Anti hypertensive drug in pharmacology
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
I now have rich content from the pharmacology textbooks. Let me synthesize a comprehensive answer from the retrieved content.
Antihypertensive Drugs — Pharmacology
Classification of Antihypertensive Drugs
Antihypertensive agents are divided into several major classes based on their mechanism of action:
1. Diuretics
| Subclass | Examples | Mechanism |
|---|---|---|
| Thiazides | Hydrochlorothiazide, Chlorthalidone | Inhibit Na⁺/Cl⁻ cotransporter in distal tubule → reduce plasma volume |
| Loop diuretics | Furosemide | Block Na⁺/K⁺/2Cl⁻ in thick ascending limb |
| K⁺-sparing | Spironolactone (MRA), Amiloride | Aldosterone antagonism or ENaC blockade |
Key points:
- Thiazides are first-line for essential hypertension and isolated systolic hypertension
- Mineralocorticoid receptor antagonists (MRAs) are preferred in hyperaldosteronism and heart failure
- In elderly: use low antihypertensive doses (not maximum diuretic doses) to avoid hypokalemia, hyperglycemia, and hyperuricemia
2. ACE Inhibitors (ACEIs)
Examples: Captopril, Enalapril, Lisinopril, Ramipril
Mechanism: Inhibit angiotensin-converting enzyme → ↓ Angiotensin II → vasodilation + ↓ aldosterone
Preferred in:
- Heart failure
- Diabetes mellitus (especially with proteinuria/microalbuminuria)
- Post-MI, coronary artery disease
- Renal dysfunction, end-stage renal disease with proteinuria
- Previous stroke (with diuretics)
- Left ventricular hypertrophy
- Peripheral artery disease
Side effects: Dry cough (bradykinin accumulation), angioedema, hyperkalemia, teratogenicity
3. Angiotensin Receptor Blockers (ARBs)
Examples: Losartan, Valsartan, Candesartan, Telmisartan
Mechanism: Block AT₁ receptors → prevent angiotensin II effects without bradykinin accumulation
Preferred in: Same as ACEIs (used when ACEI causes cough); also metabolic syndrome, diabetes with proteinuria
Note: ACEIs and ARBs should not be combined (dual RAAS blockade increases adverse effects without added benefit)
4. Calcium Channel Blockers (CCBs)
| Subclass | Examples | Key Use |
|---|---|---|
| Dihydropyridines | Amlodipine, Nifedipine, Felodipine | Systemic vasodilation, hypertension, angina |
| Non-dihydropyridines | Verapamil, Diltiazem | Rate control in atrial fibrillation, angina |
Mechanism: Block L-type Ca²⁺ channels in vascular smooth muscle → vasodilation (dihydropyridines) or cardiac rate reduction (non-DHP)
Preferred in:
- Isolated systolic hypertension
- Asymptomatic atherosclerosis
- Stable angina (with β-blockers)
- Atrial fibrillation rate control (non-DHPs)
- Elderly patients (safe and effective)
- Metabolic syndrome, diabetes mellitus
5. Beta-Blockers (β-Blockers)
Examples: Metoprolol, Atenolol, Bisoprolol, Carvedilol, Labetalol
Mechanism: Block β₁ (cardiac) and/or β₂ adrenergic receptors → ↓ HR, ↓ CO, ↓ renin release
Preferred in:
- Post-MI, coronary artery disease
- Chronic heart failure (bisoprolol, carvedilol)
- Angina pectoris
- Aortic aneurysm
- Prevention of atrial fibrillation
Contraindicated/caution in:
- Obstructive airway disease (asthma/COPD) — can cause bronchospasm
- Considered less useful in the elderly unless chronic heart failure is present
6. Centrally Acting Agents (Alpha-2 Agonists)
Examples: Methyldopa, Clonidine
Mechanism: Stimulate central α₂ receptors → ↓ sympathetic outflow → ↓ BP and HR
Key uses:
- Methyldopa: drug of choice in pregnancy-related hypertension
- Clonidine: hypertensive urgency, opioid withdrawal
7. Direct Vasodilators
Examples: Hydralazine, Minoxidil, Sodium Nitroprusside, Diazoxide
Mechanism: Direct relaxation of arteriolar smooth muscle
| Drug | Use |
|---|---|
| Hydralazine | Pregnancy hypertension, hypertensive emergency |
| Minoxidil | Refractory/severe hypertension; most powerful oral agent |
| Sodium Nitroprusside | Hypertensive emergencies (IV) |
Note: Powerful vasodilators like minoxidil are rarely needed and reserved for resistant cases. They cause reflex tachycardia and sodium retention — must be combined with β-blocker and diuretic.
8. Alpha-1 Blockers
Examples: Prazosin, Doxazosin, Terazosin
Mechanism: Block α₁ adrenergic receptors → vasodilation
Special use: BPH with hypertension (doxazosin)
Side effect: First-dose orthostatic hypotension
Drug Selection by Compelling Indication
| Condition | Preferred Drug(s) |
|---|---|
| Diabetes + proteinuria | ACEI or ARB |
| Heart failure | ACEI/ARB + BB + diuretic + MRA |
| Post-MI | ACEI/ARB + BB |
| Angina | BB + CCB |
| Elderly | Thiazide, CCB |
| Pregnancy | Methyldopa, Labetalol, Hydralazine |
| Hyperaldosteronism | MRA (Spironolactone) |
| Isolated systolic HTN | ACEI/ARB, CCB, Diuretics |
| Atrial fibrillation (rate) | BB, non-DHP CCB |
| Metabolic syndrome | ACEI/ARB, CCB |
| Renal dysfunction | ACEI or ARB |
| Peripheral artery disease | ACEI, CCB |
| Aortic aneurysm | BB |
First-Line Recommendation (JNC / ACC/AHA Guidelines)
The four preferred first-line classes for essential hypertension are:
- ACE Inhibitors / ARBs
- Calcium Channel Blockers
- Thiazide Diuretics
- Beta-Blockers (particularly with compelling indications)
Blood pressure lowering per se is the most important goal — differences in outcome between drug classes are minimal when BP is equally controlled. Drug choice is individualized based on comorbidities and tolerability.
