Normal Labour comprehensive review with exam ready notes
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Here are comprehensive, exam-ready notes on Normal Labour:
Normal Labour — Comprehensive Exam-Ready Notes
1. DEFINITION
Labour = sufficient uterine contractions in frequency, intensity, and duration to produce progressive effacement and dilation of the cervix.
- Onset is clinically defined as painful, regular uterine contractions associated with cervical effacement and dilation
- Precise onset is often uncertain — the latent phase blurs into prelabour
Key mediators of labour initiation: Prostaglandins — particularly PGE₂ and PGF₂α — plus oxytocin, oestrogen/progesterone ratio changes, and inflammatory cytokines.
2. PRELABOUR CHANGES
| Sign | Notes |
|---|---|
| Lightening | Fetal head descends into pelvis; fundal height drops; pelvic pressure increases; may occur gradually over weeks or acutely |
| Cervical ripening | Effacement and early dilation begin in the third trimester before contractions are felt |
| Bloody show | Blood-tinged mucous plug passage; true labour usually within 3 days |
| Braxton-Hicks (false labour) | Irregular, short, painless contractions; aid cervical effacement |
Nulliparas show greater effacement in prelabour; multiparas show greater dilation (Hendricks et al.). Mean cervical dilation in the last 3 days before labour: 1.8 cm nullipara / 2.2 cm multipara.
3. STAGES OF LABOUR
| Stage | From | To |
|---|---|---|
| 1st stage | Onset of regular contractions | Full cervical dilation (10 cm) |
| 2nd stage | Full dilation | Delivery of the infant |
| 3rd stage | Delivery of infant | Delivery of the placenta |
| 4th stage* | Delivery of placenta | ~1 hour postpartum (uterine contraction) |
*Not universally accepted as an official stage.
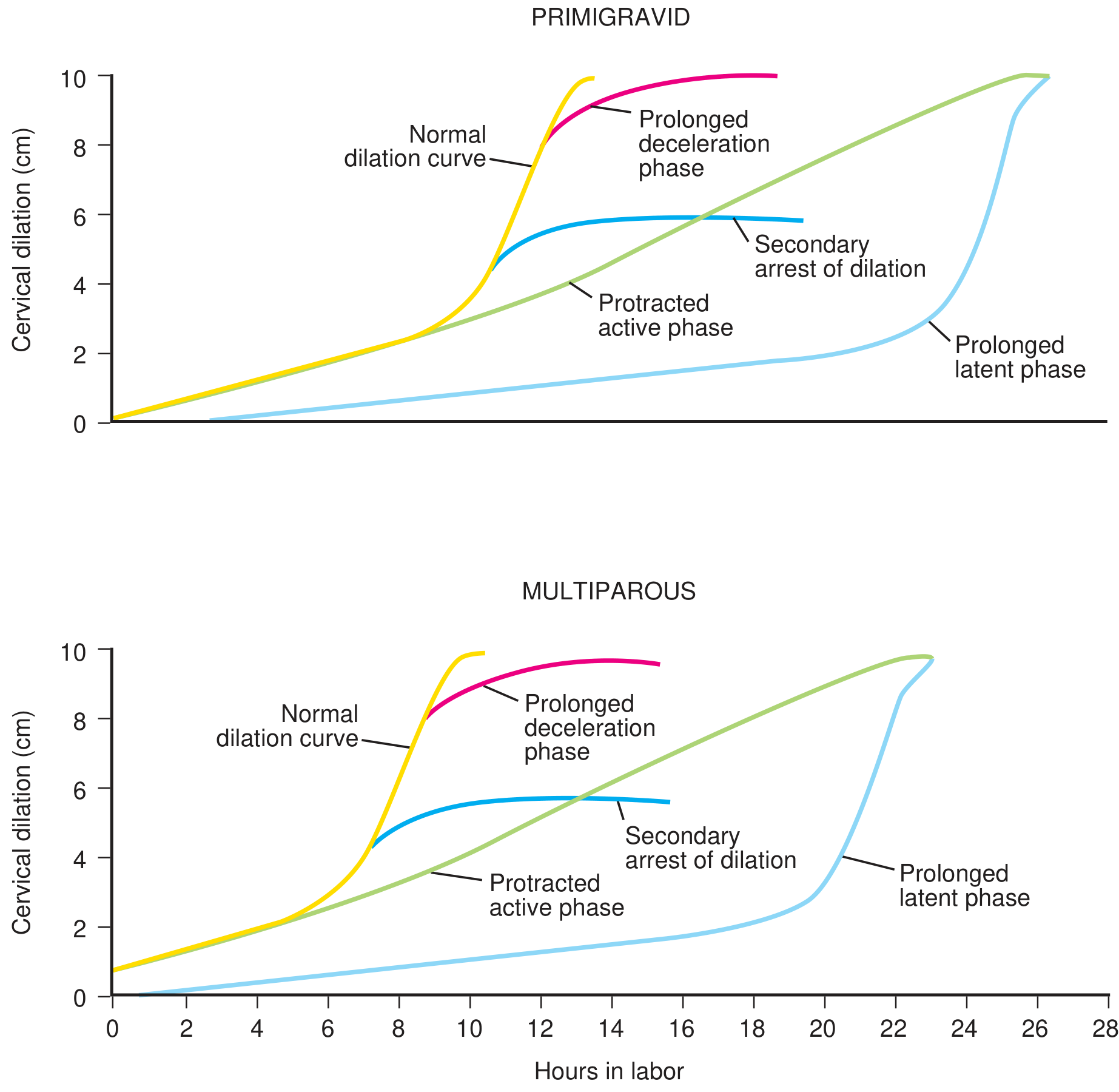
4. FIRST STAGE — FRIEDMAN CURVE
The relationship of cervical dilation to time follows a sigmoid (S-shaped) curve described by Friedman.
