Everything about liver trauma in surgery with reference to standard textbooks
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
I now have extensive, comprehensive content from multiple authoritative surgical textbooks. Let me synthesize everything into a thorough, well-structured answer.
Liver Trauma in Surgery
1. Epidemiology and Mechanisms
The liver is one of the most commonly injured intra-abdominal organs. After blunt trauma, the liver is injured in 22.2% of cases (2012 NTDB data). After penetrating trauma, it is the most commonly injured solid organ, present in 26.1% of cases.
Mechanisms of blunt hepatic trauma include:
- Direct compression causing parenchymal damage
- Shearing forces tearing hepatic tissue and disrupting vascular and ligamentous attachments
- Deceleration forces (especially relevant in high-grade injuries)
High-velocity missile injuries cause massive tissue destruction with high mortality. The liver's large size and fixed position beneath the right costal margin make it vulnerable to both direct blows and crush injuries.
— Sabiston Textbook of Surgery, p. 692; Mulholland and Greenfield's Surgery, p. 1279–1280
2. Types of Liver Injury
Blunt or penetrating trauma may cause:
- Intraparenchymal lacerations and haematomas — usually elliptical or linear in shape
- Subcapsular haematoma — blood collection between the liver capsule and parenchyma
- Capsular rupture with intraperitoneal haemorrhage
- Major vascular injury — lacerations involving major branches of the hepatic veins or IVC; relatively rare but life-threatening
Secondary complications include: ischaemia, necrosis, abscess formation, haemobilia, focal fibrosis, calcification, and lobar/segmental atrophy.
— Grainger & Allison's Diagnostic Radiology, p. 661
3. AAST Liver Injury Grading Scale (2018 Revision)
The American Association for the Surgery of Trauma (AAST) Organ Injury Scale is the universally adopted grading system, updated in 2018 to include radiologic (CT) vascular injury criteria.
| Grade | AIS | Imaging/Operative Criteria |
|---|---|---|
| I | 2 | Subcapsular hematoma <10% surface area; capsular tear; parenchymal laceration <1 cm depth |
| II | 2 | Subcapsular hematoma 10–50% surface area; intraparenchymal hematoma <10 cm; laceration 1–3 cm depth, <10 cm length |
| III | 3 | Subcapsular hematoma >50% surface area; ruptured subcapsular or parenchymal hematoma; intraparenchymal hematoma >10 cm; laceration >3 cm depth |
| IV | 4 | Parenchymal disruption involving 25–75% of hepatic lobe, or 1–3 Couinaud segments; active bleeding contained within parenchyma or extending into peritoneum |
| V | 5 | Parenchymal disruption >75% of a hepatic lobe; juxtahepatic venous injuries (retrohepatic IVC/central major hepatic veins) |
| VI | — | Hepatic avulsion (lethal) |
Advance one grade for multiple injuries, up to Grade III.
— Sabiston Textbook of Surgery, p. 693; Mulholland and Greenfield's Surgery, p. 1280–1281; Current Surgical Therapy 14e, p. 1342
4. Initial Evaluation and Diagnostics
Primary Survey and Resuscitation
- Follow ATLS primary survey (Airway, Breathing, Circulation)
- Early blood product resuscitation for suspected ongoing haemorrhage
- Patients with peritonitis or ongoing hypotension with suspected abdominal haemorrhage belong in the operating room
FAST Examination
- The Focused Assessment with Sonography in Trauma (FAST) detects free intraperitoneal fluid (hemoperitoneum)
- Positive FAST + haemodynamic instability = operative intervention
- Positive FAST alone does not mandate surgery
- US demonstrates free fluid or thrombus but does not reliably identify all lacerations
CT Scan (Gold Standard)
Contrast-enhanced CT (with arterial and portal venous phases) is the imaging modality of choice for haemodynamically stable patients. It provides:
- Accurate delineation of injury architecture and grade
- Amount of intraperitoneal blood
- Detection of active arterial extravasation ("blush")
- Identification of pseudoaneurysms or arteriovenous fistulas
- Assessment of hilar/vascular involvement
- Associated injury inventory
Unenhanced CT reveals large lacerations as low-attenuation regions; fresh thrombus (higher attenuation than blood) indicates nearby haemorrhage. Arterial phase is mandatory for detection of vascular injuries.
