Pontine haemorrhage
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
pontine hemorrhage CT MRI brainstem
Loading Image
Loading Image
Excellent. I now have everything needed for a comprehensive answer.
Pontine Haemorrhage
Epidemiology & Aetiology
Pontine haemorrhage accounts for approximately 5–10% of all spontaneous intracerebral haemorrhages (ICH). The principal causes are:
- Arterial hypertension (most common) — causes lipohyalinosis and fragility of small penetrating arteries
- Vascular malformations — cavernous malformations (more common in younger patients; tend to be subependymal, more discrete, and do not rupture into the ventricle, but carry a risk of rebleeding)
- Clotting disorders, trauma
Hypertensive pontine haemorrhages tend to be deep, rather diffuse, frequently rupture into the fourth ventricle, occur in older individuals, and carry a poor prognosis. Cavernous angioma bleeds are typically more superficial and have a better prognosis but tend to recur.
— Plum and Posner's Diagnosis and Treatment of Stupor and Coma; Localization in Clinical Neurology, 8e
Pathophysiology & Anatomy
Haemorrhage typically arises from ruptured paramedian pontine arterioles at the midpontine level, near the junction of the basis pontis and tegmentum. Blood dissects in all directions in a relatively symmetric fashion. Rupture into the fourth ventricle is frequent, causing acute obstructive hydrocephalus; extension into the medulla is rare.
The damage is caused as much by direct tissue destruction as by mass effect (unlike hemispheric haemorrhages where mass effect predominates). This is why recovery, even if the patient survives, is often limited.
— Plum and Posner's Diagnosis and Treatment of Stupor and Coma, p. 296–327
Clinical Classification
Primary pontine haemorrhages have been divided into three clinical types:
| Type | Frequency | Features |
|---|---|---|
| Classic (massive) | ~60% | Bilateral basal-tegmental involvement; quadriparesis, coma, hyperthermia, tachycardia → death |
| Hemipontine | ~20% | Unilateral basis pontis + tegmentum; hemiparesis, preserved consciousness, skew deviation, unilateral absent corneal reflex, dysarthria, facial nerve palsy; good functional recovery |
| Dorsolateral tegmental | ~20% | Gaze paresis or CN VI palsy, skew deviation, facial nerve palsy, contralateral extremity and ipsilateral facial sensory loss, ataxia, motor sparing, preserved consciousness; good recovery |
— Localization in Clinical Neurology, 8e, p. 1393
Clinical Features — Classic (Massive) Pontine Haemorrhage
Onset is abrupt, typically when the patient is awake and active, often without prodrome. A few patients complain of sudden occipital headache, vomiting, or slurred speech before losing consciousness.
Consciousness
- Coma begins abruptly in the majority; ~50% of patients present in coma
The "Classic Tetrad"
- Pinpoint (miotic) pupils (2–3 mm), but reactive to light (requires magnifying glass to detect the reaction) — due to bilateral interruption of descending sympathetic fibres with preserved parasympathetic outflow via CN III
- Absent oculovestibular/oculocephalic responses (horizontal eye movements lost)
- Quadriplegia (often flaccid acutely, or decerebrate posturing)
- Irregular/ataxic breathing — Cheyne-Stokes, apneustic, or gasping patterns
Other findings
- Ocular bobbing (or its variants) — rapid downward jerk with slow return
- Hyperthermia (38.5–40°C) within hours, seen in nearly all survivors beyond a few hours — due to involvement of thermoregulatory pathways
- Bradycardia
- If the haemorrhage extends into the midbrain: pupils may become asymmetric or dilate to midposition
Clinical Data (80 patients with pontine haemorrhage)
| Finding | % |
|---|---|
| Coma at presentation | 50% |
| Respiratory disturbance | 46% |
| Bradycardia | 43% |
| Hyperthermia | 40% |
| Pinpoint pupils | 29% |
| Hemiplegia | 43% |
| Tetraplegia | 28% |
| Decerebrate posturing | 20% |
— Plum and Posner's Diagnosis and Treatment of Stupor and Coma, Table 4.18
Less Severe Forms
With CT/MRI, milder presentations are now recognised — tegmental haemorrhages lateral to the midline producing predominantly unilateral dorsal pontine syndromes: one-and-a-half syndrome, internuclear ophthalmoplegia (INO), CN V and VII palsies, with variable long-tract signs. These result from rupture of distal tegmental branches of long circumferential arteries from the basilar trunk.