4a. Latent Phase
- Onset of contractions → beginning of active phase
- Little cervical dilation; major cervical changes are softening, effacement, and positioning
- Upper limits of normal:
- Nullipara: < 20 hours
- Multipara: < 14 hours
- Latent phase mean duration (Friedman data): ~6.4 hr nullipara / ~4.8 hr multipara
4b. Active Phase
- Begins at ~3–4 cm dilation (Friedman era) or ~6 cm (contemporary data — Zhang et al.)
- Rapid, near-linear cervical dilation
- Minimum rates (Friedman criteria):
- Nullipara: ≥ 1.2 cm/hr
- Multipara: ≥ 1.5 cm/hr
- Modern data (Zhang) show contemporary active phase begins later and progresses more slowly than Friedman described
Exam tip: Active phase arrest = no dilation for ≥ 2 hours in the active phase.
Friedman Table — Duration of First Stage:
| Nullipara (mean ± SD) | Multipara (mean ± SD) | |
|---|---|---|
| Latent phase | 6.4 ± 5.1 hr | 4.8 ± 4.9 hr |
| Active phase | 4.6 ± 3.6 hr | 2.4 ± 2.2 hr |
| Total 1st stage | 11.0 ± 8.7 hr | 7.2 ± 7.1 hr |
5. FETAL DESCENT
- Descent begins well before the second stage (often in late first stage)
- Rate of descent increases in late first stage and continues linearly into the second stage
- Maximum rate of descent (Friedman):
- Nullipara: 3.3 ± 2.3 cm/hr
- Multipara: 6.6 ± 4.4 cm/hr
Station is measured in relation to the ischial spines (−5 to +5 cm scale, or in thirds):
- Negative = above spines (not engaged)
- Zero = at the level of spines (engaged)
- Positive = below spines
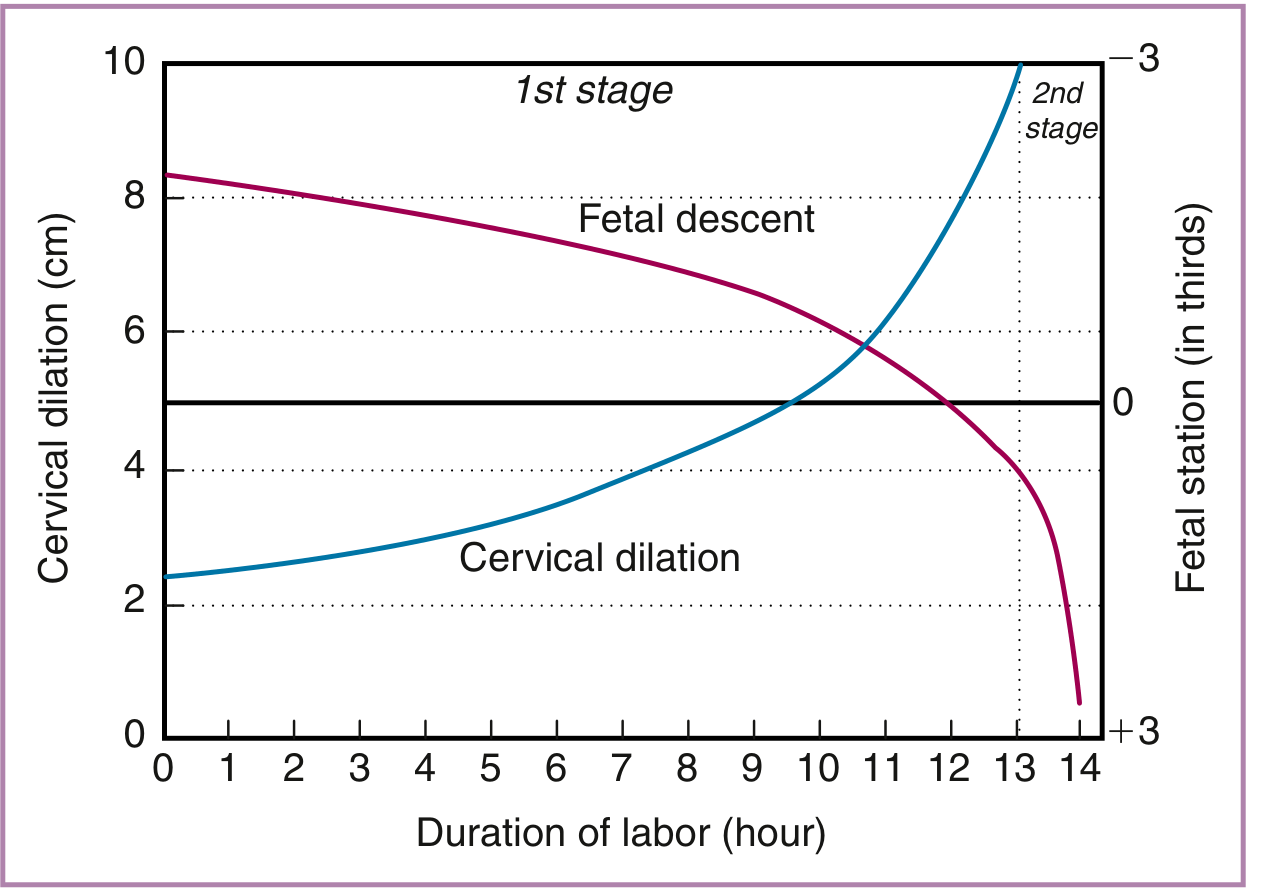
Zhang fetal descent data (median, 5th–95th percentile):
| Station | Time interval (hr) |
|---|---|
| −2 to −1 | 7.9 (0.9–65) |
| −1 to 0 | 1.8 (0.1–23) |
| 0 to +1 | 1.4 (0.1–13) |
| +1 to +2 | 0.4 (0.04–3.8) |
| +2 to +3 | 0.1 (0.02–0.9) |
6. SECOND STAGE
- Full dilation → delivery of the infant
- Fetal head descent + rotation occur throughout this stage
- Historically limited to 2 hours; modern evidence does not support rigid time cutoffs when there is fetal well-being and progress
Current guidance on second-stage limits:
- If no serious FHR abnormalities, mother is well hydrated and comfortable, and there is any progress in descent/rotation → no mandatory operative delivery
- Prolonged second stage (epidural): Nullipara > 3 hr; Multipara > 2 hr — associated with increased chorioamnionitis, severe perineal lacerations, uterine atony, PPH
- Rouse et al.: 55% of women with second stage ≥ 3 hours still delivered vaginally
7. THIRD STAGE
- Delivery of infant → delivery of placenta
