Abdominal hysterectomy in detail
abdominal hysterectomy surgical technique steps procedure
abdominal hysterectomy anatomy uterus surgical approach
Abdominal Hysterectomy — Comprehensive Overview
Definition & Types
| Type | What is Removed |
|---|---|
| Total Abdominal Hysterectomy (TAH) | Uterus + cervix |
| Subtotal (Supracervical) Hysterectomy | Uterus only, cervix preserved |
| TAH + Bilateral Salpingo-Oophorectomy (TAH-BSO) | Uterus + cervix + both tubes + both ovaries |
| Radical (Wertheim's) Hysterectomy | Uterus + cervix + parametrium + upper vaginal cuff + pelvic lymph nodes |
Indications
- Uterine leiomyomas (fibroids) — most common indication
- Dysfunctional / abnormal uterine bleeding refractory to medical therapy
- Adenomyosis
- Uterine prolapse
- Chronic pelvic pain (endometriosis, pelvic inflammatory disease)
- Obstetric emergencies: uterine atony, rupture, placenta accreta spectrum
- Endometrial carcinoma
- Cervical carcinoma (early stage — Wertheim's)
- Ovarian carcinoma (as part of debulking)
- Gestational trophoblastic neoplasia
Contraindications / Relative Cautions
- Desire for future fertility (relative — must counsel)
- Severe cardiorespiratory comorbidity (anaesthetic risk)
- Coagulopathy (must correct pre-operatively)
- Active pelvic infection (delay if possible)
- Very large uterus or dense adhesions (may influence route choice)
Pre-operative Assessment & Preparation
Workup
- Full history and examination; pelvic exam + bimanual
- Pap smear / cervical cytology (if not recent)
- Endometrial biopsy where indicated
- Ultrasound pelvis (size, vascularity, adnexae)
- MRI pelvis for suspected malignancy, fibroids, endometriosis
- CBC, coagulation profile, blood group & crossmatch (2 units PRBC)
- Renal function, LFT, ECG, CXR (per anaesthetic protocol)
- Ureteral imaging if anatomy likely distorted
Counselling
- Route of hysterectomy and reason for abdominal approach
- Permanent loss of fertility
- Risks: bleeding, infection, urinary/bowel/vascular injury, DVT/PE, anaesthetic risk
- Ovarian conservation vs. removal (discuss hormone implications)
- Recovery time (~6 weeks)
- Consent documentation
Pre-operative Measures
- Bowel preparation (selective use; not routine per ERAS)
- Antibiotic prophylaxis: Cefazolin 2 g IV (or clindamycin + gentamicin if penicillin allergy) within 60 min of incision
- VTE prophylaxis: compression stockings + LMWH (e.g., enoxaparin 40 mg SC)
- Urinary catheterisation (Foley, 14–16 Fr) in theatre
- Positioning: supine with slight Trendelenburg
Anaesthesia
- General anaesthesia (most common) ± epidural for post-op analgesia
- Spinal/regional in selected cases
- Nasogastric tube: selective use
Surgical Anatomy (Key Relations)
| Structure | Relevance |
|---|---|
| Ureters | Cross under uterine artery ("water flows under the bridge") at the level of the internal os — most common site of ureteral injury |
| Bladder | Adherent anteriorly to lower uterine segment / cervix — must be dissected off |
| Rectum | Posterior to cervix / posterior vaginal fornix |
| Uterine artery | Branch of internal iliac; enters broad ligament at level of cervix |
| Ovarian ligament & tube | Part of broad ligament; ligated if conserving adnexae |
| Infundibulopelvic (IP) ligament | Contains ovarian vessels; ligated if removing ovaries |
| Round ligament | First structure ligated; provides entry into broad ligament |
| Cardinal ligament (Mackenrodt's) | Main uterine support; clamped close to cervix |
| Uterosacral ligaments | Posterior support; identify to avoid rectal injury |
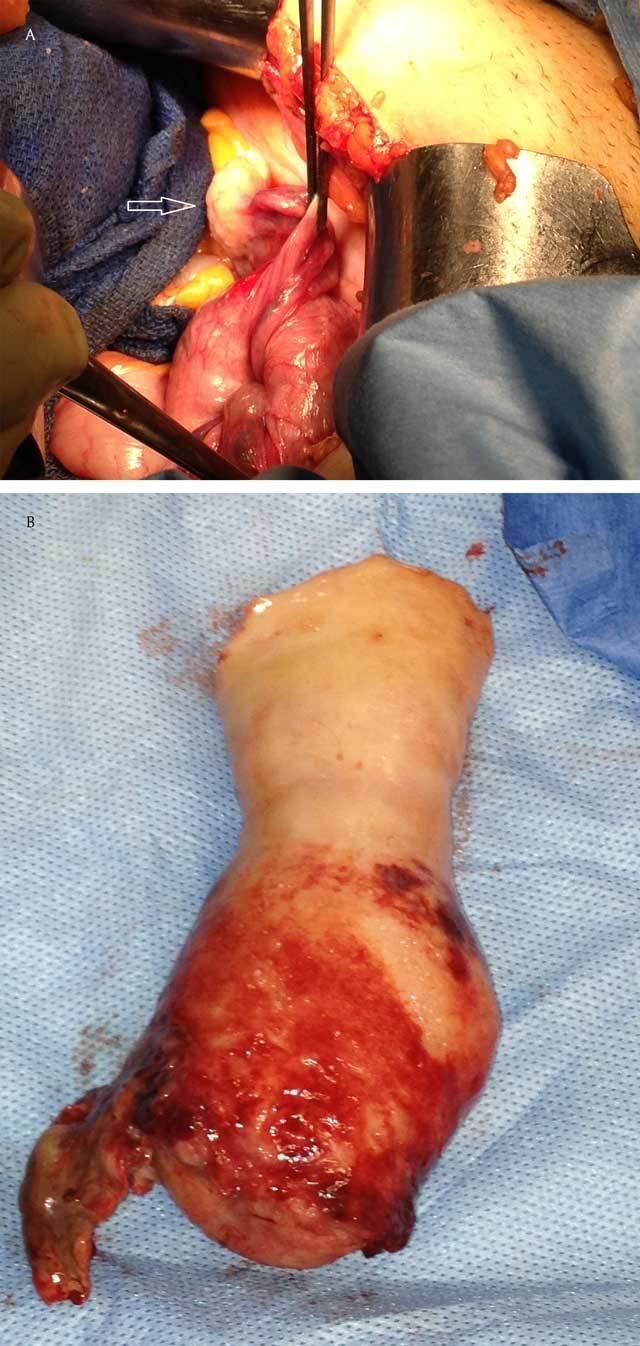
Incision
| Incision | Use Case |
|---|---|
| Pfannenstiel (transverse, 2 cm above pubic symphysis) | Most common; cosmetically superior; adequate access for most benign cases |
| Midline subumbilical (vertical) | Preferred for malignancy, very large uterus, obesity, re-operation, emergency |
| Maylard / Cherney | Modified transverse; better lateral access if needed |
Step-by-Step Surgical Technique (Total Abdominal Hysterectomy)
Step 1 — Entry & Exploration
- Make Pfannenstiel or midline incision through skin, subcutaneous fat.
- Incise anterior rectus sheath; separate rectus muscles (Pfannenstiel) or divide linea alba (midline).
- Open peritoneum carefully to avoid bowel/bladder injury.
- Explore abdomen: note uterine size, consistency, adhesions, adnexae, lymph nodes (in malignancy).
- Pack bowel cephalad with moist laparotomy packs; place self-retaining retractor (e.g., Balfour or O'Sullivan-O'Connor).
Step 2 — Uterine Elevation & Round Ligaments
- Grasp uterine fundus with Bonney's forceps (or similar clamps) to elevate and antiflex.
- Clamp, cut, and ligate the round ligaments bilaterally — this opens the broad ligament leaves and provides access to the retroperitoneal space.
Step 3 — Opening the Broad Ligament / Retroperitoneal Space
- Incise the anterior leaf of the broad ligament extending towards the bladder reflection.
- Identify and palpate the ureter in the retroperitoneal space (runs along the medial leaf of the broad ligament).
Step 4 — Adnexal Decision
- Clamp, cut, and ligate the utero-ovarian ligament and fallopian tube close to the uterus (using Heaney or Kocher clamps, 3-clamp technique).
- Suture-ligate the pedicle (0-Vicryl or equivalent).
- Identify the infundibulopelvic (IP) ligament containing the ovarian vessels.
- Create a window in the avascular area of the broad ligament lateral to the IP ligament.
- Clamp, cut, and doubly ligate the IP ligament (3-clamp technique; suture ligate to prevent slippage of ovarian vessels).
Step 5 — Bladder Flap
- Identify the vesicouterine peritoneal fold (bladder reflection) anteriorly.
- Incise the peritoneum transversely.
- Use sharp and blunt dissection to push the bladder inferiorly off the lower uterine segment and cervix.
- This is the most critical step to prevent bladder injury and to expose the uterine vessels.
Step 6 — Uterine Vessels (Uterine Artery Ligation)
- Skeletonise the uterine vessels at the level of the internal os by pushing the bladder down.
- Place Heaney clamps perpendicular to the uterus at the level of the internal cervical os (one clamp on each side).
- Cut and suture-ligate the uterine artery and vein pedicle (transfixion suture — 0 or 1 Vicryl).
- This is the most haemorrhagic step; confirm haemostasis.
