Q. Orbital Decompression in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances
orbital decompression surgery indications techniques complications
orbital decompression thyroid eye disease Graves orbitopathy surgical approaches
orbital decompression anatomy walls surgical approach diagram
orbital decompression medial wall lateral wall floor endoscopic transantral approach
proptosis exophthalmos thyroid eye disease orbital anatomy CT scan
ORBITAL DECOMPRESSION — Comprehensive 50-Mark Answer
1. DEFINITION
2. SURGICAL ANATOMY OF THE ORBIT
┌─────────────────────────────────────────────────────────┐
│ BONY ORBIT — OVERVIEW │
│ │
│ ROOF (Superior wall): │
│ • Orbital plate of frontal bone │
│ • Lesser wing of sphenoid (posteriorly) │
│ • Relations: Anterior cranial fossa, frontal sinus │
│ │
│ MEDIAL WALL (Thinnest — 0.2–0.4 mm): │
│ • Lacrimal bone → lamina papyracea (ethmoid) → │
│ lesser wing of sphenoid │
│ • Relations: Ethmoidal air cells, sphenoid sinus │
│ │
│ FLOOR (Second thinnest): │
│ • Maxillary, zygomatic, palatine bones │
│ • Infraorbital canal runs through it │
│ • Relations: Maxillary sinus (roof) │
│ │
│ LATERAL WALL (Strongest): │
│ • Zygomatic bone + greater wing of sphenoid │
│ • Relations: Temporalis fossa, middle cranial fossa │
│ │
│ APEX: │
│ • Optic canal (CN II + ophthalmic artery) │
│ • Superior orbital fissure (CN III, IV, V1, VI, │
│ ophthalmic veins) │
│ • Inferior orbital fissure (V2, infraorbital vessels)│
└─────────────────────────────────────────────────────────┘
Key Surgical Landmarks at the Orbital Apex
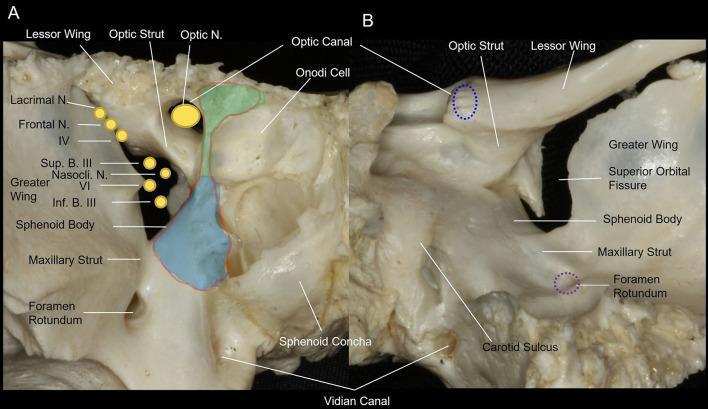
Volume Considerations
| Wall Removed | Volume Gained (approx.) | Proptosis Reduction |
|---|---|---|
| Medial wall alone | 2–3 mL | 2–4 mm |
| Floor alone | 2–3 mL | 2–4 mm |
| Medial + Floor (balanced) | 4–6 mL | 4–6 mm |
| Lateral wall (deep) | 2–4 mL | 2–4 mm |
| Three-wall (medial + floor + lateral) | 6–10 mL | 5–8 mm |
| Four-wall | 8–12 mL | 6–10 mm |
| Fat decompression alone | ~2 mL | 2–4 mm |
3. INDICATIONS
A. Primary Indications (Emergency / Urgent)
- Dysthyroid Optic Neuropathy (DON) — compressive optic neuropathy from enlarged EOM at orbital apex threatening vision
- Exposure keratopathy with corneal breakdown — severe lagophthalmos unresponsive to medical care
- Globe subluxation — proptosis so severe the lids cannot close over the globe
B. Elective Indications
- Disfiguring / cosmetically unacceptable proptosis — Graves' ophthalmopathy (most common indication overall)
- Chronic orbital congestion — venous congestion, chemosis, periorbital edema
- Orbital tumors — selected benign tumors (e.g., cavernous hemangioma, sphenoid wing meningioma) where decompression provides access and volume
- Orbital pseudotumor / idiopathic orbital inflammation — refractory cases
- Traumatic orbital hemorrhage — acute decompression (lateral canthotomy ± bony decompression)
- Post-enucleation socket syndrome — implant-related or volume augmentation
- Fibrous dysplasia — when optic nerve is compressed
C. EUGOGO Severity-Based Indications
| Severity | Indication |
|---|---|
| Mild | Not usually indicated unless refractory |
| Moderate-severe | Elective decompression after inflammation controlled |
| Sight-threatening (DON) | Emergency: IV methylprednisolone → if no response in 2 weeks → urgent decompression |
4. CLINICAL EVALUATION & WORKUP
History
- Duration and rate of proptosis progression
- Visual acuity changes, color vision loss (red desaturation = early DON)
- Diplopia, gaze restriction
- Thyroid history, medications, smoking (doubles risk of TED)
Clinical Assessment
- Hertel exophthalmometry — baseline proptosis (normal <21 mm; asymmetry >2 mm significant)
- Visual acuity + color vision (Ishihara/red cap test)
- Pupillary reflexes — RAPD indicates DON
- Visual fields — Humphrey perimetry
- Slit lamp — corneal exposure, chemosis
- EOM assessment — duction range, diplopia charting
- IOP in upgaze (raised IOP indicates EOM restriction)
Investigations
- CT orbits (axial + coronal) — bony anatomy for surgical planning, muscle enlargement, "Coca-Cola bottle" sign at apex
- MRI orbits — soft tissue characterization, optic nerve sheath enhancement in DON
- Thyroid function tests (TSH, T3, T4, TSI/TRAb)
- Visual evoked potentials — latency delay in DON
- CAS (Clinical Activity Score) — guides timing (surgery preferred when CAS <3, i.e., inactive phase)
Imaging Findings in TED
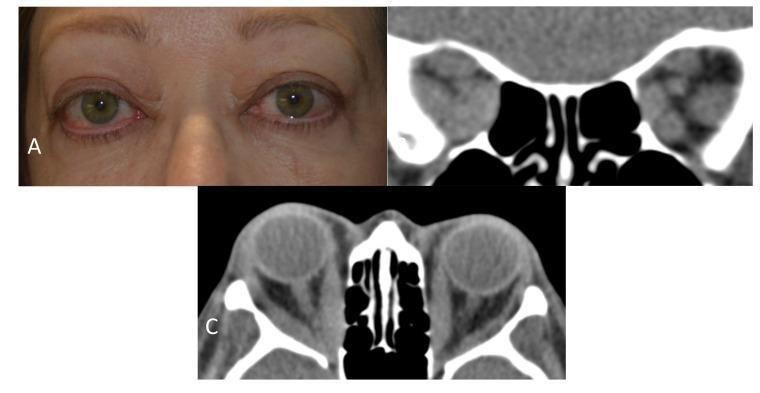
5. CONTRAINDICATIONS
| Absolute | Relative |
|---|---|
| Active orbital cellulitis | Active phase TED (CAS ≥3) — increased risk of worsening diplopia |
| Uncorrected coagulopathy | Single-eyed patient (relative) |
| Uncontrolled thyroid disease | Prior orbital surgery/radiotherapy |
| — | Severe sinusitis (for endonasal approach) |
6. SURGICAL APPROACHES — CLASSIFICATION
ORBITAL DECOMPRESSION APPROACHES
│
├─── A. BONY WALL DECOMPRESSION
│ │
│ ├── 1. MEDIAL WALL
│ │ ├── Endoscopic endonasal (transethmoidal)
│ │ ├── Transconjunctival (Lynch incision)
│ │ └── Transcaruncular
│ │
│ ├── 2. FLOOR (INFERIOR WALL)
│ │ ├── Transantral (Caldwell-Luc) — Ogura technique
│ │ ├── Transconjunctival (infraciliary / fornix)
│ │ └── Endoscopic endonasal (via maxillary antrostomy)
│ │
│ ├── 3. LATERAL WALL
│ │ ├── Lateral orbitotomy (Kronlein / Stallard-Wright)
│ │ ├── Swinging eyelid / transconjunctival lateral
│ │ └── Endoscopic (keyhole) lateral
│ │
│ └── 4. COMBINED / THREE-WALL / FOUR-WALL
│
└─── B. FAT DECOMPRESSION
├── Transconjunctival (medial + lateral fat)
└── Combined with bony decompression
7. DETAILED SURGICAL TECHNIQUES
7.1 Endoscopic Endonasal Medial Wall Decompression (Most Common — Gold Standard)
- General anesthesia (or LA with sedation); head 15–20° elevation; topical decongestant (oxymetazoline)
- Total ethmoidectomy (anterior + posterior) — creates the medial sinus cavity
- Identify the lamina papyracea — medial orbital wall
- Remove lamina papyracea from posterior lacrimal crest anteriorly to the posterior ethmoid/sphenoid junction posteriorly; superiorly up to the frontoethmoidal line (fovea ethmoidalis), inferiorly to the orbital floor
- Incise periorbita in a posterior-to-anterior direction, in a 'T' or grid pattern — allows orbital fat to prolapse
- Extent of decompression can be extended to include the sphenoid face and medial optic canal strut (for DON)
- Hemostasis with bipolar cautery; no packing usually required