A locked-in syndrome is rarely the presentation because the haematoma typically dissects symmetrically, destroying dorsal structures. However, a bleed restricted to the basis pontis can produce this acutely.
Diagnosis
The diagnosis is usually clinically straightforward. Almost no other lesion produces the combination of:
- Sudden coma
- Periodic or ataxic breathing
- Pinpoint pupils
- Absent oculovestibular responses
- Quadriplegia
The main differential is opiate overdose (also causes pinpoint pupils), but opiates do not cause absent eye signs or flaccid quadriplegia. Naloxone can be given diagnostically if any doubt exists.
Imaging: Non-contrast CT is the first-line investigation — the haematoma appears as a hyperdense lesion in the pons. MRI (T2*/SWI) is more sensitive for small tegmental bleeds and cavernous malformations.
Imaging
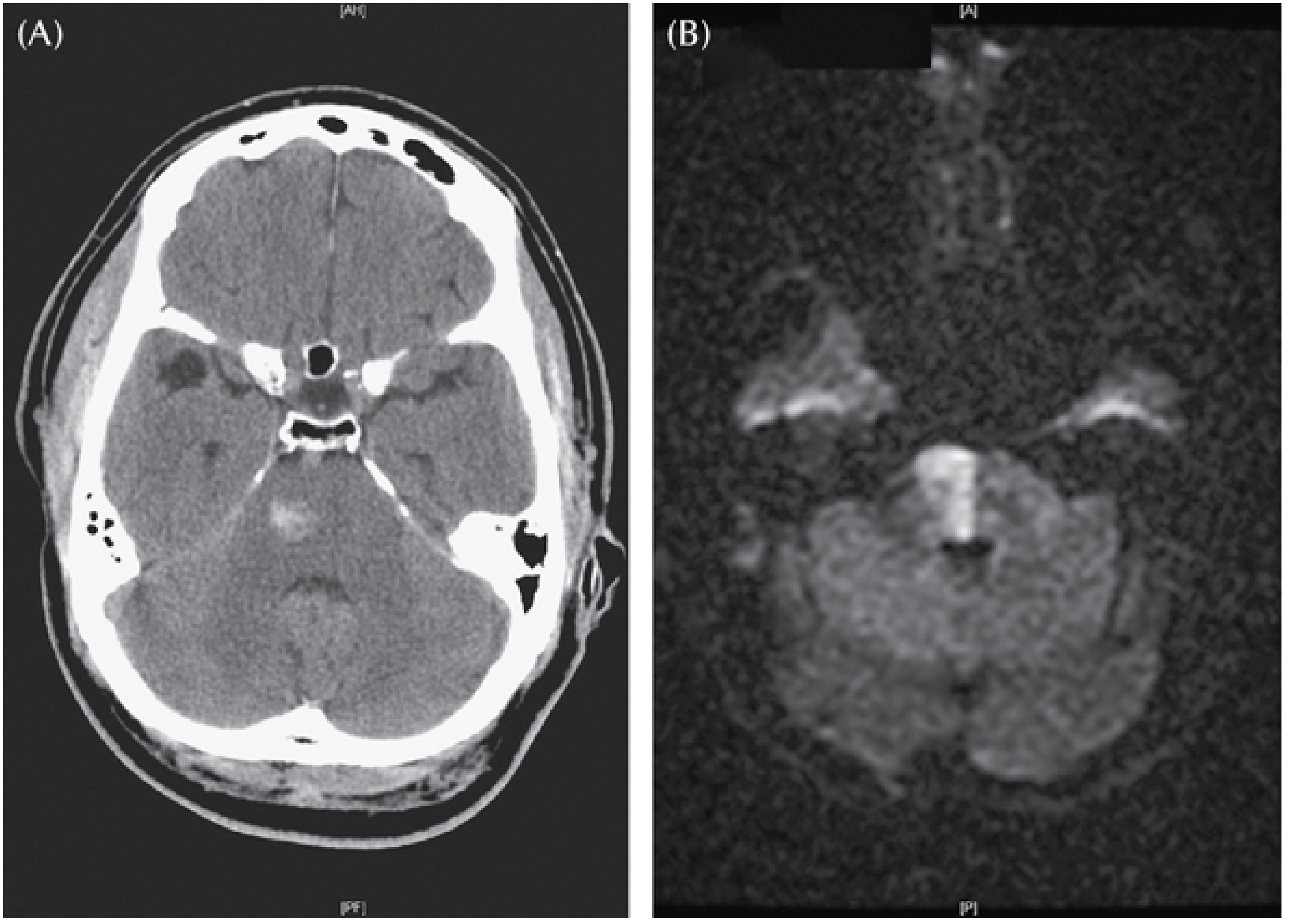
(A) Non-contrast CT showing a small haemorrhage into the right pontine base and tegmentum in a hypertensive patient presenting with left hemiparesis and dysarthria. (B) DWI MRI of a medial pontine infarct for comparison. — Plum and Posner's, Fig. 4.8
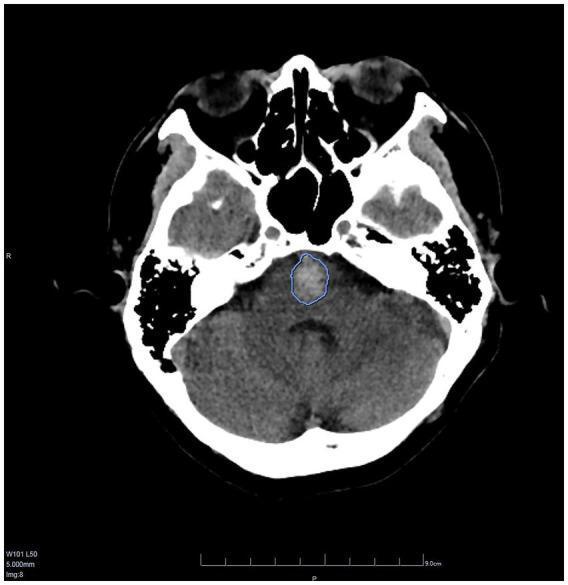
Axial CT at the level of the posterior fossa showing a primary pontine haemorrhage — the hyperdense lesion is centrally located within the pons. AI-assisted volumetric contour (blue) estimates 3.97 mL.
Poor Prognostic Features
The following predict a very poor outcome:
- Coma on presentation
- Absence of oculocephalic reflexes
- Absent corneal reflexes
- No motor responses
- Hypertension + tachycardia
- Hyperthermia (core ≥39°C)
- Acute obstructive hydrocephalus
- Intraventricular blood extension
— Localization in Clinical Neurology, 8e
Management
