Management of polyartharlgia with anti u1 rnp strongly positive
Management of polyartharlgia with anti u1 rnp strongly positive
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have sufficient information to provide a comprehensive, well-referenced answer. Here is the management of polyarthralgia with strongly positive anti-U1 RNP:
Polyarthralgia with Strongly Positive Anti-U1 RNP
What This Means Clinically
A strongly positive anti-U1 RNP antibody in the context of polyarthralgia points toward Mixed Connective Tissue Disease (MCTD). Anti-U1 RNP is always present in MCTD and is its serologic hallmark. The full diagnostic picture of MCTD includes:
- Raynaud phenomenon
- Polyarthralgia / arthritis / swollen joints
- Swollen ("sausage") fingers
- Esophageal dysmotility
- Muscle weakness
- ANA in a speckled/particulate pattern (reflecting high-titer RNP)
MCTD is distinguished from simple overlap syndromes because it requires high-titer anti-RNP without anti-Sm antibodies. — Andrews' Diseases of the Skin, p. 290
Management of Polyarthralgia in MCTD
1. Mild Disease (Arthralgia / Arthritis Without Organ Involvement)
| Agent | Role |
|---|---|
| NSAIDs | First-line for arthralgias, joint pain, fever, and mild serositis. COX-2 inhibitors offer similar efficacy. Use cautiously given risk of hepatic/renal toxicity in CTDs. |
| Hydroxychloroquine (HCQ) 5 mg/kg/day | Recommended for all MCTD/SLE-spectrum patients. Effective for arthralgias, arthritis, rash, alopecia, and malaise. Reduces flares and long-term damage. Also reduces thrombosis risk if antiphospholipid antibodies are co-present. |
| Low-dose corticosteroids | For inflammatory arthritis not controlled by NSAIDs/HCQ — short-course prednisone to bridge until steroid-sparing therapy takes effect. |
— The Washington Manual of Medical Therapeutics, p. 957
2. Moderate-to-Severe Inflammatory Arthritis / Myositis
| Agent | Dosage | Notes |
|---|---|---|
| Prednisone | 1 mg/kg/day PO | Effective for inflammatory features (arthritis, myositis). Taper once controlled (reduce ~10% every 7–10 days). |
| Methotrexate | 10–25 mg/week PO or SC | Primary steroid-sparing agent for musculoskeletal manifestations. Add folic acid 1 mg/day to reduce side effects. |
| Azathioprine | 1–2.5 mg/kg/day | Alternative steroid-sparing DMARD; also used for skin/serositis manifestations. |
— Andrews' Diseases of the Skin, p. 303; Washington Manual, p. 957–958
3. Refractory or Life-Threatening Disease
- Rituximab: For features refractory to conventional therapy — 80% response rate for MCTD-associated thrombocytopenia; also used in severe SLE-overlap. — Andrews' Diseases of the Skin, p. 303
- IV pulse methylprednisolone (500–1000 mg/day × 3–5 days): For rapidly progressive or life-threatening manifestations.
Important Co-management Considerations
| Issue | Action |
|---|---|
| Osteoporosis prevention | Start early — MCTD itself carries independent osteoporosis risk, compounded by long-term steroids. Use bisphosphonates + calcium/Vitamin D. |
| Raynaud phenomenon | Calcium channel blockers (e.g., nifedipine); avoid cold exposure. |
| Pulmonary screening | Pulmonary arterial hypertension (PAH) and ILD are the most serious complications and major causes of death — screen with PFTs and echocardiography. |
| Renal monitoring | Renal disease is less common than in scleroderma but should be monitored. |
| Steroid-sparing strategy | Introduce DMARD early to minimize cumulative steroid dose. |
— Andrews' Diseases of the Skin, p. 301–303
Key Principle
The lupus-like features of MCTD (including arthritis) are the most responsive to treatment; scleroderma-like features are least likely to improve. The overall prognosis of MCTD is better than scleroderma, largely due to lower renal involvement. — Andrews' Diseases of the Skin, p. 303
Summary for polyarthralgia specifically: Start with hydroxychloroquine ± NSAIDs. Add low-dose prednisone for active inflammatory flares and bridge to methotrexate as the preferred steroid-sparing DMARD. Monitor closely for systemic evolution (PAH, ILD, myositis) as MCTD can differentiate into SLE, systemic sclerosis, or polymyositis over time.
