Bronchail artery embolization full details for md exams 15marks answer
Reading File
Finding Sources
Finding Sources
Searching PubMed
"bronchial artery embolization" AND hemoptysis
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Searching the Web
bronchial artery embolization procedure technique indications complications MD exam review
Reading Web Page
bronchial artery embolization indications technique complications outcomes
Searching Images
bronchial artery embolization angiography hemoptysis procedure
I now have all the material needed for a comprehensive MD exam answer. Let me compose it.
Bronchial Artery Embolization (BAE)
A Complete MD Examination Answer (15 Marks)
1. Introduction
Bronchial Artery Embolization (BAE) is a minimally invasive, catheter-based endovascular procedure used to achieve hemostasis in patients with massive or recurrent hemoptysis by selectively occluding the hypertrophied bronchial arteries responsible for bleeding. It is now considered the first-line definitive procedure for managing significant hemoptysis before definitive surgery.
"Bronchial artery embolization is considered a first-line definitive procedure for managing hemoptysis. Bronchial artery embolization may control brisk bleeding in 75–90% of patients, permitting the definitive surgical procedure to be done more safely if it is appropriate." — Harrison's Principles of Internal Medicine 22E (2025)
2. Anatomy of the Bronchial Arteries
Understanding normal and variant anatomy is critical for successful BAE.
| Feature | Details |
|---|---|
| Origin | Usually arise from the descending thoracic aorta between T5–T6 |
| Normal number | Typically 2 on the left, 1 on the right (total 2–4) |
| Right bronchial artery | Commonly arises as an intercostobronchial trunk (ICBT) shared with the 3rd right intercostal artery |
| Left bronchial arteries | Usually arise directly from the aorta |
| Ectopic origins | Can arise from subclavian, internal mammary, thyrocervical trunk, inferior phrenic arteries (~30%) |
| Spinal artery of Adamkiewicz | Arises between T8–L2, often shares a common trunk with the right bronchial artery — must be identified to avoid spinal cord ischemia |
In disease states (TB, bronchiectasis, fungal infections), bronchial arteries become hypertrophied, tortuous, and develop pathological shunts with pulmonary circulation.
3. Indications for BAE
Primary Indications:
- Massive hemoptysis: classically defined as >200–600 mL/24 hours (or any amount threatening life)
- Moderate hemoptysis (100–300 mL/24 hr) not responding to conservative management
- Recurrent hemoptysis preventing normal activity despite conservative measures
- First-line treatment before elective/semi-elective surgery
Common Underlying Causes:
- Pulmonary tuberculosis (most common cause in developing countries)
- Bronchiectasis (including cystic fibrosis)
- Aspergilloma / invasive fungal infections
- Lung abscess
- Lung carcinoma (central tumors)
- Pulmonary arteriovenous malformations
- Rasmussen's aneurysm (mycotic pulmonary artery aneurysm in TB cavities)
- Dieulafoy disease (tortuous dysplastic submucosal artery)
Contraindications:
- Allergy to contrast (relative — can premedicate)
- Severe coagulopathy (relative — correct first)
- Spinal artery arising from the target bronchial artery (requires superselective catheterization)
- Hemoptysis from pulmonary artery source (requires different approach)
4. Pre-Procedure Evaluation
Clinical Assessment:
- Airway stabilization is the absolute first priority
- Hemodynamic resuscitation (large-bore IV, blood products)