REBOA
For haemodynamically unstable patients, a Resuscitative Endovascular Balloon Occlusion of the Aorta (REBOA) inflated in Zone I (between left subclavian and celiac trunk) may temporise haemorrhage as a bridge to definitive control. Deflation should be expeditious to prevent distal ischaemia.
— Current Surgical Therapy 14e, p. 1341–1342; Grainger & Allison's Diagnostic Radiology, p. 661; Mulholland and Greenfield's Surgery, p. 1282
CT images of liver injury:
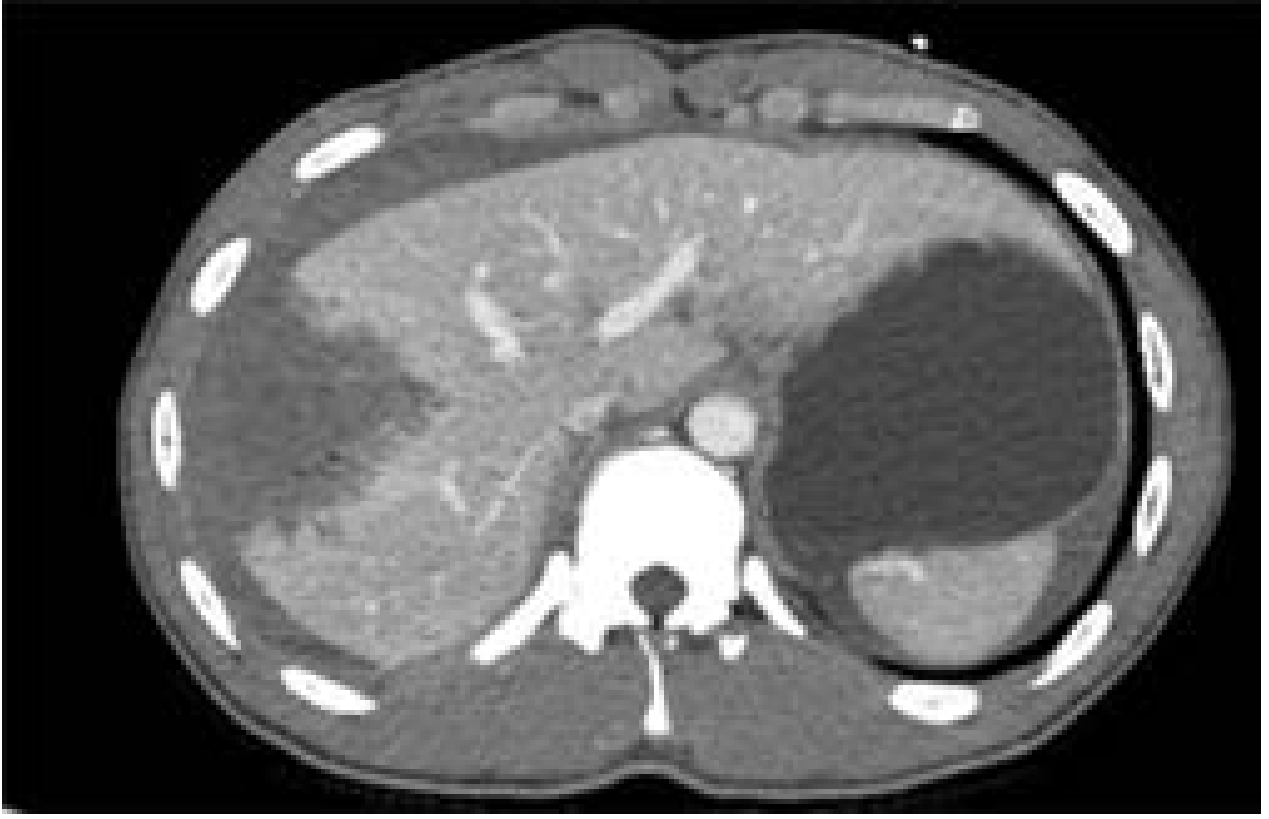
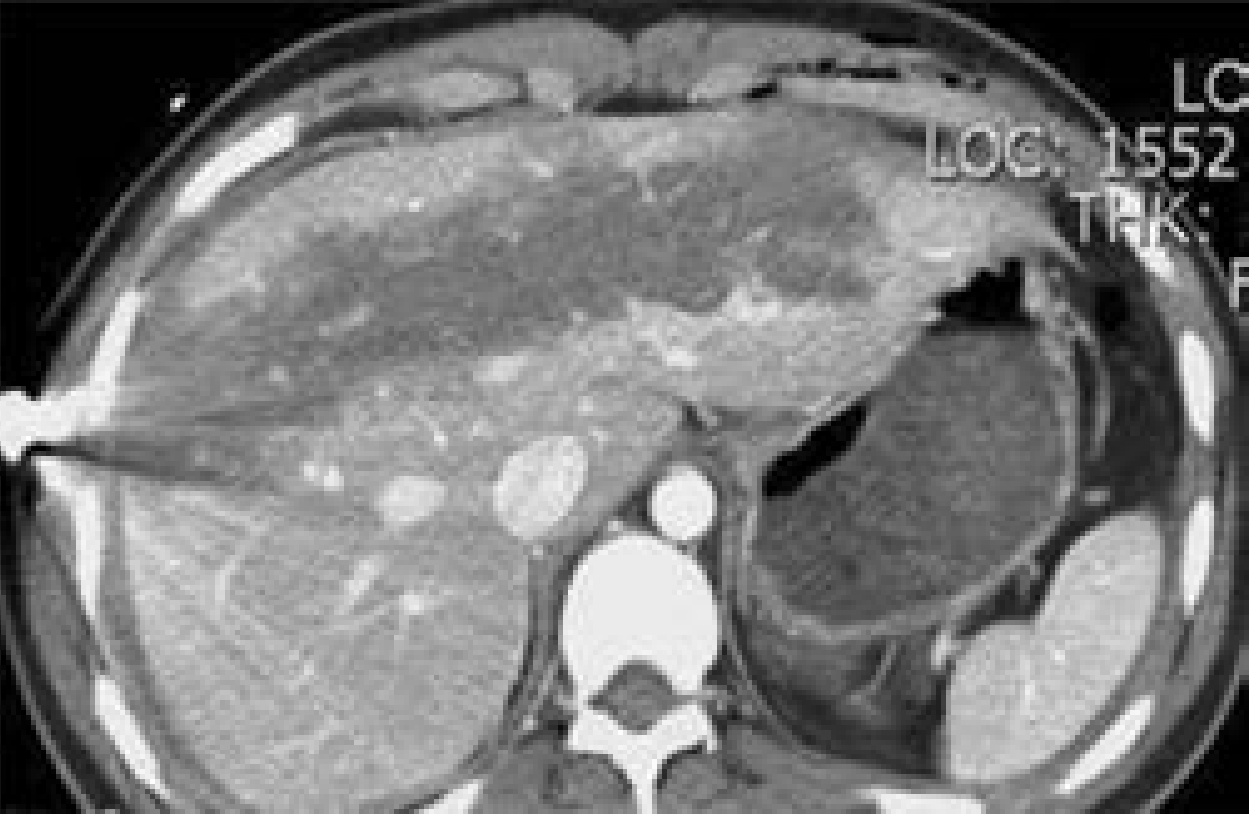
CT images of liver injury — Current Surgical Therapy 14e, p. 1342
