Liver tumours
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I have enough rich content to compile a comprehensive answer. Let me now synthesize everything.
Liver Tumours
Classification
| Category | Tumour |
|---|---|
| Benign | Cavernous haemangioma, Hepatocellular adenoma, Focal nodular hyperplasia (FNH) |
| Primary malignant | Hepatocellular carcinoma (HCC), Cholangiocarcinoma, Hepatoblastoma, Angiosarcoma |
| Secondary (metastatic) | Most common overall — colorectal, breast, lung, pancreas |
Benign Tumours
1. Cavernous Haemangioma
The most common benign liver tumour, with a prevalence of 2–20% in the general population. There is a 5:1 female predominance. They are vascular malformations — congenital, enlarging by ectasia rather than angiogenesis — and are surrounded by a pseudocapsule. Growth compresses rather than invades surrounding structures.
- Symptoms: The vast majority are asymptomatic. Symptoms (pain, fullness, nausea) are linked to stretch of Glisson's capsule. Giant haemangiomas (>10 cm) can rarely cause biliary obstruction or Budd-Chiari syndrome.
- Diagnosis: Imaging (US, CT, MRI) is usually diagnostic without biopsy.
- Kasabach-Merritt syndrome: Rare — thrombocytopenia from platelet sequestration within the lesion.
- Management: Observation for asymptomatic lesions. If surgery is needed, enucleation or parenchymal resection are both options.
- Key distinction: Must be differentiated from metastatic tumours radiographically or intraoperatively.
— Current Surgical Therapy 14e | Robbins & Kumar Basic Pathology
2. Hepatocellular Adenoma (HA)
A benign tumour arising in a noncirrhotic liver, most commonly in reproductive-age women.