- The ureter lies ~1.5 cm lateral at this point — the "water under the bridge" principle.
Step 7 — Cardinal and Uterosacral Ligaments
- Progressively clamp, cut, and ligate the cardinal ligament (Mackenrodt's ligament) in sequential bites down to the level of the cervicovaginal junction.
- Similarly ligate the uterosacral ligaments posteriorly (stay close to cervix to avoid rectal injury).
Step 8 — Vaginal Vault Opening (Total Hysterectomy)
- Confirm position of cervicovaginal junction by palpating the Foley balloon.
- Place straight clamps (Kocher or Heaney) on the lateral vaginal angles.
- Use a scalpel or heavy scissors to circumscribe and incise the vagina around the cervix.
- The uterus and cervix are removed en bloc.
- Send specimen for histopathology.
Step 9 — Vaginal Vault Closure
- Achieve haemostasis at the vault angles (angle sutures are critical — the vaginal angle suture includes the uterosacral and cardinal ligament remnants to support the vault and prevent vault prolapse).
- Close the vaginal vault with interrupted or continuous locking sutures (0 or 1 Vicryl).
- The vault may be left open (for drainage) or closed — both are acceptable; current evidence favours closure.
- Some surgeons reattach uterosacral ligaments to the vault angles (McCall culdoplasty) to reduce future vault prolapse.
Step 10 — Peritonealisation & Abdominal Closure
- Peritonealisation (re-suturing the pelvic peritoneum) is not routinely required — evidence shows no benefit and may increase adhesion formation.
- Remove packs; confirm haemostasis; count swabs and instruments (WHO surgical safety checklist).
- Close the abdomen in layers:
- Peritoneum: optional (0 Vicryl continuous)
- Rectus sheath / linea alba: 1 or loop-PDS continuous (mass closure for midline)
- Subcutaneous fat: if >2 cm, approximate with 2-0 Vicryl
- Skin: subcuticular Monocryl or staples
Haemostasis Technique — 3-Clamp Method
- Clamp 1 — distal (specimen side)
- Clamp 2 — proximal (patient side, haemostatic)
- Clamp 3 — safety clamp (most proximal)
- Cut between Clamp 1 and Clamp 2
- Ligate pedicle under Clamp 2 with transfixion suture
- Remove Clamp 3
Complications
Intra-operative
| Complication | Notes |
|---|---|
| Haemorrhage | Uterine/ovarian vessels; internal iliac injury |
| Ureteral injury | Kinking, ligation, transection — at uterine artery level or IP ligament; incidence ~0.5–1% |
| Bladder injury | During bladder flap dissection; higher risk with previous caesarean sections; incidence ~1–2% |
| Bowel injury | Small/large bowel; higher risk with adhesions, endometriosis |
| Vascular injury | Iliac vessels |
Post-operative
| Complication | Notes |
|---|---|
| Primary haemorrhage / vault haematoma | Within 24–48 h |
| Urinary retention | Remove catheter at 24–48 h |
| Urinary tract infection | Common; treat with antibiotics |
| Wound infection / dehiscence | 3–5% |
| Vault cellulitis / infection | Pelvic infection; treat with IV antibiotics |
| DVT / Pulmonary Embolism | VTE prophylaxis mandatory; LMWH for 28 days in malignancy |
| Ureteral fistula / vesicovaginal fistula | Delayed presentation (10–14 days post-op) |
| Ovarian remnant syndrome | If oophorectomy attempted but remnant left |
| Vault prolapse | Late complication; vault support sutures help prevent |
| Psychological / sexual effects | Counsel pre-operatively |
| Bowel obstruction | Adhesions |
Wertheim's Radical Hysterectomy (for Cervical Cancer)
- Pelvic lymphadenectomy (obturator, external iliac, internal iliac nodes)
- Resection of parametrium (medial half)
- Removal of upper 2–3 cm of vagina
- Complete ureteral dissection ("unroofing" the ureter from the ureteric tunnel)
- Significantly higher risk of ureteral injury, bladder dysfunction, lymphoedema
ERAS (Enhanced Recovery After Surgery) Protocols
| Element | Recommendation |
|---|---|
| Pre-op carbohydrate loading | Up to 2 h before surgery |
| Avoid prolonged fasting | Clear fluids until 2 h, solids until 6 h |
| Minimise opioids | Multimodal analgesia (NSAIDs, paracetamol, TAP block) |
| Early mobilisation | Day 0 or Day 1 |
| Early oral intake | Day 0 or Day 1 |
| Catheter removal | 24–48 h post-op |
| Avoid routine drains | Not recommended unless specific indication |
| Discharge | Day 2–3 (benign cases) |
Post-operative Care
- Analgesia: IV paracetamol + NSAIDs (if no contraindication) ± patient-controlled analgesia or epidural; reduce opioids
- Fluids: IV fluids until tolerating orally; transition to oral early
- Ambulation: Encourage early mobilisation (Day 1)
- Diet: Light diet when bowel sounds return; usually Day 1–2
- Catheter: Remove at 24–48 h (after vault closure confirmed)
- Wound care: Inspect at 24–48 h
- VTE: Continue LMWH (enoxaparin) for 7–28 days depending on indication
- Discharge: Typically Day 2–4 (benign); longer for malignancy or complications
- Follow-up: 4–6 weeks post-op; pathology review
- Restrictions: No heavy lifting, intercourse, or strenuous activity for 6 weeks
Comparison of Hysterectomy Routes
| Feature | Abdominal (TAH) | Vaginal | Laparoscopic (TLH) | Robotic |
|---|---|---|---|---|
| Incision | Laparotomy | None | 3–4 ports | 3–4 ports |
| Hospital stay | 3–5 days | 1–3 days | 1–2 days | 1–2 days |
| Recovery | 4–6 weeks | 2–4 weeks | 2–3 weeks | 2–3 weeks |
| Blood loss | More | Less | Less | Least |
| Access to upper abdomen | Excellent | Poor | Good | Good |
| Cost | Lower | Lowest | Higher | Highest |
| Best for | Large uterus, malignancy, adhesions | Prolapse, small uterus | Most benign cases | Complex/obese |
Key Teaching Points
- "Water under the bridge" — uterine artery crosses over the ureter at the internal os; always identify the ureter before clamping.
- Adequate bladder mobilisation is the cornerstone of safe pelvic dissection.
- Angle sutures at the vault corners must incorporate the cardinal and uterosacral ligament stumps to prevent future vault prolapse.
- The 3-clamp technique ensures no pedicle is ever ligated without a safety clamp.
- Intraoperative ureteral injury — if suspected, give IV indigo carmine or methylene blue and check for efflux from ureteral orifices via cystoscopy.
- Retroperitoneal dissection and ureteral identification is mandatory whenever anatomy is distorted (fibroids, endometriosis, malignancy, adhesions).