— Goodman & Gilman's The Pharmacological Basis of Therapeutics, pp. 754–1433 | Katzung's Basic and Clinical Pharmacology, 16th Ed., p. 1655
Cardiovascular tone drugs in pharmacology
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have rich content from both Katzung's Pharmacology and Ganong's Physiology. Let me synthesize a comprehensive answer on cardiovascular tone drugs.
Drugs Affecting Cardiovascular Tone
"Cardiovascular tone" refers to the baseline level of tension/contraction in blood vessel walls and the heart that determines vascular resistance, blood pressure, and cardiac output. Drugs affecting cardiovascular tone act on the neurohumoral, endothelial, and direct smooth muscle mechanisms that regulate this tone.
Physiological Basis of Cardiovascular Tone
Cardiovascular tone is maintained by:
| Mechanism | Vasoconstrictors | Vasodilators |
|---|---|---|
| Neural | Sympathetic (norepinephrine → α₁) | Parasympathetic (ACh → NO) |
| Humoral | Angiotensin II, Vasopressin (ADH), Endothelin-1, Thromboxane A₂ | Bradykinin, ANP, BNP, Prostacyclin, NO |
| Metabolic/Local | ↑ O₂, ↓ CO₂, cold, serotonin | ↓ O₂, ↑ CO₂, ↑ K⁺, adenosine, heat, lactate |
| Endothelial | Endothelin-1 | Nitric oxide (NO), Prostacyclin (PGI₂) |
Baroreceptors in the carotid sinus and aortic arch monitor BP and relay signals to the rostral ventrolateral medulla (RVLM) → modulate sympathetic output to the heart and vasculature.
Classification of Drugs Affecting Cardiovascular Tone
A. VASOCONSTRICTORS (Drugs that ↑ Tone / ↑ BP)
1. Adrenergic Agonists (Sympathomimetics)
| Drug | Receptor | Effect |
|---|---|---|
| Norepinephrine | α₁ > α₂, β₁ | Potent vasoconstriction, ↑ SVR, ↑ BP; used as vasopressor in shock |
| Epinephrine | α₁, α₂, β₁, β₂ | ↑ HR, ↑ CO, vasoconstriction at high doses; vasodilation (β₂) at low doses |
| Phenylephrine | α₁ selective | Pure vasoconstriction; reflex bradycardia; used in hypotension |
| Dopamine | D₁, β₁, α₁ (dose-dependent) | Low dose: renal vasodilation (D₁); moderate: ↑ CO (β₁); high: vasoconstriction (α₁) |
| Dobutamine | β₁ > β₂ | ↑ Inotropy, ↑ CO; mild vasodilation; used in heart failure/cardiogenic shock |
Mechanism: Bind α₁ adrenoceptors on vascular smooth muscle → IP₃/DAG → ↑ intracellular Ca²⁺ → contraction → vasoconstriction
2. Renin-Angiotensin System (Angiotensin II)
- Angiotensin II is a potent vasoconstrictor acting via AT₁ receptors → vascular smooth muscle contraction + aldosterone release
- Drugs that block this system reduce tone (see vasodilators below)
3. Vasopressin (ADH) / Terlipressin
- Acts on V₁ receptors on vascular smooth muscle → vasoconstriction
- Used in: vasodilatory shock (sepsis), GI varices hemorrhage (terlipressin)
4. Endothelin Receptor Agonists
- Endothelin-1 is the most potent endogenous vasoconstrictor; acts via ETA receptors
B. VASODILATORS (Drugs that ↓ Tone / ↓ BP)
1. Nitrates / Nitric Oxide Donors
| Drug | Mechanism | Use |
|---|---|---|
| Nitroglycerine (GTN) | Releases NO → ↑ cGMP → smooth muscle relaxation | Angina, acute MI, hypertensive emergency |
| Isosorbide dinitrate/mononitrate | Same as GTN | Chronic angina, heart failure |
| Sodium Nitroprusside | Directly releases NO | Hypertensive emergency, acute heart failure |
Mechanism: NO activates guanylyl cyclase → ↑ cGMP → ↓ intracellular Ca²⁺ → vasodilation (predominantly venous at low doses, arterial at high doses)
2. ACE Inhibitors
- Examples: Enalapril, Lisinopril, Ramipril, Captopril
- Mechanism: Block conversion of Ang I → Ang II; also prevent degradation of bradykinin (a vasodilator)
- Effect: ↓ Vasoconstriction + ↑ vasodilation via bradykinin
- Bradykinin inhibition by ACE contributes significantly to the antihypertensive action
3. Angiotensin Receptor Blockers (ARBs)
- Examples: Losartan, Valsartan, Candesartan
- Block AT₁ receptors → prevent Ang II-mediated vasoconstriction
- Unlike ACEIs, do NOT accumulate bradykinin → no cough
4. Calcium Channel Blockers (CCBs)
- Mechanism: Block L-type Ca²⁺ channels in vascular smooth muscle and/or cardiac myocytes
- Dihydropyridines (Amlodipine, Nifedipine): predominantly vascular → peripheral vasodilation
- Non-dihydropyridines (Verapamil, Diltiazem): cardiac + vascular → ↓ HR + ↓ contractility