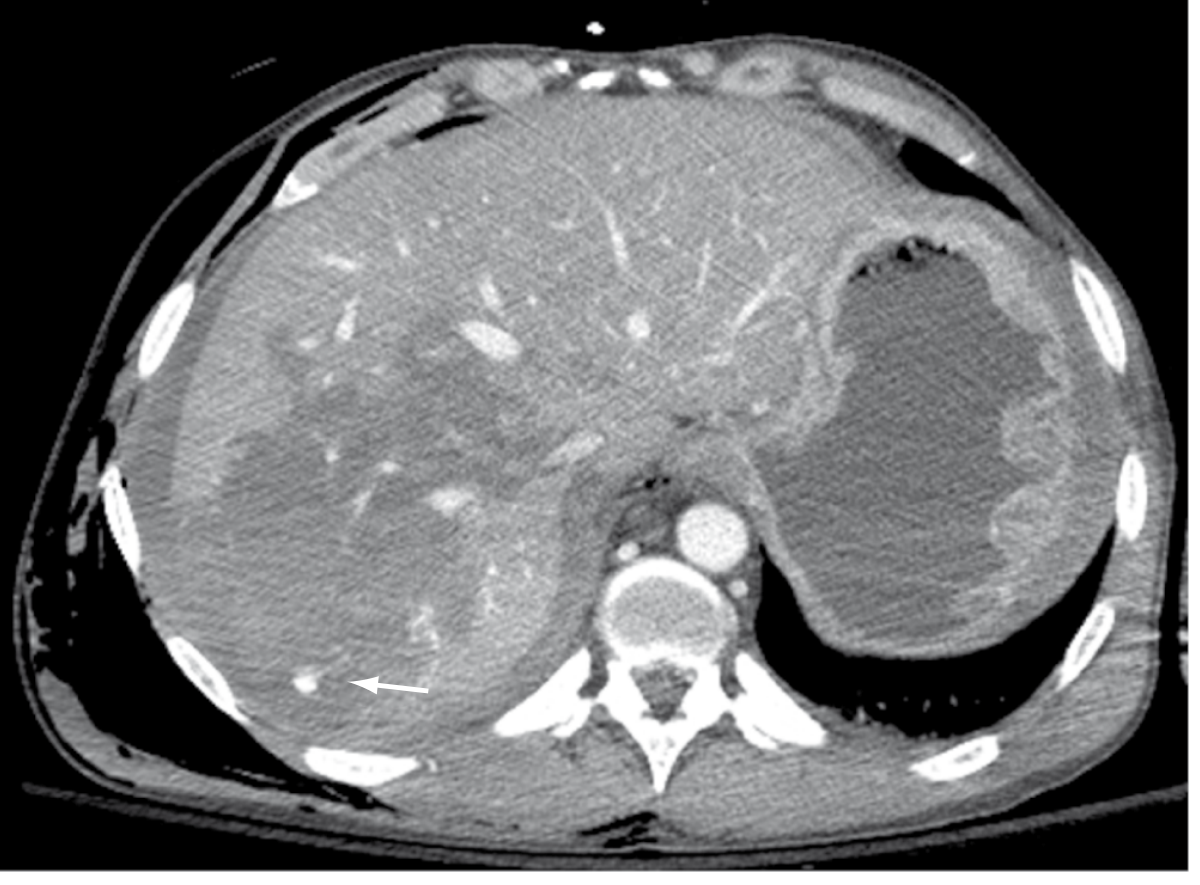
Grade IV liver laceration with active extravasation (arrow) — Sabiston Textbook of Surgery, p. 692
5. Non-Operative Management (NOM)
NOM has become the management of choice for haemodynamically stable patients and represents the dominant shift in hepatic trauma care over the past four decades.
Historical Evolution
- 1960–1975: Aggressive operative management (resection, hepatic artery ligation, T-tube choledochostomy); morbidity/mortality 27–65%
- 1976: Lucas and Ledgerwood shifted focus to bleeding control → mortality fell to 22%
- Modern era: NOM with selective angioembolisation; dramatically reduced in-hospital mortality
Criteria for NOM
- Haemodynamic stability or response to initial resuscitation
- No peritonitis
- Absence of associated injuries mandating laparotomy
- Grade I–III injuries are highly amenable; Grade IV–V can be attempted in stable patients
Failure Rates of NOM by Grade
Higher-grade injuries carry significantly higher NOM failure rates:
- Grade I–II: failure rate <5%
- Grade III: ~10%
- Grade IV: 14–30%
- Grade V: 50–75%
Failure is defined as haemodynamic deterioration, transfusion requirement, or development of peritonitis requiring operative conversion.
