow — for H-type TEF (shows fistula); CONTRAINDICATED in EA (aspiration risk)
- Preoperative CXR + echo — assess aortic arch anatomy (right-sided arch = more complications)
7. ASSOCIATED ANOMALIES — VACTERL/VATER
| Letter | Anomaly |
|---|---|
| V | Vertebral anomalies (absent/hemivertebrae) |
| A | Anorectal malformations (imperforate anus) |
| C | Cardiac defects (most common — ~55%; VSD, ASD, TOF) |
| T | Tracheoesophageal fistula |
| E | Esophageal atresia |
| R | Renal/urogenital anomalies (~50%) |
| L | Limb defects (radial ray abnormalities, ~45%) |
Cardiac anomalies are the most common associated defects (~55%), followed by urogenital (~50%), musculoskeletal/limb (~45%), then craniofacial.
8. PREOPERATIVE MANAGEMENT
- NPO — nothing by mouth
- Prone/upright positioning — 30–45° head-up, to reduce aspiration
- Sump tube (Replogle tube) — continuous suction applied to the proximal esophageal pouch to drain saliva and prevent aspiration
- IV fluids and broad-spectrum antibiotics
- Avoid endotracheal intubation/positive pressure ventilation if possible — PPV forces air through TEF into stomach → worsens gastric distension → impairs diaphragmatic excursion → respiratory compromise
- If intubation unavoidable: advance ETT past the TEF (below fistula) or use Fogarty balloon catheter to occlude fistula
- Emergency gastrostomy may be needed if severe gastric distension compromises ventilation (gastrostomy placed to water seal)
- Pre-op echocardiogram to determine aortic arch side — right-sided arch changes surgical approach
9. SURGICAL MANAGEMENT
Standard Operation (Type C — most common)
- Divide azygos vein to expose the fistula
- Ligate and divide the TEF (tracheal end closed with absorbable interrupted sutures)
- Mobilize proximal esophageal pouch
- Primary end-to-end esophageal anastomosis
Right thoracotomy is favored when aortic arch is left-sided (normal). Left-sided arch → left thoracotomy.
Historical milestones:
- 1939 — Ladd and Leven: first successful repair (ligated fistula + gastrostomy + delayed repair)
- 1941 — Dr. Cameron Haight (Ann Arbor): first successful primary anastomosis — still the current gold standard approach
H-type (Type E) TEF:
- Diagnosed by bronchoscopy + barium swallow
- Surgical approach: cervical incision (near thoracic inlet)
- Bronchoscopic guidewire cannulation aids in locating the fistula
10. LONG-GAP EA
- Gap defined as: ≥2 vertebral body lengths between pouches
- Management options:
- Suture distal esophagus to prevertebral fascia with hemoclip marker → as infant grows, esophagus lengthens
- Delayed primary anastomosis — typically at ~4–6 months when adequate growth has occurred
- Gastrostomy for enteral feeding in the interim
- Cervical esophagostomy if aspiration cannot be controlled
- Esophageal replacement (stomach, colon, or small intestine conduit) at ~1–2 years if primary anastomosis not feasible
11. COMPLICATIONS
Intraoperative:
- Tracheal injury
Early postoperative:
- Anastomotic leak — most common early complication
- Anastomotic stricture (later) — most common long-term complication; needs dilation
- Recurrent TEF
- Wound infection
Late:
- Gastroesophageal reflux (GERD) — very common (up to 40–70%); due to esophageal dysmotility + poor LES function
- Esophageal dysmotility — impaired peristalsis (intrinsic esophageal innervation is abnormal)
- Tracheomalacia — weak/floppy tracheal cartilage → characteristic "seal bark" cough, respiratory symptoms — present in ~75% of EA/TEF patients
- Failure to thrive
- Barrett's esophagus — long-term risk from chronic GERD
12. PROGNOSIS — WATERSTON CLASSIFICATION (Risk Stratification)
| Class | Criteria | Prognosis |
|---|---|---|
| A | Birth weight >2500g, healthy | Excellent |
| B | BW 1800–2500g or BW >2500g with moderate pneumonia/anomaly | Good |
| C | BW <1800g OR any weight with severe pneumonia/severe anomaly | Guarded |
Modern outcomes are excellent (>90% survival) for Class A patients. Prognosis is mainly determined by associated cardiac anomalies and prematurity.
