Hb-8.6, creat- 0.5, ESR-95, Urine PCR-12, urine alb-3+, CECT abd- bilateral pleural effusion, L1 wedge compression fracture, renal biopsy- membranous nephropathy
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Here is a comprehensive analysis of this clinical case:
Clinical Case Analysis
Patient Summary
| Parameter | Value | Interpretation |
|---|---|---|
| Hemoglobin | 8.6 g/dL | Normocytic anemia |
| Creatinine | 0.5 mg/dL | Low — preserved/hyperfiltration state |
| ESR | 95 mm/hr | Markedly elevated |
| Urine PCR | 12 (g/g or mg/mg) | Nephrotic-range proteinuria |
| Urine albumin | 3+ | Heavy albuminuria |
| CECT Abd | Bilateral pleural effusion, L1 wedge compression | Fluid overload; vertebral lesion |
| Renal biopsy | Membranous nephropathy | Confirmed glomerulopathy |
Diagnosis: Membranous Nephropathy (MN) with Nephrotic Syndrome
The clinical picture is classic: heavy proteinuria (PCR 12), hypoalbuminemia (implied by 3+ urine albumin and pleural effusions), normal-to-low creatinine, and biopsy-confirmed membranous nephropathy.
Biopsy Findings in MN
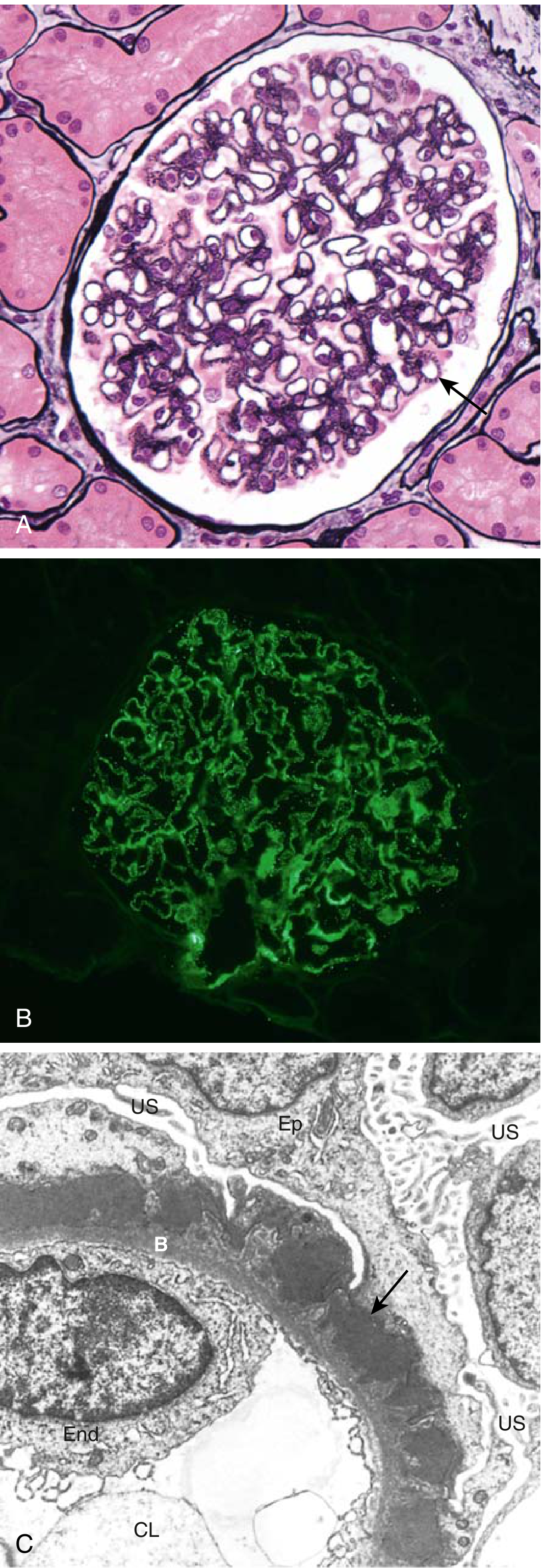
Fig. 12.6 — Membranous nephropathy. (A) Diffuse GBM thickening without cellular proliferation (silver stain). (B) Granular IgG deposits by immunofluorescence. (C) Subepithelial electron-dense deposits (arrow) with podocyte foot-process effacement.
Histologically, MN shows:
- Light microscopy: Diffuse capillary wall thickening without hypercellularity
- Immunofluorescence: Granular IgG + complement along GBM
- Electron microscopy: Subepithelial deposits → "spike and dome" pattern
— Robbins & Kumar Basic Pathology, p. 506
Critical Flag: The L1 Wedge Compression Fracture
This finding demands a thorough secondary cause workup. The combination of:
- Markedly elevated ESR (95)
- Vertebral compression fracture (L1)
- Nephrotic syndrome
...raises serious concern for an underlying malignancy causing secondary MN.