Monitoring During NOM
- ICU admission for high-grade injuries
- Serial haemoglobin/haematocrit monitoring
- CT scan follow-up for grade III–V injuries to exclude pseudoaneurysm or progression
— Sabiston Textbook of Surgery, p. 693; Current Surgical Therapy 14e, p. 1342–1343; Mulholland and Greenfield's Surgery, p. 1282
6. Angioembolisation
Angioembolisation has become a critical adjunct to NOM and is used for:
- CT evidence of active arterial extravasation ("blush")
- Pseudoaneurysm or arteriovenous fistula on CT
- Patients who can be temporarily stabilised with volume resuscitation but are not candidates for immediate laparotomy
Technique: Selective hepatic artery embolisation using coils or gelfoam, targeting the bleeding vessel while preserving hepatic parenchyma. Bilaterally or centrally positioned pseudoaneurysms may require more proximal embolisation.
Haemodynamic instability mandates expeditious operative management; angiography/embolisation is reserved for patients who can be stabilised.
— Current Surgical Therapy 14e, p. 1342–1343
7. Operative Management
Indications for Surgery
- Haemodynamic instability not responding to resuscitation
- Peritonitis
- Active haemorrhage demonstrated on CT in an unstable patient
- Failure of NOM
- Associated injuries mandating laparotomy (hollow viscus injury, diaphragmatic injury)
Initial Operative Steps (Damage Control Philosophy)
Access is via a midline laparotomy. The abdomen is entered and:
- Manual compression of the liver and packing of the perihepatic space for temporary haemostasis
- Rapid assessment of the entire abdomen for life-threatening associated injuries
- Pringle manoeuvre (manual or tourniquet compression of the hepatoduodenal ligament) to reduce hepatic inflow
Haemostatic Techniques
Simple Bleeding Control (Low-Grade Injuries, Grades I–III)
- Direct pressure and packing — the most reliable initial manoeuvre
- Electrocautery or argon beam coagulator for surface bleeding
- Topical haemostatic agents (Gelfoam, oxidised cellulose, thrombin, fibrin glue)
- Suture hepatorrhaphy — mattress sutures across lacerations to compress parenchymal vessels
- Omental pack — plugging the laceration with a pedicle of omentum
Deeper Bleeding Control
- Hepatotomy with selective vascular ligation: the laceration is opened (finger fracture technique) to directly identify and ligate bleeding vessels and bile ducts
- Balloon tamponade (Foley catheter inflated within a tract for penetrating wounds)
- Pringle manoeuvre: occlusion of the portal triad (portal vein + hepatic artery) by compression or tourniquet; safe for up to 60 minutes (longer with ischaemic preconditioning)
Hepatic Resection
- Debridement resection (non-anatomic resection): removal of devitalised tissue while following the injury plane; most common resective technique in trauma
- Anatomic hepatic resection (lobectomy, segmentectomy): reserved for selected cases with massive injury along a segmental or lobar plane; technically demanding and time-consuming — generally avoided in unstable patients
Juxtahepatic Venous Injuries (Grade V Vascular)
Among the most lethal injuries in trauma surgery. Options include:
- Atriocaval shunt (Schrock shunt): a large-bore tube inserted via the right atrial appendage and advanced into the IVC to divert IVC blood flow while the retrohepatic IVC/hepatic vein injury is repaired
- Veno-venous bypass
- Total hepatic vascular isolation: hepatic inflow occlusion (Pringle) combined with suprahepatic and infrahepatic IVC clamping
- Mortality remains >50% despite these approaches
Damage Control Surgery (DCS)
For physiologically compromised patients (coagulopathy, acidosis, hypothermia — the "lethal triad"):
- Phase I — Haemorrhage and contamination control only; pack perihepatic spaces; temporary abdominal closure (damage control laparotomy)
- Phase II — ICU resuscitation: correction of coagulopathy, acidosis, hypothermia; massive transfusion protocol
- Phase III — Planned re-exploration at 24–72 hours for pack removal, definitive repair, and abdominal closure
The concept was promoted by Feliciano, Mattox, and Jordan and is now standard of care for physiologically unstable patients with high-grade liver injuries.