- Normal duration: < 30 minutes (typically 5–15 min)
- Signs of placental separation: fundal rise, cord lengthening, gush of blood, uterus becomes globular
- Active management (oxytocin + controlled cord traction) reduces PPH risk
8. UTERINE CONTRACTIONS IN ACTIVE LABOUR
| Parameter | Value |
|---|---|
| Frequency | Every 2–3 minutes |
| Duration | ~60 seconds |
| Intensity (IUPC) | Mean 40 mmHg above baseline |
| Adequate relaxation between contractions | Essential for fetal oxygenation through intervillous space |
Contractions 1–2 minutes apart may indicate increased intrauterine pressure — rule out abruptio placentae.
9. CARDINAL MOVEMENTS OF LABOUR (Second Stage)
Mnemonic: "Every Darn Fetus Internally Rotates, Exits Externally"
- Engagement — biparietal diameter at the pelvic inlet
- Descent — throughout labour, most active in 2nd stage
- Flexion — chin to chest; reduces presenting diameter
- Internal rotation — occiput rotates to anterior (OA)
- Extension — head delivers under pubic symphysis
- External rotation (restitution) — shoulders align with AP diameter
- Expulsion — delivery of shoulders and body
10. LABOUR ABNORMALITIES (High-Yield Exam Contrasts)
| Abnormality | Definition | Nullipara | Multipara |
|---|---|---|---|
| Prolonged latent phase | Latent phase exceeding upper limit | > 20 hr | > 14 hr |
| Protracted active phase dilation | Slow but continuing dilation | < 1.2 cm/hr | < 1.5 cm/hr |
| Arrest of active phase | No dilation for ≥ 2 hr | ≥ 2 hr | ≥ 2 hr |
| Protracted descent | Slow fetal descent | < 1.0 cm/hr | < 2.0 cm/hr |
| Arrest of descent | No descent for ≥ 1 hr | ≥ 1 hr | ≥ 1 hr |
11. FETAL HEART RATE MONITORING
Baseline FHR
- Normal: 110–160 bpm
- Tachycardia: > 160 bpm; bradycardia: < 110 bpm
Variability
- Normal (moderate): 6–25 bpm fluctuation — most reassuring
- Absent/minimal variability: concerning for hypoxia or CNS depression
Accelerations
- Transient FHR rise ≥ 15 bpm above baseline lasting ≥ 15 seconds
- Reassuring; basis of reactive NST (≥ 2 accelerations)
Decelerations
| Type | Cause | Pattern | Significance |
|---|---|---|---|
| Early | Head compression (vagal) | Mirror of contraction; nadir at peak | Benign |
| Variable | Cord compression | Abrupt, V/U/W shaped; variable timing | Most common (50–80%); mild-moderate usually benign |
| Late | Uteroplacental insufficiency | Delayed start/nadir after contraction peak | Non-reassuring; indicates fetal compromise |
Variable decelerations are the most common deceleration type, occurring in 50–80% of deliveries.