5. Kinins (Pharmacological Context)
- Bradykinin and Kallidin → act on B₂ receptors → stimulate NO and PGI₂ release → vasodilation
- ACE inhibitors amplify kinin levels → contributes to vasodilatory antihypertensive effect
- B₂ receptor antagonists (e.g., Icatibant) → used in hereditary angioedema
- Kallikrein inhibitors (Ecallantide, Aprotinin, Lanadelumab) → reduce kinin synthesis
6. Natriuretic Peptides
| Peptide | Source | Action |
|---|---|---|
| ANP (Atrial Natriuretic Peptide) | Cardiac atria (released by stretch) | ↑ Na⁺ excretion, vasodilation, ↓ renin/aldosterone/ADH |
| BNP (Brain/B-type) | Ventricular myocardium | Same as ANP; marker + treatment for heart failure |
| CNP | Endothelium | Local vasodilation |
Drugs based on this system:
- Nesiritide (recombinant BNP) — venodilation + natriuresis in acute heart failure
- Sacubitril (neprilysin inhibitor, combined with valsartan as LCZ696/Entresto) — prevents ANP/BNP breakdown → prolongs vasodilatory/natriuretic effects
7. Endothelin Receptor Antagonists
| Drug | Receptor | Use |
|---|---|---|
| Bosentan | ETA + ETB | Pulmonary arterial hypertension (PAH) |
| Ambrisentan | ETA selective | PAH |
| Macitentan | ETA + ETB | PAH |
Mechanism: Block ET-1 binding → prevent vasoconstriction of pulmonary vasculature
8. Phosphodiesterase Inhibitors
| Drug | PDE subtype | Effect |
|---|---|---|
| Sildenafil, Tadalafil | PDE-5 | ↑ cGMP in pulmonary vessels → vasodilation; PAH + erectile dysfunction |
| Milrinone | PDE-3 | ↑ cAMP in heart + vasculature → ↑ inotropy + vasodilation; acute heart failure |
9. Alpha-1 Blockers
- Prazosin, Doxazosin, Terazosin
- Block α₁ adrenoceptors → prevent norepinephrine-mediated vasoconstriction
- Used in hypertension + BPH
10. Direct Vasodilators
- Hydralazine — arteriolar dilation (mechanism unclear, possibly NO-related); used in heart failure, pregnancy HTN
- Minoxidil — opens K⁺_ATP channels → hyperpolarization → vasodilation; severe/resistant HTN
- Diazoxide — opens K⁺_ATP channels; hypertensive emergencies
11. Prostacyclin Analogues
| Drug | Use |
|---|---|
| Epoprostenol (PGI₂) | PAH (IV) |
| Iloprost | PAH (inhaled) |
| Treprostinil | PAH (IV, SC, inhaled) |
Mechanism: Prostacyclin (PGI₂) produced by endothelium inhibits platelet aggregation + promotes vasodilation via ↑ cAMP in vascular smooth muscle
Summary Table
| Drug Class | Tone Effect | Key Drugs | Main Clinical Use |
|---|---|---|---|
| Sympathomimetics | ↑ (vasopressor) | Norepinephrine, Phenylephrine, Dopamine | Shock, hypotension |
| Inotropes | ↑ CO | Dobutamine, Milrinone | Cardiogenic shock, HF |
| Nitrates/NO donors | ↓ (venodilation) | Nitroglycerine, Nitroprusside | Angina, HTN emergency |
| ACE Inhibitors | ↓ | Enalapril, Ramipril | HTN, HF, post-MI |
| ARBs | ↓ | Losartan, Valsartan | HTN, HF, diabetic nephropathy |
| CCBs | ↓ | Amlodipine, Diltiazem | HTN, angina, arrhythmia |
| Alpha blockers | ↓ | Prazosin, Doxazosin | HTN, BPH |
| Endothelin antagonists | ↓ pulmonary | Bosentan, Ambrisentan | PAH |
| PDE-5 inhibitors | ↓ pulmonary | Sildenafil, Tadalafil | PAH, erectile dysfunction |
| PDE-3 inhibitors | ↑ CO + ↓ tone | Milrinone | Acute HF |
| Prostacyclin analogues | ↓ pulmonary | Epoprostenol, Iloprost | PAH |
| Natriuretic peptides | ↓ | Nesiritide, Sacubitril | Acute/chronic HF |
| Direct vasodilators | ↓ | Hydralazine, Minoxidil | Resistant HTN, HF |
| Vasopressin/Terlipressin | ↑ | Vasopressin | Vasodilatory shock, varices |
— Katzung's Basic and Clinical Pharmacology, 16th Ed. | Ganong's Review of Medical Physiology, 26th Ed.
Cardiovascular tone of drugs in pharmacology
Asking for Preferences
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Loading Image
Loading Image
Excellent — I have rich content and two highly relevant diagrams directly from Katzung's. Now I'll deliver a comprehensive, well-illustrated answer.
Cardiovascular Tone — Drugs in Pharmacology
Cardiovascular tone = the baseline level of tension in vascular smooth muscle that determines:
- Arteriolar tone → Peripheral vascular resistance (PVR) → arterial blood pressure
- Venous tone → Venous capacitance → ventricular preload (right ventricular diastolic wall stress)
Both directly affect myocardial wall stress, cardiac output, and oxygen demand.