- Basal lamella — separates anterior from posterior ethmoids
- Skull base (fovea ethmoidalis) — superior limit
- Maxillary line — inferior limit (where floor begins)
- Optic canal bulge — posterior medial wall landmark
7.2 Transantral (Caldwell-Luc / Ogura) Approach — Floor Decompression
- Sublabial incision above upper premolars
- Antrostomy through anterior maxillary wall
- Enter maxillary sinus; identify orbital floor (roof of antrum)
- Resect orbital floor (periosteum stripped, bone removed), preserving infraorbital nerve
- Incise periorbita to allow fat herniation into antrum
7.3 Transconjunctival Approach (Floor + Medial Wall)
- Conjunctival incision in lower fornix (retroseptal or preseptal route)
- Dissect to orbital floor
- Subperiosteal dissection exposing floor and medial wall
- Remove floor bone (medial to infraorbital nerve) and lamina papyracea
- Incise periorbita; allow prolapse
- Conjunctival closure with absorbable sutures
7.4 Lateral Wall Decompression (Kronlein / Stallard-Wright Approach)
- Lateral canthotomy + cantholysis (inferior crus of lateral canthal tendon)
- "Swinging eyelid" flap — lateral lower lid reflected inferiorly
- Lateral periosteal incision; periosteum elevated
- Remove outer cortex of lateral orbital wall with drill/chisel
- Remove greater wing of sphenoid (posterior to sphenozygomatic suture) using high-speed drill — this provides the greatest volume relief
- Periorbita optionally incised
- Reattach lateral canthal tendon; close in layers
7.5 Balanced (Medial + Floor) Decompression (Standard for Most TED Cases)
7.6 Fat Decompression
7.7 Four-Wall (Maximum) Decompression
8. ALGORITHM: MANAGEMENT OF THYROID EYE DISEASE REQUIRING DECOMPRESSION
THYROID EYE DISEASE (TED)
│
▼
Assess Activity (CAS) & Severity
│
┌─────┴───────────────────────┐
▼ ▼
Mild TED Moderate–Severe TED
(CAS <3, cosmesis) │
│ ┌─────┴──────────┐
▼ ▼ ▼
Conservative Active Phase Inactive Phase
(lubricants, (CAS ≥3) (CAS <3)
selenium, │ │
sunglasses) IV methylpred- Surgical
nisolone decompression
± orbital RT │
│ ┌────┴──────┐
▼ ▼ ▼
No response Cosmetic DON/Corneal
in 2 weeks Proptosis Emergency
│ │ │
▼ ▼ ▼
URGENT Balanced 2–3 wall
DECOMPRESSION Medial+ + optic canal
(vision- Floor decompression
threatening) approach URGENT
│
▼
POST-DECOMPRESSION REHABILITATION
(Allow 6 months stabilization)
│
▼
Strabismus Surgery (if diplopia persists)
│
▼
Eyelid Surgery (ptosis repair, lid retraction)
│
▼
Blepharoplasty / Aesthetic Refinement
9. SPECIFIC APPROACH SELECTION FLOWCHART
INDICATION FOR DECOMPRESSION
│
┌────┴─────────────────────────────────┐
▼ ▼
Vision-threatening DON Elective Proptosis
│ │
Urgent 2–3 wall + ┌──────────┴──────────┐
optic canal ▼ ▼
decompression Diplopia already No pre-existing
(endoscopic ± present pre-op diplopia
lateral) │ │
▼ ▼
Avoid medial Balanced
wall alone; decompression
consider fat (medial + floor)
or lateral or fat-only if
approach fat-predominant
▼
Very large proptosis?
(>28 mm, globe sublux)
│
▼
3–4 wall maximum
decompression
10. OPTIC CANAL DECOMPRESSION
- Complete total ethmoidectomy + sphenoidotomy
- Identify optic canal bulge on the lateral sphenoid wall
- Using diamond drill, thin and remove the medial optic canal wall (the "optic strut")
- Do NOT incise the optic nerve sheath routinely (optic nerve sheath fenestration is a separate procedure)
- The carotid artery lies medial — extreme caution required
11. COMPLICATIONS
Intraoperative
| Complication | Cause | Prevention |
|---|---|---|
| CSF leak | Skull base breach (fovea ethmoidalis) | Identify skull base early, stay below |
| Orbital hemorrhage | Periorbital vessel injury | Careful periorbita incision, bipolar ready |
| Optic nerve injury | Posterior over-dissection, optic strut drill | Intraoperative neuromonitoring |
| Infraorbital nerve injury | Floor decompression | Preserve nerve periosteum |
| Carotid artery injury | Sphenoid/optic canal work | Preop CT, diamond drill |
| Nasolacrimal duct injury | Anterior medial wall dissection | Identify posterior lacrimal crest |
Early Postoperative
| Complication | Incidence | Management |
|---|---|---|
| Periorbital ecchymosis/edema | Very common | Ice packs, head elevation |
| Diplopia (new-onset) | 15–30% (medial approach) | Most resolve; prism glasses; later strabismus surgery |
| Orbital emphysema | Rare | Avoid nose blowing; resolves spontaneously |
| Sinusitis/infection | 2–5% | Nasal saline, antibiotics if needed |
| Lower lid ectropion | Transconjunctival/Caldwell-Luc | Lid support, massage; revision surgery |
Late Complications
| Complication | Notes |
|---|---|
| Persistent diplopia | 20–30% after medial wall decompression; most managed with strabismus surgery |
| Globe hypotony | Uncommon |
| Enophthalmos | Over-decompression (rare) |
| Intracranial hemorrhage | Rare but life-threatening; described by Badilla & Dolman, Orbit 2008 — cited in Management of Thyroid Eye Disease, p. 32 |
| Worsening proptosis | If performed in active phase TED |
| Nasal adhesions/synechiae | Common post-endoscopic; managed with saline irrigation |
- Wait 6+ months for stabilization
- Prism correction during recovery
- Botulinum toxin injection to over-acting muscle
- Strabismus surgery (recession/resection) when stable
12. OUTCOMES
Proptosis Reduction
- Medial wall alone: 2–4 mm
- Medial + Floor: 4–6 mm
- 3-wall: 5–8 mm
- Fat decompression: 2–4 mm
Visual Recovery in DON
- 80–90% of patients show visual improvement after surgical decompression for DON
- Best results when performed early (before irreversible axonal loss)
- IV methylprednisolone non-responders → surgery salvages vision in ~85% (EUGOGO)
13. COMPARISON OF APPROACHES
| Parameter | Endoscopic Medial | Transconjunctival Floor | Lateral Wall | Fat Decompression |
|---|---|---|---|---|
| Access | Endonasal | Conjunctival | External/lateral | Transconjunctival |
| External scar | None | None | Minimal (canthal) | None |
| Proptosis reduction | 2–4 mm | 2–4 mm | 2–4 mm | 2–4 mm |
| Combined (med+floor) | 4–6 mm | — | — | — |
| Diplopia risk | 15–30% | 10–20% | Low (<10%) | Low (<5%) |
| Best indication | TED, endonasal | Combined approach | Fat-predominant TED | Mild–moderate; cosmetic |
| DON suitability | Excellent (+ optic canal) | Poor | Moderate | Poor |
| Learning curve | High | Moderate | Moderate | Low |
14. SPECIFIC TEXTBOOK CONTRIBUTIONS
Scott Brown's Otorhinolaryngology (8th ed.)