- Medical (supportive): Airway protection, blood pressure control, treatment of hyperthermia, management of hydrocephalus (external ventricular drain if obstructive hydrocephalus develops)
- Surgery: Generally has no role in hypertensive brainstem haemorrhages. However, for superficially located cavernous angiomas, surgical evacuation is sometimes appropriate — these have a better prognosis and a tendency to rebleed
— Plum and Posner's Diagnosis and Treatment of Stupor and Coma, p. 326
Key Differential Diagnoses
| Condition | Distinguishing features |
|---|---|
| Opiate overdose | Pinpoint pupils, but no absent eye movements, no quadriplegia; reverses with naloxone |
| Cerebellar haemorrhage with brainstem extension | May mimic, but usually occipital headache, ataxia prominent early |
| Basilar artery occlusion | Ischaemic (DWI positive), progressive onset, no blood on CT |
| Metabolic encephalopathy | Preserved pupillary reflexes even when deeply comatose |
Tramadol poisioning
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have comprehensive information from multiple authoritative sources. Let me compile the full answer.
Tramadol Poisoning
Pharmacology (Relevant to Toxicity)
Tramadol is a synthetic centrally-acting analgesic with a dual mechanism:
- Weak μ-opioid receptor agonism — via its active metabolite O-desmethyltramadol (M1), produced by CYP2D6. M1 has greater μ-receptor affinity than the parent drug and an elimination half-life of ~9 hours.
- Serotonin and norepinephrine reuptake inhibition (SNRI-like) — contributes to both analgesia and toxicity
Because of the reliance on CYP2D6 for active metabolite generation, toxicity varies significantly with genetic polymorphisms (ultrarapid metabolisers are at greater risk), liver/renal disease, and co-prescribed CYP inhibitors/inducers.
Tramadol itself has a half-life of 5–7 hours (oral); M1 ~9 hours.