Molecular Basis of Vascular Smooth Muscle Tone
Contraction Pathway (↑ Tone)
Ca²⁺ enters via L-type channels → binds Calmodulin → forms Ca²⁺-Calmodulin complex → activates Myosin Light Chain Kinase (MLCK)* → phosphorylates Myosin-LC → interacts with Actin → CONTRACTION
Relaxation Pathway (↓ Tone)
Four major drug-targetable mechanisms:
| Mechanism | Second Messenger | Drug Examples |
|---|---|---|
| ↑ cGMP → dephosphorylates myosin-LC | cGMP | Nitrates, Nitroprusside, Sildenafil |
| ↓ intracellular Ca²⁺ | — | Calcium Channel Blockers |
| Membrane stabilisation (↑ K⁺ permeability → hyperpolarization) | K⁺ | Minoxidil, Nicorandil |
| ↑ cAMP → inactivates MLCK | cAMP | β₂ agonists, Milrinone (PDE-3 inhibitor) |
Figure 1 — Vascular Smooth Muscle Contraction & Relaxation
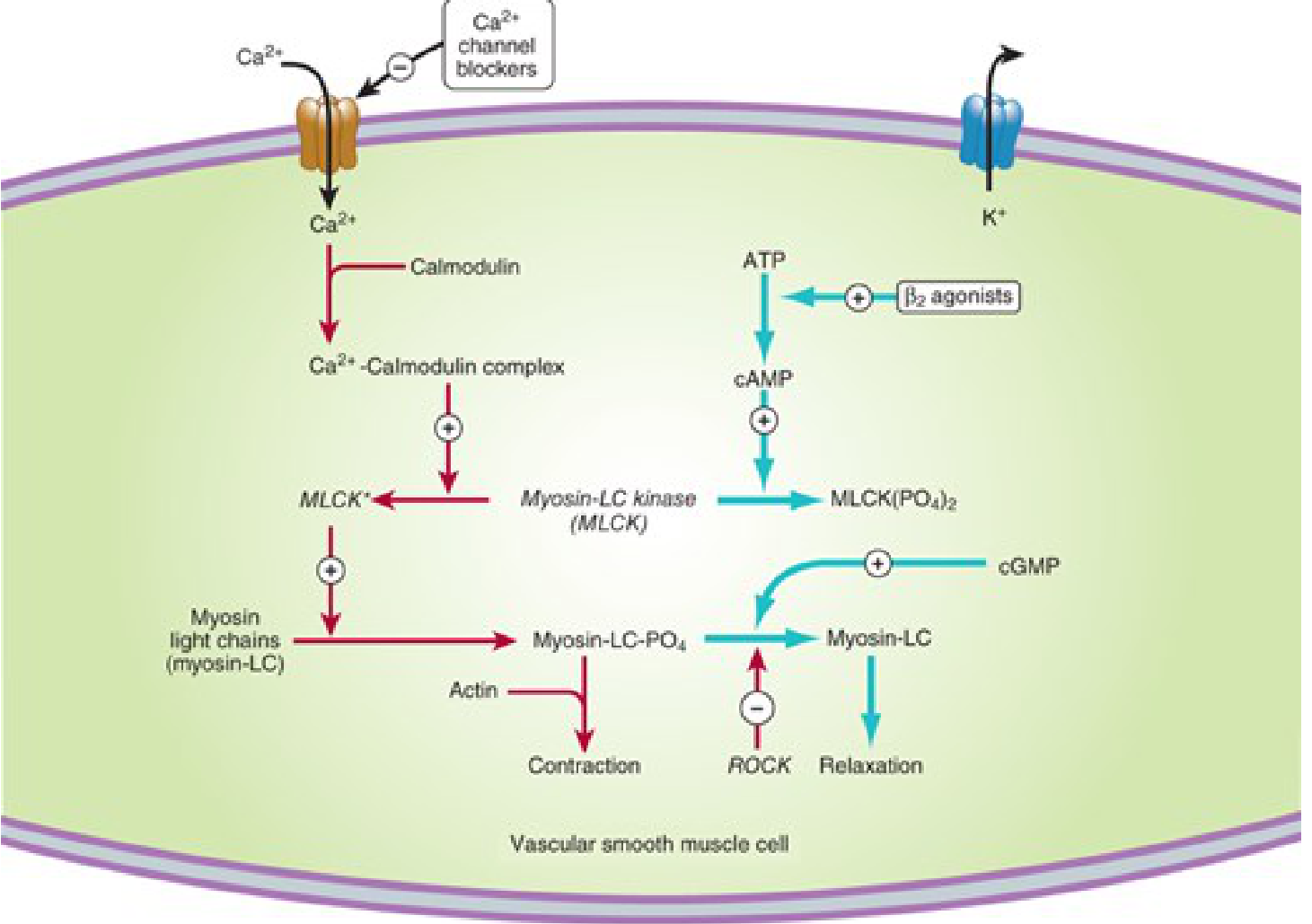
Red arrows = contraction pathway. Blue arrows = relaxation pathways. Ca²⁺ channel blockers block Ca²⁺ entry. β₂ agonists ↑ cAMP → inactivate MLCK. cGMP promotes dephosphorylation of myosin-LC. ROCK kinase inhibits relaxation.
Figure 2 — Nitric Oxide / Nitrate Mechanism in Vascular Smooth Muscle
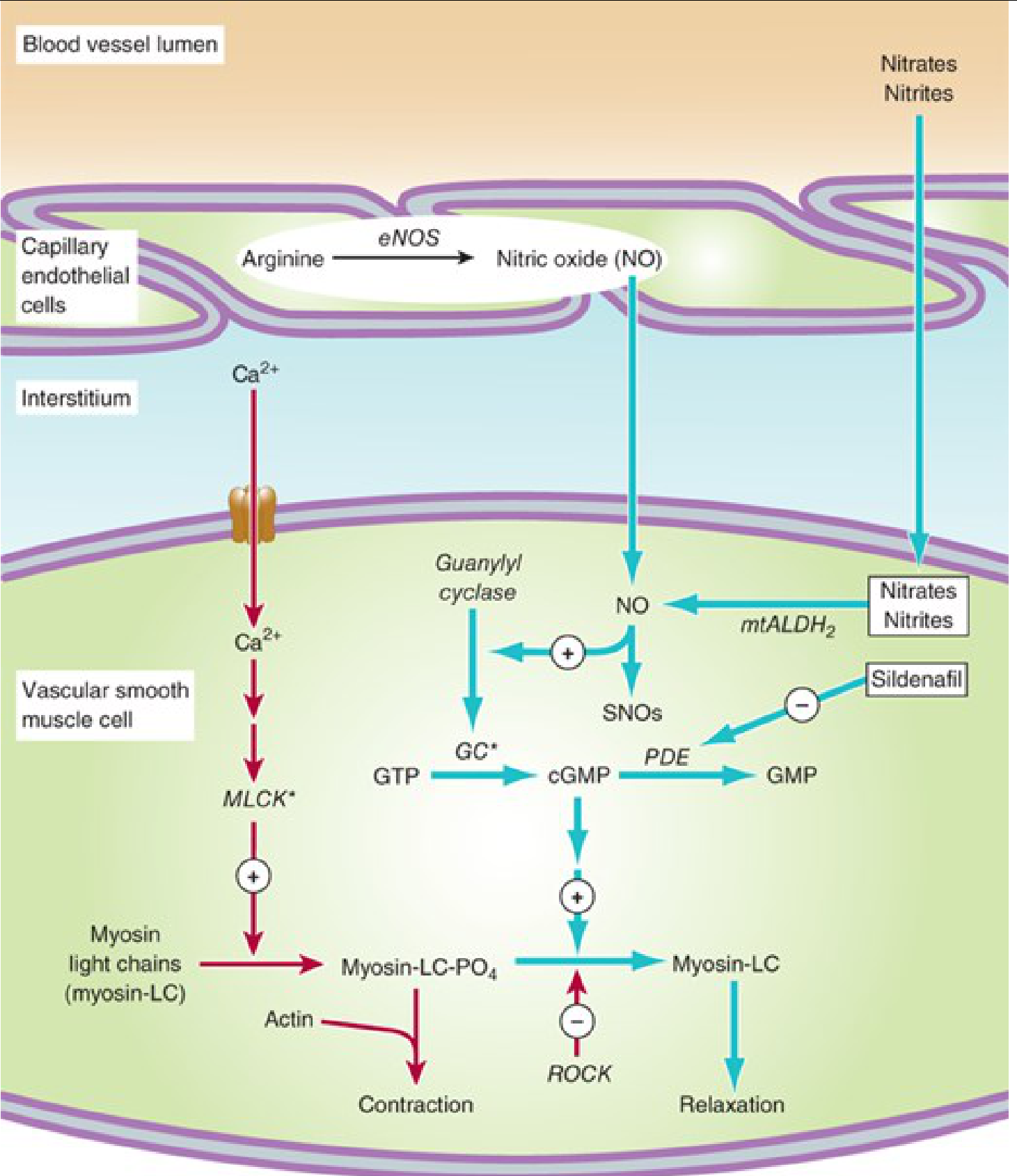
Endothelium generates NO via eNOS (from arginine). Nitrates/Nitrites also release NO via mtALDH₂. NO → activates Guanylyl Cyclase → ↑ cGMP → myosin-LC dephosphorylation → RELAXATION. Sildenafil blocks PDE (the enzyme that breaks down cGMP), thereby prolonging vasodilation.
Classification of Drugs by Effect on Cardiovascular Tone
A. DRUGS THAT INCREASE TONE (Vasopressors / Vasoconstrictors)
1. Adrenergic Agonists (Sympathomimetics)
| Drug | Receptor | Cardiovascular Effect |
|---|---|---|
| Norepinephrine | α₁ > α₂, β₁ | ↑ SVR (vasoconstriction), ↑ BP, ↑ contractility |
| Epinephrine | α₁, α₂, β₁, β₂ | Low dose → β₂ vasodilation; high dose → α₁ vasoconstriction |
| Phenylephrine | α₁ selective | Pure vasoconstriction → reflex bradycardia |
| Dopamine | D₁, β₁, α₁ (dose-dependent) | Low: renal vasodilation; mid: ↑ CO; high: vasoconstriction |
| Dobutamine | β₁ > β₂ | ↑ Inotropy, ↑ CO; mild peripheral vasodilation |
Mechanism (vasoconstriction): α₁ activation → Gq → IP₃/DAG → ↑ intracellular Ca²⁺ → MLCK activation → contraction
2. Vasopressin (ADH) / Terlipressin
- Receptor: V₁ (vascular smooth muscle)
- Effect: Potent vasoconstriction → ↑ SVR
- Use: Vasodilatory (septic) shock; variceal hemorrhage (terlipressin)
3. Angiotensin II
- Acts via AT₁ receptors → vasoconstriction + aldosterone release
- Not used pharmacologically as a vasopressor per se, but underlies RAAS-mediated tone
- Pharmacological relevance: Blocked by ACEIs and ARBs (see below)
4. Endothelin-1 (ET-1)
- Most potent endogenous vasoconstrictor
- Stimulated by: Angiotensin II, catecholamines, hypoxia, thrombin, oxidized LDL, shear stress
- Inhibited by: NO, ANP, prostacyclin, PGE₂
B. DRUGS THAT DECREASE TONE (Vasodilators)
1. Organic Nitrates / Nitric Oxide Donors
(Increase cGMP mechanism)
| Drug | Route | Duration | Use |
|---|---|---|---|
| Nitroglycerin (GTN) | SL, IV, transdermal | Short | Acute angina, acute MI, hypertensive emergency |
| Isosorbide Dinitrate | Oral, SL | Intermediate | Chronic angina, heart failure |
| Isosorbide Mononitrate | Oral | Long | Chronic angina prophylaxis |
| Sodium Nitroprusside | IV only | Very short | Hypertensive emergency, acute HF |
Key points:
- Predominantly venodilation at lower doses → ↓ preload