Maternal changes in pregnancy in detail
maternal physiological changes pregnancy cardiovascular respiratory
maternal changes pregnancy renal hematological endocrine gastrointestinal
pregnancy cardiac output blood volume hemodynamic changes trimester
pregnancy weight gain uterus skin musculoskeletal anatomical changes
Maternal Changes in Pregnancy — Comprehensive Overview
1. Cardiovascular System
Blood Volume
| Parameter | Change | Magnitude | Timing |
|---|---|---|---|
| Total blood volume | ↑ | +40–50% (1200–1600 mL) | Begins wk 6, peaks 30–34 wks |
| Plasma volume | ↑ | +50% (~1200 mL) | |
| Red cell mass | ↑ | +20–30% (~300 mL) | |
| Net effect | Haemodilution | "Physiological anaemia of pregnancy" |
- Plasma volume expands disproportionately more than red cell mass → haematocrit falls (normal in pregnancy: Hb ≥10.5 g/dL; Hct ~32–34%)
- Purpose: meets metabolic demands, reduces blood viscosity, provides buffer against delivery blood loss
Cardiac Output (CO)
- CO increases 30–50% above pre-pregnancy values by mid-second trimester
- Begins at 6 weeks gestation, peaks at 20–28 weeks (CO ~6–7 L/min vs. baseline ~4.5 L/min)
- Mechanism: ↑ stroke volume (early) + ↑ heart rate (later; +15–20 bpm)
- Position effect: CO decreases 25–30% in the supine position (aortocaval compression by gravid uterus — supine hypotensive syndrome)
- During labour: CO rises further (+30% in first stage, +45% in second stage, +65% immediately post-delivery due to autotransfusion)
Heart Rate
- Rests +10–20 bpm above baseline by third trimester
- Palpitations are common and physiological
Blood Pressure
| Phase | Systolic | Diastolic |
|---|---|---|
| First trimester | Slightly ↓ | ↓ 5–10 mmHg |
| Second trimester (nadir ~20 wks) | Lowest | Lowest (↓10–15 mmHg) |
| Third trimester | Returns toward normal | Returns toward normal |
- Fall caused by: progesterone-mediated peripheral vasodilatation + relaxin + formation of low-resistance uteroplacental circulation
- SVR falls by ~30–35%
Structural/Cardiac Changes
- Heart displaced upward and laterally (by elevated diaphragm) → apparent cardiomegaly on CXR
- Mild eccentric hypertrophy and increased ventricular wall thickness
- Physiological systolic flow murmurs in 90% (due to high CO + low viscosity); Grade I–II over left sternal border — normal
- S3 gallop may be heard — normal in pregnancy
- ECG: left axis deviation, T-wave flattening/inversion in lead III, ST changes — all physiological
2. Respiratory System
Anatomical Changes
- Diaphragm rises 4 cm due to enlarging uterus
- Thoracic cage flares: subcostal angle increases from ~68° to ~103°; chest circumference increases 6 cm
- Overall thoracic volume relatively preserved despite diaphragmatic elevation
Lung Volumes
| Parameter | Change | Amount |
|---|---|---|
| Tidal Volume (TV) | ↑↑ | +40–45% (500 → 700 mL) |
| Respiratory Rate | Minimal ↑ or unchanged | +1–2 breaths/min |
| Minute Ventilation | ↑↑ | +40–50% |
| Functional Residual Capacity (FRC) | ↓ | −20% |
| Expiratory Reserve Volume (ERV) | ↓ | −20% |
| Residual Volume (RV) | ↓ | −20% |
| Total Lung Capacity (TLC) | ↓ slightly | −5% |
| Inspiratory Capacity | ↑ | +5% |
| FEV₁ / VC ratio | Unchanged | — |
Blood Gas Changes
- Chronic respiratory alkalosis (compensated):
- PaCO₂ falls: 40 → 28–32 mmHg (progesterone stimulates respiratory centre)
- pH: 7.40–7.45
- Renal compensation: HCO₃⁻ falls to 18–22 mEq/L
- PaO₂ rises slightly: ~105 mmHg
Clinical Effects
- Dyspnoea of pregnancy in 60–70% (even at rest) — due to progesterone-driven hyperventilation; NOT pathological
- Increased risk of hypoxia during apnoea/intubation (reduced FRC = reduced O₂ reserve)
- Nasal congestion, rhinitis — due to oestrogen-induced mucosal hyperaemia
- Voice changes (oedema of larynx)
3. Haematological System
Red Cell Changes
- Physiological anaemia: Hb may fall to 10.5–11 g/dL (WHO threshold for anaemia in pregnancy: Hb <11 g/dL first/third trimester, <10.5 g/dL second trimester)
- MCV rises slightly (macrocytosis from folate demands)
- ESR rises significantly — unreliable marker of infection in pregnancy
White Cell Changes
- Leucocytosis is normal: WBC 9,000–12,000/μL; during labour up to 25,000/μL
- Predominantly neutrophilia; lymphocyte count relatively unchanged
Platelets
- Gestational thrombocytopaenia: mild fall in platelet count (~10%); counts >100,000 are usually benign
- Platelet function enhanced (hypercoagulable state)
Coagulation — Hypercoagulable State
| Factor | Change |
|---|---|
| Fibrinogen | ↑↑ 50–100% (most important) — 400–600 mg/dL |
| Factors VII, VIII, X, XII | ↑ |
| Protein S | ↓ (anticoagulant — reduced) |
| Protein C | Unchanged or mild ↑ |
| Antithrombin III | Slightly ↓ |
| Plasminogen activator inhibitor-1,2 | ↑ (fibrinolysis inhibited) |
| Factors XI, XIII | ↓ slightly |
- Purpose: prepares for haemostasis at delivery
- Risk: 5-fold increase in VTE during pregnancy; 10–20-fold increase in puerperium
Iron Metabolism
- Increased demand: ~1000 mg total (fetus 300 mg, expansion of red cell mass 500 mg, other 200 mg)
- Serum ferritin falls; serum transferrin rises
- Supplemental iron required (elemental iron 60 mg/day)
4. Renal System
Structural Changes
- Kidneys enlarge by ~1 cm in length (renal hypertrophy, increased vascularity)
- Physiological hydronephrosis and hydroureter — bilateral (right > left), due to:
- Progesterone-mediated smooth muscle relaxation of ureters
- Mechanical compression by gravid uterus + right-sided dextrorotation
- Right ovarian vein compression
- Bladder: elevated and anteriorly displaced; increased frequency of micturition
Renal Haemodynamics
| Parameter | Change |
|---|---|
| Renal Plasma Flow (RPF) | ↑ 50–80% |
| GFR (Glomerular Filtration Rate) | ↑ 40–65% |
| Serum Creatinine | ↓ (0.4–0.8 mg/dL) — normal values lower in pregnancy |
| Serum Urea | ↓ |
| Serum Uric Acid | Falls (first/second trimester), rises in third trimester |
Tubular Changes
- Glycosuria in 1–10% — physiological; GFR overwhelms tubular reabsorption capacity; does not indicate diabetes
- Proteinuria: up to 300 mg/24h is normal (vs. non-pregnant: up to 150 mg)
- Sodium retention enhanced (aldosterone and progesterone effects)
- Total body water increases by ~8 L (plasma, interstitial, amniotic fluid)
- Nocturia common: redistribution of oedema fluid when recumbent
5. Gastrointestinal System
Upper GI
- Nausea and vomiting (morning sickness): 50–90% of pregnancies; peaks at 8–12 weeks; driven by hCG; resolves by 16 weeks in most
- Hyperemesis gravidarum: severe vomiting requiring hospitalisation (<1%)
- Oesophageal sphincter tone reduced (progesterone) + intragastric pressure increased → gastro-oesophageal reflux (GORD) — very common; treat with antacids/PPI
- Gastric emptying delayed in labour (anaesthetic risk — aspiration)
- Gastric acid secretion reduced in first/second trimester; rises in third
Lower GI
- Intestinal motility reduced (progesterone effect on smooth muscle)
- Constipation — common; oral iron worsens it
- Haemorrhoids — due to venous engorgement + constipation + increased intra-abdominal pressure
- Appendix displaced upward and laterally to right iliac fossa → McBurney's point becomes unreliable in pregnancy
Liver & Gallbladder
- Liver: anatomically displaced upward; liver function tests marginally altered:
- Alkaline phosphatase (ALP) doubles (from placental isoenzyme — normal)
- Albumin falls (haemodilution + reduced synthesis); normal: 2.8–3.5 g/dL
- ALT, AST, bilirubin: unchanged (elevation is pathological)
- Fibrinogen and clotting factors: rise (see coagulation)
- Gallbladder: reduced motility + bile stasis (progesterone) + ↑ cholesterol saturation → gallstone formation risk increased (2x); "pregnancy is a lithogenic state"
- Spider naevi and palmar erythema: normal due to ↑ oestrogen
6. Endocrine System
The Placenta as an Endocrine Organ
| Hormone | Source | Function |
|---|---|---|
| hCG | Syncytiotrophoblast | Maintains corpus luteum; peaks wk 8–10; basis of pregnancy tests |
| Progesterone | Corpus luteum (1st trimester) → Placenta | Uterine quiescence, smooth muscle relaxation, immunosuppression |
| Oestrogen (oestriol) | Placenta + fetal adrenal/liver | Uterine growth, breast development, fluid retention |
| Human Placental Lactogen (hPL) | Placenta | Insulin resistance, lipolysis, glucose sparing for fetus |
| Relaxin | Corpus luteum, placenta | Ligament laxity, cervical softening, vasodilation |
Thyroid
- Total T3 and T4 rise (due to ↑ TBG from oestrogen) — but free T3 and T4 remain normal
- TSH: may dip in first trimester (hCG has mild TSH-like activity)
- Thyroid gland enlarges mildly (increased vascularity + iodine demand)
- Iodine requirement increases; supplementation recommended
Adrenal
- Cortisol rises 2–3x (increased CBG from oestrogen + increased ACTH); free cortisol also rises
- Causes: striae gravidarum, glucose intolerance, immunosuppression