- Risk factors: Historically associated with oral contraceptive use (high-oestrogen pills); now more strongly linked to obesity and metabolic syndrome. Oestrogen stimulates growth.
- Driver mutations: β-catenin gain-of-function mutations, among others.
- Morphology: Ranges from sheets of normal-appearing hepatocytes to tumours with significant cytologic atypia. No portal tracts; arterial supply only.
- Complications:
- Rupture → life-threatening intraabdominal haemorrhage
- Malignant transformation (especially in β-catenin-mutated adenomas)
- Rupture risk is increased during pregnancy (high maternal and fetal mortality)
- Management: Lesions >5 cm or those with β-catenin mutations are typically resected. OCP cessation is advised. Surveillance imaging for smaller lesions.
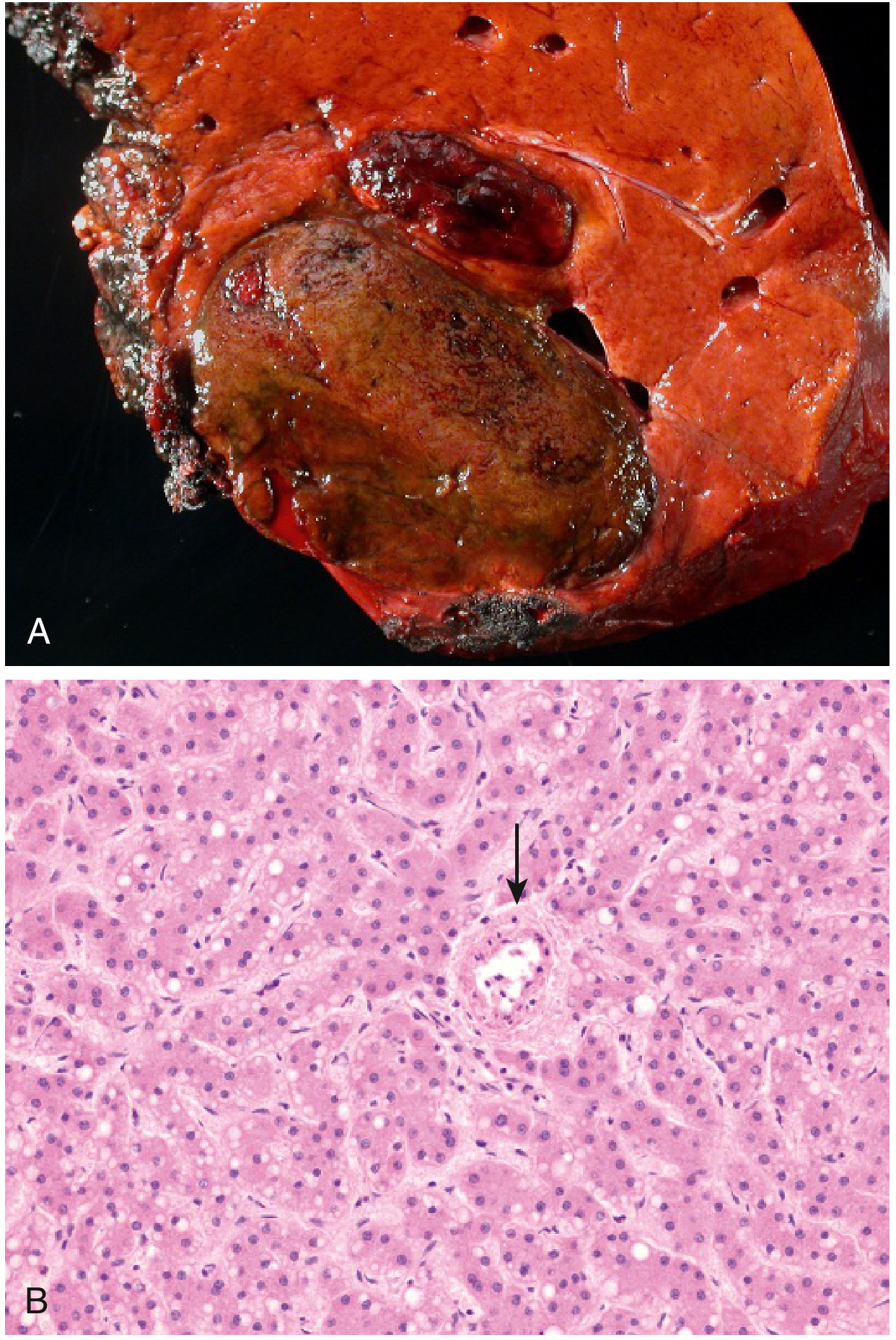
— Robbins & Kumar Basic Pathology, p. 626
3. Focal Nodular Hyperplasia (FNH)
- A well-demarcated, poorly encapsulated nodule in an otherwise normal liver, most frequent in young to middle-aged adults.
- Hallmark: Central gray-white stellate scar with fibrous septa radiating to periphery; large abnormal vessels and ductular reactions within the central scar.
- Clinically benign; rarely requires resection. Surgery may be considered when it cannot be distinguished from hepatic adenoma or fibrolamellar HCC on imaging.
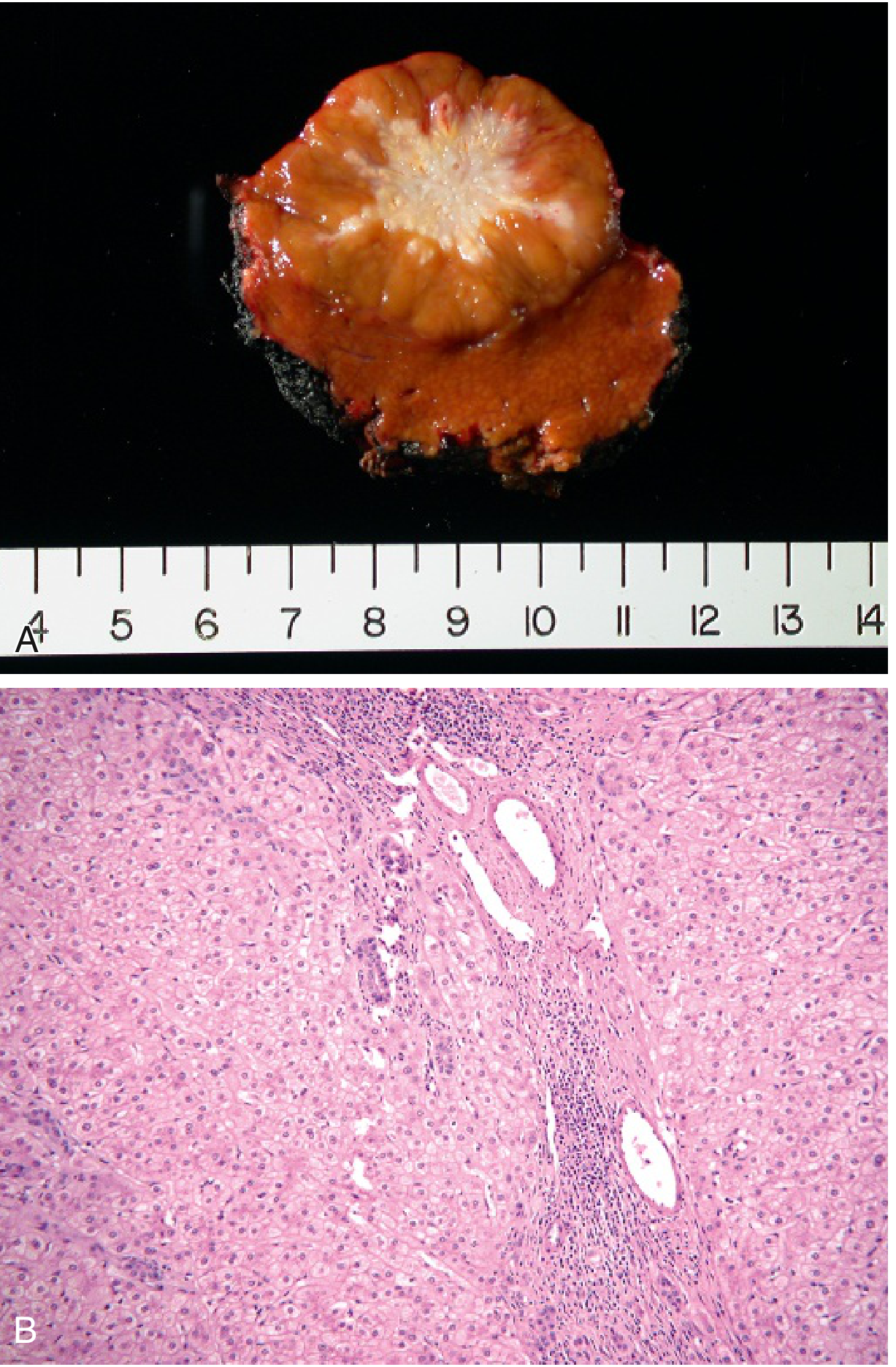
— Robbins & Kumar Basic Pathology
Primary Malignant Tumours
1. Hepatocellular Carcinoma (HCC)
The most common primary malignant liver tumour worldwide, accounting for ~5% of all cancers globally.
Epidemiology
- Highest incidence in Southeast Asia, Korea, Taiwan, and sub-Saharan Africa (endemic HBV), with peak onset at 20–40 years. In these regions, ~50% of cases arise without cirrhosis.