- Arteriolar dilation at higher doses → ↓ afterload
- Arterioles and precapillary sphincters dilate least due to reflex compensation and variable NO release
- Tolerance develops with continuous nitrate use (nitrate-free interval required)
- Nitroprusside does not develop tolerance (unlike nitroglycerin)
2. Calcium Channel Blockers (CCBs)
(↓ intracellular Ca²⁺ mechanism)
| Subtype | Drugs | Primary Site | Tone Effect |
|---|---|---|---|
| Dihydropyridines (DHP) | Amlodipine, Nifedipine, Felodipine | Vascular smooth muscle | ↓ Arteriolar tone → ↓ SVR |
| Non-DHP | Verapamil, Diltiazem | Cardiac > Vascular | ↓ HR + ↓ contractility + ↓ tone |
Mechanism: Block L-type Ca²⁺ channels → ↓ Ca²⁺ influx → ↓ MLCK activity → relaxation
Uses: Hypertension, angina, arrhythmias (non-DHP), pulmonary arterial hypertension
3. ACE Inhibitors (ACEIs)
(RAAS blockade + bradykinin-mediated vasodilation)
- Examples: Captopril, Enalapril, Lisinopril, Ramipril, Perindopril
- Mechanism:
- Block ACE → ↓ Angiotensin II → ↓ vasoconstriction + ↓ aldosterone
- Also block bradykinin degradation → ↑ bradykinin → ↑ NO + PGI₂ → vasodilation
- Uses: HTN, HF, post-MI, diabetic nephropathy
- Side effect: Dry cough (bradykinin accumulation), angioedema
4. Angiotensin Receptor Blockers (ARBs)
(AT₁ receptor blockade)
- Examples: Losartan, Valsartan, Candesartan, Telmisartan
- Mechanism: Block AT₁ → prevent Ang II vasoconstriction without bradykinin accumulation
- No cough (unlike ACEIs)
5. Beta-Blockers
(Reduction of cardiac tone and sympathetic-driven vasoconstriction)
- Examples: Metoprolol (β₁), Atenolol (β₁), Carvedilol (α₁ + β₁ + β₂), Labetalol (α₁ + β)
- Mechanism: ↓ HR, ↓ contractility, ↓ CO → ↓ BP; also ↓ renin release
- Carvedilol and labetalol additionally block α₁ → direct vasodilation
- Uses: HTN, angina, HF, post-MI, arrhythmias
6. Potassium Channel Openers
(Membrane hyperpolarization mechanism)
| Drug | Use |
|---|---|
| Minoxidil | Severe/resistant hypertension; oral |
| Nicorandil | Angina (also has nitrate component) |
| Diazoxide | Hypertensive emergency |
Mechanism: Open ATP-sensitive K⁺ channels → K⁺ efflux → membrane hyperpolarization → reduced Ca²⁺ entry → vasodilation
Side effects: Reflex tachycardia, sodium/water retention → must combine with β-blocker + diuretic
7. PDE Inhibitors
(↑ cAMP or ↑ cGMP mechanism)
| Drug | PDE Target | Effect |
|---|---|---|
| Sildenafil, Tadalafil | PDE-5 | ↑ cGMP → pulmonary vasodilation |
| Milrinone | PDE-3 | ↑ cAMP → ↑ inotropy + ↓ SVR (vasodilation) |
| Theophylline | Non-selective PDE | Bronchodilation + mild CV effects |
8. Alpha-1 Blockers
(Block α₁-mediated vasoconstriction)
- Examples: Prazosin, Doxazosin, Terazosin
- Prevent norepinephrine binding → arteriolar and venous dilation
- Side effect: First-dose orthostatic hypotension
9. Centrally Acting Agents (α₂ Agonists)
(↓ Central sympathetic outflow)
| Drug | Use |
|---|---|
| Clonidine | HTN, hypertensive urgency, opioid withdrawal |
| Methyldopa | Drug of choice in pregnancy HTN |
| Moxonidine | HTN (imidazoline receptor agonist) |
Mechanism: Stimulate α₂ receptors in RVLM (rostral ventrolateral medulla) → ↓ sympathetic outflow → ↓ HR + ↓ vascular tone
10. Endothelin Receptor Antagonists
| Drug | Receptors Blocked | Use |
|---|---|---|
| Bosentan | ETA + ETB | Pulmonary arterial hypertension (PAH) |
| Ambrisentan | ETA selective | PAH |
| Macitentan | ETA + ETB | PAH |