13. KEY RELATED TERMS (OSCE-Relevant)
| Term | Definition |
|---|---|
| Tracheoesophageal septum | Embryological structure that divides the primitive foregut into trachea and esophagus |
| Replogle tube | Double-lumen sump tube used for continuous suction of proximal esophageal pouch |
| Polyhydramnios | Excess amniotic fluid due to fetal inability to swallow; associated with EA |
| Tracheomalacia | Structural weakness of tracheal cartilage → "seal bark" cough; complication of EA/TEF |
| GERD (Gastroesophageal Reflux Disease) | Very common long-term complication; due to esophageal dysmotility + short distal segment |
| Long-gap EA | Gap >2 vertebral bodies between esophageal pouches; precludes primary anastomosis |
| H-type fistula (Type E TEF) | TEF without atresia; fistula resembles letter "H"; subtle presentation, no atresia |
| Foregut | The primitive embryological structure giving rise to esophagus, trachea, stomach, and proximal duodenum |
| Sonic Hedgehog (SHH) signaling | Critical pathway for tracheoesophageal separation; defects implicated in EA |
| Azygos vein | Divided during thoracotomy to expose the fistula |
| Extrapleural dissection | Classic surgical approach for EA/TEF; avoids entering pleural space |
| Anastomotic stricture | Most common long-term surgical complication; managed with endoscopic dilation |
| Waterston classification | Risk stratification scheme for EA/TEF based on birth weight and associated anomalies |
| Pouch sign | Prenatal US finding — dilated upper esophagus posterior to trachea = diagnostic of EA |
| VACTERL association | Vertebral, Anorectal, Cardiac, Tracheo-Esophageal, Renal, Limb — nonrandom cluster of anomalies |
QUICK-RECALL SUMMARY FOR OSCE
"3 Cs at first feed" + excessive drooling + failure to pass NG tube = EA until proven otherwise
- Most common type: Type C (EA + distal TEF) = 85%, gas in abdomen ✅
- Pure EA (Type A) = no gas in abdomen ❌, most prominent polyhydramnios
- H-type (Type E) = subtle, recurrent chest infections, no atresia
- Gold standard diagnosis: coiled NG tube on CXR
- NEVER do oral contrast in suspected EA (aspiration risk)
- Pre-op: Replogle suction + prone positioning + IV antibiotics
- Surgery: right extrapleural thoracotomy → ligate TEF → primary anastomosis
- Most common complication: anastomotic stricture (long-term), GERD, tracheomalacia
- Key association: VACTERL — cardiac anomalies most common (~55%)
- Prognosis determined by: cardiac defects + prematurity + pneumonia
CLINICAL AND PHARMACOLOGICAT. CHARACTERISIICS OF ANTTANGINAL DRUGS. INCOMING CONTROL. 1 OPTION 1. Specify the drug used to stop an attack of angina pectoris 2. 3 Anaprilin Nitrong Nitroglycerine Finoptin 5. Sustak-forte 2. After what period of time does nitroglycerin relieve pain in angina pectoris? 1 hour 2. 3 within 5 minutes within 30 minutes within 20 minutes 5. within 40 minutes 3. The mechanism of antianginal action of nitrosorbitol is associated Decreased contractile function of the myocardium 2. 3. Reflex expansion of the coronary arteries 4. Decreased preload on the myocardium An increase in total peripheral resistance 5 Decrease in BCC 4. The mechanism of antianginal action of B-blockers is explained 1. Decreased myocardial oxygen demand by reducing the work of the heart Reflex expansion of the coronary arteries 3 Myotropic coronary vasodilation 4. Decreased afierload on the myocardium 5. Decreased preload on the myocardium 5. The use of B-blockers in the amount of antianginal drugs is contraindicated in 1. Bronchial asthma 2 arterial hypertension Extrasystoles Atrial fibrillation atrial flutter 6.B-agonists, expanding the coronary arteries, simultaneously