Malignancies account for 6–22% of secondary MN cases. These include carcinoma of the bronchus, breast, colon, stomach, ovary, kidney, pancreas, and prostate, as well as Hodgkin disease. — Brenner and Rector's The Kidney
In cancer-associated MN, the kidney biopsy may show increased inflammatory infiltrate (>8 cells/glomerulus) in 92% of cases vs. 25% in idiopathic MN. IgG subclass analysis shows IgG1/IgG2 predominance (versus IgG4 in primary/idiopathic MN).
Key antibody-guided distinction:
- Anti-PLA2R positive → primary (idiopathic) MN
- Anti-PLA2R negative + THSD7A positive → strongly suspect malignancy. In one cohort, 8 of 40 THSD7A-positive MN patients developed malignancy within a median of 3 months of diagnosis.
Differential Diagnosis for Secondary MN
| Cause | Clues in this patient |
|---|---|
| Malignancy | High ESR, vertebral compression fracture (lytic/pathological?) |
| SLE / Autoimmune | Elevated ESR, anemia |
| Chronic HBV/HCV | Need serological testing |
| Drugs/Toxins | Drug history required |
| Syphilis / Malaria | Endemic area history |
Workup to Perform
- Anti-PLA2R antibody (serum) — differentiates primary from secondary MN
- Anti-THSD7A antibody — if PLA2R negative
- ANA, anti-dsDNA, complement (C3/C4) — exclude SLE (class V lupus nephritis)
- HBsAg, anti-HCV, VDRL/RPR — infectious causes
- Serum protein electrophoresis / SIFE — paraproteinemia, myeloma
- Bone scan / MRI spine — characterize L1 lesion (lytic vs. osteoporotic vs. pathological fracture)
- Low-dose CT chest — lung malignancy
- Age-appropriate cancer screening — colonoscopy, PSA, mammography
- Serum albumin — quantify nephrotic state and thrombotic risk stratification
Management
Non-Immunosuppressive (All Patients)
- RAAS blockade (ACE inhibitor or ARB): reduces proteinuria and BP — first-line regardless of BP
- Sodium restriction <2 g/day
- Statins: for dyslipidemia (elevated total cholesterol, LDL in nephrotic syndrome)
- Anticoagulation: Albumin <2.8 g/dL is the strongest predictor of venous thromboembolism. With albumin 2–3 g/L → aspirin 81 mg; albumin <2 g/L or PCR >10 g/g → consider prophylactic anticoagulation
- Loop diuretics: for edema and pleural effusions
— National Kidneys Foundation Primer on Kidney Diseases, 8e; Comprehensive Clinical Nephrology, 7th ed.
Risk Stratification for Immunosuppression
| Risk Category | Criteria | Approach |
|---|---|---|
| Low risk | Proteinuria ≤4 g/day, normal eGFR | Conservative; no immunosuppression |
| Moderate risk | Proteinuria 4–8 g/day, stable eGFR | Rituximab (preferred) or alkylating agent + steroid |
| High risk | Proteinuria >8 g/day or declining eGFR | Cyclophosphamide + glucocorticoids (Ponticelli protocol) |
This patient has PCR of 12 → high-risk category.
Immunosuppressive Options
-
Ponticelli Protocol (cyclophosphamide + steroids): IV methylprednisolone 1 g on days 1–3 of months 1, 3, 5; oral methylprednisolone on remaining days; alternating with oral cyclophosphamide 2.5 mg/kg/day in months 2, 4, 6. Superior long-term outcomes; cyclophosphamide preferred over chlorambucil.
-
Rituximab (anti-CD20): Now the favored first-line agent in moderate-risk patients and many high-risk patients. The MENTOR trial showed rituximab superior to cyclosporine for sustained remission. The RI-CYCLO trial directly compared rituximab to the cyclophosphamide regimen — evidence continues to evolve.
However, if secondary MN due to malignancy is confirmed, immunosuppression should be withheld until the underlying malignancy is treated. Tumor resection/remission can lead to complete remission of nephrotic syndrome.
Summary
This patient most likely has secondary membranous nephropathy given the high ESR, vertebral compression fracture, and nephrotic-range proteinuria. A malignancy workup is urgent and mandatory before initiating immunosuppression. Anti-PLA2R testing is the pivotal next step: a negative result with IgG1/IgG2 predominance on biopsy greatly increases suspicion for cancer-associated MN.