- In Western countries, HCC incidence tripled recently, driven largely by HCV. Almost 90% of Western cases emerge after established cirrhosis; peak onset is after 60 years.
- Male predominance: 3:1 (low-incidence areas) to 8:1 (high-incidence areas).
Risk Factors
| Factor | Notes |
|---|---|
| Chronic HBV | Vertical transmission; carcinogenesis can occur without cirrhosis |
| Chronic HCV | Almost always in setting of cirrhosis |
| Aflatoxin | Mycotoxin from Aspergillus spp.; contaminates food crops; synergises with HBV |
| Alcohol | Synergises with HBV, HCV, and smoking |
| NAFLD/Metabolic syndrome | Expected to surpass HCV as leading risk factor in the US |
| Hereditary haemochromatosis, α1AT deficiency | Inherited disorders |
| Wilson disease | Lesser risk |
Pathogenesis
Chronic liver injury → persistent inflammation, cytokines, growth factors → hepatocyte proliferation → acquisition of mutations in oncogenes and tumour suppressor genes. Cirrhosis and carcinogenesis are parallel processes driven by the same chronic injury.
Morphology
- Gross: Unifocal, multifocal, or diffusely infiltrating masses. Tumours are often soft, haemorrhagic, and have areas of necrosis. A nodule-in-nodule pattern suggests evolving cancer.
- Histology: Well-differentiated tumours resemble hepatocytes; may show trabecular or pseudo-glandular (acinar) patterns. Bile production is a useful diagnostic feature.
- Fibrolamellar HCC: A distinct variant — occurs in younger patients, those without cirrhosis, and without chronic liver disease. Better prognosis.
- Vascular invasion: Very common; portal vein invasion → intrahepatic spread.
- Metastases: Lung, peritoneum, bone, spleen, adrenal gland, brain.