Causes of Fetal Bradycardia (exam list)
Cord prolapse/compression, hypothermia, tetanic contractions, paracervical block, epidural/spinal analgesia, maternal seizure, rapid descent, vigorous vaginal exam, congenital heart disease, fetal heart block, severe hypoxia.
12. ANALGESIA IN LABOUR
| Method | Notes |
|---|---|
| Epidural | Most effective; prolongs second stage; may reduce maternal expulsive sensation |
| Spinal | Single shot; used for operative delivery |
| Combined spinal-epidural (CSE) | Rapid onset + titratable |
| Systemic opioids (morphine, fentanyl) | Partial pain relief; cross placenta — neonatal respiratory depression possible |
| Entonox (N₂O) | 50:50 nitrous oxide/oxygen; mild analgesia; patient-controlled |
13. QUICK-FIRE EXAM FACTS
- Labour is defined by painful contractions + cervical change (not contractions alone)
- Cervical dilation of 10 cm = full dilation = end of 1st stage
- Active phase begins at 6 cm by contemporary data (Zhang) vs. 3–4 cm (Friedman)
- Nullipara latent phase limit = 20 hr; multipara = 14 hr
- Active phase arrest = no dilation for ≥ 2 hours with adequate contractions
- Bloody show → labour typically within 3 days
- Normal contraction frequency in active labour = every 2–3 min
- Placenta should deliver within 30 minutes of birth (3rd stage)
- Most common deceleration = variable (cord compression)
- Late decelerations = uteroplacental insufficiency — never benign
- Mueller-Hillis maneuver = fundal pressure + vaginal exam to assess head descent (helps predict cephalopelvic proportion)
Sources: Creasy & Resnik's Maternal-Fetal Medicine (Thorp & Grantz, "Normal Labor and Its Limits"), p. 936–943 | Textbook of Family Medicine 9e ("Normal Labor and Delivery"), p. 502–510
Act as a PhD scholar and give me review of pregnancy induced hypertension from standard textbooks with citations and bibliography at end.
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Pregnancy-Induced Hypertension: A Comprehensive Scholarly Review
Abstract
Pregnancy-induced hypertension (PIH), encompassing gestational hypertension, preeclampsia, and eclampsia, constitutes one of the most clinically significant complications of human gestation. Affecting approximately 2–10% of all pregnancies globally, PIH remains a leading cause of maternal and perinatal morbidity and mortality. Its pathogenesis, rooted in aberrant placentation and systemic endothelial dysfunction, has been substantially illuminated over the past three decades, yet definitive aetiology remains elusive. This review synthesises current knowledge from authoritative medical textbooks regarding classification, epidemiology, pathophysiology, clinical manifestations, diagnostic criteria, complications, and management of PIH, with particular attention to molecular mechanisms and therapeutic considerations.
1. Introduction
Hypertension in pregnancy represents a heterogeneous spectrum of disorders that share the common feature of elevated arterial blood pressure arising during gestation. The term "pregnancy-induced hypertension" (PIH) refers specifically to hypertensive disorders that are a direct consequence of pregnancy itself and resolve in the postpartum period, thereby distinguishing them from chronic or pre-existing hypertension. The clinical and scientific importance of PIH cannot be overstated: it contributes substantially to preterm delivery, placental abruption, fetal growth restriction, and in its most severe manifestations, maternal seizures, end-organ failure, and death.¹
The disorder has long been recognised — eclampsia was described in ancient Hippocratic texts — yet its molecular underpinnings began to be understood only in the modern era of vascular biology and molecular medicine. Understanding PIH demands an integrative command of obstetrics, vascular physiology, immunology, and pharmacology.