— Tintinalli's Emergency Medicine; Katzung's Basic and Clinical Pharmacology, 16e; Rosen's Emergency Medicine
Dose-Related Toxicity
| Dose | Clinical Effects |
|---|---|
| Therapeutic (50–100 mg QID) | Analgesia; nausea, dizziness, orthostatic hypotension |
| Moderate overdose | Lethargy, nausea, tachycardia, seizures |
| >500 mg | Coma, hypertension, respiratory depression, apnea |
— Tintinalli's Emergency Medicine, p. 1277
Clinical Features of Tramadol Poisoning
1. Opioid Toxidrome (Partial)
Tramadol produces a partial opioid toxidrome (via M1):
- CNS depression — lethargy → stupor → coma
- Respiratory depression — slow, shallow breathing; hypercarbia, hypoxia, cyanosis
- Miosis — though less consistently than pure opioids
- Nausea, vomiting, ileus, urinary retention
Important caveat: Naloxone only partially reverses tramadol toxicity (the SNRI component is not reversed), and naloxone may paradoxically increase seizure risk.
2. Seizures (hallmark feature)
- Common and distinctive — tramadol lowers the seizure threshold
- Usually generalised tonic-clonic, typically single and self-limiting
- Naloxone does not prevent tramadol-induced seizures
- Anticonvulsants (benzodiazepines) are the treatment of choice
- Risk is amplified by co-administration of other seizure-threshold-lowering drugs (antidepressants, antipsychotics, bupropion)
3. Serotonin Syndrome
A serious and potentially life-threatening complication — can occur:
- In isolated tramadol overdose
- Much more commonly when combined with SSRIs, SNRIs, MAOIs, TCAs, linezolid, dextromethorphan
Features of serotonin syndrome:
- Altered mentation (anxiety → agitation → delirium)
- Hyperthermia
- Autonomic instability (tachycardia, hypertension, diaphoresis)
- Hyperreflexia and clonus (especially lower limbs)
- Muscle rigidity
- Deaths have been reported
Naloxone is not effective for serotonin syndrome.
4. CNS Excitation
At toxic doses, tramadol paradoxically produces CNS excitation:
- Agitation, tremor, hallucinations
- This is the SNRI and serotonergic component, distinct from the opioid component
5. Other Complications
- Hypoglycaemia — tramadol is associated with higher rates of hypoglycaemia than codeine, especially within the first 30 days of use (mechanism unclear)
- Rhabdomyolysis, compartment syndrome, myoglobinuric renal failure (especially post-seizure)
- Acute lung injury (non-cardiogenic pulmonary oedema) — uncommon; presents with tachypnoea, hypoxia, bilateral infiltrates
- Dependence and withdrawal — reported with chronic use; tramadol should be tapered before discontinuation
— Tintinalli's Emergency Medicine; Lippincott Illustrated Reviews Pharmacology; Rosen's Emergency Medicine
High-Risk Drug Interactions
| Combination | Risk |
|---|---|
| Tramadol + SSRIs/SNRIs | Serotonin syndrome |
| Tramadol + MAOIs | Serotonin syndrome (potentially fatal) |
| Tramadol + TCAs | Serotonin syndrome + lowered seizure threshold |
| Tramadol + bupropion | Markedly lowered seizure threshold |
| Tramadol + linezolid | Serotonin syndrome |
| Tramadol + CYP2D6 inhibitors (e.g. fluoxetine, paroxetine) | Reduced M1 formation → unpredictable opioid effect |
Special Populations
- Paediatric patients: Tramadol is contraindicated in children <12 years and for post-tonsillectomy/adenoidectomy pain in patients <18 years (same FDA black box warning as codeine). Contraindicated in breastfeeding mothers due to risk of infant death.