- Emphasizes the endoscopic approach as the approach of choice for medial wall decompression
- Describes the "balanced decompression" concept — combining medial + floor to reduce diplopia
- Covers skull base anatomy in detail for surgical safety
- Notes the role of pre-operative CT planning and intraoperative navigation systems
Cummings Otolaryngology (7th ed.)
- Provides stepwise endoscopic decompression technique
- Discusses the "fat-sparing" vs "fat-decompression" debate
- Emphasizes the CAS scoring system (Mourits criteria) for timing surgery
- Covers the role of orbital radiotherapy as adjunct to decompression
Stell & Maran's Head and Neck Surgery
- Classic description of transantral (Ogura) approach
- Emphasizes anatomical basis of orbital surgery
- Discusses historical context of four-wall decompression (Tessier approach)
Dhingra's Diseases of ENT (8th ed.)
- Concise overview suitable for undergraduates/postgraduates
- Classifies indications into urgent vs elective
- Describes the Caldwell-Luc/transantral approach in accessible language
- Notes proptosis grading using Hertel exophthalmometer
Hazarika's ENT
- Covers orbital decompression in the context of orbital complications of sinusitis
- Emphasizes the role of the ENT surgeon in managing orbital apex syndrome
- Describes endoscopic medial wall decompression technique
Zakir Hussain's ENT
- Focuses on clinical features leading to decompression (Chandler classification of orbital complications)
- Indications table clearly presented
- Management algorithm for periorbital cellulitis vs orbital involvement
15. RECENT ADVANCES
1. Endoscopic Navigation-Assisted Decompression
- Electromagnetic / optical navigation (BrainLab, StealthStation) allows real-time 3D localization during endoscopic orbital surgery
- Reduces risk of skull base breach and carotid injury during optic canal decompression
- Particularly useful for revision cases with altered anatomy
2. Teprotumumab (IGF-1R Inhibitor) — Medical Alternative to Surgery
- FDA-approved (2020) for active moderate–severe TED
- Monoclonal antibody against IGF-1R; reduces proptosis by mean 2.82 mm (vs 0.54 mm placebo) in pivotal OPTIC trial
- For DON: early efficacy data from Sears et al. (Am J Ophthalmol Case Rep 2021) — reduces need for emergency decompression
- Represents a paradigm shift — medical treatment can achieve results previously requiring surgery
- Does not replace surgery for severe/vision-threatening cases
3. Tocilizumab (IL-6 Receptor Inhibitor)
- Used as first-line treatment for DON secondary to Graves' orbitopathy by Pascual-Camps et al. (Orbit 2018 — cited in Management of Thyroid Eye Disease, p. 32)
- May reduce the need for surgical decompression in select patients
4. Fat Decompression Renaissance
- Increasing popularity for fat-predominant TED (younger patients, lower diplopia risk)
- Combined endoscopic fat decompression + bony wall decompression under same anesthetic
- Kazim et al. (Br J Ophthalmol 2000) demonstrated reversal of DON following fat-only decompression in selected cases
5. "Balanced" Decompression Refinements
- Tying Garip Kuebler et al. (Eye, 2020): Evaluation of medical vs surgical decompression in DON — confirmed surgery superior for established DON
- Combination of medial + floor ("balanced") with selective periorbita preservation reduces new-onset diplopia from ~30% to ~15%
6. Robotic / Exoscope-Assisted Surgery
- Emerging use of exoscopes (VITOM, ORBEYE) providing 3D magnification without laparoscope limitations
- Robotic-assisted endoscopic orbital surgery: early case series showing feasibility
7. Customized 3D CT Planning
- Patient-specific volumetric analysis guides wall selection and expected proptosis reduction
- 3D printing of orbital models for preoperative planning in complex cases
8. Lateral Wall "Deep" Decompression
- Goldberg technique: greater wing of sphenoid removal behind the equator of the globe
- Provides 3–4 mm additional reduction with low diplopia risk
- Gaining popularity as a first-line approach in patients with significant pre-existing diplopia
9. Combined Immunosuppression + Decompression Protocol
- EUGOGO 2021 recommendations: IV methylprednisolone pulse therapy (500 mg × 6 weeks + 250 mg × 6 weeks) → reassess → if DON persists or recurs → surgical decompression
- Orbital radiotherapy (20 Gy in 10 fractions) combined with steroids for active DON (Gold et al., Ophthalmic Plast Reconstr Surg 2018)
16. ORBITAL DECOMPRESSION FOR NON-TED CONDITIONS
Orbital Subperiosteal Abscess (Chandler Grade IV)
- Medial subperiosteal abscess — endoscopic drainage through the medial wall approach (essentially decompression of the orbit)
- Combined with ethmoidectomy for the primary sinus source
- Indication for immediate decompression when visual acuity drops or RAPD appears
Orbital Apex Syndrome
- Combined optic nerve and cranial nerve compression at apex
- Decompression via endoscopic optic canal decompression ± lateral orbitotomy
Traumatic Retrobulbar Hematoma
- Emergency decompression via lateral canthotomy + inferior cantholysis (first step)
- If IOP remains >40 mmHg with visual compromise → bony decompression (lateral wall)
Fibrous Dysplasia
- Optic canal compression → endoscopic optic canal decompression with diamond drill
17. POSTOPERATIVE CARE
- Head elevation (30–45°) — reduces periorbital edema
- Topical nasal decongestants — for endoscopic cases
- Nasal saline irrigation — begins 24 hours post-op
- Systemic corticosteroids (short course) — reduce orbital edema
- Eye lubricants / taping — corneal protection immediately post-op
- Avoid nose blowing — prevents emphysema (2–3 weeks)
- Endoscopic debridement at 1 week — remove crusting, synechiae
- Ophthalmic follow-up — visual acuity, color vision, IOP monitoring
- Rehabilitation sequence: Wait 6 months → strabismus surgery → eyelid surgery
18. QUICK REFERENCE SUMMARY TABLE
| Feature | Detail |
|---|---|
| Most common indication | Thyroid Eye Disease (Graves' orbitopathy) |
| Most urgent indication | Dysthyroid Optic Neuropathy (DON) |
| Gold standard approach | Endoscopic endonasal medial wall decompression |
| Most proptosis reduction | Three-wall or four-wall decompression |
| Lowest diplopia risk | Fat decompression or lateral wall alone |
| Key tool for planning | CT orbits (axial + coronal) |
| Timing in TED | Inactive phase (CAS <3) for elective; any time for DON |
| Key complication to avoid | CSF leak (skull base), carotid injury (optic canal work) |
| Latest medical alternative | Teprotumumab (FDA approved 2020) |
| Rehabilitation sequence | Decompression → Strabismus → Lid surgery |
REFERENCES
- Scott Brown's Otorhinolaryngology Head & Neck Surgery (8th ed.) — Vol. 3: Rhinology and Anterior Skull Base
- Cummings Otolaryngology — Head and Neck Surgery (7th ed.) — Chapter on Orbital Surgery
- Stell & Maran's Head and Neck Surgery (5th ed.) — Orbital Surgery chapter
- Dhingra PL. Diseases of Ear, Nose and Throat (8th ed.) — Chapter 33: Complications of Sinusitis
- Hazarika P. Textbook of ENT and Head & Neck Surgery — Orbital Complications chapter
- Zakir Hussain. Clinical ENT — Orbital Complications, Chandler Classification
- Management of Thyroid Eye Disease (EUGOGO Guidelines) — p. 24, 32
- Ogura JH, Walsh TE. The Transantral Orbital Decompression Operation. Laryngoscope 1962.