cause Bronchospasm Increase in myocardial oxygen demand by increasing heart rate and force of heart contraction Increase OPS 4. Reduce BCC 5. Raise blood pressure 7. What drug is used to prevent myocardia! infarction in unstable angina, relying on its antiplatelet effect Sustak 2 Validol Finoptin Aspirin 5 * specify the drug that is contrandatad in the presence of bronchial asthma in a petita 2. Isadrin Anaprilin Sustak 5. Nitroglycerine 9. Name an antianginal drug, the action of which is associated with blocking slow calcium channels Nitroglycerine 2. Validol 3. Erinite 4. Sustak 5. Finoptin 10. Name an antianginal drug. the action of which is associated with blocking slow calcium channels Nitroglycerine 2. 3 4 5. Validol Erinite Sustak Finoptin Give me only one correct option of these mcqs
Quick Summary Table
| Q | Answer | Drug |
|---|---|---|
| 1 | 3 | Nitroglycerine |
| 2 | 2 | Within 5 minutes |
| 3 | 3 | Decreased preload |
| 4 | 1 | ↓ O₂ demand |
| 5 | 1 | Bronchial asthma |
| 6 | 2 | ↑ O₂ demand |
| 7 | 4 | Aspirin |
| 8 | 3 | Anaprilin (Propranolol) |
| 9 | 5 | Finoptin (Verapamil) |
| 10 | 5 | Finoptin (Verapamil) |
What questions can mam ask in this topic Esophageal Atresia (EA) & Tracheoesophageal Fistula (TEF) in ospe
Predicted OSCE Questions — Esophageal Atresia & TEF
🔴 SPOT DIAGNOSIS / IMAGE-BASED (Very High Yield)
- "What is the diagnosis in this neonate?" (Shows CXR with coiled NG tube in upper chest + gas in abdomen)
- "What type of EA/TEF is shown in this diagram?" (Shows Type C — most likely asked)
- "Label the diagram of EA types A, B, C, D, E"
- "What does the arrow point to in this X-ray?" (Coiled orogastric tube / blind pouch)
- "What is the significance of gas seen in the abdomen on this CXR?" (Confirms distal TEF)
- "What is absent in this X-ray of a neonate with EA Type A?" (No abdominal gas)
🟠 SHORT ANSWER / DEFINE
- Define Esophageal Atresia
- Define Tracheoesophageal Fistula
- What is H-type fistula?
- What is the most common type of EA? (Type C — 85%)
- What is long-gap EA?
- What is polyhydramnios and why does it occur in EA?
- What is the "pouch sign" on prenatal ultrasound?
- What is tracheomalacia?
- What is VACTERL association? Expand the abbreviation.
- What is the Waterston classification?
🟡 MECHANISM / EMBRYOLOGY
- At what week of gestation does EA/TEF occur? (~4th week)
- What embryological structure fails to form properly in EA? (Tracheoesophageal septum)
- What is the embryological origin of the esophagus and trachea? (Both from primitive foregut)
- Why does polyhydramnios occur in EA? (Fetus cannot swallow amniotic fluid)
- Why is there gas in the abdomen in Type C EA? (Air passes trachea → TEF → distal esophagus → stomach)
- Why is there NO gas in the abdomen in Type A EA? (No fistula — air cannot reach stomach)
- What signaling pathway is implicated in EA? (Sonic Hedgehog signaling)
🟢 CLINICAL PRESENTATION
- What are the "3 Cs" of EA presentation?
- What is the first sign noticed by the nurse/mother in EA? (Excessive frothy salivation)
- What happens when the first feed is given to a neonate with EA?
- How does H-type TEF differ in presentation from EA? (Subtle, postneonatal, no atresia, recurrent chest infections)
- What is the diagnostic hallmark at the bedside for EA? (Failure to pass NG/OG tube — coils at ~10 cm)
- Which lobe is most commonly affected by aspiration pneumonia in EA? (Right upper lobe)
- Why does abdominal distension occur in Type C EA?
🔵 DIAGNOSIS
- What is the first investigation you would do in a neonate with suspected EA? (Pass NG tube + CXR)
- What will CXR show in EA + distal TEF (Type C)?
- What will CXR show in pure EA (Type A)?