— Sabiston Textbook of Surgery, p. 693–695; Current Surgical Therapy 14e, p. 1343–1346; Mulholland and Greenfield's Surgery, p. 1282–1284
8. Complications
| Complication | Notes |
|---|---|
| Rebleeding | Most common cause of NOM failure; peaks in first 48–72 hours |
| Bile leak / biloma | From disrupted intrahepatic bile ducts; managed with ERCP + stenting or percutaneous drainage |
| Haemobilia | Classically: Quincke's triad (RUQ pain, jaundice, GI haemorrhage); treated with angioembolisation |
| Hepatic abscess / infected necrosis | Devitalised parenchyma prone to infection; requires CT-guided or surgical drainage |
| Hepatic necrosis | From extensive injury or hepatic artery ligation |
| Biliary peritonitis | Bile leakage into peritoneum requiring operative washout |
| Hepatic arteriovenous fistula | Late complication; presents with bruit, high-output failure; treat with embolisation |
| Post-traumatic haemorrhage | Delayed rupture of subcapsular haematoma |
| Coagulopathy | Consumption and hypothermia; managed with massive transfusion protocol |
| Abdominal compartment syndrome | Following massive resuscitation; managed with decompressive laparotomy |
— Sabiston Textbook of Surgery, p. 695; Grainger & Allison's Diagnostic Radiology, p. 661; Current Surgical Therapy 14e, p. 1346
9. Special Situations
Penetrating Liver Trauma
- Gunshot wounds carry higher mortality than stab wounds due to greater energy transfer
- The liver is the most commonly injured organ in penetrating abdominal trauma
- Stable patients with gunshot wounds to the abdomen may be managed with NOM in select centres with serial clinical monitoring