- Liao SL et al. Transcanicular Orbital Decompression. Am J Ophthalmol 2006;141(5):810–818.
- Choe CH et al. Comparison of lateral and medial orbital decompression. Ophthalmic Plast Reconstr Surg 2011;27(1):4–11.
- Kazim M et al. Reversal of dysthyroid optic neuropathy following orbital fat decompression. Br J Ophthalmol 2000;84(6):600–605.
- Gold KG et al. Orbital radiotherapy combined with corticosteroid treatment for TED-compressive optic neuropathy. Ophthalmic Plast Reconstr Surg 2018.
- Sears CM et al. Early efficacy of teprotumumab for dysthyroid optic neuropathy. Am J Ophthalmol Case Rep 2021.
- Pascual-Camps I et al. Tocilizumab as first treatment option in optic neuropathy secondary to Graves' orbitopathy. Orbit 2018.
- Garip Kuebler A et al. Evaluation of medical and surgical decompression in patients with dysthyroid optic neuropathy. Eye (Lond) 2020;34(9):1702–1709.
- Badilla J, Dolman PJ. Intracranial hemorrhage complicating orbital decompression. Orbit 2008.
- EUGOGO (European Group on Graves' Orbitopathy) Guidelines 2021.
Q. Allergic Fungal Sinusitis in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances
allergic fungal sinusitis diagnosis criteria pathophysiology
allergic fungal rhinosinusitis surgical treatment FESS recurrence management
allergic fungal sinusitis CT scan MRI findings hyperdense mucin
Bent Kuhn criteria allergic fungal sinusitis fungi Aspergillus Bipolaris eosinophilic mucin
allergic fungal sinusitis nasal polyps histopathology Charcot Leyden crystals eosinophils
ALLERGIC FUNGAL SINUSITIS (AFRS) — Comprehensive 50-Mark Answer
1. DEFINITION
- Type I (IgE-mediated) and Type III (immune complex–mediated) hypersensitivity to inhaled fungal antigens
- Accumulation of characteristic eosinophilic allergic mucin containing non-invasive fungal hyphae within the paranasal sinuses
- Associated nasal polyposis, atopy, and a chronic relapsing course
"The affected patients present with chronic (perennial) sinusitis relatively unresponsive to antibiotics. Many have nasal polyps, and all have congested nasal mucosae and sinuses full of mucoid material. The histologic hallmarks are local eosinophilia and Charcot-Leyden crystals." (Harrison's Principles of Internal Medicine, 21st ed., p. 6271)
2. HISTORICAL BACKGROUND
| Year | Milestone |
|---|---|
| 1976 | Safirstein — first described the condition as "aspergillosis simulating allergic bronchopulmonary aspergillosis in the sinuses" |
| 1981 | Millar, Johnston & Lamb — described eosinophilic mucin in sinuses |
| 1983 | Katzenstein et al. — coined the term "Allergic Aspergillus Sinusitis" |
| 1989 | Robson et al. — expanded to non-Aspergillus fungi (dematiaceous) |
| 1994 | Bent & Kuhn — established the classic 5-criterion diagnostic criteria |
| 1996 | deShazo — proposed alternative diagnostic criteria |
| 2001 | Waxman et al. — raised the concept of eosinophilic mucin rhinosinusitis (EMRS) |
3. EPIDEMIOLOGY
- Represents 5–10% of all chronic rhinosinusitis cases requiring surgery
- Predominantly affects young immunocompetent atopic individuals (mean age 20–30 years)
- Geographic distribution: Subtropical and tropical regions with high humidity and fungal spore burden — common in South and Southeast Asia, Southern USA (Mississippi/Tennessee River valleys), Middle East
- High prevalence in India, Pakistan — Bipolaris species most common rather than Aspergillus
- Gender: Slight male predominance in some series
- Socioeconomic: Develops more in areas with higher ambient fungal spore counts
- Strong association with asthma (50–60%), allergic rhinitis (>90%), and nasal polyposis
4. ETIOLOGY — CAUSATIVE FUNGI
A. Dematiaceous (Melanized) Fungi — Most Common Overall
| Genus | Species | Geographic Predominance |
|---|---|---|
| Bipolaris | B. spicifera, B. hawaiiensis | India, Pakistan, Southeast Asia, Southern USA |
| Curvularia | C. lunata | Tropical regions |
| Alternaria | A. alternata | Worldwide |
| Exserohilum | E. rostratum | Southern USA |
| Helminthosporium | Various | — |
B. Hyaline Fungi
| Genus | Species | Notes |
|---|---|---|
| Aspergillus | A. fumigatus, A. flavus | Western countries, historically first described |
| Fusarium | Various | Less common |
Key point: In India and the Indian subcontinent, Bipolaris and Curvularia species account for the majority of AFRS cases, not Aspergillus (Hazarika; Zakir Hussain)
Mnemonic: "B-C-A-E" (Bipolaris, Curvularia, Alternaria, Exserohilum)
5. PATHOPHYSIOLOGY
5.1 Immunological Basis
INHALED FUNGAL SPORES
│
▼
ATOPIC HOST (defective mucociliary clearance,
altered innate immunity, IgE predisposition)
│
├─── TYPE I HYPERSENSITIVITY (IgE-mediated)
│ • Mast cell degranulation
│ • Histamine, leukotrienes → mucosal edema, mucus hypersecretion
│ • Elevated total serum IgE + specific anti-fungal IgE
│
└─── TYPE III HYPERSENSITIVITY (Immune complex–mediated)
• Elevated fungal-specific IgG
• Complement activation → tissue damage
• Eosinophil recruitment
5.2 The Vicious Cycle of AFRS
Atopic individual
│
▼
Fungal antigen inhalation
│
▼
IgE-mediated mucosal inflammation
│
▼
Mucosal edema → ostial obstruction
│
▼
Sinus stasis → fungal proliferation
│
▼
Eosinophil recruitment → MBP release
│
▼
Further mucosal damage + allergic mucin accumulation
│
▼
Polyp formation + sinus expansion
│
▼
Bone remodeling / erosion (pressure necrosis — NOT invasion)
│
└─── feeds back to worsening obstruction ────►
5.3 Role of Eosinophils and Allergic Mucin
- Major Basic Protein (MBP) from eosinophil granules is directly toxic to respiratory epithelium
- Charcot-Leyden crystals — derived from eosinophil lysophospholipase; pathognomonic of eosinophilic mucin
- Allergic mucin = thick, tenacious, peanut butter–like material composed of:
- Eosinophils (intact and degranulated)
- Charcot-Leyden crystals
- Non-invasive fungal hyphae (sparse)
- Fibrin matrix, mucus glycoproteins
- Concentrated heavy metals (iron, manganese) → accounts for CT hyperdensity and MRI signal void
6. CLASSIFICATION OF FUNGAL RHINOSINUSITIS
FUNGAL RHINOSINUSITIS
│
├─── NON-INVASIVE (intact basement membrane — no tissue invasion)
│ │
│ ├── 1. SAPROPHYTIC (Fungal colonization) — surface growth only
│ │
│ ├── 2. FUNGAL BALL (Mycetoma) — dense hyphal mat, usually maxillary,
│ │ NO eosinophilic mucin, NO allergy, middle-aged/elderly
│ │
│ └── 3. ALLERGIC FUNGAL RHINOSINUSITIS (AFRS)