- Why is oral contrast (barium swallow) CONTRAINDICATED in EA? (Risk of aspiration)
- When is barium swallow used in EA/TEF? (Only for H-type TEF diagnosis)
- Why must echocardiography be done in all EA cases? (Most common associated anomaly is cardiac)
- What prenatal ultrasound findings suggest EA?
- What is the role of bronchoscopy in EA/TEF?
🟣 MANAGEMENT / SURGICAL
- What is the immediate preoperative management of EA? (NPO, Replogle tube suction, prone positioning, IV antibiotics)
- What is a Replogle tube?
- Why should positive pressure ventilation be avoided in EA + distal TEF? (Air goes into stomach via TEF → gastric distension → respiratory compromise)
- What is the definitive surgical treatment of Type C EA? (Right extrapleural thoracotomy → ligate TEF → primary esophageal anastomosis)
- Who performed the first successful primary anastomosis for EA? (Dr. Cameron Haight, 1941)
- What is the surgical approach if the patient has a right-sided aortic arch?
- What is the management of long-gap EA?
- What structure is divided during thoracotomy to expose the TEF? (Azygos vein)
- What is the management of isolated TEF (H-type)? (Cervical incision)
⚫ COMPLICATIONS
- What is the most common EARLY complication after EA repair? (Anastomotic leak)
- What is the most common LATE/LONG-TERM complication? (Anastomotic stricture — needs dilation)
- What is tracheomalacia and how does it present? ("Seal bark" cough, respiratory symptoms)
- Why is GERD so common after EA repair?
- What is the long-term cancer risk in EA survivors? (Barrett's esophagus → esophageal adenocarcinoma from chronic GERD)
🔶 MOST LIKELY OSPE STATION FORMATS
| Station Type | Most Likely Question |
|---|---|
| X-ray station | Identify coiled NG tube, comment on gas, give diagnosis + type |
| Diagram station | Label/identify EA type from anatomical drawing |
| Case vignette | Neonate with frothy secretions + failed NG tube → diagnosis, first investigation, management |
| Short answer | Expand VACTERL / define Replogle tube / mechanism of polyhydramnios |
| Embryology | Week of development, structure involved, why gas present/absent |
⭐ TOP 5 GUARANTEED QUESTIONS
- Most common type of EA? → Type C (85%)
- Why gas in abdomen on CXR? → Air via distal TEF
- Expand VACTERL → Vertebral, Anorectal, Cardiac, TE fistula, Esophageal atresia, Renal, Limb
- First bedside sign/test? → Fail to pass NG tube (coils at thoracic inlet)
- Immediate management? → Replogle suction + prone position + NPO + IV antibiotics
Tip for OSPE: Always mention Type C when asked about "most common" and always link gas in abdomen = TEF present, no gas = pure EA (Type A). These two points come up in almost every EA OSPE question.
Give also the answers like you only gave questions give answers too
OSCE Q&A — Esophageal Atresia & TEF (Complete with Answers)
🔴 SPOT DIAGNOSIS / IMAGE-BASED
A: Esophageal Atresia with distal Tracheoesophageal Fistula — Type C EA/TEF (most common type, ~85%)
A: Type C — Esophageal atresia with distal tracheoesophageal fistula
A:
- A = Pure/isolated EA (no fistula)
- B = EA + proximal TEF
- C = EA + distal TEF ← most common
- D = EA + both proximal and distal TEF
- E = TEF without EA (H-type fistula)
A: The coiled orogastric tube in the proximal esophageal blind pouch (upper mediastinum/thoracic inlet) — confirming esophageal atresia
A: It confirms the presence of a distal tracheoesophageal fistula. Air from the trachea passes through the fistula into the distal esophagus and then into the stomach, producing visible abdominal gas
A: No abdominal gas — because there is no fistula connecting the trachea to the distal esophagus, so air cannot reach the stomach
🟠 SHORT ANSWER / DEFINE
A: A congenital interruption of esophageal continuity resulting in a blind-ending proximal esophageal pouch, preventing normal passage of food/fluid from the mouth to the stomach
A: An abnormal communication (fistula) between the trachea and the esophagus — can occur with or without esophageal atresia
A: It is Type E TEF — a tracheoesophageal fistula without esophageal atresia. The fistula resembles the letter "H" anatomically. It presents subtly (often postneonatally) with feeding-induced choking, coughing, and recurrent chest infections
A: Type C — Esophageal atresia with distal tracheoesophageal fistula — occurs in approximately 85% of all EA cases
A: When the gap between the upper and lower esophageal pouches is ≥2 vertebral body lengths, making primary end-to-end anastomosis impossible at birth. Requires delayed repair at 4–6 months
A: Polyhydramnios = excess amniotic fluid during pregnancy. In EA, the fetus cannot swallow amniotic fluid (blocked by the blind esophageal pouch), so fluid accumulates. It is most prominent in pure EA (Type A) — present in ~86% of isolated EA cases