Mechanism: Block ET-1 → prevent vasoconstriction of pulmonary vasculature → ↓ pulmonary vascular resistance
11. Prostacyclin Analogues
| Drug | Route | Use |
|---|---|---|
| Epoprostenol (PGI₂) | IV | PAH |
| Iloprost | Inhaled | PAH |
| Treprostinil | IV / SC / Inhaled | PAH |
Mechanism: PGI₂ → ↑ cAMP in vascular smooth muscle → MLCK inactivation → vasodilation + inhibit platelet aggregation
12. Hydralazine (Direct Vasodilator)
- Mechanism: Direct arteriolar dilation (possibly via NO release or K⁺ channel opening)
- Uses: Resistant HTN, HF (combined with nitrates), hypertension in pregnancy
- Side effect: Reflex tachycardia, lupus-like syndrome with long-term use
Neurohumoral Regulators of Vascular Tone — Summary (Braunwald's Heart Disease)
| Category | Vasoconstrictors | Vasodilators |
|---|---|---|
| Catecholamines | Norepinephrine (α₁) | Epinephrine (β₂), Dopamine |
| Peptides | Angiotensin II, Vasopressin (AVP), Endothelin-1, Neuropeptide Y, Urotensin II | Bradykinin, Adrenomedullin, Apelin, ANP, BNP |
| Gases | — | Nitric Oxide (EDRF) |
| Lipids | Thromboxane A₂ | Prostacyclin (PGI₂), PGE₂ |
Master Summary Table
| Drug Class | Tone Effect | Mechanism | Key Drugs |
|---|---|---|---|
| Organic Nitrates | ↓ (veno > arterio) | ↑ cGMP via NO | GTN, Isosorbide, Nitroprusside |
| Calcium Channel Blockers | ↓ | ↓ Ca²⁺ influx → ↓ MLCK | Amlodipine, Verapamil |
| ACE Inhibitors | ↓ | ↓ Ang II + ↑ Bradykinin | Enalapril, Ramipril |
| ARBs | ↓ | Block AT₁ receptor | Losartan, Valsartan |
| Beta-Blockers | ↓ (cardiac tone) | ↓ HR, CO, renin | Metoprolol, Carvedilol |
| Alpha-1 Blockers | ↓ | Block α₁ receptors | Prazosin, Doxazosin |
| K⁺ Channel Openers | ↓ | Hyperpolarization | Minoxidil, Nicorandil |
| PDE-5 Inhibitors | ↓ (pulmonary) | ↑ cGMP | Sildenafil, Tadalafil |
| PDE-3 Inhibitors | ↓ + ↑ CO | ↑ cAMP | Milrinone |
| ET Antagonists | ↓ (pulmonary) | Block ET-1 | Bosentan, Ambrisentan |
| Prostacyclin analogues | ↓ (pulmonary) | ↑ cAMP | Epoprostenol, Iloprost |
| Hydralazine | ↓ (arterial) | Direct; K⁺/NO | Hydralazine |
| Central α₂ agonists | ↓ | ↓ Sympathetic outflow | Clonidine, Methyldopa |
| Sympathomimetics | ↑ (vasopressors) | α₁ → ↑ Ca²⁺ | Norepinephrine, Phenylephrine |
| Vasopressin | ↑ | V₁ receptor | Vasopressin, Terlipressin |
— Katzung's Basic and Clinical Pharmacology, 16th Ed., pp. 304–308 | Braunwald's Heart Disease, 15th Ed. | Goodman & Gilman's Pharmacological Basis of Therapeutics
Hypotension drugs for pharmacology
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content from Tintinalli's Emergency Medicine, Goodman & Gilman's, Harrison's, and Goldman-Cecil Medicine. Here is the full synthesized answer:
Drugs for Hypotension — Pharmacology
Hypotension is managed differently depending on its cause and type:
| Type | Cause | Primary Drug Approach |
|---|---|---|
| Distributive (septic, anaphylactic, neurogenic) | Loss of vascular tone | Vasopressors |
| Cardiogenic | Pump failure | Inotropes ± vasopressors |
| Hypovolemic | Volume loss | IV fluids first, then vasopressors |
| Obstructive | Tension pneumothorax, PE | Treat cause; vasopressors as bridge |
| Orthostatic / Neurogenic | Autonomic failure, chronic | Fludrocortisone, Midodrine, Droxidopa |
Key principle: Adequate volume resuscitation must be attempted first. Vasopressors are initiated when hypotension and tissue hypoperfusion persist despite fluids (target MAP ≥ 65 mmHg).