2. Classification
The following classification is widely adopted in contemporary clinical and academic practice:¹,²
| Category | Defining Features |
|---|---|
| Gestational hypertension | New-onset BP ≥ 140/90 mmHg after 20 weeks gestation; no proteinuria; resolves postpartum |
| Preeclampsia | Gestational hypertension + proteinuria (> 300 mg/24 h) and/or signs of end-organ damage |
| Eclampsia | Preeclampsia + generalised tonic-clonic seizures not attributable to other causes |
| Superimposed preeclampsia | New-onset proteinuria or worsening hypertension in a woman with pre-existing chronic hypertension |
| Chronic hypertension | Present before 20 weeks gestation or persisting > 6 weeks postpartum |
Preeclampsia is further stratified clinically into:
- Without severe features — BP 140–159/90–109 mmHg with proteinuria, without end-organ involvement
- With severe features — BP ≥ 160/110 mmHg, or evidence of renal, hepatic, neurological, or haematological compromise²
3. Epidemiology
The incidence of hypertensive disorders of pregnancy is approximately 85 per 1,000 deliveries in the United States, with significant regional variation.² Preeclampsia alone affects 3–5% of pregnant women worldwide, with eclampsia complicating approximately 1 in 2,000 deliveries in developed nations.³ Hypertension overall is observed in up to 8–10% of pregnancies.¹,⁴
Principal risk factors for preeclampsia include:
- Nulliparity — the single greatest population-attributable fraction, accounting for 32.3% (95% CI: 27.4–37.0%) of cases²
- Extremes of reproductive age (adolescence and advanced maternal age)
- Prior preeclampsia
- Multiple gestation and molar pregnancy
- Chronic hypertension — 25% of such women develop superimposed preeclampsia²
- Pregestational diabetes mellitus — 20% overall risk; up to 70% in White's class F and R²
- Chronic renal disease
- Systemic lupus erythematosus and antiphospholipid syndrome
- Obesity and hypercholesterolaemia¹
- In vitro fertilisation and family history of preeclampsia²
Eclampsia demonstrates a clear socioeconomic gradient, being substantially more prevalent in women with limited access to quality obstetric care.²
4. Pathophysiology
4.1 The Placental Origin Hypothesis
The fundamental pathophysiological event in preeclampsia is inadequate trophoblastic invasion of the maternal spiral arteries.³,⁵ In a normal pregnancy, extravillous trophoblasts migrate into the decidua and myometrium, remodelling the musculoelastic walls of spiral arteries into wide, low-resistance vascular sinusoids capable of delivering the high-volume, low-pressure blood flow required by the growing placenta.
In preeclampsia, this invasion is superficial and incomplete. The spiral arteries retain their musculoelastic walls, remain narrow and high-resistance, and fail to transform into adequate conduits. The result is chronic uteroplacental ischaemia.³
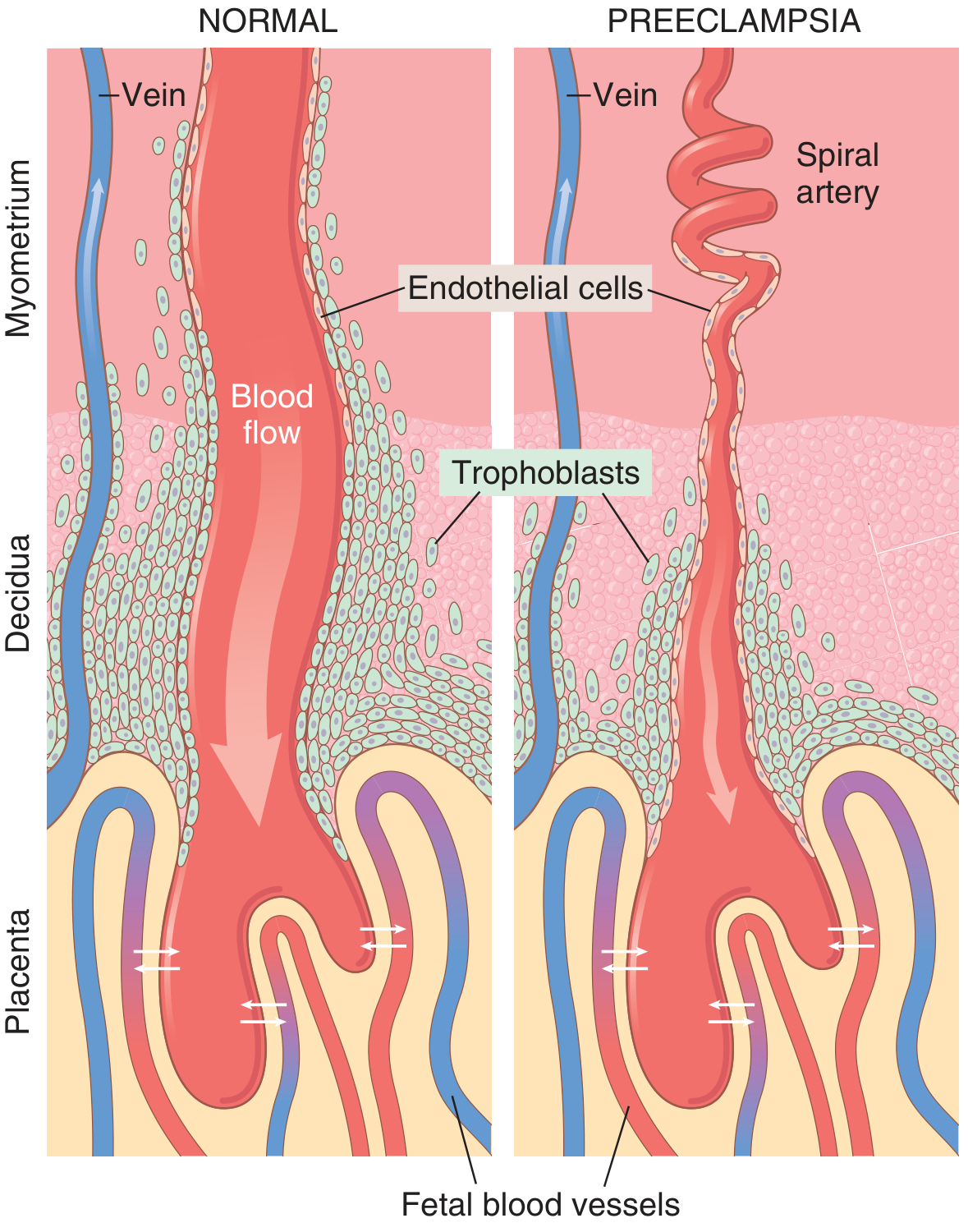
4.2 Antiangiogenic Factors and Endothelial Dysfunction
The ischaemic placenta releases circulating factors into the maternal bloodstream that produce widespread maternal endothelial dysfunction, the proximate cause of the systemic manifestations of preeclampsia.³,⁵