A: It is the appearance of a dilated upper esophagus seen as a fluid-filled structure posterior to the trachea on prenatal ultrasound. It is considered diagnostic of esophageal atresia
A: A structural weakness/softening of the tracheal cartilage leading to tracheal collapse during breathing. It is a common complication of EA/TEF, present in ~75% of cases. It causes a characteristic "seal bark" cough and recurrent respiratory symptoms
A:
- V — Vertebral anomalies
- A — Anorectal malformations (imperforate anus)
- C — Cardiac defects ← most common (~55%)
- T — Tracheo-Esophageal fistula
- E — Esophageal Atresia
- R — Renal/urogenital anomalies
- L — Limb defects (radial ray abnormalities)
A: A risk stratification system for EA/TEF based on 3 factors:
- Class A: Birth weight >2500g, otherwise healthy → Excellent prognosis
- Class B: BW 1800–2500g OR BW >2500g with moderate pneumonia/anomaly → Good prognosis
- Class C: BW <1800g OR any weight with severe pneumonia/severe anomaly → Guarded prognosis
🟡 MECHANISM / EMBRYOLOGY
A: During the 4th week of gestation — when the primitive foregut is dividing to form the separate trachea and esophagus
A: The tracheoesophageal septum — normally this septum divides the primitive foregut into the anterior trachea and posterior esophagus. Its failure leads to EA and/or TEF
A: Both arise from the primitive foregut (endodermal tube). A diverticulum forms off the anterior aspect of the proximal foregut and elongates caudally forming the laryngotracheal groove
A: The fetus normally swallows amniotic fluid. In EA, the blind proximal pouch blocks swallowing, so amniotic fluid cannot pass into the GI tract, causing it to accumulate → polyhydramnios
A: In Type C, the distal esophagus communicates with the trachea via a fistula. During inspiration, air travels: trachea → TEF → distal esophagus → stomach → visible gas on X-ray
A: In pure EA (Type A), there is no fistula at all. Neither end of the esophagus connects to the trachea, so air cannot enter the stomach → gasless abdomen on X-ray
A: Sonic Hedgehog (SHH) signaling — critical for proper separation of the respiratory tract from the primitive foregut. Defects in SHH signaling are implicated in EA/TEF formation. Mutations in N-myc, Sox2, and CHD7 are found in syndromic cases
🟢 CLINICAL PRESENTATION
A:
- Coughing
- Choking
- Cyanosis
All occurring at the first feed
A: Excessive frothy salivation — saliva pools in the blind proximal pouch and overflows from the mouth and nose. This is often the very first sign before any feeding attempt
A: The feed cannot pass into the stomach (blocked by blind pouch) → immediate regurgitation, coughing, choking, and cyanosis. Milk may be aspirated into the lungs → aspiration pneumonia
A:
- No atresia — NG tube passes normally, neonate can feed
- Presentation is subtle and delayed (often postneonatal)
- Symptoms: feeding-induced coughing/choking, recurrent aspiration pneumonia, recurrent chest infections
- Often diagnosed late, sometimes in infancy or even childhood
A: Failure to pass an orogastric (NG) tube — the tube coils back at approximately 10 cm from the lips (at the thoracic inlet/blind pouch). This is the single most important bedside finding
A: The right upper lobe — due to the anatomical position and aspiration of saliva/secretions from the blind pouch
A: Air from the trachea continuously passes through the distal TEF into the stomach → progressive gastric distension → abdominal distension. This can also compress the diaphragm and worsen breathing
🔵 DIAGNOSIS
A: Pass an orogastric tube and take a chest X-ray (CXR). If the tube coils at the thoracic inlet on X-ray → confirms EA
A:
- Coiled orogastric tube in the upper mediastinum (proximal blind pouch)
- Gas present in the abdomen (stomach + bowel) — confirms distal TEF
- May show aspiration pneumonia (right upper lobe)
A:
- Coiled orogastric tube in the upper mediastinum
- No abdominal gas — gasless abdomen (no fistula to carry air to stomach)
A: Because contrast material will be aspirated into the lungs from the blind proximal pouch → aspiration pneumonitis/pneumonia → can be fatal in a neonate
A: Only in H-type TEF (Type E) — where there is no atresia and the patient can swallow. A small amount of dilute contrast under fluoroscopy can reveal the fistulous communication between esophagus and trachea
A: Because cardiac anomalies are the most common associated defect (~55% of EA patients). Cardiac defects also largely determine prognosis. Echo also identifies the side of the aortic arch, which determines the surgical approach (right thoracotomy for left arch, etc.)