- CYP2D6 ultrarapid metabolisers: Produce excessive M1 → greater opioid toxicity
- Epilepsy: Tramadol is relatively contraindicated
- Renal/hepatic impairment: Impaired clearance of tramadol and M1
— Rosen's Emergency Medicine
Diagnosis
- Diagnosis is primarily clinical
- Standard urine opioid immunoassays typically do NOT detect tramadol (these screens detect morphine-class opioids; tramadol requires specific GC/MS or HPLC confirmation)
- Check: blood glucose, renal function, CK (if seizures occurred), ECG, paracetamol level (if co-ingestion suspected)
- Consider serotonin syndrome criteria (Hunter or Sternbach)
Management
Immediate Priorities
- Airway, Breathing, Circulation — respiratory depression is the primary cause of death
- Oxygen and bag-valve-mask ventilation as needed
- Endotracheal intubation if respiratory depression is unresponsive to naloxone or if acute lung injury is suspected
Decontamination
- Activated charcoal (1 g/kg PO) — may be given within 1–2 hours of ingestion in a cooperative patient with intact airway reflexes; with caution given CNS depression risk
Naloxone
- Partially reverses tramadol's opioid effects (respiratory depression, sedation)
- Does NOT prevent or treat seizures
- May precipitate/worsen seizures — use cautiously, titriting to respiratory effect rather than full reversal
- Dosing (Tintinalli's, Table 186-3):
| Route | Dose |
|---|---|
| IV (spontaneously breathing, opioid-naive) | 0.4 mg |
| IV (opioid-dependent) | 0.04 mg (titrate) |
| IV (apnoeic/cyanotic) | 2 mg, repeat every 3 min up to 10 mg |
| IM/SC | 2 mg |
| Intranasal | 2 mg (1 mg each nostril) |
- Duration of naloxone (20–90 min) is often shorter than tramadol/M1 half-life → naloxone infusion may be required
Seizures
- Benzodiazepines (e.g. diazepam, lorazepam) are first-line
- Tramadol seizures are usually single and self-limiting — routine prophylactic anticonvulsants are not necessary
- Avoid physostigmine; naloxone does not help
Serotonin Syndrome
- Discontinue tramadol and all serotonergic drugs
- Benzodiazepines for agitation and muscle rigidity
- Active cooling for hyperthermia
- Cyproheptadine (serotonin antagonist) as adjunct
- Severe cases: consider chlorpromazine or methysergide; intubation, paralysis, and ICU care if temperature >41°C or haemodynamic instability
Disposition
- All significant tramadol overdoses warrant monitoring for a minimum of 6–8 hours (longer for extended-release formulations)
- Admit for seizures, respiratory compromise, persistent altered consciousness, or serotonin syndrome features
Summary: What Makes Tramadol Poisoning Unique
| Feature | Unlike typical opioids |
|---|---|
| Seizures | Common; naloxone ineffective; benzodiazepines needed |
| Serotonin syndrome | Can occur even in isolation; key risk with serotonergic drugs |
| Naloxone response | Only partial reversal; may worsen seizures |
| Urine screen | Usually negative on standard opiate immunoassay |
| CNS excitation | Can occur alongside or instead of depression |
| Hypoglycaemia | Unexpected complication |
— Tintinalli's Emergency Medicine, Rosen's Emergency Medicine, Lippincott Pharmacology, Katzung's Basic and Clinical Pharmacology, 16e
Tell me about vacutainers and which sample to send in them and which vacutainor we have to keep moving in order to prevent clotting
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Searching Images
vacutainer blood collection tube colors chart EDTA heparin citrate
I now have comprehensive data from the textbooks. Let me compile the full answer.
Vacutainers — Complete Guide
A vacutainer (evacuated blood collection tube) is a sterile glass or plastic tube with a pre-measured vacuum that draws a fixed volume of blood. The coloured stopper identifies the additive inside and therefore what the tube is used for.
Tube Colours, Additives & Uses
| Stopper Colour | Additive | Mechanism | Samples Sent |
|---|---|---|---|
| Yellow (SPS) | Sodium polyanetholsulfonate (SPS) | Inhibits complement, phagocytes, and some antibiotics | Blood cultures (microbiology) |
| Yellow (ACD) | Acid citrate dextrose (ACD) | Citrate chelates calcium; dextrose preserves cells | Blood bank (group & screen, crossmatch), HLA typing, DNA studies, paternity testing |
| Light Blue | Sodium citrate (3.2%) | Chelates calcium → reversibly inhibits coagulation | Coagulation studies: PT, APTT, INR, fibrinogen, D-dimer, mixing studies |
| Red (plain glass) | None | Blood clots naturally → serum obtained after centrifugation | Serum chemistry, serology, drug levels, therapeutic drug monitoring |
| Gold / Red-Black (SST) | Clot activator + gel | Gel separates serum from clot after centrifugation | Serum chemistry (most routine biochemistry) |
| Green | Lithium heparin or sodium heparin | Inactivates thrombin and Factor Xa → prevents clotting; yields plasma | Plasma chemistry (urgent biochemistry, electrolytes, LFTs, renal function) |
| Lavender / Purple | EDTA (K₂ or K₃) | Chelates calcium → irreversibly prevents clotting | Haematology: FBC/CBC, blood film, HbA1c, ESR, reticulocytes, flow cytometry |
| Pink | EDTA | Same as lavender | Blood bank / transfusion (cross-match in some institutions) |
| Grey | Sodium fluoride + potassium oxalate | Fluoride inhibits glycolysis (enolase enzyme); oxalate chelates calcium | Glucose, lactate (prevents glycolysis-related glucose drop) |
| Royal Blue | None, or sodium heparin, or EDTA (trace-metal free) | Prevents trace metal contamination | Toxicology, heavy metal testing (zinc, lead, mercury, copper) |
| Black | Sodium citrate (3.8%) | Same as light blue but higher citrate concentration | ESR (Westergren method) |
Which Tube Must Be Kept Moving (Gently Inverted)?