- Aldosterone rises markedly (4–5x) → Na+ and water retention
- DHEA-S falls (increased 16α-hydroxylation)
Pancreas
- Insulin resistance increases progressively (from hPL, cortisol, progesterone, TNF-α)
- First trimester: enhanced insulin sensitivity (lower fasting glucose)
- Second/third trimester: diabetogenic state — fasting glucose lower, post-prandial glucose higher
- β-cell hyperplasia compensates; failure → Gestational Diabetes Mellitus (GDM)
- Fasting hypoglycaemia more common (glucose diverted to fetus)
Pituitary
- Enlarges ~135% (lactotroph hyperplasia under oestrogen stimulation)
- Prolactin rises progressively (up to 10x); suppresses ovulation
- FSH, LH: suppressed throughout pregnancy
- Sheehan's syndrome risk: pituitary infarction if severe postpartum haemorrhage occurs (enlarged gland is vulnerable)
Parathyroid & Calcium Metabolism
- Fetal demand for calcium: ~25–30 g throughout pregnancy
- PTH initially falls then rises in third trimester
- 1,25-dihydroxyvitamin D (calcitriol) rises — increases gut calcium absorption
- Total serum calcium falls (albumin fall), but ionised calcium remains normal
- Urinary calcium excretion increases
7. Musculoskeletal System
- Relaxin causes ligamentous laxity throughout the body:
- Sacroiliac joints, symphysis pubis (widens 3–4 mm — diastasis symphysis pubis)
- Lumbar spine hyperlordosis increases → lower back pain (50–70% of pregnant women)
- Hip joints: slightly widened
- Centre of gravity shifts anteriorly → compensatory lordosis + waddling gait
- Diastasis recti: separation of rectus abdominis muscles
- Carpal tunnel syndrome: due to oedema compressing median nerve; common in third trimester
- Generalised joint laxity → pelvic girdle pain, pubic symphysis pain
8. Skin Changes
Pigmentation (Melanocyte-stimulating hormone + oestrogen + progesterone)
- Linea nigra: darkening of linea alba from pubis to umbilicus
- Chloasma (Melasma): "mask of pregnancy" — hyperpigmentation of face (cheeks, forehead, upper lip); fades post-delivery
- Darkening of areolae, nipples, vulva, perineum, axillae
- Pre-existing nevi may darken
Striae Gravidarum (Stretch Marks)
- Pink/purple linear marks on abdomen, breasts, thighs — due to stretching + cortisol-induced collagen disruption
- Fade to silver-white post-delivery but persist
Vascular Changes (due to ↑ oestrogen)
- Spider angiomas (telangiectasiae) — face, neck, arms; regress after delivery
- Palmar erythema — bilateral, blanching
- Varicose veins — legs, vulva (↑ venous pressure + progesterone relaxation of vessel walls)
- Gum hyperaemia → pregnancy gingivitis; can develop epulis (pyogenic granuloma of gums)
Hair and Nails
- Hair growth: enters prolonged anagen (growth) phase during pregnancy; hair feels thicker
- Post-partum telogen effluvium: 2–4 months after delivery, excessive shedding as hair re-enters telogen phase; usually self-limiting
- Nails may soften or develop transverse grooving (Beau's lines)
9. Neurological Changes
- Peripheral nervous system: nerve compression syndromes common (carpal tunnel, meralgia paraesthetica — lateral femoral cutaneous nerve compression)
- Cerebral blood flow maintained despite vasodilation
- Sleep disturbance (positional discomfort, nocturia, restless legs)
- Restless Legs Syndrome: more common in pregnancy (iron/folate deficiency contribution)
- Cognitive changes ("pregnancy brain") — subjective memory and concentration lapses; supported by some objective studies
- Increased seizure threshold in first trimester; decreased in third trimester
10. Immunological Changes
- Pregnancy represents a state of immune tolerance to the semi-allogeneic fetus:
- Shift from Th1 (cellular immunity) to Th2 (humoral immunity) dominance
- Regulatory T cells (Tregs) expanded at the maternal-fetal interface
- NK cell activity at decidua modified
- Clinical implications:
- Autoimmune diseases with Th1 pathogenesis (e.g., rheumatoid arthritis, multiple sclerosis) often improve in pregnancy
- Autoimmune diseases with Th2 pathogenesis (e.g., SLE) may worsen
- Susceptibility to intracellular pathogens (Listeria, Toxoplasma, Mycobacteria, viruses) increased
- Reduced cell-mediated immunity → severe varicella, influenza in pregnancy
11. The Uterus
| Feature | Non-Pregnant | Term Pregnancy |
|---|---|---|
| Weight | 60–70 g | 1000–1200 g |
| Capacity | 10 mL | 5–10 L |
| Length | 7.5 cm | 30–35 cm |
| Blood flow | ~50 mL/min | 750–800 mL/min (10% of CO) |
- Decidualisation of endometrium
- Braxton Hicks contractions from mid-trimester (painless, irregular)
- Uterine body (fundal) muscle thickens in first trimester → thins in third (stretching)
- Lower uterine segment (LUS) forms from isthmus at 12 weeks, fully formed by 28 weeks
- Cervical changes: Goodell's sign (softening), Hegar's sign (softening of isthmus), Chadwick's sign (bluish discolouration)
12. Breast Changes
| Timing | Changes |
|---|---|
| 4–6 weeks | Fullness, heaviness, tingling, tenderness |
| 6–8 weeks | Enlargement begins; Montgomery's tubercles enlarge |
| 8–12 weeks | Nipple and areola darken; secondary areola appears |
| 16 weeks | Colostrum secretion begins |
| Throughout | Increased vascularity (visible veins — Haller's sign) |
| Near term | Breasts fully prepared for lactation (prolactin, oxytocin) |
13. Weight Gain in Pregnancy
Recommended Total Weight Gain (Institute of Medicine)
| Pre-pregnancy BMI | Recommended Gain |
|---|---|
| Underweight (<18.5) | 12.5–18 kg |
| Normal (18.5–24.9) | 11.5–16 kg |
| Overweight (25–29.9) | 7–11.5 kg |
| Obese (≥30) | 5–9 kg |
| Twin pregnancy (normal BMI) | 17–25 kg |
Distribution of Weight Gain (~12.5 kg total)
| Component | Weight |
|---|---|
| Fetus | 3.3–3.5 kg |
| Placenta | 0.65 kg |
| Amniotic fluid | 0.8 kg |
| Uterus | 0.9 kg |
| Breasts | 0.4 kg |
| Blood volume expansion | 1.25 kg |
| Extracellular fluid | 1.5 kg |
| Maternal fat stores | 3.5 kg |
14. Metabolic Changes
| Parameter | Change |
|---|---|
| Basal Metabolic Rate (BMR) | ↑ 15–20% by term |
| Total caloric need | +300 kcal/day (second/third trimester) |
| Fasting glucose | ↓ slightly (glucose diverted to fetus) |
| Postprandial glucose | ↑ (insulin resistance) |
| Serum cholesterol | ↑ 50% (needed for hormone synthesis) |
| Triglycerides | ↑ 3x |
| Serum albumin | ↓ (haemodilution + transfer) |
| Total protein | ↓ |
| Body fat | Accumulates first/second trimester; mobilised in third |
- Accelerated starvation: fasting ketosis develops more rapidly in pregnancy (fetus has priority on glucose) — "facilitated anabolism" in fed state, "accelerated catabolism" in fasted state
Summary Table: Key Physiological Changes at a Glance
| System | Key Change | Direction |
|---|---|---|
| Blood volume | Total ↑ 40–50%; plasma ↑ 50%; RBC ↑ 20% | ↑ |
| Cardiac output | +30–50% by 28 weeks | ↑ |
| Blood pressure | Falls mid-trimester, returns to normal | ↓ then → |
| SVR | −30–35% | ↓ |
| Tidal volume | +40–45% | ↑ |
| PaCO₂ | 28–32 mmHg (physiological alkalosis) | ↓ |
| FRC | −20% | ↓ |
| GFR | +40–65% | ↑ |
| Serum creatinine | Falls to 0.4–0.8 mg/dL | ↓ |
| Fibrinogen | +50–100% | ↑ |
| Haemoglobin | Falls (physiological anaemia) | ↓ |
| Insulin resistance | Progressive from 2nd trimester | ↑ |
| ALP | Doubles (placental isoenzyme) | ↑ |
| Albumin | Falls | ↓ |
| Thyroid hormones (total) | ↑ (free T3/T4 normal) | ↑ (total) |
Give me 5 questions regarding twin pregnancy that can be asked in a competitive exams like USMLE, PLAB, NZREX
twin pregnancy complications management zygosity chorionicity
twin to twin transfusion syndrome monochorionic dichorionic twin pregnancy
Twin Pregnancy — 5 Competitive Exam Questions
Question 1
- A) Dichorionic diamniotic (DCDA)
- B) Monochorionic diamniotic (MCDA)
- C) Monochorionic monoamniotic (MCMA)
- D) Dizygotic twin pregnancy
- E) Conjoined twins
| Feature | DCDA | MCDA | MCMA |
|---|---|---|---|
| Membrane insertion | Lambda (λ) / Twin-peak sign | T-sign | No membrane |
| Membrane thickness | Thick | Thin | Absent |
| Placentae | 2 (or fused) | 1 | 1 |
| Amniotic sacs | 2 | 2 | 1 |
| Zygosity | Di or monozygotic | Monozygotic | Monozygotic |
- Lambda/twin-peak sign: chorionic villi project into the inter-twin membrane base (DCDA) — thick membrane
- T-sign: amnion reflected directly off placenta with no intervening villi (MCDA) — thin membrane
- MCDA twins are at risk for Twin-to-Twin Transfusion Syndrome (TTTS), cord entanglement (less so), and IUGR
- D is incorrect — dizygotic twins are always DCDA
- E (conjoined twins) arise from MCMA pregnancies with incomplete division
Question 2
- A) Immediate caesarean section
- B) Serial ultrasound monitoring every 2 weeks
- C) Referral to a fetal medicine unit for laser photocoagulation
- D) Amnioreduction of Twin A's sac
- E) Intracordal potassium chloride injection to Twin B
| Quintero Stage | Criteria |