A:
- Absent/small stomach bubble (fetus cannot swallow)
- Polyhydramnios (especially in 3rd trimester — 86.7% at ≥27 weeks)
- "Pouch sign" — dilated upper esophagus posterior to trachea = diagnostic
A:
- Identifies the exact location of the fistula
- Evaluates the degree of tracheomalacia
- Excludes a secondary/second fistula
- Essential in recurrent TEF and H-type fistula (guidewire cannulation helps locate it)
🟣 MANAGEMENT / SURGICAL
A: (ABCDE approach)
- NPO — nothing by mouth
- Replogle tube (double-lumen sump tube) — continuous suction to drain the blind pouch
- Prone/upright positioning — 30–45° head-up to reduce aspiration
- IV fluids + broad-spectrum IV antibiotics
- Avoid PPV (positive pressure ventilation) if possible
- Echocardiography + preop CXR to assess anatomy
A: A double-lumen sump tube inserted into the proximal blind esophageal pouch with continuous suction applied to drain pooled saliva and secretions, preventing aspiration pneumonia preoperatively
A: PPV forces air preferentially through the path of least resistance — the TEF — into the stomach instead of the lungs → massive gastric distension → diaphragm elevation → respiratory compromise → may be fatal. It also inadequately ventilates the lungs
A:
- Right extrapleural thoracotomy (or thoracoscopic approach)
- Divide the azygos vein to expose the TEF
- Ligate and divide the TEF (tracheal end closed with absorbable interrupted sutures)
- Mobilize the proximal esophageal pouch
- Primary end-to-end esophageal anastomosis
A: Dr. Cameron Haight in Ann Arbor, Michigan — performed the first successful primary anastomosis in 1941, which remains the current standard of care
A: A left thoracotomy may be used (instead of the usual right thoracotomy). A right-sided aortic arch is also associated with increased risk of arch anomalies and postoperative complications
A:
- Gastrostomy for enteral feeding
- Suture distal esophagus to prevertebral fascia with hemoclip marker → allows esophageal lengthening as infant grows
- Replogle tube for continuous suction of proximal pouch
- Delayed primary anastomosis at ~4–6 months once adequate esophageal length is achieved radiographically
- If still not possible → esophageal replacement with stomach/colon/small intestine at 1–2 years
A: The azygos vein — dividing it exposes the underlying tracheoesophageal fistula
A: Surgical repair via cervical incision (near the thoracic inlet). Rigid bronchoscopy with Fogarty/guidewire cannulation is used to locate the fistula before surgical division
⚫ COMPLICATIONS
A: Anastomotic leak — occurs at the site where the two esophageal ends are joined. Can lead to mediastinitis, pleural effusion, or sepsis
A: Anastomotic stricture — narrowing at the anastomotic site due to scarring. Presents with dysphagia and feeding difficulties. Managed with endoscopic balloon dilation
A: Weakness of tracheal cartilage → trachea collapses during expiration. Present in ~75% of EA/TEF patients. Classic presentation:
- "Seal bark" / brassy cough
- Recurrent respiratory infections
- Episodes of cyanosis/apnea
- Worsens with respiratory infections
A: Because:
- The distal esophagus has abnormal/absent peristalsis (intrinsic innervation is defective)
- Lower esophageal sphincter (LES) is dysfunctional
- The mobilized short distal esophagus creates tension → poor LES competence Results in 40–70% incidence of GERD post-repair