A. VASOPRESSORS (Acute / ICU Setting)
1. Norepinephrine (Noradrenaline) — FIRST-LINE for Septic Shock
- Receptors: α₁ > α₂ (dominant), β₁
- Mechanism: α₁ → potent vasoconstriction → ↑ SVR; β₁ → ↑ inotropy + chronotropy
- Dose: Initial 0.02–0.05 mcg/kg/min IV; titrate up to 1.0 mcg/kg/min
- Pharmacokinetics: Half-life 1–2 min; metabolised via MAO + COMT
- Uses: Septic shock (first choice), post-cardiac arrest, mixed shock
- Adverse effects: Peripheral ischaemia, tissue necrosis (extravasation), tachyarrhythmia, ↓ splanchnic flow at high doses
- Admin: Preferred via central venous catheter
2. Epinephrine (Adrenaline) — Second-line Septic Shock / First-line Anaphylaxis / Cardiac Arrest
- Receptors: α₁, α₂, β₁, β₂ (non-selective)
- Mechanism:
- Low dose: β₂ predominates → vasodilation + bronchodilation
- High dose: α₁ predominates → vasoconstriction + ↑ SVR
- β₁ → ↑ HR, ↑ contractility, ↑ CO
- Dose:
- Cardiac arrest: 1 mg IV/IO every 3–5 min
- Anaphylaxis: 0.3–0.5 mg IM (auto-injector) or IV infusion
- Septic shock: 0.05–2 mcg/kg/min IV; titrate
- Bradycardia: 2–10 mcg/min IV
- Half-life: <5 min
- Uses: Anaphylaxis, cardiac arrest (VF/VT/asystole), adjunct in septic shock when NE insufficient, symptomatic bradycardia
- Adverse effects: Tachyarrhythmias, myocardial ischaemia, ↓ splanchnic blood flow, pulmonary hypertension, lactic acidosis
- Formulations:
- 1:10,000 (0.1 mg/mL) — for IV bolus in cardiac arrest
- 1:1000 (1 mg/mL) — for IM anaphylaxis / IV infusion diluted
3. Dopamine — No Longer First-Line; Selective Use Only
- Receptors: Dose-dependent
- Low (2–5 mcg/kg/min): D₁ → renal/mesenteric vasodilation
- Moderate (5–10 mcg/kg/min): β₁ → ↑ HR, ↑ CO
- High (>10 mcg/kg/min): α₁ → vasoconstriction
- Dose: 2–20 mcg/kg/min IV; max 50 mcg/kg/min
- Half-life: ~2 min; metabolised by MAO (75%) and converted to NE (25%)
- Uses: Haemodynamic support in MI, trauma, heart failure — only when NE unsuitable (bradycardia + low risk of tachyarrhythmia)
- Why downgraded: Randomised trials show ↑ tachyarrhythmia vs norepinephrine; ↑ mortality in cardiogenic shock subgroup
- Adverse effects: Tachyarrhythmia, ectopic beats, nausea, tissue ischaemia
4. Phenylephrine — Pure α₁ Agonist
- Receptors: α₁ selective
- Mechanism: Pure vasoconstriction → ↑ SVR → ↑ MAP; no β activity → causes reflex bradycardia
- Dose: IV bolus 40–100 mcg over 20–30 s (peri-anaesthesia); or IV infusion
- Half-life: Alpha phase ~5 min; terminal phase 2–3 h
- Uses: Hypotension under anaesthesia, peri-intubation hypotension, SVT (raises BP to trigger vagal response)
- Not recommended in septic shock (less effective than NE; ↓ cardiac output)
- Adverse effects: Reflex bradycardia, low CO, renal/mesenteric/myocardial ischaemia, hypertension
5. Vasopressin (ADH) — Add-on in Septic Shock
- Receptor: V₁ (vascular smooth muscle) → direct vasoconstriction; V₂ (renal) → antidiuresis
- Mechanism: Non-adrenergic vasoconstriction; does not cause tachycardia or increase myocardial oxygen demand
- Dose: Up to 0.03 units/min IV (added to norepinephrine)
- Uses:
- Adjunct to NE in septic shock to ↑ MAP or ↓ NE dose
- Vasodilatory shock (vasopressin deficiency present)
- NOT recommended as single initial vasopressor in sepsis
- No longer part of the ACLS cardiac arrest algorithm
- Adverse effects: Splanchnic ischaemia, coronary vasoconstriction, skin necrosis, hyponatraemia
6. Dobutamine — First-Line INOTROPE in Cardiogenic Shock
- Receptors: β₁ > β₂, minimal α
- Mechanism: ↑ Inotropy (β₁) + ↑ CO; β₂ → peripheral vasodilation (↓ afterload) — unlike NE
- Dose: 2–20 mcg/kg/min IV
- Uses: Cardiogenic shock, acute decompensated heart failure
- Can be combined with NE in mixed distributive + cardiogenic shock
- Adverse effects: Tachycardia, arrhythmias; long-term use deleterious in CHF (↑ catecholamine toxicity, receptor downregulation)
7. Milrinone — Inodilator (PDE-3 Inhibitor)
- Mechanism: Inhibits PDE-3 → ↑ cAMP → ↑ inotropy + ↑ chronotropy + ↓ SVR (vasodilation)
- Use: Acute decompensated HF; cardiogenic shock (especially when β-blocker cannot be stopped)
- Advantage: Works independent of β-receptors (useful when receptors are downregulated in chronic HF)
- Adverse effects: Hypotension (vasodilation), arrhythmias
B. DRUGS FOR ORTHOSTATIC / CHRONIC HYPOTENSION
Orthostatic hypotension = excessive fall in BP on standing due to autonomic failure (Shy-Drager syndrome, Parkinson's, diabetes, drugs).