|---|---|
| I | DVP discordance (≥8 cm recipient, ≤2 cm donor); bladder visible in both |
| II | Donor bladder non-visualised |
| III | Abnormal Doppler (absent/reversed EDF in donor UA, or ductus venosus changes in recipient) |
| IV | Hydrops in either twin |
| V | Death of one or both twins |
- TTTS affects 10–15% of MCDA pregnancies (Targeted Neonatal Echocardiography, p. 24) via unidirectional flow through placental arteriovenous anastomoses
- Donor twin (Twin B): hypovolaemia, oligohydramnios ("stuck twin"), IUGR, absent EDF
- Recipient twin (Twin A): fluid overload, polyhydramnios, cardiac dysfunction, risk of hydrops
- Treatment of choice for Stage II–IV TTTS before 26 weeks: Selective Laser Photocoagulation of Communicating Vessels (SLPCV) — improves survival and reduces cardiovascular morbidity
- Amnioreduction (D) is a temporising measure, not preferred over laser where available
- Caesarean (A) at 20 weeks is not viable
- KCl injection (E) is selective feticide — not indicated here
Question 3
- A) Emergency caesarean section must be performed once Twin A is delivered
- B) Internal podalic version (IPV) and breech extraction is an acceptable option for Twin B after vaginal delivery of Twin A
- C) External cephalic version (ECV) should be performed for Twin B before Twin A is delivered
- D) Caesarean delivery of Twin B is always required if it remains breech
- E) Oxytocin must be stopped after delivery of Twin A to prevent uterine hyperstimulation
| Twin B Presentation | Options |
|---|---|
| Cephalic | Await spontaneous descent and deliver |
| Breech (with experienced operator) | Breech extraction OR IPV + breech extraction |
| Transverse | IPV (to footling breech) + breech extraction |
| Any (operator preference) | ECV to cephalic after Twin A delivers |
- IPV (reaching into the uterus to grasp a fetal foot and convert to breech) is a well-established technique for Twin B delivery by experienced operators
- Caesarean for Twin B after vaginal Twin A (option A/D) is not routinely indicated — combined deliveries (vaginal Twin A + caesarean Twin B) occur in ~4% of cases but are not the goal
- ECV of Twin B (option C) can be attempted after Twin A delivers but is less reliable and carries cord prolapse risk; IPV + breech extraction is generally preferred
- After delivery of Twin A: oxytocin infusion should be commenced or continued (not stopped — E is wrong) to maintain uterine tone and hasten delivery of Twin B; aim to deliver within 30 minutes
- The cervix remains fully dilated; the goal is to deliver Twin B promptly
Question 4
- A) Twin-to-Twin Transfusion Syndrome
- B) Selective Intrauterine Growth Restriction (sIUGR)
- C) Cord entanglement
- D) Discordant fetal anomalies
- E) Vanishing twin syndrome
| Complication | MCMA | MCDA | DCDA |
|---|---|---|---|
| Cord entanglement | Yes — unique risk | No (separated by amnion) | No |
| TTTS | Yes (shared chorion) | Yes (most common here) | No |
| sIUGR | Yes | Yes | Less common |
| Conjoined twins | Yes (incomplete division) | No | No |
| Vanishing twin | First trimester, any | Any | Any |
- TTTS (A) and sIUGR (B) occur in monochorionic pregnancies (both MCMA and MCDA) due to shared placental circulation — they are NOT specific to MCMA
- Cord entanglement is the defining danger of MCMA twins → risk of sudden fetal death
- Management: inpatient admission from 26–28 weeks, intensive CTG monitoring, elective delivery at 32–34 weeks (before spontaneous labour increases cord tension risk)
- Vanishing twin (E) is a first-trimester phenomenon, not specific to MCMA
Question 5
- A) Termination of the entire pregnancy
- B) Observation — trisomy 21 fetuses often spontaneously demise
- C) Selective fetal reduction using intracardiac potassium chloride (KCl) injection into the affected fetus
- D) Amniotic fluid drainage from the affected twin's sac
- E) Await delivery and provide neonatal palliation
| Feature | DCDA | MCDA/MCMA |
|---|---|---|
| Placental circulation | Separate — no shared anastomoses | Shared anastomoses |
| KCl use | Safe — does not cross to co-twin | CONTRAINDICATED — KCl crosses placental anastomoses and kills both twins |
| Alternative in MC twins | Cord occlusion (bipolar diathermy, radiofrequency ablation) | Same |
- In DCDA twins: KCl injection into the fetal heart or umbilical vein is safe and effective; the agent cannot reach the co-twin as there are no shared vascular connections
- In monochorionic twins: KCl is absolutely contraindicated; cord occlusion techniques must be used instead
- Most studies show selective reduction is safer when performed in the first trimester (NIPS guidelines, p. 8)
- Risk of miscarriage of the entire pregnancy following selective reduction: ~5–7%
- Observation (B) is inappropriate — trisomy 21 fetuses do not reliably demise spontaneously and waiting increases procedural risk
Summary of High-Yield Concepts Tested
| Q | Topic | Exam Favourite |
|---|---|---|
| 1 | Chorionicity determination — T-sign vs. lambda sign | ⭐⭐⭐⭐⭐ |
| 2 | TTTS Quintero staging + laser treatment | ⭐⭐⭐⭐⭐ |
| 3 | Intrapartum management of twin labour | ⭐⭐⭐⭐ |
| 4 | MCMA-specific risk — cord entanglement | ⭐⭐⭐⭐ |
| 5 | Selective fetal reduction — KCl vs. cord occlusion | ⭐⭐⭐⭐⭐ |
Give me 5 questions regarding maternal changes in pregnancy that can be asked in a competitive exams like USMLE, PLAB, NZREX
maternal physiological changes pregnancy cardiovascular hemodynamic blood volume cardiac output
pregnancy coagulation respiratory renal changes physiological adaptations clinical significance
Maternal Changes in Pregnancy — 5 Competitive Exam Questions
Question 1
- A) Pre-existing essential hypertension masked by pregnancy
- B) Anaemia causing reduced peripheral vascular resistance
- C) Physiological fall in systemic vascular resistance due to progesterone and uteroplacental circulation
- D) Reduced cardiac output from aortocaval compression
- E) Inappropriate antidiuretic hormone secretion causing haemodilution
| Factor | Effect |
|---|---|
| Progesterone | Direct smooth muscle relaxant → peripheral vasodilation |
| Relaxin | Stimulates nitric oxide → vasodilation |
| Uteroplacental circulation | Low-resistance vascular bed added to maternal circulation |
| Oestrogen | Promotes endothelial prostacyclin production |
- SVR falls by 30–35% — this is the primary driver of the blood pressure fall
- Cardiac output simultaneously rises 30–50% (starting at 6 weeks) to compensate — Management of Cardiovascular Disease and Pregnancy, p. 12
- Heart rate rises +10–20 bpm to further compensate
- Her Hb of 10.8 g/dL reflects physiological anaemia (plasma volume expands ~50%, red cell mass only ~20%) — but this is not the cause of her low BP
- Aortocaval compression (D) occurs in the supine position, not in general
- There is no proteinuria or features of pre-eclampsia; this is entirely physiological
Question 2
| Parameter | Result | Normal (non-pregnant) |
|---|---|---|
| pH | 7.44 | 7.35–7.45 |
| PaCO₂ | 30 mmHg | 35–45 mmHg |
| PaO₂ | 104 mmHg | 80–100 mmHg |
| HCO₃⁻ | 20 mEq/L | 22–28 mEq/L |
- A) Acute metabolic alkalosis due to vomiting
- B) Acute respiratory alkalosis — pathological, requires urgent investigation for pulmonary embolism
- C) Compensated respiratory alkalosis — normal finding in pregnancy
- D) Partially compensated metabolic acidosis
- E) Normal blood gas — no abnormality detected
| ABG Feature | Explanation |
|---|---|
| ↓ PaCO₂ (28–32 mmHg) | Progesterone directly stimulates the medullary respiratory centre → hyperventilation → CO₂ blown off |
| ↑ Tidal volume (+40–45%) | Primary driver of hyperventilation (not respiratory rate) |
| ↓ HCO₃⁻ (18–22 mEq/L) | Renal compensation — kidneys excrete bicarbonate to normalise pH |
| pH 7.40–7.45 | Fully compensated → pH remains normal |
| ↑ PaO₂ (~104–108 mmHg) | Consequence of hyperventilation |
- The dyspnoea of pregnancy (felt by 60–70% of women) is caused by this progesterone-driven hyperventilation — it is physiological, not a sign of PE
- Clinical trap: If this ABG were seen in a non-pregnant patient, the low HCO₃⁻ + low PaCO₂ might suggest metabolic acidosis with respiratory compensation — but in pregnancy, the primary event is respiratory alkalosis
- PaO₂ of >100 mmHg with normal pH and no hypoxia makes PE (B) very unlikely here
- FRC falls 20% in pregnancy — this reduces oxygen reserve and explains why pregnant women desaturate rapidly during apnoea (important for anaesthetists)
Question 3
| Test | Result | Lab Reference Range (non-pregnant) |
|---|---|---|
| Haemoglobin | 11.4 g/dL | 12–16 g/dL |
| MCV | 88 fL | 80–100 fL |
| Serum creatinine | 0.5 mg/dL | 0.6–1.2 mg/dL |
| Serum albumin | 3.0 g/dL | 3.5–5.0 g/dL |
| Alkaline phosphatase (ALP) | 180 U/L | 44–147 U/L |
| Platelet count | 155 × 10⁹/L | 150–400 × 10⁹/L |