A: Barrett's esophagus — from chronic uncontrolled GERD → metaplasia of esophageal squamous epithelium to columnar epithelium → risk of esophageal adenocarcinoma. Long-term endoscopic surveillance is needed
⭐ MOST IMPORTANT 10 Q&As (Memorize These)
| # | Question | Answer |
|---|---|---|
| 1 | Most common type of EA | Type C — EA + distal TEF (85%) |
| 2 | Why gas in abdomen? | Air via distal TEF into stomach |
| 3 | Why NO gas in abdomen? | Pure EA (Type A) — no fistula |
| 4 | First bedside test/sign | Fail to pass NG tube (coils at 10 cm) |
| 5 | Expand VACTERL | Vertebral, Anorectal, Cardiac, TE fistula, Esophageal atresia, Renal, Limb |
| 6 | Most common associated anomaly | Cardiac defects (~55%) |
| 7 | Immediate management | Replogle suction + prone + NPO + IV antibiotics |
| 8 | Why avoid PPV? | Air goes into stomach via TEF → gastric distension |
| 9 | Most common late complication | Anastomotic stricture |
| 10 | First successful repair | Dr. Cameron Haight, 1941 (primary anastomosis) |
Golden Rule for OSCE:
- Gas in abdomen + coiled NG tube = Type C EA ✅
- No gas in abdomen + coiled NG tube = Type A (pure) EA ✅
- No atresia + recurrent chest infections = H-type (Type E) TEF ✅
Tell me again about differnetial diagnosis
Differential Diagnosis of Esophageal Atresia / TEF
The 3 Main Presenting Scenarios
Scenario 1 — Neonate with excessive salivation + choking/cyanosis at first feed + NG tube won't pass
Scenario 2 — Neonate with respiratory distress + recurrent chest infections (H-type)
Scenario 3 — Prenatal — absent stomach bubble + polyhydramnios
DIFFERENTIAL DIAGNOSIS TABLE
| Condition | How it Mimics EA/TEF | How to Differentiate |
|---|---|---|
| Choanal Atresia | Neonatal respiratory distress, cyanosis, feeding difficulty, excessive secretions | Cyanosis improves with crying (mouth breathing); pass catheter through NOSE — fails; NG tube passes easily into stomach ✅ |
| Congenital Esophageal Stenosis | Difficulty feeding, regurgitation, dysphagia | NG tube passes but with resistance; barium swallow shows narrowing not blind pouch; no gas issue |
| Laryngeal Cleft | Choking/coughing with feeds, aspiration, recurrent pneumonia — mimics H-type TEF | Direct laryngoscopy/bronchoscopy shows posterior laryngeal cleft; NG tube passes normally |
| Pharyngeal Incoordination / Dysphagia | Choking at feeding, aspiration | Video fluoroscopic swallow study (VFSS) normal anatomy; NG tube passes; no structural defect |
| Duodenal Atresia | Vomiting after feeds, polyhydramnios prenatally | "Double bubble" sign on X-ray (stomach + duodenum); NG tube passes; vomiting is bilious (if below ampulla); stomach bubble IS present on prenatal US |
| Pyloric Stenosis | Vomiting in neonate (though usually 2–6 weeks) | Projectile non-bilious vomiting at 2–6 weeks; NG tube passes; ultrasound shows pyloric muscle thickening; olive-shaped mass palpable |
| Intestinal Atresia (Jejunal/Ileal) | Polyhydramnios prenatally, abdominal distension, vomiting | Multiple dilated loops on X-ray; NG tube passes; bowel obstruction picture |
| Diaphragmatic Hernia | Respiratory distress, bowel sounds in chest, cyanosis | CXR shows bowel loops in chest; scaphoid abdomen; heart displaced; NG tube passes into stomach |
| Vascular Ring | Stridor, feeding difficulty, recurrent chest infections | Barium swallow shows external esophageal compression; CT angiography confirms; no atresia |