Investigations to be done before starting methotrexate ?
Investigations to be done before starting methotrexate ?
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Loading Image
Excellent — comprehensive data gathered. Here is the full answer:
Investigations Before Starting Methotrexate
1. Haematology
| Test | Rationale |
|---|---|
| CBC with differential + platelets | MTX causes myelosuppression. Significant pre-existing anaemia, leukopenia, or thrombocytopenia are absolute contraindications. |
2. Renal Function
| Test | Rationale |
|---|---|
| Serum creatinine, BUN | MTX is renally excreted; renal dysfunction causes drug accumulation → toxicity. GFR should be calculated in all patients (formal eGFR if at risk). Renal impairment is a relative contraindication requiring dose reduction. |
3. Liver Function
| Test | Rationale |
|---|---|
| AST, ALT, Alkaline phosphatase, serum albumin | MTX is hepatotoxic — causes toxic hepatitis and liver fibrosis/cirrhosis with chronic use. Active hepatic disease is a relative contraindication. |
| Hepatitis B surface antigen (HBsAg), anti-HBc | MTX can trigger HBV reactivation — a potentially fatal complication. |
| Hepatitis C antibody | Active hepatitis C increases risk of MTX hepatotoxicity. |
4. Infection Screening
| Test | Rationale |
|---|---|
| Tuberculin skin test (PPD) or IGRA (QuantiFERON-Gold) | MTX is a long-term immunosuppressant; latent TB must be ruled out / treated before initiating. Strongly recommended by the CDC for any long-term immunosuppressive therapy. |
| HIV screening (where indicated) | HIV infection increases risk of opportunistic infections and MTX toxicity. |
5. Reproductive / Pregnancy
| Test | Rationale |
|---|---|