All tubes containing anticoagulant additives must be gently inverted immediately after collection to mix the additive with blood and prevent clotting.
The tube that must be kept moving most critically is the EDTA tube (Lavender/Purple)
- EDTA binds calcium and prevents clotting, but only if thoroughly mixed with blood
- If not mixed immediately, micro-clots form → falsely low platelet count, falsely elevated WBC, and invalid CBC results
- Must be inverted 8 times gently (not shaken — shaking causes haemolysis)
Required inversions for each tube type (Tietz, Table 4-2):
| Tube | Inversions Required |
|---|---|
| Blood culture (yellow/SPS) | 8 |
| Royal blue (no additive) | 0 |
| Clear discard tube | 0 |
| Light blue (sodium citrate) | 3–4 |
| Gold/red SST (serum separator) | 5 |
| Red (serum, clot activator) | 5 |
| Green (heparin) | 8 |
| Lavender/Purple (EDTA) | 8 |
| Gray (fluoride/oxalate) | 8 |
The EDTA (lavender) tube is the classic answer — it must be gently rocked/inverted continuously (or at least 8 times immediately after filling) to prevent clotting, as its mechanism depends entirely on complete contact between EDTA and blood.
"It is critical that complete mixing of any additive with the blood collected be accomplished as quickly as possible. Completing a blood collection within 2 minutes of starting, and getting the tubes mixed correctly as soon as possible, helps to prevent clotting in anticoagulated tubes." — Tietz Textbook of Laboratory Medicine, 7th Edition
Order of Draw (When Multiple Tubes Are Collected)
The correct order prevents carryover contamination of additives between tubes:
| Order | Tube | Reason |
|---|---|---|
| 1st | Blood cultures (yellow/SPS) | Must be sterile; collected first |
| 2nd | Light blue (citrate) | Coagulation tests — must be filled to exactly 90% (1:9 ratio of anticoagulant:blood); EDTA contamination would prolong PT/APTT |
| 3rd | Red / Gold SST | Serum tubes |
| 4th | Green (heparin) | Plasma chemistry |
| 5th | Lavender (EDTA) | Haematology |
| Last | Grey (fluoride) | Glycolytic inhibitor |
General rule: No additive → mild anticoagulant → strong anticoagulant
Important note on the blue (citrate) tube: Must be filled to >90% of the marked line. If underfilled, the anticoagulant:blood ratio increases → artificially prolonged PT and APTT. In patients with haematocrit >55–60%, the amount of citrate must be reduced (less plasma available), otherwise coagulation times will be falsely prolonged.