│ — immunological, eosinophilic mucin, atopic host ← [TODAY'S TOPIC]
│
└─── INVASIVE (tissue invasion present)
│
├── 4. ACUTE INVASIVE (Fulminant) — immunocompromised, <4 weeks,
│ vascular invasion, necrosis — LIFE-THREATENING
│
├── 5. CHRONIC INVASIVE — immunocompetent/mild immunocompromise,
│ >12 weeks, slow progression, indolent
│
└── 6. GRANULOMATOUS INVASIVE — Sudan, India, Pakistan; Aspergillus
flavus; granuloma formation; limited geographic distribution
7. DIAGNOSTIC CRITERIA
7.1 Bent & Kuhn Criteria (1994) — GOLD STANDARD
| # | Criterion | Detail |
|---|---|---|
| 1 | Type I hypersensitivity | Positive skin-prick test OR elevated serum specific IgE to fungal antigens |
| 2 | Nasal polyposis | Endoscopic or radiological evidence |
| 3 | Characteristic CT findings | Heterogeneous hyperdense areas within opacified sinuses |
| 4 | Eosinophilic mucin | WITHOUT tissue invasion (histopathological confirmation) |
| 5 | Positive fungal smear/culture | From surgically removed mucin |
7.2 Supporting (Minor) Criteria
- Unilateral disease (initially)
- Bony erosion on imaging
- Elevated total serum IgE (>1000 IU/mL)
- Elevated specific IgG (precipitins) to offending fungus
- Charcot-Leyden crystals on histology
- Positive fungal culture
7.3 deShazo Criteria (1996) — Alternative
- Eosinophilic mucin without fungal invasion into sinus tissue
- Positive fungal stain of sinus contents
- Nasal polyposis
- Characteristic CT/MRI findings
8. CLINICAL FEATURES
8.1 Symptoms
- Nasal obstruction — unilateral > bilateral (early); bilateral (late) — most common
- Anosmia / hyposmia — loss of smell from polyposis
- Chronic nasal discharge — thick, tenacious, dark (brown/green/black) — "peanut butter–like"
- Facial pain/pressure — dull, episodic
- Headache — from sinus pressure
- Proptosis / periorbital swelling — orbital extension (medial orbital wall involvement)
- Visual changes — diplopia, reduced acuity (orbital apex involvement in advanced cases)
- Facial deformity / swelling — late sign; bony expansion causing mid-face widening
- Epistaxis — from polyp surface or erosion
8.2 Signs on Examination
- Nasal polyps — multiple, bilateral (may be unilateral early)
- Allergic mucin — thick, rubbery, dark brown/green mucin visible in middle meatus
- Mucosal hypertrophy — pale, boggy
- Septal deviation — pressure effect from expanding polyposis
- "Cheesy" material — characteristic mucin extruding from sinuses
- Signs of atopy: allergic shiners, Dennie-Morgan lines, nasal crease
- Wheezing (associated asthma)
- Mid-facial expansion — widening of nasal bridge (suggests ethmoid expansion)
- Proptosis — if orbital wall eroded
8.3 Associated Conditions
- Allergic rhinitis — virtually 100%
- Asthma — 50–60%
- Aspirin sensitivity — AERD (Aspirin-Exacerbated Respiratory Disease) / Samter's triad in some
- Atopic dermatitis — less common
9. INVESTIGATIONS
9.1 Laboratory Tests
| Investigation | Finding in AFRS | Significance |
|---|---|---|
| Total serum IgE | Markedly elevated (often >1000 IU/mL; up to >10,000) | Confirms atopy; monitors disease activity/recurrence |
| Specific IgE (RAST/ImmunoCAP) | Elevated to specific fungus | Confirms type I hypersensitivity; identifies causative organism |
| Skin-prick test | Positive to fungal antigens | Bent & Kuhn criterion #1 |
| Specific IgG (precipitins) | Elevated | Type III hypersensitivity; supports diagnosis |
| Absolute eosinophil count | Elevated (peripheral eosinophilia) | Reflects systemic atopy |
| Fungal culture | Positive (Bipolaris, Aspergillus, etc.) | Identifies causative organism |
| KOH mount | Fungal hyphae in mucin | Rapid bedside confirmation |
| Fungal stain (GMS/PAS) | Non-invasive hyphae in mucin | Histopathological confirmation |
| CBC | Eosinophilia | — |
| Total protein / albumin | Usually normal | Excludes immunodeficiency |
9.2 Radiology
CT Paranasal Sinuses (GOLD STANDARD for imaging)
- Views: Axial + coronal ± sagittal reconstruction
- Classic findings:
- Complete sinus opacification — one or more sinuses
- Heterogeneous hyperdensity — central hyperdense areas (the "double density" sign) representing iron/manganese in allergic mucin
- Bone thinning and remodeling (pressure erosion, NOT invasion)
- Expansion of sinuses — sinuses appear enlarged due to chronic pressure
- Orbital wall displacement — medial orbital wall pushed laterally (proptosis)
- Skull base thinning — in severe cases
- Unilateral predominance — early disease is often unilateral
MRI Paranasal Sinuses
- T1-weighted: Variable signal (iso to hypointense centrally)
- T2-weighted: Characteristic "signal void" or very low signal centrally — PATHOGNOMONIC
- Due to concentrated iron, manganese, and high protein in allergic mucin
- T1 + Gadolinium: Peripheral mucosal enhancement; central non-enhancement
- Differentiates AFRS from:
- Mucocoele (T2 hyperintense)
- Fungal ball (T2 moderate signal)
- Invasive fungal disease (enhancing central component)
Imaging of AFRS
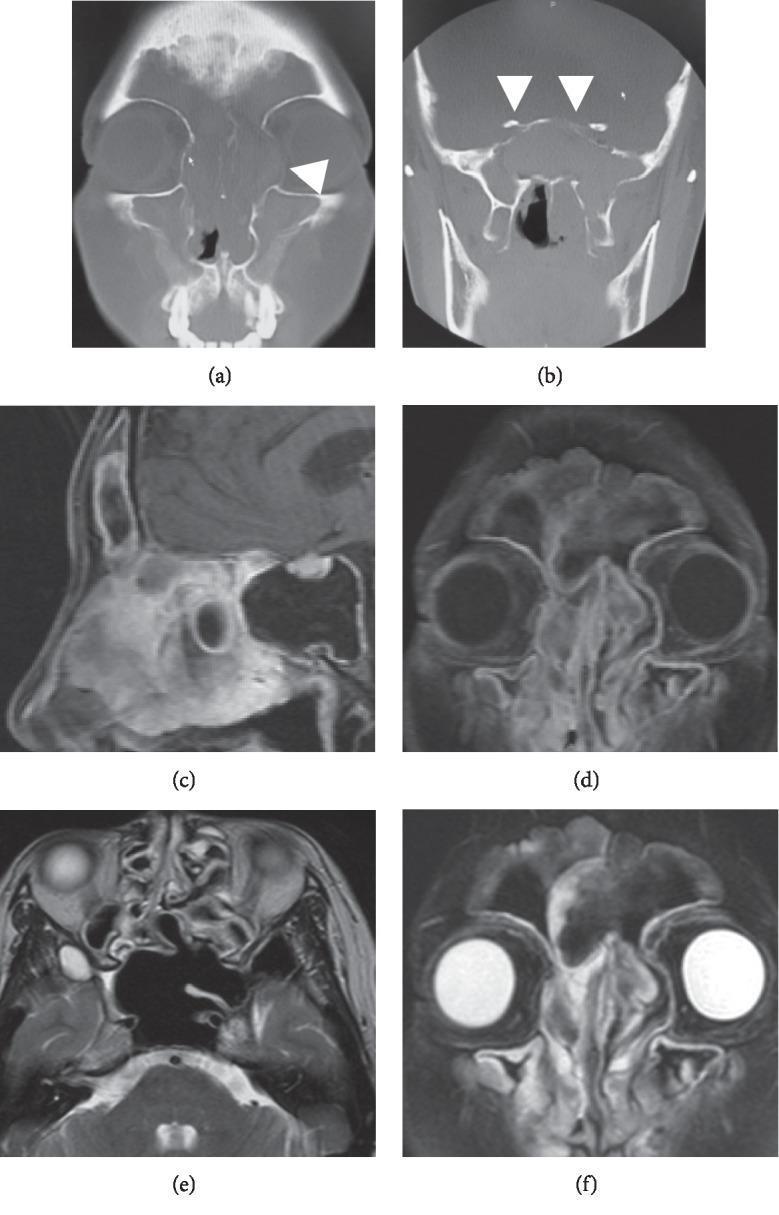
10. HISTOPATHOLOGY
Gross Pathology
- Allergic mucin — thick, tenacious, peanut butter–like; dark brown, green, or black
- Nasal polyps — pale, gelatinous, multiple
- NO evidence of tissue necrosis or vascular invasion (distinguishes from invasive disease)
Microscopy (H&E Stain)
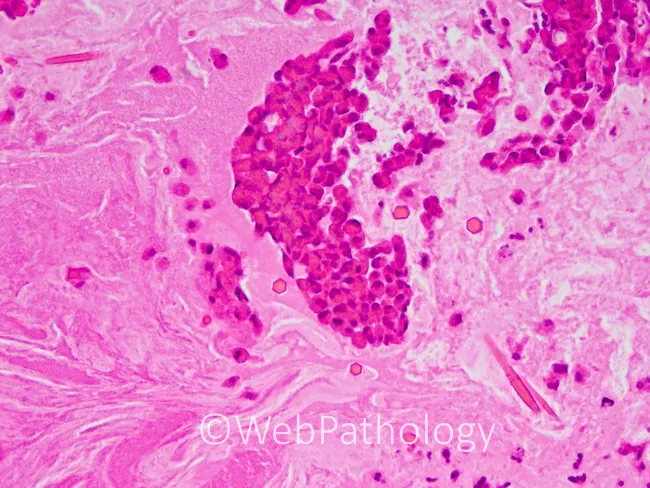
Histological Hallmarks
- Eosinophilic mucin — amorphous, laminated matrix
- Charcot-Leyden crystals — hexagonal/diamond-shaped eosinophilic structures from eosinophil lysophospholipase (Harrison's 21st ed., p. 6271)
- Intact basement membrane — NO tissue invasion (mandatory for diagnosis)
- Sparse non-invasive fungal hyphae — seen with GMS (Grocott-Methenamine Silver) or PAS stain
- Inflammatory cells: Eosinophils >> neutrophils, plasma cells, histiocytes
Special Stains
| Stain | Purpose | Appearance |
|---|---|---|
| GMS (Grocott Methenamine Silver) | Fungal hyphae | Black hyphae on green background |
| PAS (Periodic Acid-Schiff) | Fungal hyphae | Magenta/red hyphae |
| H&E | Overall architecture | Eosinophilic mucin, Charcot-Leyden crystals |
| Mucicarmine | Mucin confirmation | Red mucin |
11. DIFFERENTIAL DIAGNOSIS
| Condition | Key Distinguishing Features |
|---|---|
| Chronic rhinosinusitis with polyps (non-fungal) | No eosinophilic mucin, no fungal elements, IgE normal |