| Urine / serum β-hCG (women of childbearing age) | MTX is Category X — teratogenic and embryocidal. Pregnancy is an absolute contraindication. Contraception counselling is mandatory. |
6. Chest X-ray
| Test | Rationale |
|---|---|
| CXR (PA view) | MTX pneumonitis is an idiosyncratic, potentially life-threatening toxicity. A baseline CXR establishes a reference for comparison if pulmonary symptoms develop. Also screens for active pulmonary TB. — Berek & Novak's Gynecology; Fitzpatrick's Dermatology |
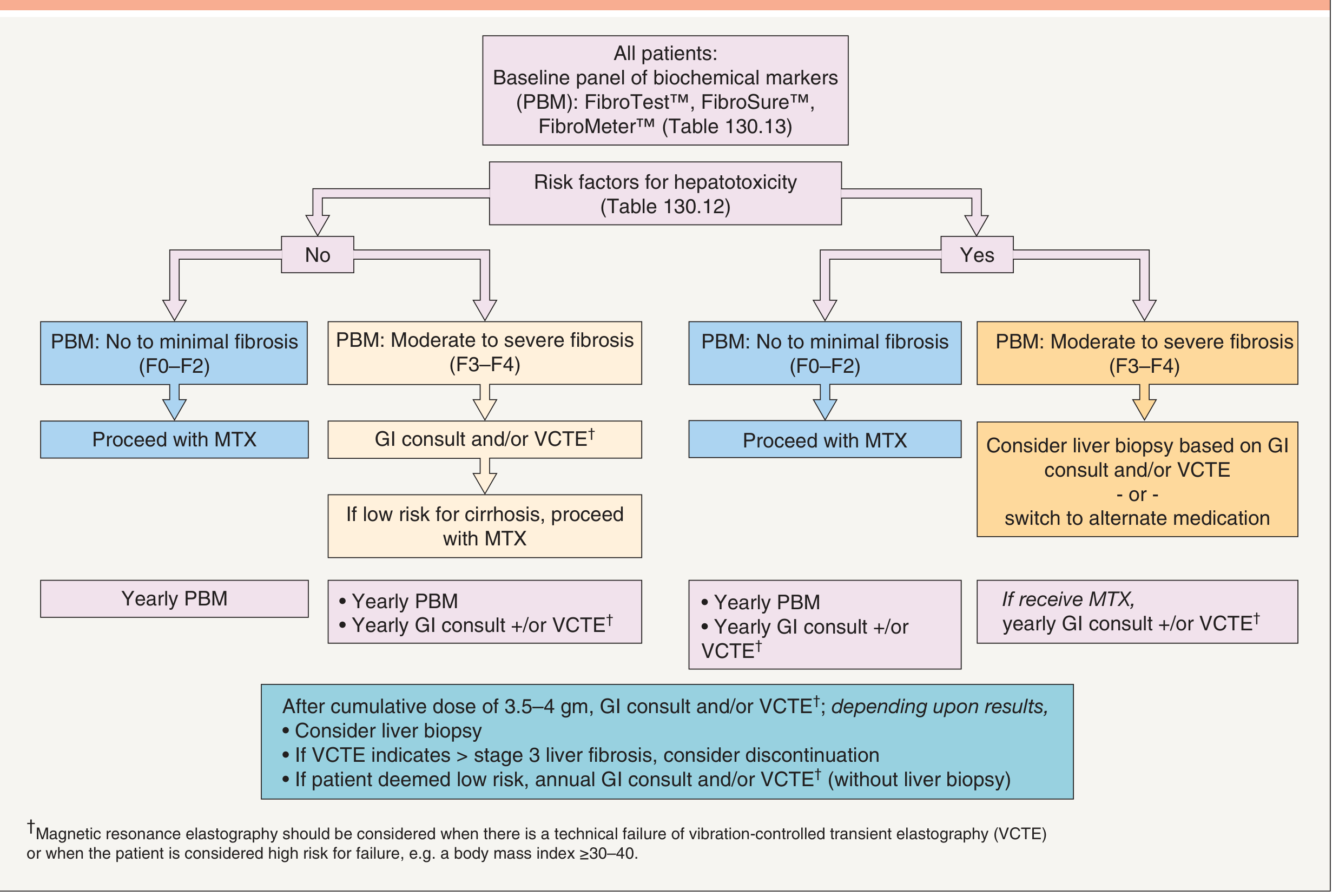
7. Hepatic Fibrosis Assessment (Risk-stratified)
Before starting, assess for hepatic fibrosis risk factors (obesity, diabetes, excessive alcohol, pre-existing liver disease) and perform a panel of biochemical markers (PBM) — FibroTest / FibroSure / FibroMeter:
- If no risk factors + PBM shows F0–F2: Proceed with MTX
- If no risk factors + PBM shows F3–F4: GI consult ± VCTE (vibration-controlled transient elastography) first
- If risk factors present + F3–F4 fibrosis on PBM: Consider liver biopsy or switch to alternative agent
Summary Checklist
| # | Investigation |
|---|---|
| 1 | CBC with differential + platelets |
| 2 | Serum creatinine + BUN (± eGFR) |
| 3 | LFTs — AST, ALT, ALP, albumin |
| 4 | Hepatitis B serology (HBsAg, anti-HBc) |
| 5 | Hepatitis C antibody |
| 6 | HIV (where indicated) |
| 7 | TB screening — PPD or IGRA (QuantiFERON-Gold) |
| 8 | Urine/serum β-hCG (women of childbearing potential) |
| 9 | Chest X-ray |
| 10 | Hepatic fibrosis markers (PBM) if risk factors present |
After baseline, start with a test dose of 2.5–5 mg to assess tolerability, then check CBC and LFTs at 7–14 days before escalating. Always co-prescribe folic acid 1 mg/day to reduce haematologic and GI toxicity. — Fitzpatrick's Dermatology, p. 5505–5506
This is a shared conversation. Sign in to Orris to start your own chat.