- Patient positioning: lateral decubitus with bleeding side down to protect contralateral lung
Imaging:
- Chest X-ray: First test; localizes lesion but blood soiling may obscure pathology
- Multidetector CT Angiography (MDCT-A): Preferred pre-procedure imaging
- Identifies source of bleeding with high sensitivity
- Delineates normal and abnormal bronchial/non-bronchial systemic arteries
- Maps ectopic origins and determines laterality
- Identifies Rasmussen's aneurysm, pseudoaneurysms, AV malformations
- Flexible Bronchoscopy: Used to localize bleeding to a specific lobe/segment, guides the angiographer; must be performed with excellent suction prepared
Laboratory: Coagulation profile, renal function (creatinine/GFR for contrast dosing), CBC, blood group & crossmatch
Consent: Written informed consent discussing risks, benefits, alternatives
Fasting: 6 hours for solids, 2 hours for clear liquids
5. Step-by-Step Procedure Technique
Step 1: Vascular Access
- Right common femoral artery is preferred access (5/6 Fr sheath over Seldinger technique)
- Brachial or radial access for ectopic bronchial arteries from subclavian branches (higher morbidity)
- Systemic heparin administered intra-arterially (2000–3000 units, weight-based)
Step 2: Aortography
- A 5 Fr catheter (Cobra, Simmons, or Judkins Right) is advanced to the descending thoracic aorta
- Preliminary aortogram at T5–T6 level to identify bronchial artery origins and ectopic vessels
- Identify all bronchial and non-bronchial systemic collateral arteries (internal mammary, intercostal, inferior phrenic)
Step 3: Selective Catheterization
- Selective catheterization of each bronchial artery
- Digital Subtraction Angiography (DSA) performed to identify:
- Hypertrophy and tortuosity of arteries
- Hypervascularity and neovascularity
- Contrast extravasation (active bleeding — pathognomonic but seen in only 5–10%)
- Bronchopulmonary shunting
- Spinal artery branches (anterior spinal artery — must be excluded before embolization)
Step 4: Superselective Catheterization
- A 3 Fr coaxial microcatheter (e.g., Progreat) advanced beyond the origin of the spinal artery to reduce risk of spinal cord ischemia
- Critical safety step when the anterior spinal artery is identified sharing a trunk with the target vessel
Step 5: Embolization
- Embolic material injected slowly under fluoroscopic guidance until radiographic stasis (cessation of flow) is achieved
- Bilateral embolization performed if bleeding is bilateral or site not localized
- If bleeding persists after BAE, pulmonary angiography performed at the same setting to exclude pulmonary artery source
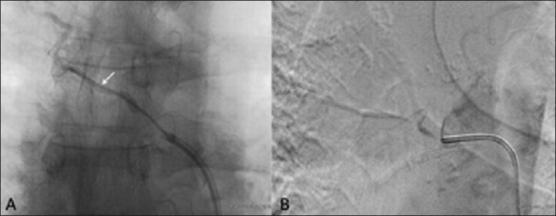
6. Embolic Agents
| Agent | Description | Preferred Use |
|---|---|---|
| Polyvinyl alcohol (PVA) particles | 300–500 µm (most common); 500–1000 µm if rapid shunting | First-line agent for most cases |
| Gelatin sponge (Gelfoam) | Temporary occlusion; used in earlier era; cheaper | Temporary adjunct |
| n-Butyl-2-cyanoacrylate (NBCA/Glue) | Permanent liquid embolic | Large tortuous arteries with rapid flow (bronchiectasis); recurrent bleeds with recanalization |
| Metallic coils | Permanent; flow-independent; lower spinal cord ischemia risk | Large vessel occlusion; sealing after distal particle embolization |
| Detachable coils | Enhanced precision | Tortuous or high-risk anatomy near spinal arteries |
Important: Gelfoam as sole agent is associated with high recanalization and recurrence. Coils alone are discouraged proximally as they prevent re-access for re-embolization.