What are the malignancy workup to be done
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Here is a focused, textbook-grounded malignancy workup for this patient with biopsy-confirmed membranous nephropathy:
Malignancy Workup in Membranous Nephropathy
Why This Patient is High-Risk
Based on Table 42.7 (Brenner & Rector's The Kidney):
| Risk Feature | This Patient |
|---|---|
| Age >65 years | Not specified — relevant if elderly |
| Smoking history >20 pack-years | To be elicited |
| Family history of malignancy | To be elicited |
| History of thromboembolism | Possible (pleural effusion) |
| Anti-PLA2R absent | Must be checked |
| IgG1/IgG2 predominance on biopsy | Must be reviewed |
| >8 inflammatory cells/glomerulus | Must be reviewed |
Additionally: L1 wedge compression fracture + ESR 95 — both strongly suggest a lytic/metastatic lesion or myeloma rather than an osteoporotic collapse.
Workup — Organized by System
1. Serology (First-Line — Mandatory)
| Test | Purpose |
|---|---|
| Serum anti-PLA2R antibody | Negative → favors secondary/malignancy-associated MN; positive → favors primary MN |
| Serum anti-THSD7A antibody | If PLA2R negative; 7 of 25 THSD7A-positive MN patients had malignancy |
| Serum protein electrophoresis (SPEP) | Detect M-band (myeloma, paraproteinemia) |
| Serum immunofixation electrophoresis (SIFE) | More sensitive for monoclonal protein |
| Urine immunofixation (UIFE) | Bence Jones proteins (myeloma) |
| Serum free light chains (κ/λ ratio) | Multiple myeloma workup |
| LDH, uric acid, β2-microglobulin | Lymphoma/hematologic malignancy markers |
The L1 compression fracture combined with high ESR and nephrotic syndrome mandates myeloma exclusion as a priority.
2. Imaging
| Test | Target |
|---|---|
| Low-dose CT chest | Lung carcinoma (most common cancer in MN — especially smokers) |
| CT abdomen & pelvis (already done — review for masses) | Renal cell, gastric, pancreatic, colorectal, ovarian, prostate |
| MRI spine (L1 lesion) | Characterize: lytic metastasis vs. osteoporotic fracture vs. myeloma deposit |
| Skeletal survey / Whole-body low-dose CT | If myeloma suspected — bony lesions |
| PET-CT | If occult malignancy strongly suspected after initial workup is negative |
3. Gender- and Age-Appropriate Cancer Screening
| Test | Cancer Screened |
|---|---|
| PSA (males) | Prostate carcinoma |
| Mammography (females) | Breast carcinoma |
| Colonoscopy / Fecal occult blood test | Colorectal carcinoma |
| Pap smear / TVS (females) | Cervical / ovarian / endometrial |
| CECT abdomen (review) | Renal cell carcinoma, hepatocellular carcinoma |
4. Tumor Markers (Targeted)
| Marker | Cancer |
|---|---|
| CEA | Colorectal, gastric, lung |
| CA 19-9 | Pancreatic, gastric |
| CA-125 | Ovarian |
| AFP | Hepatocellular carcinoma |
| PSA | Prostate |
| β-hCG | Testicular |
| CYFRA 21-1 / NSE | Lung (squamous/SCLC) |
5. Hematologic Malignancy Workup
Given ESR 95 + vertebral compression + anemia (Hb 8.6):
| Test | Purpose |
|---|---|
| Bone marrow biopsy | If myeloma/lymphoma suspected |
| Peripheral blood smear | Rouleaux (myeloma), blast cells (leukemia) |
| Serum calcium | Hypercalcemia of malignancy |
| Serum LDH | Lymphoma |
| CT neck/chest/abdomen/pelvis | Lymphadenopathy (lymphoma) |
6. Biopsy Re-review (Pathology)
Ask pathologist to report:
- IgG subclass staining — IgG1/IgG2 (malignancy) vs. IgG4 (primary MN)
- Inflammatory cell count per glomerulus — >8 cells favors malignancy-associated MN (92% sensitivity)
- Anti-PLA2R staining on tissue — negative staining supports secondary cause
- NELL1 staining — newer antigen identified in malignancy-associated MN
Key Principle
"A search for malignancy should be undertaken in older patients with newly diagnosed MN after excluding other secondary causes. The presence of anti-THSD7A antibody, absence of anti-PLA2R antibody, and/or predominant IgG1 and IgG2 deposits are other reasons to exclude malignancies thoroughly." — Brenner and Rector's The Kidney
"In those patients in whom no malignancy is found on initial screening, follow-up vigilance for possible cancer should be maintained because cancers have been detected up to 5 years after the initial diagnosis of MN." — Brenner and Rector's The Kidney
Priority Order for This Patient
Given the L1 wedge fracture + ESR 95 + Hb 8.6 + nephrotic MN:
- Myeloma screen first — SPEP, SIFE, UIFE, serum free light chains, serum calcium, skeletal survey/MRI spine
- Anti-PLA2R + anti-THSD7A — serologic pivot point
- Low-dose CT chest — lung malignancy
- PSA/mammography — gender-appropriate
- Colonoscopy / tumor markers — if above negative
- Bone marrow biopsy — if paraprotein found or myeloma suspected
70 yr male
Finding Sources
Finding Sources
Reading File
Reading File
With age confirmed as 70 years male, here is the refined, focused workup:
Malignancy Workup — 70-Year-Old Male with MN
Why Age Matters Here
"In patients older than 60 years, MN is associated with a malignancy in 20–30% of patients." — Brenner and Rector's The Kidney
"The standardized incidence ratio of cancer in male patients with MN was 9.8 compared to the general population. The most common malignancies were lung and prostate. In almost half the patients, the tumor was asymptomatic and detected only because of diagnostic workup for MN." — Brenner and Rector's The Kidney
"Patients with MN who developed cancer were significantly older (65 vs. 52 years; p<0.001)."