Two antiangiogenic proteins have been particularly implicated:
- Soluble fms-like tyrosine kinase-1 (sFlt-1) — a circulating decoy receptor that sequesters and antagonises VEGF (vascular endothelial growth factor) and PlGF (placental growth factor), thereby impairing endothelial maintenance and nitric oxide production
- Soluble endoglin (sEng) — antagonises TGF-β signalling in endothelial cells, compounding vascular dysfunction
Both sFlt-1 and soluble endoglin are measurably elevated in maternal circulation prior to the clinical onset of preeclampsia, offering potential predictive utility.³,⁵
Endothelial dysfunction results in:
- Reduced production of vasodilators: prostacyclin (PGI₂) and prostaglandin E₂
- Increased production of the vasoconstrictor thromboxane A₂ (TXA₂)
- The net result is systemic vasoconstriction, hypertension, and platelet activation³
4.3 Vascular Reactivity and Angiotensin II Hypersensitivity
A hallmark of preeclampsia is heightened vascular sensitivity to endogenous pressors. Normal pregnancy is a high-output, low-resistance state in which responsiveness to angiotensin II is markedly blunted — normotensive pregnant women require approximately 2.5 times as much angiotensin II to raise blood pressure by a given increment compared to non-pregnant women.²
In preeclampsia, this protective refractoriness is lost. Preeclamptic women are significantly more sensitive to angiotensin II than either normal pregnant or non-pregnant women, and this hypersensitivity may be detectable as early as 14 weeks of gestation — weeks before hypertension becomes clinically manifest.² Similarly, enhanced pressor responses to norepinephrine and vasopressin are documented. These findings point to a fundamental alteration in the vascular smooth muscle or endothelial milieu, rather than simple elevation of circulating pressors.
4.4 Inflammatory and Immunological Mechanisms
Experimental evidence supports a role for systemic inflammatory activation in preeclampsia. Elevated levels of tumour necrosis factor-α (TNF-α) and interleukin-6 (IL-6) are found in the circulation of preeclamptic women, likely triggered by the ischaemic placenta.⁵ An immunological dimension is further suggested by the protective effect of prior paternal antigen exposure (explaining the predisposition of nulliparae and the reduced risk in parous women with the same partner) and by the prompt resolution of all features following delivery of the placenta.³
4.5 Haemodynamic Profile
The haemodynamic sequence in preeclampsia has been characterised:¹
- Early phase: cardiac output is elevated and peripheral resistance begins to rise above normal
- Established disease: cardiac output falls as peripheral resistance rises substantially, producing a paradoxically contracted intravascular volume (despite oedema)
- Central venous pressures are typically normal; pulmonary capillary wedge pressure is variable
This contracted plasma volume is clinically important — overly aggressive diuresis is hazardous and vasopressors must be used with caution.