| Fungal ball (Mycetoma) | No allergy, no polyps, no eosinophilic mucin; dense hyphal ball; middle-aged/elderly |
| Acute invasive fungal sinusitis | Immunocompromised; tissue necrosis, vascular invasion; rapidly progressive |
| Chronic invasive fungal sinusitis | Tissue invasion on histology; immunocompromised/diabetic |
| Granulomatous invasive sinusitis | Granuloma formation; Sudan/India subcontinent; Aspergillus flavus |
| Antrochoanal polyp | Single large polyp; no eosinophilic mucin; originates from maxillary sinus |
| Inverting papilloma | Unilateral; CT shows bony sclerosis; no allergy |
| Mucocoele | T2 hyperintense; no eosinophilic mucin; no fungal elements |
| Sinonasal malignancy | Bony destruction (aggressive, irregular); enhances on MRI |
| Wegener's granulomatosis | c-ANCA positive; septal perforation; granulomatous on biopsy |
12. MANAGEMENT
12.1 Overview: Multimodal Approach
DIAGNOSIS CONFIRMED (Bent & Kuhn criteria)
│
▼
PREOPERATIVE PREPARATION
• Control atopy (antihistamines, nasal steroids)
• Treat acute exacerbation (short oral steroid course)
• CT/MRI for surgical planning
• Allergy testing → identify specific fungal antigen
│
▼
SURGICAL TREATMENT (PRIMARY)
Functional Endoscopic Sinus Surgery (FESS)
+ Complete evacuation of allergic mucin
+ Polypectomy
+ Wide sinus opening for access/drainage
│
▼
IMMEDIATE POSTOPERATIVE MEDICAL THERAPY
• Topical intranasal corticosteroids (MAINSTAY)
• Saline irrigation
• Systemic steroids (short taper)
• Consider antifungal (refractory/relapsing)
• Allergen immunotherapy
│
▼
LONG-TERM SURVEILLANCE
• Endoscopic follow-up (3, 6, 12 months then annually)
• Monitor serum IgE levels
• Early management of recurrence
12.2 SURGICAL TREATMENT — FESS
"We recommend polypectomy and sinus washout as the optimal means of symptom control and inducing remission; however, relapse is frequent." (Diagnosis and Management of Aspergillosis, p. 9 — Strong recommendation, moderate-quality evidence)
Surgical Steps:
- General or monitored local anesthesia; head elevated 15–20°
- Nasal endoscopy — assess polyp extent and mucin distribution
- Uncinectomy → Middle meatal antrostomy (maxillary sinus)
- Total ethmoidectomy (anterior + posterior) — most commonly involved
- Frontal sinusotomy (Draf I/II/IIB/III depending on extent)
- Sphenoidotomy (if sphenoid involved)
- Complete evacuation of allergic mucin — the cardinal step; all mucin must be removed
- Polypectomy — microdebrider-assisted for polyps
- Wide sinus openings (Draf III/Lothrop for frontal sinus in bilateral severe disease)
- Intraoperative cultures and histopathology from all specimens
Special Surgical Considerations:
- Bony erosion — does NOT necessarily mean invasion; pressure erosion is common; repair/reconstruction of skull base/orbital walls may be needed intraoperatively
- Orbital decompression — if orbital wall eroded and proptosis present
- Neurosurgical involvement — if intracranial extension (rare) suspected on imaging
Ethmoid > Maxillary > Sphenoid > Frontal
12.3 POSTOPERATIVE MEDICAL MANAGEMENT
A. Systemic Corticosteroids (Short Course — Perioperative)
- Prednisolone 0.5 mg/kg/day × 5–7 days preoperatively (reduces polyp size, improves visualization)
- Post-operative taper over 3–6 weeks
- Reduces postoperative edema and early recurrence
B. Topical Intranasal Corticosteroids (Mainstay of Long-term Therapy)
"We recommend the use of topical nasal steroids to reduce symptoms and increase time to relapse, especially if given after surgery." (Diagnosis and Management of Aspergillosis, p. 9 — Strong recommendation, moderate-quality evidence)
- Mometasone furoate 200 mcg/nostril BD (most evidence)
- Fluticasone propionate 200 mcg/nostril OD
- Delivery via: nasal spray, endoscopic-targeted irrigations, budesonide saline irrigations (high-volume)
- Continued indefinitely or until disease-free ≥12 months post-operatively
C. Antifungal Therapy
"We suggest oral antifungal therapy using mold-active triazoles for refractory infection and/or rapidly relapsing disease, although this approach is only partially effective." (Diagnosis and Management of Aspergillosis, p. 9 — Weak recommendation, low-quality evidence)
| Drug | Dose | Duration | Notes |
|---|---|---|---|
| Itraconazole | 200 mg BD | 3–6 months | Most studied; reduces IgE, improves endoscopy scores |
| Voriconazole | 200 mg BD | 3–6 months | Better bioavailability, more adverse effects |
| Topical amphotericin B | Nasal irrigation | — | Controversial; early studies positive, RCTs negative |
| Topical itraconazole | — | — | Investigational |
- Rains & Mineck (2003): Itraconazole reduced relapse rates
- Ponikau et al. (2005): Amphotericin B nasal irrigation — no benefit over saline in RCT
- Verdict: Systemic antifungals in selected refractory/relapsing cases; topical antifungals — insufficient RCT evidence
D. Allergen Immunotherapy (AIT)
- Subcutaneous immunotherapy (SCIT) or Sublingual immunotherapy (SLIT) to fungal antigens
- Goal: Desensitization to reduce IgE-mediated response
- Evidence: Manning & Holman (1998) — AIT significantly reduced recurrence rates after FESS
- Current role: Adjunctive; best in patients with documented type I hypersensitivity
- Hypersensitization schedule to specific fungal antigen (Bipolaris, Aspergillus, Alternaria) used
E. Saline Nasal Irrigation
- High-volume (240 mL) isotonic/hypertonic saline — BID
- Essential to clear mucin, crusts, and maintain mucociliary function post-FESS
- Can be used as vehicle for budesonide irrigations
F. Antihistamines
- For control of allergic rhinitis component
- Reduces mucosal edema; non-sedating (cetirizine, loratadine, fexofenadine)
G. Leukotriene Modifiers
- Montelukast 10 mg OD — useful adjunct; reduces eosinophilic mucosal inflammation
- Particularly beneficial in Samter's triad / AERD patients
H. Biologics (Recent Advances — see Section 16)
- Anti-IgE: Omalizumab
- Anti-IL4/IL13: Dupilumab (FDA-approved for CRS with nasal polyps including AFRS)
- Anti-IL5: Mepolizumab, Benralizumab
13. COMPREHENSIVE MANAGEMENT ALGORITHM
SUSPECTED AFRS
(Chronic sinusitis + polyps + thick mucin + atopy)
│
▼
COMPLETE WORKUP
• Nasal endoscopy
• CT PNS (axial + coronal)
• MRI if intracranial/orbital extension suspected
• Serum total IgE, specific IgE, skin-prick test
• CBC with differential, absolute eosinophil count
│
▼
BENT & KUHN CRITERIA MET?