Diagnosis & Staging
- In chronic liver disease, CT and MRI are typically diagnostic without biopsy: look for arterial hyperenhancement + portal venous washout.
- LI-RADS 5 = diagnostic of HCC.
- Tumour markers: AFP (alpha-fetoprotein) is the primary marker; CA 19-9 and CEA if diagnosis is unclear.
- Staging: AJCC, Barcelona Clinic Liver Cancer (BCLC), and Okuda staging systems. BCLC also incorporates degree of chronic liver disease.
Management
| Approach | Indication |
|---|---|
| Liver transplantation | Advanced cirrhosis + limited HCC (Milan criteria); removes field defect; treatment of choice when applicable |
| Surgical resection | Limited underlying liver disease (Child-Pugh A, no portal hypertension); negative margins; anatomic resections preferred (portal territory-based) as HCC spreads along portal venous tributaries |
| Ablation | Medically unfit for surgery; small tumours (<2–3 cm): outcomes comparable to resection |
| TACE (transarterial chemoembolisation) | Locoregional control; also used as bridge to transplantation |
| Systemic therapy | Advanced disease (sorafenib, lenvatinib, atezolizumab + bevacizumab) |
- Perioperative mortality for major hepatectomy is <5% in Child-Pugh A without portal hypertension, but increases significantly in Child-Pugh B/C.
- No proven adjuvant systemic therapy exists for resected HCC currently.
— Current Surgical Therapy 14e, pp. 418–420 | Robbins & Kumar Basic Pathology
2. Cholangiocarcinoma (CCA)
Arises from biliary epithelium. Less common than HCC.
- Intrahepatic CCA: Arises from intrahepatic bile ducts; presents as a hepatic mass.
- Perihilar (Klatskin tumour) and distal CCA: At the biliary confluence and distal CBD respectively.
- Risk factors: Primary sclerosing cholangitis (PSC), liver flukes (Opisthorchis, Clonorchis), choledochal cysts, chronic HBV/HCV.
- Often presents late; poor prognosis. Surgical resection is the only curative option.
3. Hepatoblastoma
- The most common primary liver malignancy in children (usually <5 years of age).
- Two main subtypes: epithelial (most common) and mixed epithelial-mesenchymal.
- Treated with chemotherapy + surgical resection. Complete resection before drug resistance develops is essential for survival.
— Robbins, Cotran & Kumar Pathologic Basis of Disease
Metastatic Tumours
The most common hepatic malignancy overall — metastases far outnumber primary tumours. Common primaries:
- Colorectal cancer (most surgically resectable)
- Breast, lung, pancreas, stomach, carcinoid/NETs
Metastases typically appear as multiple nodules. Curative resection is possible for colorectal liver metastases in selected patients (5-year survival ~40%).
Diagnostic Imaging Summary
| Lesion | Key Imaging Feature |
|---|---|
| Haemangioma | Peripheral nodular enhancement → centripetal fill-in (CT/MRI) |
| FNH | Central scar with spoke-wheel vascularity on arterial phase |
| Hepatic adenoma | Arterial enhancement; may contain fat or haemorrhage |
| HCC | Arterial hyperenhancement + portal venous washout (LI-RADS 5) |
| Cholangiocarcinoma | Peripheral rim enhancement; biliary dilatation |
| Metastases | Multiple lesions; "target" or "ring" enhancement |
Sources: Robbins & Kumar Basic Pathology (9780323790185) | Current Surgical Therapy 14e (9780323796835) | Robbins, Cotran & Kumar Pathologic Basis of Disease (9780443264528)
Now made it concise for my usmle step 1 exam
Liver Tumours — USMLE Step 1