5. Systemic Consequences and Organ Pathology
Vasospasm, ischaemia, and microangiopathy in preeclampsia/eclampsia affect virtually every organ system:³
5.1 Renal Effects
- Glomerular endotheliosis — pathognomonic histological lesion: swelling of glomerular endothelial cells with protein deposits in the basement membrane, narrowing the capillary lumen
- Decreased renal blood flow and GFR (contrary to the physiological increase of normal pregnancy)
- Proteinuria (> 300 mg/24 h), resulting from disruption of the glomerular filtration barrier
- Oliguria and, in severe cases, acute kidney injury
5.2 Hepatic Effects
- Hepatocellular necrosis and periportal haemorrhage from vasospasm
- Elevated liver transaminases (AST, ALT)
- Right upper quadrant or epigastric pain (hepatic capsule distension)
- Subcapsular haematoma and hepatic rupture — rare but catastrophic
5.3 Haematological Effects
- Thrombocytopenia — platelet consumption secondary to endothelial activation and microthrombi
- Microangiopathic haemolytic anaemia (MAHA) — fragmentation of erythrocytes across fibrin strands in small vessels
- In severe cases, disseminated intravascular coagulation (DIC)
5.4 Neurological Effects
- Severe headache, visual disturbances (scotomata, blurred vision), hyperreflexia
- Cerebral oedema and vasoconstriction
- Eclamptic seizures — generalised tonic-clonic; mechanism incompletely understood but involves posterior reversible encephalopathy (cerebral vasospasm and auto-regulatory breakthrough)
- Cortical blindness and intracranial haemorrhage in severe cases
5.5 Placental and Fetal Effects
- Placental infarction (ischaemic necrosis from chronic hypoperfusion)
- Retroplacental haemorrhage and abruption
- Increased syncytial epithelial knots on placental villi
- Acute atherosis — fibrinoid necrosis of decidual vessels with lipid-laden macrophage accumulation (the vascular hallmark on pathology)³
- Fetal growth restriction, oligohydramnios, fetal distress
6. HELLP Syndrome
HELLP — Haemolysis, Elevated Liver enzymes, Low Platelets — is a particularly severe variant of preeclampsia occurring in approximately 10% of women with severe preeclampsia.³
Diagnostic criteria:
- Microangiopathic haemolysis (elevated LDH, abnormal peripheral blood smear)
- AST and ALT > 70 U/L¹
- Platelet count < 100,000/μL¹
HELLP can occur without antecedent hypertension or proteinuria, making early recognition challenging. It carries high risk of DIC, hepatic rupture, placental abruption, and maternal death. Delivery is the definitive treatment.
7. Diagnostic Criteria
Preeclampsia is diagnosed when:
- BP ≥ 140/90 mmHg on two occasions ≥ 4 hours apart, after 20 weeks gestation in a previously normotensive woman, plus
- Proteinuria > 300 mg/24 hours (or protein/creatinine ratio ≥ 0.3), or
- In the absence of proteinuria: thrombocytopenia (< 100,000/μL), renal insufficiency (creatinine > 1.1 mg/dL), impaired liver function (transaminases twice the upper limit of normal), pulmonary oedema, or new-onset headache unresponsive to analgesia²
Severe features are defined by:
- BP ≥ 160/110 mmHg on two occasions ≥ 4 hours apart
- Creatinine > 1.1 mg/dL or doubling of baseline
- Platelets < 100,000/μL
- Transaminases ≥ twice the upper limit of normal
- Pulmonary oedema
- New-onset headache or visual disturbances²
8. Management
8.1 General Principles
The only curative intervention in preeclampsia is delivery of the placenta. All clinical decisions must balance maternal safety against fetal maturity. The fundamental management framework is:²
- Term (≥ 37 weeks) with any preeclampsia: prompt delivery
- Late preterm (34–36⁶⁄₇ weeks) with severe features: deliver
- Early preterm (< 34 weeks) without contraindications: expectant management with close surveillance to extend gestation and allow antenatal corticosteroid administration (betamethasone)
Absolute contraindications to expectant management:
Eclampsia, pulmonary oedema, DIC, uncontrollable severe hypertension, abnormal fetal testing, placental abruption, stillbirth, nonviable fetus²
8.2 Antihypertensive Therapy
Drug initiation is recommended when:
- Systolic BP > 160 mmHg or diastolic BP > 105 mmHg⁴
Acute (inpatient) control:
- Hydralazine 5–10 mg IV/IM, repeated at 20-minute intervals⁴
- Labetalol 20 mg IV, escalating to 40 mg at 10 min if BP uncontrolled⁴
Chronic/outpatient management:
- α-Methyldopa 250 mg twice daily — centrally acting α₂ agonist; established safety profile in pregnancy⁴
- Labetalol 100 mg twice daily — combined α₁/β-blocker⁴
- Nifedipine 30 mg once daily — calcium channel blocker; reasonable evidence of safety⁴
Contraindicated agents:
- ACE inhibitors and angiotensin receptor blockers — unequivocal adverse fetal effects (renal agenesis, fetal hypotension, oligohydramnios)⁴