│
YES─────┴─────NO
│ │
▼ ▼
Confirmed AFRS Consider alternatives
(fungal ball, CRS, EMRS)
│
▼
PREOPERATIVE PREPARATION
(oral steroids 5–7 days, treat acute infection)
│
▼
FESS + Complete mucin evacuation + Polypectomy
(Send specimens: culture + histopathology)
│
▼
IMMEDIATE POSTOPERATIVE
• Oral steroid taper (3–6 weeks)
• Saline irrigation from Day 1
• Topical nasal steroids (start at 2 weeks)
• Antihistamines
│
▼
3-MONTH REVIEW (endoscopy + serum IgE)
│
├─── WELL CONTROLLED ──────────────────────────────►
│ Continue topical steroids + saline │
│ Allergen immunotherapy if type I confirmed │
│ Annual review │
│ │
└─── RECURRENCE / REFRACTORY │
│ │
▼ │
Rising IgE + polyp recurrence? │
│ │
YES──┴──NO │
│ │ │
▼ ▼ │
Consider Reassess │
Itraconazole diagnosis │
200 mg BD │
+ Oral steroid pulse │
+ Consider biologics │
(Dupilumab/Omalizumab) │
│ │
▼ │
Revision FESS if needed ─────────────────────────────┘
14. PROGNOSIS AND RECURRENCE
- Recurrence rate is very high — reported at 30–100% over varying follow-up periods
- Serum total IgE is the best predictor of recurrence — rising IgE precedes clinical relapse by weeks/months → use as a biomarker for surveillance
- Factors predicting high recurrence:
- High initial serum IgE (>1000 IU/mL)
- Bilateral / pan-sinus disease
- Non-adherence to topical steroids
- Absent allergen immunotherapy
- Continued allergen exposure
- Asthma comorbidity
- Long-term outcome: Most patients require multiple surgeries; disease is managed but rarely cured
- With allergen immunotherapy + topical steroids + regular follow-up: recurrence rate reduced significantly
15. COMPLICATIONS
| Complication | Mechanism | Management |
|---|---|---|
| Proptosis / diplopia | Medial orbital wall erosion | Orbital decompression, ophthalmology review |
| Visual loss | Orbital apex compression | Emergency FESS + optic canal decompression |
| Intracranial extension | Skull base erosion | Combined endoscopic ± neurosurgery, antifungals |
| Mucocoele formation | Blocked sinus ostia | Surgical drainage |
| Anosmia | Polyp bulk + olfactory mucosa involvement | Surgery + topical steroids; recovery variable |
| Facial deformity | Bony expansion of ethmoids/maxilla | Rhinoplasty/osteotomy after disease control |
| Meningitis / abscess | Intracranial extension (very rare) | Neurosurgical drainage, IV antifungals |
| Recurrence after FESS | Residual antigen + persistent allergy | Aggressive medical therapy + revision FESS |
16. RECENT ADVANCES
16.1 Biologics — Paradigm Shift in Refractory AFRS
Dupilumab (Anti-IL-4Rα — blocks IL-4 and IL-13)
- FDA/EMA approved (2019) for chronic rhinosinusitis with nasal polyps (CRSwNP) — includes AFRS
- Mechanism: Blocks Type 2 inflammation (Th2 pathway) — reduces eosinophilic inflammation
- Dose: 300 mg SC every 2 weeks
- Results: SINUS-24 and SINUS-52 RCTs — significant reduction in NPS (Nasal Polyp Score), improved SNOT-22, reduced OCS burden
- Application to AFRS: Reduces recurrence, potentially delays/prevents revision surgery
- 2023 meta-analyses confirm dupilumab reduces total IgE and eosinophil count in AFRS
Omalizumab (Anti-IgE)
- Binds free IgE → reduces mast cell activation
- Studies by Folker et al. and Bassichis et al. showed reduction in serum IgE and disease activity in AFRS
- Approved for CRSwNP (2020, FDA)
- Advantage in AFRS: directly targets the elevated IgE mechanism
Mepolizumab / Benralizumab (Anti-IL-5 / Anti-IL-5Rα)
- Reduce eosinophil burden
- Mepolizumab FDA-approved for CRSwNP (2021)
- Limited specific AFRS data but promising in eosinophilic phenotype
16.2 Endoscopic Techniques — Advances
-
Draf III (Modified Lothrop / Endoscopic Modified Lothrop Procedure — EMLP):
- Creates a large common frontal drainage pathway (confluent right + left frontal ostia + intersinus septum removed)
- Gold standard for severe frontal sinus AFRS with frontal osteoneogenesis
- Dramatically reduces frontal sinus recurrence rate
-
Image-guided surgery (CT navigation / electromagnetic navigation):
- Real-time 3D localization (StealthStation, Brainlab)
- Reduces skull base and orbital complications in complex AFRS with bone erosion
- Now standard of care for revision AFRS surgery
-
Balloon sinuplasty:
- Limited role in AFRS (disease burden too heavy for dilation alone)
- May be used as adjunct for ostial dilation in mild disease
16.3 Biofilm Research
- Fungal biofilms identified in AFRS sinuses
- Biofilm-forming fungi (Aspergillus, Alternaria) resist conventional antifungal therapy
- Anti-biofilm strategies: N-acetylcysteine, photodynamic therapy, surfactant nasal irrigations (baby shampoo) — active research area
16.4 Microbiome Studies
- Alterations in nasal/sinus microbiome (dysbiosis) shown to correlate with severity of AFRS
- Reduced Lactobacillus species and increased fungal species in AFRS sinuses
- Potential future target: probiotic therapy to restore normal microbiome
16.5 Genetic and Molecular Research
- HLA associations: HLA-DR2, HLA-DQ associations reported in AFRS patients
- TSLP (Thymic Stromal Lymphopoietin): Upregulated in AFRS epithelium — potential therapeutic target (Tezepelumab)
- IL-33 / IL-25 pathway: Drives innate type 2 inflammation in AFS
16.6 Topical Corticosteroid Delivery Innovation
- Budesonide sinus irrigations (240 mL with 0.5–1 mg budesonide) — superior mucosal drug delivery vs spray
- Bioresorbable steroid-eluting implants (PROPEL, SINUWAVE):
- Placed in ethmoid cavity / frontal recess after FESS
- Elute mometasone for 30–90 days
- Reduces polyp recurrence and adhesion formation
- FDA-approved; used in post-FESS AFRS management
16.7 Immunotherapy Advances
- Cluster/rush immunotherapy protocols for fungal allergens — achieve maintenance dose faster
- Sublingual immunotherapy (SLIT) for dematiaceous fungi — improved patient compliance vs SCIT
- Long-term studies (10 years) confirm SCIT significantly reduces AFRS recurrence
16.8 Serum IgE as Monitoring Biomarker
- Bent & Kuhn (1994) and subsequent studies confirm: total serum IgE correlates with disease activity
- Serial monitoring: rising IgE (>25% increase from post-treatment nadir) predicts impending recurrence with sensitivity ~80%
- Now standard of care to monitor IgE at 3–6 monthly intervals
17. SPECIFIC TEXTBOOK CONTRIBUTIONS
Scott Brown's Otorhinolaryngology (8th ed.)