Step-wise Pharmacotherapy
1. Fludrocortisone — First Step
- Class: Synthetic mineralocorticoid
- Mechanism: ↑ Renal Na⁺ and water reabsorption → ↑ intravascular volume → ↑ BP
- Dose: 0.1 mg/day (low dose)
- Onset: Requires ≥7 days for significant clinical effect
- Adverse effects: Supine hypertension, hypokalaemia, oedema
2. Midodrine — Selective α₁ Agonist (Oral)
- Class: Prodrug → converted peripherally to desglymidodrine (active α₁ agonist)
- Mechanism: Arteriolar + venous constriction → ↑ SVR + ↑ venous return → ↑ standing BP
- Dose: 5–10 mg three times daily
- Timing: Taken before getting out of bed, before lunch, and not within 3–4 hours of bedtime (avoid supine hypertension)
- Use: Symptomatic orthostatic hypotension, POTS (postural tachycardia syndrome)
3. Droxidopa — Norepinephrine Precursor
- Mechanism: Oral synthetic amino acid → decarboxylated to norepinephrine by AAAD in sympathetic postganglionic terminals and non-neuronal tissues → restores NE levels
- Dose: 100–600 mg three times daily
- Use: Neurogenic orthostatic hypotension (Parkinson's, MSA, pure autonomic failure)
- Same timing rule as midodrine (avoid supine hypertension)
4. Pyridostigmine
- Mechanism: Acetylcholinesterase inhibitor → ↑ cholinergic transmission in autonomic ganglia → ↑ sympathetic output on demand (only when standing) → less risk of supine hypertension
- Dose: 30–60 mg 2–3×/day
- Advantage: Does not worsen supine hypertension
5. Atomoxetine (NE Reuptake Inhibitor)
- Mechanism: Blocks NE reuptake → ↑ NE at neurovascular junction
- Dose: 10–18 mg twice daily
- Use: Orthostatic hypotension with preserved peripheral sympathetic function (e.g., MSA)
6. Octreotide (Somatostatin Analogue)
- Mechanism: Splanchnic vasoconstriction → reduces postprandial blood pooling
- Use: Postprandial hypotension
- Dose: 0.2–0.4 mcg/kg SC
7. Acarbose (α-glucosidase inhibitor)
- Mechanism: ↓ GI absorption of glucose → ↓ insulin-mediated vasodilation → ↓ postprandial BP drop
- Use: Postprandial hypotension (especially in diabetics)
8. Caffeine
- Mechanism: Adenosine receptor antagonist → inhibits adenosine-mediated vasodilation
- Use: Mild orthostatic hypotension, postprandial hypotension (morning coffee strategy)
Summary Table — Vasopressors & Inotropes
| Drug | Receptor | ↑ SVR | ↑ CO | Main Use |
|---|---|---|---|---|
| Norepinephrine | α₁↑↑, β₁↑ | +++ | + | Septic shock (1st line) |
| Epinephrine | α₁, β₁, β₂ | ++ | +++ | Anaphylaxis, cardiac arrest |
| Dopamine | D₁, β₁, α₁ | + (high dose) | ++ | Selected shock (limited use) |
| Phenylephrine | α₁ | +++ | – (↓ reflex) | Anaesthesia hypotension |
| Vasopressin | V₁ | ++ | 0 | Add-on in septic shock |
| Dobutamine | β₁ > β₂ | – | +++ | Cardiogenic shock |
| Milrinone | PDE-3 | – (vasodilation) | ++ | Acute decompensated HF |
Summary Table — Orthostatic Hypotension Drugs
| Drug | Mechanism | Route | Use |
|---|---|---|---|
| Fludrocortisone | Mineralocorticoid → ↑ volume | Oral | Chronic neurogenic OH |
| Midodrine | α₁ agonist (prodrug) | Oral | OH, POTS |
| Droxidopa | NE precursor | Oral | Neurogenic OH |
| Pyridostigmine | AChE inhibitor | Oral | OH without supine HTN |
| Atomoxetine | NE reuptake inhibitor | Oral | OH with intact sympathetics |
| Octreotide | Splanchnic vasoconstriction | SC | Postprandial hypotension |
| Acarbose | ↓ Glucose absorption | Oral | Postprandial hypotension |
— Tintinalli's Emergency Medicine, pp. 175–177 | Goodman & Gilman's Pharmacological Basis of Therapeutics, pp. 1905–1917 | Harrison's Principles of Internal Medicine, 22nd Ed. | Goldman-Cecil Medicine, p. 4061
This is a shared conversation. Sign in to Orris to start your own chat.