This patient is high-risk by age alone — workup is mandatory and thorough.
Priority Workup for This 70M
Step 1 — Serology (Pivot Tests)
| Test | Rationale |
|---|---|
| Serum anti-PLA2R | Negative → strongly favors secondary/malignancy-associated MN |
| Serum anti-THSD7A | If PLA2R negative; 7/25 THSD7A+ MN patients had cancer |
| Serum + urine immunofixation (SIFE/UIFE) | Myeloma — critical given L1 vertebral compression + ESR 95 + anemia in a 70M |
| Serum free light chains (κ/λ) | Myeloma / AL amyloid |
| SPEP | M-band detection |
| Serum calcium | Hypercalcemia of malignancy / myeloma |
| LDH, β2-microglobulin | Lymphoma, myeloma tumor burden |
Step 2 — Gender/Age-Specific in a 70-Year-Old Male
| Test | Cancer Targeted | Priority |
|---|---|---|
| PSA (total + free) | Prostate carcinoma (most common cancer in elderly males with MN) | ⭐ HIGH |
| Low-dose CT chest | Lung carcinoma (commonest cancer associated with MN; especially if smoker) | ⭐ HIGH |
| CECT abdomen (already done — re-review) | Renal cell carcinoma, gastric, pancreatic, hepatocellular | ⭐ HIGH |
| Upper GI endoscopy / CECT stomach | Gastric carcinoma (lung, gastric, renal, prostate = top 4 in MN) | MODERATE |
| Colonoscopy or CT colonography | Colorectal carcinoma | MODERATE |
Step 3 — Vertebral Lesion Workup (L1 Compression)
In a 70-year-old male with ESR 95, Hb 8.6, and nephrotic syndrome, this fracture is potentially pathological — not osteoporotic.
| Test | Purpose |
|---|---|
| MRI whole spine | Characterize L1 lesion: lytic/infiltrative = myeloma/metastasis vs. benign |
| Whole-body low-dose CT or Skeletal survey | Bony metastases / myeloma lesions |
| PET-CT | If MRI inconclusive or occult primary suspected |
| Bone marrow biopsy | If myeloma strongly suspected (paraprotein detected) |
Step 4 — Targeted Tumor Markers in a 70M
| Marker | Cancer |
|---|---|
| PSA | Prostate |
| CEA | Colorectal, gastric, lung |
| CA 19-9 | Pancreatic, gastric |
| AFP | Hepatocellular carcinoma |
| NSE / CYFRA 21-1 | SCLC / squamous lung |
| LDH + β2-microglobulin | Lymphoma / myeloma |
Step 5 — Biopsy Pathology Re-review
Request the pathologist to specifically report:
- IgG subclass — IgG1/IgG2 (malignancy) vs. IgG4 (primary MN)
- Inflammatory cell count/glomerulus — >8 cells in 92% of cancer-associated MN
- PLA2R tissue staining — negative = secondary
- NELL1 antigen (newer marker linked to malignancy-associated MN)
Summary Algorithm for This Patient
70M + MN + L1 compression + ESR 95 + Hb 8.6
│
▼
Anti-PLA2R + THSD7A serology
│
┌────┴──────┐
Negative Positive (IgG4, PLA2R+)
│ │
Malignancy Primary MN
workup URGENT conservative Rx
│
▼
Myeloma screen (SIFE, free light chains, MRI spine)
+
PSA + CT chest (lung)
+
CECT abdomen review (renal cell, gastric, pancreatic)
+
Colonoscopy / upper GI scope
80% of cancers in MN are detected before or at the time of renal biopsy — this patient is already at that window. The L1 fracture + anemia + elevated ESR constellation in a 70M makes multiple myeloma and metastatic prostate/lung carcinoma the top differential diagnoses to actively exclude.
— Brenner and Rector's The Kidney, 2-Volume Set
This is a shared conversation. Sign in to Orris to start your own chat.