| Tracheomalacia (isolated) | "Seal bark" cough, stridor, respiratory difficulty | Bronchoscopy shows floppy trachea; NG tube passes normally; no feeding-related choking at first feed |
| Pierre Robin Sequence | Respiratory distress, feeding difficulty in neonate | Micrognathia + glossoptosis + cleft palate visible on exam; NG tube passes; airway obstruction from tongue |
| Gastroesophageal Reflux (GER) | Regurgitation, coughing, recurrent aspiration | NG tube passes; 24-hr pH monitoring confirms; common and usually non-urgent; no atresia |
| Absent stomach bubble (prenatal DDx) | EA suspected prenatally | Also consider: severe oligohydramnios, renal agenesis (Potter sequence), diaphragmatic hernia, normal variation in early pregnancy |
FOCUSING THE DIFFERENTIAL — Key Distinguishing Points
EA vs. Choanal Atresia
| Feature | EA | Choanal Atresia |
|---|---|---|
| NG tube | Fails (coils at 10 cm) | Passes into stomach ✅ |
| Nasal catheter | Passes | Fails |
| Cyanosis | With feeding | Worse at rest, better with crying |
| Abdominal gas | Absent (Type A) or present (Type C) | Normal |
EA vs. Duodenal Atresia (both cause polyhydramnios)
| Feature | EA | Duodenal Atresia |
|---|---|---|
| Prenatal US | Absent stomach bubble | "Double bubble" (stomach + duodenum visible) |
| NG tube | Fails | Passes |
| Vomiting | Can't swallow at all | Bilious/non-bilious vomiting |
| Abdominal gas | Absent (Type A) | Present but with obstruction pattern |
EA vs. H-type TEF vs. Laryngeal Cleft
| Feature | EA | H-type TEF | Laryngeal Cleft |
|---|---|---|---|
| Atresia present | ✅ Yes | ❌ No | ❌ No |
| NG tube | Fails at 10 cm | Passes normally | Passes normally |
| Presentation timing | Birth (first feed) | Delayed (weeks–months) | Delayed |
| Diagnosis | CXR | Barium swallow / bronchoscopy | Direct laryngoscopy |
| Recurrent pneumonia | Yes (from aspiration) | Yes | Yes |
EA vs. Congenital Esophageal Stenosis
| Feature | EA | Congenital Esophageal Stenosis |
|---|---|---|
| NG tube | Cannot pass | Passes with difficulty |
| Barium swallow | Contraindicated | Shows focal narrowing (not blind end) |
| Presentation | Immediate at birth | Dysphagia when solids introduced |
| CXR | Coiled tube | Normal or mild dilation above stenosis |
For H-type TEF specifically — DDx of Recurrent Aspiration Pneumonia
- H-type TEF (Type E) ← must exclude first
- Laryngeal cleft
- Laryngomalacia
- Vascular ring compressing esophagus
- Pharyngeal incoordination/dysphagia
- GERD with microaspiration
- Immune deficiency
OSCE Tips for Differential Diagnosis Questions
"Neonate, first feed, chokes/blue, NG tube won't pass" → EA until proven otherwise. First DDx to rule out is choanal atresia (nasal catheter test distinguishes them)
"Absent stomach bubble on prenatal scan + polyhydramnios" → EA vs. Duodenal Atresia vs. oligohydramnios-related. Key: if double bubble = duodenal atresia; if pouch sign = EA
"Child 6 months, recurrent chest infections, chokes with feeds, NG tube fine" → H-type TEF vs. Laryngeal Cleft vs. Vascular Ring — needs barium swallow ± bronchoscopy
"Projectile non-bilious vomiting at 3 weeks" → Pyloric Stenosis — NOT EA (different age, NG passes, classic projectile pattern)
- Choanal Atresia — distinguished by which tube fails (nasal vs. oral)
- Duodenal Atresia — distinguished by prenatal US (absent bubble vs. double bubble) and NG tube passage
What is elephant sign or test in that topic that is the first question i was asked