8.3 Seizure Prevention and Treatment — Magnesium Sulphate
Magnesium sulphate (MgSO₄) remains the agent of choice for both prophylaxis and treatment of eclamptic seizures.¹
Mechanism: Mg²⁺ acts as a physiological calcium antagonist, reducing neuronal excitability and cerebrovascular vasospasm. It has minimal antihypertensive effect but maintains uterine and fetal blood flow.¹
Standard regimen:
- Loading dose: 4–6 g IV over 15–20 minutes
- Maintenance: 1–2 g/hour IV infusion
- Monitor for toxicity: loss of deep tendon reflexes (first sign, at ~7 mEq/L), respiratory depression (> 10 mEq/L), cardiac arrest (> 15 mEq/L)
- Antidote: Calcium gluconate 1 g IV
Approximately 20% of eclamptic seizures occur postpartum, most within 48 hours of delivery — MgSO₄ should be continued for at least 24 hours postpartum in women with severe features.⁴
ACOG does not recommend routine MgSO₄ for preeclampsia without severe features, as no eclamptic seizures occurred among placebo patients in adequately powered trials.²
8.4 Fetal Surveillance
- Serial growth ultrasound (every 3–4 weeks)
- Umbilical artery Doppler velocimetry — reversed end-diastolic flow is an indication for delivery regardless of gestational age²
- Non-stress testing (NST) and biophysical profile
- Continuous electronic fetal monitoring in labour
9. Prognosis and Long-Term Sequelae
Proteinuria and hypertension typically resolve within 1–2 weeks of delivery in most cases.³ However, preeclampsia is not merely an acute obstetric event — it is a powerful predictor of long-term cardiovascular risk in the mother:
- Women with a history of preeclampsia have a 2-fold increased lifetime risk of ischaemic heart disease, stroke, and hypertension
- The shared pathophysiology (endothelial dysfunction, inflammation, abnormal lipid metabolism) suggests that preeclampsia may unmask underlying susceptibility to vascular disease rather than simply cause it
Women with a history of preeclampsia, particularly early-onset or recurrent disease, should receive long-term cardiovascular risk counselling and surveillance.
10. Prevention
- Low-dose aspirin (81 mg/day, initiated between 12–28 weeks): reduces thromboxane synthesis preferentially over prostacyclin, restoring prostaglandin balance; recommended by USPSTF for women at high risk¹
- Calcium supplementation: reduces risk in populations with low dietary calcium intake
- Low-molecular-weight heparin in antiphospholipid syndrome
- There is no evidence that dietary salt restriction, bed rest, or vitamin C/E supplementation reduces incidence
11. Conclusion
Pregnancy-induced hypertension represents a complex, multisystem disorder with a pathophysiological foundation in defective trophoblastic invasion, uteroplacental ischaemia, antiangiogenic factor release, and systemic endothelial dysfunction. Its clinical spectrum — from mild gestational hypertension to fulminant eclampsia and HELLP syndrome — demands a graduated yet vigilant management approach. Delivery remains the only definitive cure. The advent of biomarkers such as sFlt-1 and PlGF offers promise for earlier prediction and risk stratification, but their routine clinical integration remains an area of active research. The long-term cardiovascular implications for affected women make PIH a condition of significance far beyond the gestational period.
Bibliography
-
Tintinalli JE, Ma OJ, Yealy DM, et al. Tintinalli's Emergency Medicine: A Comprehensive Study Guide. 9th ed. McGraw-Hill Education; 2020. Chapter: "Preeclampsia and Eclampsia," pp. 2454–2470.
-
Resnik R, Lockwood CJ, Moore TR, et al. Creasy & Resnik's Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2022. Chapter 45: "Preeclampsia and Eclampsia," pp. 1057–1120.
-
Kumar V, Abbas AK, Aster JC. Robbins & Kumar Basic Pathology. 11th ed. Elsevier; 2023. Chapter: "Preeclampsia/Eclampsia," pp. 701–702.
-
Brunton LL, Knollmann BC, eds. Goodman & Gilman's The Pharmacological Basis of Therapeutics. 14th ed. McGraw-Hill Education; 2023. Chapter: "Pregnancy-Induced Hypertension/Preeclampsia," pp. 95–104.
-
Hall JE, Hall ME. Guyton and Hall Textbook of Medical Physiology. 14th ed. Elsevier; 2021. Chapter 83: "Pregnancy and Lactation — Preeclampsia and Eclampsia," pp. 1040–1042.
-
Marx JA, Hockberger RS, Walls RM, eds. Rosen's Emergency Medicine: Concepts and Clinical Practice. 10th ed. Elsevier; 2023. Chapter: "Pregnancy-Induced Hypertension (Preeclampsia and Eclampsia)," pp. 4741–4776.
This review was compiled from primary medical textbook sources. All clinical figures cited reflect data published in the referenced editions.
This is a shared conversation. Sign in to Orris to start your own chat.