Benign Tumours
| Tumour | Key Facts |
|---|---|
| Cavernous Haemangioma | Most common benign liver tumour. Blood-filled, vascular. Asymptomatic. Must distinguish from mets on imaging (peripheral nodular enhancement → centripetal fill-in). Do NOT biopsy. |
| Hepatocellular Adenoma | Young women + OCP use (or obesity/metabolic syndrome). Risk of rupture (haemoperitoneum) and malignant transformation (β-catenin mutation). Stop OCPs; resect if >5 cm. |
| Focal Nodular Hyperplasia (FNH) | Central stellate scar + spoke-wheel vascularity. Normal liver. Benign, no malignant potential. Ductular reactions in scar. |
Primary Malignant Tumours
Hepatocellular Carcinoma (HCC)
- Most common primary liver malignancy worldwide
- Risk factors: HBV (can cause HCC without cirrhosis), HCV (needs cirrhosis), aflatoxin (Aspergillus), alcohol, NAFLD, haemochromatosis, α1AT deficiency
- AFP is the tumour marker
- Imaging: Arterial hyperenhancement + portal venous washout → LI-RADS 5 = diagnostic
- Microscopy: Trabecular pattern, bile production, no portal tracts
- Spread: Portal vein invasion (intrahepatic); mets to lung, bone, adrenal
- Fibrolamellar HCC: Young patients, NO cirrhosis, better prognosis
Cholangiocarcinoma
- Bile duct origin
- Risk factors: PSC, liver flukes (Clonorchis, Opisthorchis), choledochal cysts
- Perihilar = Klatskin tumour
Hepatoblastoma
- Most common liver malignancy in children <5 years
- Elevated AFP
- Treat with chemo + resection
Metastatic Tumours
- Most common hepatic malignancy overall (more common than primaries)
- Sources: colorectal > breast > lung > pancreas
- Colorectal mets = most resectable
High-Yield Associations
| Buzzword | Think |
|---|---|
| Young woman + OCP → liver mass | Hepatic adenoma |
| Cirrhosis + AFP ↑ | HCC |
| Young person, no cirrhosis, HCC | Fibrolamellar HCC |
| PSC → liver cancer | Cholangiocarcinoma |
| Child <5 + AFP ↑ + liver mass | Hepatoblastoma |
| Most common benign liver tumour | Cavernous haemangioma |
| Most common liver malignancy overall | Metastatic disease |
| Central stellate scar | FNH |
| Aflatoxin | HCC (synergises with HBV) |
Alpha one atitrypsin deficiency
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
I have all the content needed for a concise USMLE Step 1 summary.
α1-Antitrypsin Deficiency — USMLE Step 1
Core Concept
Autosomal recessive disorder of protein misfolding → two distinct organ systems affected by two different mechanisms.
Genetics
| Feature | Detail |
|---|---|
| Inheritance | Autosomal recessive; codominant expression |
| Normal genotype | PiMM (90% of population) |
| Most common pathogenic mutation | PiZ allele (Glu342Lys substitution — glutamine → lysine at position 342) |
| Most severe phenotype | PiZZ → circulating α1AT levels only 10% of normal |
| Intermediate | PiMZ heterozygotes → intermediate levels |
| Prevalence of PiZZ | ~1 in 1800 (northern European descent) |
Two Mechanisms, Two Organs
| Organ | Mechanism | Result |
|---|---|---|
| Lung | Loss of function — α1AT cannot inhibit neutrophil elastase → unchecked elastase destroys alveolar walls | Panacinar emphysema (lower lobe predominance) |
| Liver | Toxic gain of function — misfolded PiZ protein accumulates in hepatocyte ER → ER stress → unfolded protein response → apoptosis | Hepatitis → cirrhosis → HCC |
Key distinction: Lung disease = lack of protein. Liver disease = too much misfolded protein.