Serum vs Plasma — Practical Points
| Serum (Red/Gold tube) | Plasma (Green/Lavender tube) | |
|---|---|---|
| How obtained | Blood clots → centrifuge → remove clot | Anticoagulant prevents clotting → centrifuge → remove cells |
| Contains fibrinogen? | No (consumed in clotting) | Yes |
| Total protein | Lower (fibrinogen removed) | Higher |
| Potassium | Higher (released from platelets during clotting) | Lower |
| Time needed | 30–60 min clotting time before centrifuge | Immediate after centrifuge |
| Required for | SPE, serology, TDM | Coagulation, CBC, urgent chemistry |
— Quick Compendium of Clinical Pathology, 5th ed.; Tietz Textbook of Laboratory Medicine, 7th ed.; Roberts and Hedges' Clinical Procedures in Emergency Medicine
Tell me about vacutainers and which sample to send in them and which vacutainor we have to keep moving in order to prevent clotting
I already covered this in my previous response — here's a concise recap:
Vacutainer Tubes — Quick Reference
| Stopper Colour | Additive | Samples Sent |
|---|---|---|
| Yellow (SPS) | Sodium polyanetholsulfonate | Blood cultures |
| Yellow (ACD) | Acid citrate dextrose | Blood bank, HLA typing, DNA studies |
| Light Blue | Sodium citrate | Coagulation: PT, APTT, INR, fibrinogen, D-dimer |
| Red (plain) | None | Serum chemistry, serology, drug levels |
| Gold / Red-Black (SST) | Clot activator + gel | Routine serum biochemistry |
| Green | Lithium/sodium heparin | Urgent plasma chemistry (electrolytes, LFTs, renal function) |
| Lavender / Purple | EDTA | FBC/CBC, blood film, HbA1c, reticulocytes, flow cytometry |
| Pink | EDTA | Blood bank / cross-match |
| Grey | Sodium fluoride + oxalate | Glucose, lactate |
| Royal Blue | Trace-metal free (no additive / heparin / EDTA) | Heavy metals, toxicology |
| Black | Sodium citrate (3.8%) | ESR (Westergren) |
Which Tube Must Be Kept Moving?
The EDTA tube (Lavender/Purple) is the classic answer — and all anticoagulant-containing tubes must be gently inverted immediately after filling.
Why?
EDTA works by chelating calcium to prevent clotting — but only if it makes complete contact with the blood. If not mixed right away, micro-clots form, giving:
- Falsely low platelet count
- Invalid CBC/FBC results
Required inversions (gentle, not shaking):
| Tube | Inversions |
|---|---|
| Blood culture (yellow) | 8 |
| Light blue (citrate) | 3–4 |
| Gold/SST | 5 |
| Red (clot activator) | 5 |
| Green (heparin) | 8 |
| Lavender/Purple (EDTA) | 8 |
| Grey (fluoride) | 8 |
The EDTA and heparin tubes require the most mixing (8 inversions). The citrate (blue) tube needs only 3–4 — over-mixing can activate platelets and affect coagulation results.
Order of Draw
Blood cultures → Blue → Red/Gold → Green → Lavender → Grey
(General rule: no additive → mild anticoagulant → strong anticoagulant)
The blue (citrate) tube must be filled to exactly 90% of its marked volume — underfilling falsely prolongs PT and APTT because the anticoagulant:blood ratio becomes too high.
— Tietz Textbook of Laboratory Medicine, 7th ed.; Quick Compendium of Clinical Pathology, 5th ed.
If we invert red vial does it affect the sample
Great practical question. The answer depends on which type of red tube you're using:
Plain Red Tube (No Additive — Glass)
Inversions: 0
Inverting this tube has no beneficial effect and could theoretically cause minor turbulence, but it does not significantly harm the sample. The blood will still clot normally on its own.
- No additive to mix in → no reason to invert
- The clot forms naturally within 30–60 minutes
- Gentle inversion won't prevent or accelerate clotting in any meaningful way
Verdict: Inverting a plain red tube is essentially harmless but pointless.
Red/Gold SST Tube (Serum Separator Tube — with Clot Activator + Gel)
Inversions: 5 (required)
This tube does contain a clot activator (silica particles or thrombin coating on the wall). Here, inversion is required — the 5 gentle inversions ensure the clot activator contacts all the blood, allowing a complete and rapid clot to form before centrifugation.
- If you don't invert → incomplete clotting → fibrin strands remain → interfere with gel separation → invalid results
- If you over-invert or shake → haemolysis → falsely elevated potassium, LDH, AST
Key Practical Points
| Action | Plain Red (no additive) | Red/Gold SST |
|---|---|---|
| Invert 5× gently | Harmless, no benefit | Required |
| Don't invert at all | Fine — clots naturally | Risk of incomplete clotting |
| Shake vigorously | Causes haemolysis | Causes haemolysis |
Bottom Line
- Plain red glass tube: inverting it won't damage the sample, but it serves no purpose
- SST (gold or red-black): must be inverted exactly 5 times — not more, not less
- Vigorous shaking of any tube is the real danger — it causes haemolysis regardless of tube type
— Tietz Textbook of Laboratory Medicine, 7th ed.; Quick Compendium of Clinical Pathology, 5th ed.
This is a shared conversation. Sign in to Orris to start your own chat.