- A) The low haemoglobin indicates iron deficiency anaemia requiring immediate IV iron infusion
- B) The low creatinine is abnormal and suggests acute kidney injury
- C) The elevated ALP indicates hepatic disease and warrants further liver investigation
- D) All these results are consistent with normal physiological changes of pregnancy
- E) The low albumin suggests nephrotic syndrome given the low creatinine
| Result | Value | Physiological Explanation |
|---|---|---|
| Hb 11.4 g/dL | Low by non-pregnant standard | Plasma volume ↑ 50% vs. RBC mass ↑ 20% → haemodilution ("physiological anaemia"); WHO threshold for anaemia in pregnancy = Hb <11 g/dL (1st/3rd trimester) — 11.4 is normal |
| Creatinine 0.5 mg/dL | Low | GFR increases 40–65% → increased creatinine clearance → lower serum creatinine; normal in pregnancy is 0.4–0.8 mg/dL |
| ALP 180 U/L | Elevated | Placental isoenzyme of ALP is produced and released; ALP doubles in pregnancy — this is normal. ALT and AST remain normal |
| Albumin 3.0 g/dL | Low | Haemodilution + increased volume of distribution + some transfer to fetus; normal in pregnancy = 2.8–3.5 g/dL |
| Platelets 155 | Normal | Mild gestational thrombocytopaenia can lower platelets slightly — still within normal range here |
- Critical clinical trap: a serum creatinine of 0.9–1.0 mg/dL (normal in non-pregnant adults) may represent significant renal impairment in a pregnant woman — always interpret lab values using pregnancy-specific reference ranges
- ALP elevation in pregnancy never warrants liver investigation unless ALT/AST/bilirubin are also elevated
- Iron deficiency requires MCV <80 fL (microcytosis) + low ferritin; this patient has normal MCV
Question 4
- A) Undiagnosed right renal calculus not visible on ultrasound
- B) Right-sided renal vein thrombosis — an obstetric emergency
- C) Physiological hydronephrosis of pregnancy, more prominent on the right side
- D) Ureteric obstruction from a pelvic mass requiring urgent CT urogram
- E) Pre-eclampsia causing renal impairment and ureteral dilation
| Cause | Detail |
|---|---|
| Progesterone | Smooth muscle relaxation → ureteral peristalsis reduced → urinary stasis |
| Mechanical compression | Gravid uterus compresses ureters at the pelvic brim |
| Right-sided predominance | Uterus undergoes dextrorotation (rotates to the right) → greater right ureteral compression; right ovarian vein crosses the ureter at a right angle and causes additional compression |
| Left side protection | Left ureter is partially shielded by the sigmoid colon cushion |
| Feature | Physiological | Pathological (Calculus/Obstruction) |
|---|---|---|
| Urinalysis | Normal | Haematuria, leucocytes |
| Pain | Mild, dull | Severe colic, loin-to-groin |
| Hydronephrosis | Bilateral (R>L), gradual | Unilateral, sudden onset |
| USS | No calculus | May show calculus/obstruction |
- This patient has no haematuria, no leucocytes, no calculus → calculus (A) unlikely
- Renal vein thrombosis (B) presents with haematuria and acute flank pain with venous engorgement, not simple hydronephrosis
- No features of pre-eclampsia (E) — no hypertension or proteinuria mentioned
- Physiological hydronephrosis increases the risk of ascending UTI/pyelonephritis in pregnancy — urinary stasis is a key reason why pregnant women are at higher risk of pyelonephritis from asymptomatic bacteriuria
Question 5
- A) Haemorrhagic shock from internal injuries masked by pregnancy
- B) Vasovagal syncope triggered by pain and anxiety
- C) Aortocaval compression by the gravid uterus causing reduced venous return
- D) Pulmonary embolism causing acute haemodynamic compromise
- E) Peripartum cardiomyopathy with positional ventricular dysfunction
- In the supine position, the gravid uterus (especially from ~20 weeks onwards, most pronounced at term) compresses:
- The inferior vena cava (IVC) → reduces venous return to the right heart → ↓ cardiac output → hypotension
- The descending aorta → reduces uteroplacental perfusion → fetal distress
| Compression | Effect |
|---|---|
| IVC compression | ↓ Venous return → ↓ preload → ↓ CO → maternal hypotension, dizziness, syncope |
| Aortic compression | ↓ Uteroplacental blood flow → fetal bradycardia, hypoxia |
- Occurs in supine position after ~20 weeks
- Immediately relieved by left lateral tilt (15–30°) or full left lateral position
- Affects ~10% of women symptomatically; subclinical IVC compression occurs in virtually all women at term in the supine position
- Fetal CTG may show late decelerations during supine positioning
| Setting | Implication |
|---|---|
| Resuscitation / trauma | NEVER lay pregnant woman (>20 wks) flat supine — always apply left lateral tilt |
| Labour | Avoid prolonged supine positioning; CTG deterioration may be positional |
| Spinal anaesthesia | Left lateral tilt mandatory after spinal block (sympathectomy + aortocaval compression → severe hypotension) |
| CPR in pregnancy | Manual uterine displacement to the left during chest compressions |
- Haemorrhagic shock (A) would not immediately resolve with positional change alone
- PE (D) does not resolve with positional change
- Vasovagal (B) does not follow a strict positional pattern and would not resolve this reliably
Summary of High-Yield Concepts Tested
| Q | System | Core Concept | Exam Weight |
|---|---|---|---|
| 1 | Cardiovascular | Physiological fall in BP — SVR reduction | ⭐⭐⭐⭐⭐ |
| 2 | Respiratory | Normal ABG in pregnancy — compensated respiratory alkalosis | ⭐⭐⭐⭐⭐ |
| 3 | Haematology / Renal / Hepatic | Interpreting pregnancy-altered lab values | ⭐⭐⭐⭐⭐ |
| 4 | Renal | Physiological hydronephrosis — right-sided predominance | ⭐⭐⭐⭐ |
| 5 | Cardiovascular | Supine hypotensive syndrome — aortocaval compression | ⭐⭐⭐⭐⭐ |
Give me 5 questions regarding postpartum hemorrhage that can be asked in a competitive exams like USMLE, PLAB, NZREX
postpartum hemorrhage causes management uterine atony 4T
postpartum hemorrhage oxytocin misoprostol tranexamic acid surgical management
Postpartum Haemorrhage — 5 Competitive Exam Questions
Question 1
- A) Retained placenta — ergometrine 0.5 mg IM
- B) Uterine atony — oxytocin 10 IU IV bolus (slow) or IM
- C) Cervical laceration — misoprostol 800 mcg sublingual
- D) Coagulopathy — fresh frozen plasma
- E) Uterine inversion — carboprost 250 mcg IM
| T | Cause | Frequency |
|---|---|---|
| Tone | Uterine atony | ~70–80% — most common |
| Tissue | Retained placenta/membranes | ~10% |
| Trauma | Lacerations, haematoma, uterine rupture, inversion | ~10% |
| Thrombin | Coagulopathy (DIC, von Willebrand's, anticoagulants) | ~1% |
- Uterine atony is the most common cause — the "soft, boggy, non-contracted uterus" is its hallmark (Venous Thromboembolism in the Context of Pregnancy, p. 15)
- The primary physiological mechanism to stop placental bed bleeding is sustained myometrial contraction, not the haemostatic system — hence uterotonics are first-line
- Risk factors for atony: prolonged labour, grand multiparity, polyhydramnios, macrosomia, multiple pregnancy, chorioamnionitis, magnesium sulphate use, uterine fibroids
- Call for help — senior obstetrician, anaesthetist, haematologist
- Uterine massage / bimanual compression
- Oxytocin 10 IU IV (slow bolus) or IM — first-line uterotonic (WHO/RCOG)
- IV access (2 large-bore), bloods (FBC, coag, crossmatch), IV fluids
- Catheterise bladder (full bladder inhibits uterine contraction)
Question 2
- A) Repeat oxytocin 10 IU IV — contraindicated in diabetes
- B) Carboprost (15-methyl PGF2α) 250 mcg IM — contraindicated in asthma
- C) Misoprostol 200 mcg IV — contraindicated in renal failure
- D) Tranexamic acid 1 g IV — contraindicated in hypertension
- E) Methylergometrine 0.2 mg IV — contraindicated in anaemia
| Step | Drug | Dose & Route | Contraindication |
|---|---|---|---|
| 1st line | Oxytocin | 10 IU IV/IM; then 40 IU infusion over 4h | Rapid IV bolus (hypotension) |
| 2nd line | Ergometrine | 0.5 mg IM/slow IV | Hypertension, pre-eclampsia, cardiovascular disease, Raynaud's |
| 3rd line | Carboprost (PGF2α) | 250 mcg IM every 15 min, max 8 doses (2 mg) | Asthma (causes severe bronchospasm), relative: hypertension, cardiac/hepatic/renal disease |
| 4th line | Misoprostol (PGE1) | 800–1000 mcg sublingual/rectal | Fewer contraindications; used when others unavailable |
| Adjunct | Tranexamic acid | 1 g IV (within 3h of delivery/bleeding onset) | Thromboembolic disease (relative) |
- Carboprost (Haemabate) is a prostaglandin F2α analogue that causes powerful uterine contraction
- Its critical contraindication is asthma — PGF2α causes potent bronchospasm and can precipitate life-threatening bronchoconstriction
- Tranexamic acid (D) is an antifibrinolytic (not a uterotonic) — it is an adjunct that reduces PPH mortality when given within 3 hours (Management of Von Willebrand Disease, p. 19); not contraindicated in hypertension