Liver Disease
- Most common inherited hepatic disorder in infants and children
- Neonates: hepatomegaly, cholestatic jaundice (10–20%)
- Adolescents/adults: chronic hepatitis, cirrhosis, HCC (2–3% of PiZZ adults)
- Only 10–15% of PiZZ individuals develop overt liver disease (other genetic/environmental factors involved)
- Treatment: Liver transplantation (curative for liver disease; does NOT help lungs)
Lung Disease
- Emphysema develops in adults
- Panacinar emphysema, lower lobe predominant (vs. upper lobe in smoking-related centriacinar emphysema)
- May also develop bronchiectasis
- Smoking dramatically accelerates lung disease (neutrophil accumulation → more elastase release)
Histology (High-Yield)
- Liver biopsy: Round-to-oval cytoplasmic globular inclusions in hepatocytes
- Stain: PAS-positive, diastase-resistant (magenta globules)
- Periportal hepatocytes affected first
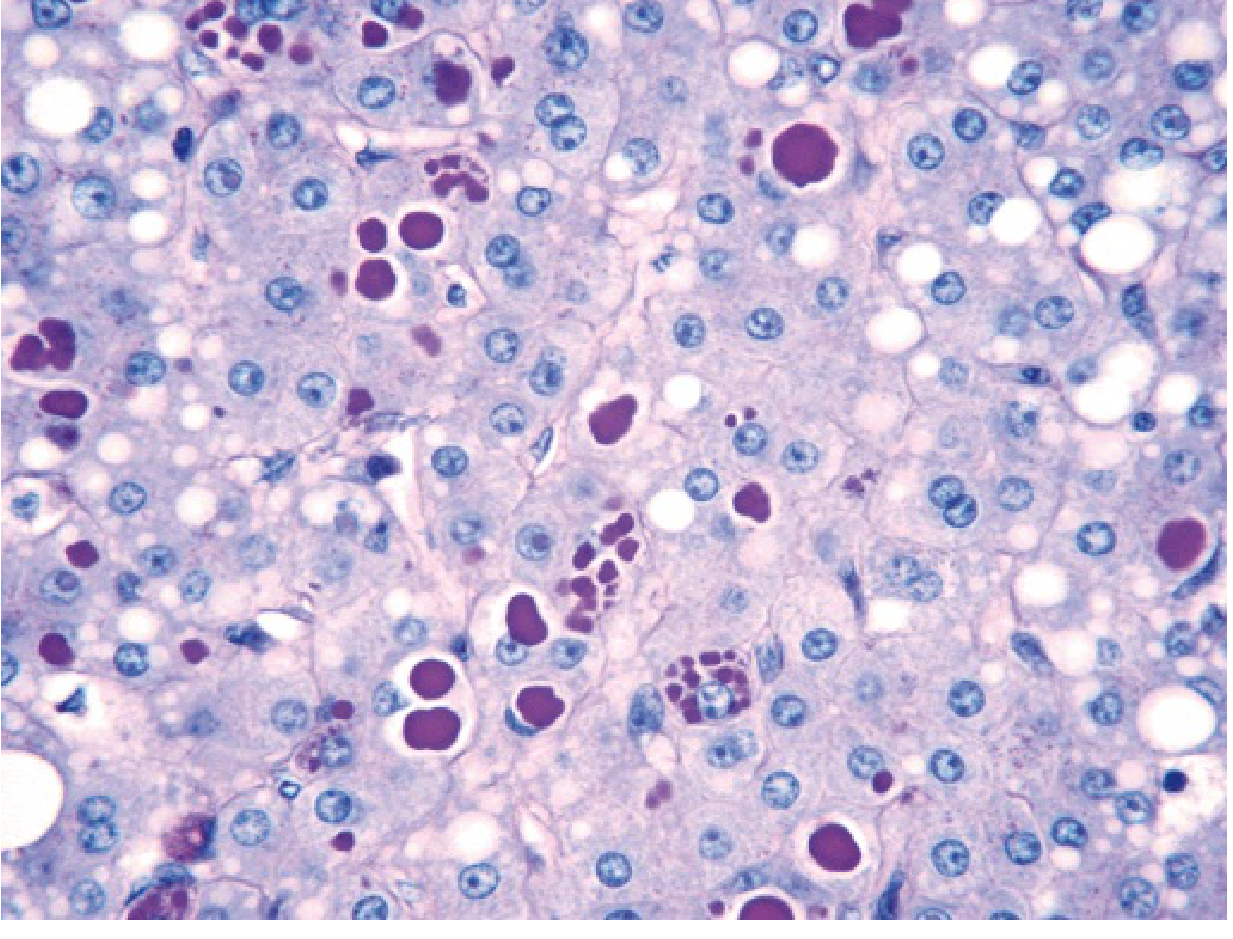
High-Yield Associations
| Buzzword | Think |
|---|---|
| Panacinar emphysema, lower lobe | α1AT deficiency |
| PAS+, diastase-resistant globules in hepatocytes | α1AT deficiency |
| Infant with cholestatic jaundice + liver disease | α1AT deficiency |
| Neonatal hepatitis → cirrhosis | α1AT deficiency |
| Protein misfolding → liver + lung disease | α1AT deficiency |
| Smoking + inherited emphysema | α1AT deficiency (smoking worsens by ↑ neutrophil elastase) |
| Most common inherited liver disease in kids | α1AT deficiency |
Sources: Robbins & Kumar Basic Pathology | Robbins, Cotran & Kumar Pathologic Basis of Disease | Murray & Nadel's Textbook of Respiratory Medicine
This is a shared conversation. Sign in to Orris to start your own chat.