Figure — Gunshot injury to the liver successfully managed nonoperatively:
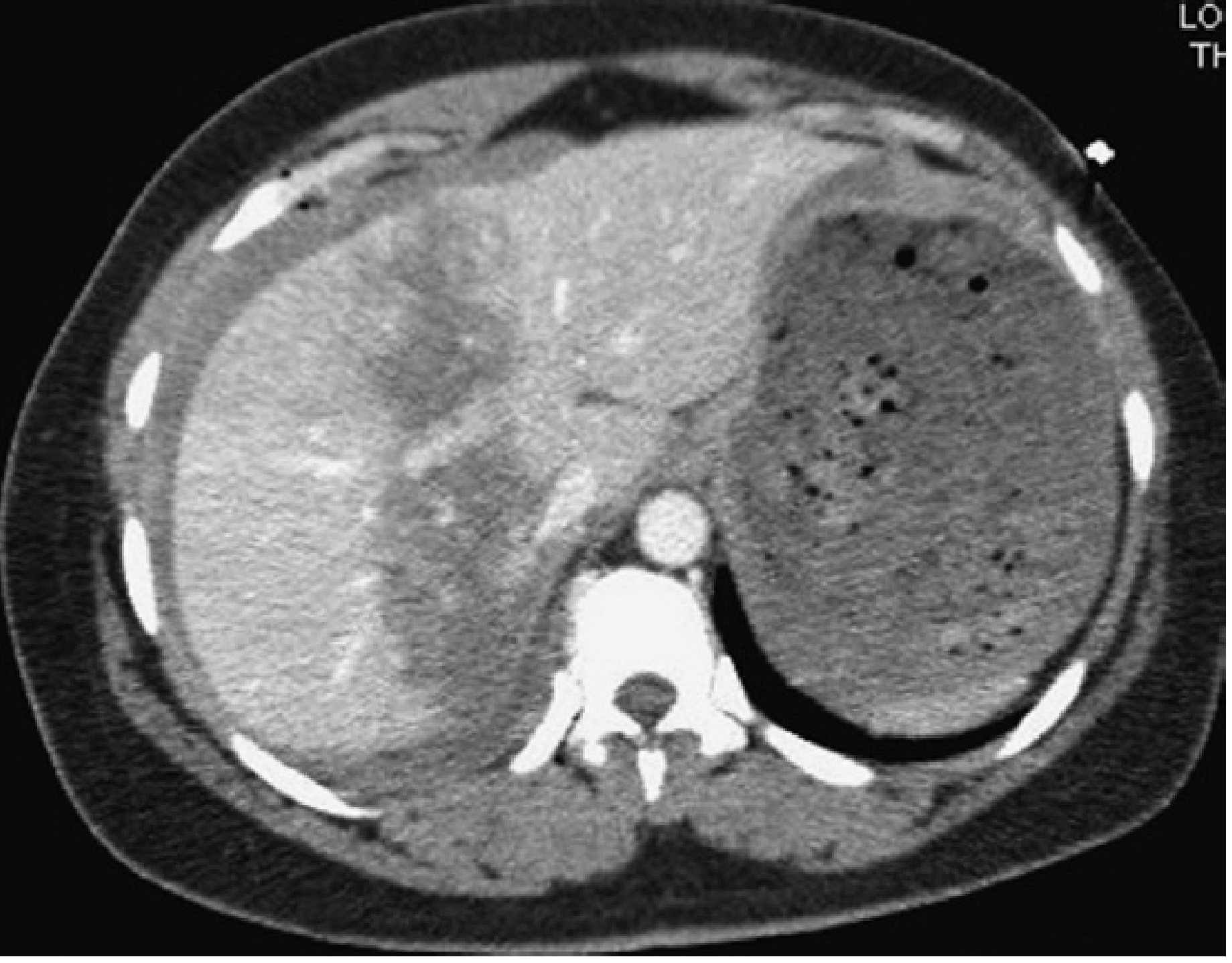 — Mulholland and Greenfield's Surgery, p. 1280
— Mulholland and Greenfield's Surgery, p. 1280

Hepatic Trauma in the Context of Coagulopathy / Anticoagulation
- Pre-existing coagulopathy (anticoagulants, liver disease, haematological disorders) significantly worsens haemorrhage control
- Early reversal agents and massive transfusion protocols are essential
Role of MRI
MRI is not used for acute hepatic trauma but is useful for monitoring established haematomas — methaemoglobin in maturing blood increases signal intensity on T1-weighted images.
10. Summary of Management Algorithm
Liver Trauma Suspected
│
▼
ATLS Primary Survey + Resuscitation
│
┌────┴────┐
Unstable Stable
│ │
FAST (+) CT Abdomen (IV contrast)
Operative ↓
Room Grade + Vascular Injury?
│ │
No blush Blush/PSA
Grade I–III Grade IV–V
↓ ↓
NOM Angioembolisation
ICU ± NOM if stable
Monitoring │
Failure / Unstable
↓
Laparotomy:
Packing → DCS → ICU
Definitive repair at 48–72h
Key References
- Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice — Chapter 36, Hepatic Injuries (p. 692–695)
- Mulholland and Greenfield's Surgery: Scientific Principles and Practice, 7e — Chapter 25 (p. 1279–1284)
- Current Surgical Therapy, 14e — Liver Trauma chapter (p. 1341–1346)
- Grainger & Allison's Diagnostic Radiology — Hepatic Trauma, Chapter 23 (p. 661)
- Moore EE et al. Organ injury scaling: spleen and liver (1994 revision). J Trauma 1995;38:323–324 (AAST OIS scale)
This is a shared conversation. Sign in to Orris to start your own chat.