- Ergometrine (already given) is contraindicated in hypertension/pre-eclampsia — not diabetes
Question 3
- A) Immediate manual removal of placenta under general anaesthesia in theatre
- B) Give a further dose of oxytocin 10 IU IV and wait another 30 minutes
- C) Attempt manual removal of placenta under regional or general anaesthesia in theatre
- D) Perform bimanual compression to expel the placenta
- E) Administer misoprostol rectally and reassess in 1 hour
| Type of Retained Placenta | Mechanism | Management |
|---|---|---|
| Trapped placenta | Placenta separated but os closed (cervical spasm) | Manual removal |
| Adherent placenta | Failed separation (partial or complete) | Manual removal ± surgical |
| Placenta accreta spectrum | Abnormal invasion into myometrium | Hysterectomy may be needed |
- Confirm placenta unseparated (cord not lengthened, uterus contracted "fundus high")
- Empty bladder (full bladder prevents placental separation)
- IV oxytocin infusion
- If not delivered by 30 minutes → theatre for manual removal of placenta (MROP) under regional (spinal/epidural) or general anaesthesia
- IV access, group and save, consent for examination under anaesthesia
- Option A is partially correct but premature — the timeline has just reached 20 minutes; at 30 minutes, theatre is indicated
- Option B is incorrect — waiting another 30 minutes (total 50 min) without action risks haemorrhage; reassess at 30 minutes
- Bimanual compression (D) cannot expel an unseparated placenta
- Grand multiparity is a risk factor for both uterine atony and retained placenta
| Type | Invasion Depth | Incidence |
|---|---|---|
| Placenta accreta | Into superficial myometrium | 75–78% |
| Placenta increta | Into myometrium | 17% |
| Placenta percreta | Through serosa/into bladder | 5–7% |
Question 4
- A) Carboprost 250 mcg IM
- B) Surgical B-Lynch suture
- C) Tranexamic acid 1 g IV and desmopressin (DDAVP) 0.3 mcg/kg IV
- D) Emergency hysterectomy
- E) Fresh frozen plasma 4 units
| Drug | Mechanism | Role in VWD |
|---|---|---|
| Tranexamic acid 1 g IV | Antifibrinolytic — inhibits plasminogen activation, prevents clot breakdown | Reduces PPH risk (RR 0.42 for secondary PPH) (Von Willebrand Disease guidelines, p. 19); first-line adjunct in all PPH including coagulopathy |
| Desmopressin (DDAVP) 0.3 mcg/kg IV/SC | Releases endogenous vWF and Factor VIII from endothelial Weibel-Palade bodies | Treatment of choice for Type 1 VWD — raises vWF and FVIII levels 3–5x within 30–60 min |
| Type | Defect | DDAVP Response | Definitive Rx |
|---|---|---|---|
| Type 1 (most common, 70–80%) | Quantitative ↓ vWF | Good response | DDAVP + tranexamic acid |
| Type 2 | Qualitative defect | Variable/poor | vWF concentrate |
| Type 3 | Absent vWF | No response | vWF/FVIII concentrate |
- FFP (E) is not indicated — fibrinogen is adequate (>2 g/L), PT is normal; FFP is used for dilutional coagulopathy/DIC with multiple factor deficiency
- B-Lynch suture (B) addresses uterine atony, not coagulopathy — uterus is well-contracted here
- Carboprost (A) addresses atony — not relevant here
Question 5
- A) Repeat B-Lynch suture with additional chromic catgut
- B) Internal iliac (hypogastric) artery ligation bilaterally
- C) Peripartum hysterectomy
- D) Uterine artery embolisation in interventional radiology suite
- E) Packing the uterine cavity with gauze and closing abdomen
| Step | Intervention | Notes |
|---|---|---|
| 1 | Uterine massage + uterotonics (oxytocin → ergometrine → carboprost → misoprostol) | Medical management |
| 2 | Tranexamic acid 1 g IV | Adjunct — antifibrinolytic |
| 3 | Intrauterine balloon tamponade (Bakri balloon) | "Uterine tamponade test" — if effective, may avoid surgery |
| 4 | Compression sutures (B-Lynch, Hayman, Cho square sutures) | Mechanical compression of myometrium |
| 5 | Uterine devascularisation — uterine artery ligation (O'Leary sutures), ovarian artery ligation, internal iliac artery ligation | Reduces pulse pressure to uterine vessels |
| 6 | Peripartum hysterectomy | Definitive life-saving procedure when all else fails |
| Option | Why Incorrect |
|---|---|
| A — Repeat B-Lynch | Already performed and failed; repeating provides no additional benefit |
| B — Internal iliac ligation | A valid step, but technically difficult, time-consuming, and 40–50% failure rate; appropriate earlier in the algorithm, not as the final step when the patient is unstable |
| D — UAE | Requires haemodynamic stability, transfer to radiology suite, and trained interventional radiologist — not appropriate in actively haemorrhaging, unstable patient in theatre |
| E — Gauze packing | Temporising measure only; gauze packing of the uterus is not a definitive treatment for refractory atonic PPH |
- Peripartum hysterectomy is the definitive, life-saving procedure for intractable PPH — it must not be delayed when the patient is deteriorating
- A subtotal (supracervical) hysterectomy is often preferred in emergency settings as it is faster and technically easier; however, if bleeding continues from the cervical stump (e.g., cervical tear or praevia), total hysterectomy is required
- Consent has been documented — this removes a potential barrier to timely action
| Component | Ratio | Rationale |
|---|---|---|
| Packed Red Blood Cells | 1 unit | Oxygen carrying capacity |
| Fresh Frozen Plasma | 1:1 with PRBC | Clotting factors |
| Platelets | 1:1:1 (per pool) | Haemostatic function |
| Cryoprecipitate | If fibrinogen <2 g/L | Fibrinogen replacement |
| Tranexamic acid | 1 g IV (repeat 1 g if bleeding >30 min or >1 g blood loss) | Antifibrinolytic |
Summary of High-Yield Concepts Tested
| Q | Topic | Core Concept | Exam Weight |
|---|---|---|---|
| 1 | Causes of PPH | 4 Ts framework — uterine atony as most common cause | ⭐⭐⭐⭐⭐ |
| 2 | Uterotonic ladder | Carboprost contraindicated in asthma; tranexamic acid as adjunct | ⭐⭐⭐⭐⭐ |
| 3 | Retained placenta | MROP at 30 minutes; placenta accreta spectrum | ⭐⭐⭐⭐ |
| 4 | Coagulopathy (Thrombin T) | VWD management — DDAVP for Type 1; tranexamic acid | ⭐⭐⭐⭐ |
| 5 | Surgical escalation | Peripartum hysterectomy as definitive treatment; MTP | ⭐⭐⭐⭐⭐ |
Give me some {around 10-20} questions from these topics: Postpartum hemorrhage Antepartum hemorrhage Twin pregnancy Don’t give the answers or highlight the answers just give the explaination in last
antepartum hemorrhage placenta previa abruptio placentae management
postpartum hemorrhage twin pregnancy complications obstetric emergencies
Mixed Obstetric MCQs — PPH · APH · Twin Pregnancy
POSTPARTUM HAEMORRHAGE
Question 1
- A) Administer ergometrine 0.5 mg IM and commence uterine massage
- B) Proceed immediately to emergency hysterectomy
- C) Insert a Bakri balloon and close the vagina
- D) Administer carboprost 250 mcg IM
- E) Examine under anaesthesia for retained placenta
Question 2
- A) Misoprostol 800 mcg sublingual
- B) Carboprost 250 mcg IM
- C) Oxytocin infusion 40 IU in 500 mL normal saline
- D) Tranexamic acid 1 g IV
- E) Dinoprostone (PGE2) suppository
Question 3
- A) Administer a second dose of oxytocin 10 IU and wait a further 30 minutes
- B) Apply fundal pressure (Credé manoeuvre) to expel the placenta
- C) Transfer to theatre for manual removal of placenta under anaesthesia
- D) Perform immediate bimanual compression
- E) Administer misoprostol 600 mcg orally and reassess in one hour
Question 4
- A) Apply a second B-Lynch compression suture
- B) Bilateral internal iliac artery ligation as a final attempt to conserve the uterus
- C) Peripartum hysterectomy
- D) Transfer to interventional radiology for uterine artery embolisation
- E) Pack the uterine cavity with gauze and reassess after 30 minutes
Question 5
- A) Fresh frozen plasma + platelet transfusion
- B) Cryoprecipitate + ergometrine 0.5 mg IM
- C) Tranexamic acid 1 g IV + desmopressin (DDAVP) 0.3 mcg/kg IV
- D) Factor VIII concentrate alone
- E) Carboprost 250 mcg IM + oxytocin infusion
ANTEPARTUM HAEMORRHAGE
Question 6
- A) Placental abruption — avoid amniocentesis
- B) Placenta praevia — avoid digital vaginal examination
- C) Vasa praevia — avoid artificial rupture of membranes
- D) Cervical polyp — avoid speculum examination
- E) Placenta praevia — avoid ultrasound examination
Question 7
- A) Placenta praevia with major haemorrhage
- B) Uterine rupture
- C) Placental abruption with concealed haemorrhage
- D) Vasa praevia with fetal haemorrhage
- E) Preterm labour with cervical laceration
Question 8
- A) Placenta accreta — uterine atony at delivery
- B) Placenta increta — uterine perforation
- C) Placenta percreta — massive haemorrhage requiring hysterectomy
- D) Placenta praevia only — cord prolapse
- E) Placenta accreta — disseminated intravascular coagulation (DIC) only
Question 9
- A) This is a major placenta praevia — immediate caesarean section is required