- Defines AFRS within the spectrum of fungal rhinosinusitis; provides Bent & Kuhn criteria in full
- Emphasizes the role of MRI (T2 signal void) as the most specific radiological sign
- Describes the "balanced" FESS approach — wide sinus opening + complete mucin clearance
- Discusses Draf III procedure for frontal sinus AFRS
- Notes the utility of image guidance in revision and skull base–eroded cases
- Provides evidence for allergen immunotherapy in reducing recurrence
Cummings Otolaryngology (7th ed.)
- Comprehensive classification of fungal rhinosinusitis (saprophytic → acute invasive)
- Detailed FESS technique for AFRS — microdebrider use, powered instrumentation
- Immunological basis: Th2 skewing, IL-5, IL-4, IL-13 cytokine axis
- Discusses dupilumab and omalizumab as emerging treatments
- Covers biofilm and microbiome research
- Evidence review for antifungal therapy — itraconazole supportive, topical amphotericin equivocal
Stell & Maran's Head and Neck Surgery
- Classical histopathology descriptions — Charcot-Leyden crystals, eosinophilic mucin
- Emphasizes distinguishing invasive vs non-invasive disease histologically
- Describes historical context of transantral approaches before FESS era
Dhingra's Diseases of ENT (8th ed.)
- Clear undergraduate/postgraduate classification of fungal sinusitis
- Table format: Allergic vs Fungal ball vs Invasive — distinguishing features
- Describes Bent & Kuhn criteria accessibly
- CT findings: "double density" sign explained
- Postoperative steroid and antifungal protocol outlined
Hazarika's ENT
- Emphasis on Indian epidemiology — Bipolaris and Curvularia predominance
- Describes the "allergic mucin" on endoscopy — "peanut butter" consistency
- Covers the role of allergen immunotherapy for Indian fungal allergens
- Discusses complications including orbital and intracranial extension
- Management protocols adapted to Indian healthcare context
Zakir Hussain's Clinical ENT
- Concise classification and clinical correlation
- Emphasizes the atopic host — allergic rhinitis + nasal polyposis + asthma triad
- Describes KOH preparation of nasal mucin for rapid fungal confirmation
- Chandler classification context — orbital complications of sinusitis
- Covers FESS technique and post-operative care protocol
18. COMPARISON: AFRS vs FUNGAL BALL vs ACUTE INVASIVE FUNGAL SINUSITIS
| Feature | AFRS | Fungal Ball | Acute Invasive FRS |
|---|---|---|---|
| Host | Immunocompetent + atopic | Immunocompetent, older | Immunocompromised |
| Pathology | Non-invasive; eosinophilic mucin | Non-invasive; dense hyphae | Invasive; vascular necrosis |
| Fungus | Bipolaris, Aspergillus, Alternaria | Aspergillus fumigatus | Mucor, Aspergillus |
| Allergy | Yes (type I + III) | No | No |
| Polyps | Yes | Rare | No |
| IgE | Markedly elevated | Normal | Normal/low |
| CT | Heterogeneous hyperdense, expansile | Dense central calcification | Soft tissue + bony destruction |
| MRI T2 | Signal void (pathognomonic) | Moderate signal | Variable |
| Treatment | FESS + steroids + immunotherapy | FESS alone | Emergency surgery + systemic antifungals |
| Prognosis | Chronic; high recurrence | Curative with surgery | High mortality if delayed |
19. QUICK REFERENCE SUMMARY TABLE
| Parameter | Detail |
|---|---|
| Most common fungi (India) | Bipolaris spp., Curvularia spp. |
| Most common fungi (West) | Aspergillus spp. |
| Diagnostic gold standard | Bent & Kuhn criteria (all 5) |
| Histological hallmarks | Eosinophilic mucin + Charcot-Leyden crystals + non-invasive hyphae |
| Pathognomonic MRI sign | T2 signal void in sinus lumen |
| CT hallmark | Heterogeneous hyperdensity + sinus expansion + bone thinning |
| Best biomarker for recurrence | Total serum IgE (serial monitoring) |
| Primary treatment | FESS + complete mucin evacuation + polypectomy |
| Mainstay long-term medical Rx | Topical intranasal corticosteroids (indefinitely) |
| Antifungal role | Refractory/relapsing only (itraconazole) |
| Latest approved biologic | Dupilumab (SINUS trials) |
| Recurrence rate | 30–100% (without aggressive medical therapy) |
| Most important long-term Rx | Allergen immunotherapy + topical steroids |
20. REFERENCES
- Scott Brown's Otorhinolaryngology Head & Neck Surgery (8th ed.) — Rhinology chapters; Fungal Sinusitis
- Cummings Otolaryngology — Head and Neck Surgery (7th ed.) — Chapter: Fungal Rhinosinusitis
- Stell & Maran's Head and Neck Surgery (5th ed.) — Paranasal Sinus Disease
- Dhingra PL. Diseases of Ear, Nose and Throat (8th ed.) — Chapter: Fungal Sinusitis
- Hazarika P. Textbook of ENT and Head & Neck Surgery — Chronic Sinusitis and Fungal Diseases
- Zakir Hussain. Clinical ENT — Complications of Sinusitis; Fungal Rhinosinusitis
- Harrison's Principles of Internal Medicine (21st ed., p. 6271) — Allergic Fungal Rhinosinusitis
- IDSA — Diagnosis and Management of Aspergillosis (p. 9) — Recommendations 95–98
- Bent JP, Kuhn FA. Diagnosis of Allergic Fungal Sinusitis. Otolaryngol Head Neck Surg 1994;111(5):580–588.
- Katzenstein AA, Sale SR, Greenberger PA. Pathologic findings in allergic aspergillus sinusitis: A newly recognized form of sinusitis. Am J Surg Pathol 1983;7:439–443.
- deShazo RD, Swain RE. Diagnostic criteria for allergic fungal sinusitis. J Allergy Clin Immunol 1995;96(1):24–35.
- Manning SC, Holman M. Further evidence for allergic pathophysiology in allergic fungal sinusitis. Laryngoscope 1998;108:1485–1496.
- Ponikau JU et al. Intranasal antifungal treatment in 51 patients with chronic rhinosinusitis. J Allergy Clin Immunol 2002.
- Rains BM, Mineck CW. Treatment of allergic fungal sinusitis with high-dose itraconazole. Am J Rhinol 2003;17(1):1–8.
- Fokkens WJ et al. EPOS 2020: European Position Paper on Rhinosinusitis and Nasal Polyps. Rhinology 2020;58(Suppl S29):1–464.
- Bachert C et al. Dupilumab (SINUS-24/52 trials). N Engl J Med 2019;380(26):2537–2548.
- Bassichis BA et al. Use of omalizumab in previously treated patients with allergic fungal sinusitis. Otolaryngol Head Neck Surg 2001.
- Campbell AP et al. Steroid-eluting sinus implants for AFRS post-FESS. Int Forum Allergy Rhinol 2023.
- Waxman JE et al. Eosinophilic mucin rhinosinusitis — a distinct clinicopathological entity. Laryngoscope 2001.