- B) This is a minor/low-lying placenta — vaginal delivery may be considered with caution
- C) This is placental abruption — deliver immediately by caesarean section
- D) This is vasa praevia — emergency caesarean section is required
- E) This is a cervical ectropion — no obstetric intervention required
Question 10
- A) HELLP syndrome
- B) Immune thrombocytopaenic purpura (ITP)
- C) Disseminated intravascular coagulation (DIC)
- D) von Willebrand disease exacerbation
- E) Thrombotic thrombocytopaenic purpura (TTP)
TWIN PREGNANCY
Question 11
- A) Monochorionic diamniotic — always monozygotic
- B) Dichorionic diamniotic — always dizygotic
- C) Dichorionic diamniotic — may be dizygotic or monozygotic
- D) Monochorionic monoamniotic — always monozygotic
- E) Dichorionic monoamniotic — may be dizygotic or monozygotic
Question 12
- Twin A: deepest vertical pocket (DVP) 10 cm, bladder visible, tricuspid regurgitation on cardiac Doppler
- Twin B: DVP 1.8 cm, bladder not visible on repeat scan, absent end-diastolic flow in umbilical artery
- A) Quintero Stage I — conservative management with fortnightly scans
- B) Quintero Stage II — amnioreduction of Twin A's sac
- C) Quintero Stage III — selective laser photocoagulation of communicating placental vessels
- D) Quintero Stage IV — emergency caesarean section
- E) Quintero Stage III — selective feticide of Twin B
Question 13
- A) Immediate delivery by caesarean section at 28 weeks
- B) Outpatient surveillance with weekly ultrasound scans
- C) Inpatient monitoring with continuous CTG, planned delivery at 32–34 weeks
- D) Laser cord coagulation of one cord to prevent further entanglement
- E) Amnioinfusion to separate the cords
Question 14
- A) Immediate emergency caesarean section for Twin B
- B) Await spontaneous conversion to longitudinal lie for up to 2 hours
- C) Perform external cephalic version (ECV), and if unsuccessful, caesarean section
- D) Internal podalic version (IPV) followed by breech extraction
- E) Administer tocolysis and reassess in 30 minutes
Question 15
- A) Intracardiac potassium chloride (KCl) injection into Twin A
- B) Radiofrequency ablation (RFA) of Twin A's umbilical cord
- C) Amniodrainage from Twin A's sac to reduce its growth
- D) Expectant management — trisomy 18 fetuses almost always spontaneously demise
- E) KCl injection is contraindicated; use bipolar cord coagulation instead
Explanations
- Stage I: DVP discordance only (≥8 cm recipient, ≤2 cm donor); both bladders visible
- Stage II: Donor bladder not visible
- Stage III: Abnormal Doppler (absent/reversed end-diastolic flow in donor UA, or abnormal ductus venosus/umbilical venous flow)
- Stage IV: Hydrops in either twin
- Stage V: Death of one or both twins
- Inpatient admission from 26–28 weeks for intensive CTG monitoring
- Planned elective caesarean section at 32–34 weeks before spontaneous labour (which increases cord tension dynamics and the risk of acute occlusion)
Diagnosis and features of pregnancy trimester wise in a flow chart
diagnosis of pregnancy signs symptoms first trimester presumptive probable positive
second third trimester pregnancy features fetal movements quickening fundal height
DIAGNOSIS + FEATURES OF PREGNANCY (TRIMESTER-WISE FLOW CHART)
START
|
v
Woman with amenorrhea / pregnancy symptoms
|
v
STEP 1: Confirm pregnancy biologically
|
|-- Urine pregnancy test (UPT, detects beta-hCG)
| |
| |-- Negative --> Re-test in 1 week if suspicion high / evaluate other causes of amenorrhea
| |
| `-- Positive --> Proceed to clinical + ultrasound dating
|
v
STEP 2: Trimester-wise evaluation
|
+----------------------------------------------------------------------------------+
| FIRST TRIMESTER (0 to 13+6 weeks) |
+----------------------------------------------------------------------------------+
| DIAGNOSIS CLUES |
| - Presumptive: amenorrhea, nausea/vomiting, breast tenderness, fatigue, |
| urinary frequency |
| - Probable: uterine enlargement, Goodell sign (soft cervix), |
| Hegar sign (soft isthmus), Chadwick sign (bluish cervix/vagina) |
| - Positive (confirmatory): |
| * TVS: gestational sac ~4.5-5 wk |
| * Yolk sac ~5-5.5 wk |
| * Fetal pole ~5.5-6 wk |
| * Cardiac activity ~6 wk |
| KEY FEATURES |
| - Organogenesis phase |
| - Uterus remains pelvic till ~12 weeks |
| - Common symptoms: morning sickness, smell aversion, mild weight fluctuation |
| EXAM/TEST FOCUS |
| - Dating scan + viability |
| - Baseline antenatal labs (CBC, blood group/Rh, infection screening, etc.) |
+----------------------------------------------------------------------------------+
|
v
+----------------------------------------------------------------------------------+
| SECOND TRIMESTER (14 to 27+6 weeks) |
+----------------------------------------------------------------------------------+
| DIAGNOSIS/FOLLOW-UP MARKERS |
| - Quickening (maternal perception of fetal movement): |
| * ~18-20 wk primigravida |
| * ~16-18 wk multigravida |
| - Fetal heart sounds on Doppler (~10-12 wk onward; routine each visit) |
| - Fundal height becomes reliable from ~20 wk (cm ≈ weeks) |
| KEY FEATURES |
| - Uterus becomes abdominal organ |
| - Nausea usually improves |
| - Maternal weight gain becomes steady |
| - Pigmentary changes: linea nigra, chloasma; striae may appear |
| EXAM/TEST FOCUS |
| - Anomaly scan (~18-22 wk) |
| - Fundal height + fetal heart tones + movement tracking at every visit |
+----------------------------------------------------------------------------------+
|
v
+----------------------------------------------------------------------------------+
| THIRD TRIMESTER (28 to term) |
+----------------------------------------------------------------------------------+
| DIAGNOSIS/FOLLOW-UP MARKERS |
| - Definite fetal movements felt regularly |
| - Fundal height continues serial growth trend |
| - Fetal lie, presentation, engagement assessed clinically + ultrasound as need |
| KEY FEATURES |
| - Increased backache, edema (physiologic mild), breathlessness, reflux |
| - Braxton Hicks contractions |
| - Frequency of micturition may reappear near term (lightening) |
| EXAM/TEST FOCUS |
| - Growth surveillance, BP/proteinuria checks |
| - Fetal wellbeing assessment (movement count, NST/BPP when indicated) |
| - Delivery planning and risk stratification |
+----------------------------------------------------------------------------------+
|
v
END POINT: Pregnancy confirmed + trimester-specific maternal/fetal monitoring
- a one-page exam revision table, or
- a cleaner visual Mermaid flowchart you can paste into notes/PPT.
2
429 RESOURCE_EXHAUSTED. {'error': {'code': 429, 'message': 'Resource exhausted. Please try again later. Please refer to https://cloud.google.com/vertex-ai/generative-ai/docs/error-code-429 for more details.', 'status': 'RESOURCE_EXHAUSTED'}}
We encountered an error while processing your request. Please try again or reach out to us for help.
2
flowchart TD
A[Woman with amenorrhea / pregnancy symptoms] --> B[Step 1: Confirm pregnancy<br/>Urine pregnancy test: beta-hCG]
B -->|Negative| B1[Re-test in 1 week if suspicion high<br/>Evaluate other causes of amenorrhea]
B -->|Positive| C[Proceed to trimester-wise evaluation]
C --> T1
subgraph T1[First Trimester 0 to 13+6 weeks]
direction TB
T1a[Presumptive signs:<br/>Amenorrhea, nausea/vomiting, breast tenderness,<br/>fatigue, urinary frequency]
T1b[Probable signs:<br/>Goodell sign, Hegar sign, Chadwick sign,<br/>uterine enlargement]
T1c[Positive / confirmatory signs:<br/>TVS gestational sac: 4.5-5 weeks<br/>Yolk sac: 5-5.5 weeks<br/>Fetal pole: 5.5-6 weeks<br/>Cardiac activity: ~6 weeks]
T1d[Focus:<br/>Dating + viability scan<br/>Baseline antenatal labs]
T1a --> T1b --> T1c --> T1d
end
T1 --> T2
subgraph T2[Second Trimester 14 to 27+6 weeks]
direction TB
T2a[Quickening:<br/>Primigravida: 18-20 weeks<br/>Multigravida: 16-18 weeks]
T2b[Fetal heart tones:<br/>Doppler detectable from ~10-12 weeks,<br/>checked each visit]
T2c[Fundal height:<br/>Reliable from ~20 weeks<br/>Fundal height in cm ≈ gestational age in weeks]
T2d[Features:<br/>Uterus becomes abdominal organ<br/>Nausea improves<br/>Linea nigra, chloasma, striae may appear]
T2e[Focus:<br/>Anomaly scan: 18-22 weeks<br/>Routine growth and fetal surveillance]
T2a --> T2b --> T2c --> T2d --> T2e
end
T2 --> T3
subgraph T3[Third Trimester 28 weeks to term]
direction TB
T3a[Regular fetal movements clearly perceived]
T3b[Serial fundal height and growth trend assessment]
T3c[Fetal lie, presentation, engagement assessment]
T3d[Features:<br/>Backache, physiologic edema, breathlessness,<br/>reflux, Braxton Hicks contractions]
T3e[Focus:<br/>BP/proteinuria monitoring<br/>Fetal wellbeing: movement count, NST/BPP if indicated<br/>Delivery planning]
T3a --> T3b --> T3c --> T3d --> T3e
end
T3 --> Z[End point:<br/>Pregnancy confirmed and monitored trimester-wise]