7. Angiographic Features of Abnormal Bronchial Arteries
- Hypertrophied, tortuous bronchial arteries (diameter >2 mm)
- Dense parenchymal blush/hypervascularity
- Neovascularity
- Bronchopulmonary vascular shunting
- Pulmonary artery filling via shunts
- Active contrast extravasation (seen in ~5–10% — direct evidence of bleeding)
- Aneurysm or pseudoaneurysm formation
8. Outcomes and Efficacy
| Outcome Measure | Rate |
|---|---|
| Technical success (cannulation + embolization of all abnormal arteries) | 81–100% |
| Immediate clinical success (complete cessation of hemoptysis) | 70–99% |
| Recurrence rate (overall) | 10–57% (most within first year) |
| Long-term recurrence | 30–60% |
| Mortality | ~2% |
| Major complications | ~0.2–1.1% |
| Minor complications | ~10–13% |
(Data from multiple systematic reviews and meta-analyses, 2021–2025)
Causes of Recurrence:
- Incomplete initial embolization (missed vessels)
- Recanalization of embolized arteries
- Recruitment of new collateral vessels
- Progression of underlying disease
- Non-bronchial systemic collateral arteries not embolized
Factors associated with higher recurrence:
- Aspergilloma (most prone)
- Non-bronchial systemic collaterals
- Bronchopulmonary shunting
- Cystic fibrosis
- Active TB reactivation / multidrug-resistant TB
"Recurrence after bronchial artery embolization is less common in the setting of malignancy and active tuberculosis but does occur and can ultimately result in patient death." — Schwartz's Principles of Surgery
9. Complications
Minor Complications (Transient, Self-limiting)
| Complication | Notes |
|---|---|
| Post-embolization syndrome | Pleuritic chest pain, fever, dysphagia, leukocytosis; lasts 5–7 days; treated symptomatically |
| Chest pain | Due to ischemia of bronchial wall |
| Low-grade fever | Inflammatory response |
| Transient dysphagia | Esophageal branch ischemia |
| Groin hematoma at access site | Conservative management |
Major Complications (Serious, Rare)
| Complication | Mechanism | Prevention |
|---|---|---|
| Spinal cord ischemia / Transverse myelitis | Most dreaded; occurs if anterior spinal artery (artery of Adamkiewicz) inadvertently embolized; right ICBT is highest risk | Superselective microcatheter placement beyond spinal branch; careful pre-embolization DSA to identify spinal branches |
| Bronchial wall necrosis | Excessive embolization | Avoid over-embolization; use appropriate particle size |
| Esophageal necrosis | Embolization of esophageal branches | Superselective technique |
| Pulmonary infarction | Embolic material migrating beyond target into pulmonary circulation via shunts | Avoid small particles (<300 µm) if significant shunting |
| Myocardial infarction | Coronary branch involvement (rare) | Careful anatomy review |
| Cerebellar infarction | Embolization of arteries adjacent to the vertebral artery | Awareness of ectopic anatomy |
| Contrast nephropathy | Dose ≤3× GFR | Adequate hydration, limit contrast volume |
10. Role in Management Algorithm
Massive Hemoptysis
↓
Airway control (ETT, bronchial blocker, double-lumen tube)
↓
Hemodynamic resuscitation
↓
MDCT Angiography (if stable) → Localize source
↓
BAE (First-line definitive treatment)
↓
┌──────────────────┬──────────────────┐
Success Failure Recurrence
(70–99%) (pulmonary artery → Repeat BAE
source → PA → Consider surgery
angiography +
embolization)
→ Surgery (salvage)
Surgery is reserved for:
- Failure of embolization
- Aspergilloma (resection)
- Iatrogenic pulmonary artery injury
- Thoracic trauma
- Tracheoinnominate artery fistula
11. Special Situations
| Situation | Management |
|---|---|
| Aspergilloma | BAE controls acute bleeding; definitive surgical resection recommended as recurrence is very high |
| Cystic fibrosis | Repeat BAE often needed; high recurrence due to diffuse bronchiectasis |
| Lung cancer (central tumor) | BAE effective short-term; rare for permanent control; surgery if resectable |
| Active TB | BAE effective; treat underlying disease; lower recurrence than fungal disease |
| Bilateral hemoptysis | Bilateral bronchial arteriography and embolization performed |
| Failed BAE | Non-bronchial systemic collaterals (ICBT, internal mammary, inferior phrenic) must be sought |
12. Post-Procedure Care
- Vital signs monitoring every 30 minutes
- Assess groin access site for hematoma; check dorsalis pedis pulse and lower limb power
- Bed rest for 4–6 hours
- Analgesia as needed
- Hemoptysis assessment at 24 hours, 2–3 weeks, 3 months, and 6 months post-BAE
13. Summary Table
| Parameter | Key Points |
|---|---|
| Definition | Endovascular occlusion of hypertrophied bronchial arteries causing hemoptysis |
| First described | Remy et al., 1974 |
| Indication | Massive/moderate/recurrent hemoptysis |
| Access | Right common femoral artery (preferred) |
| Preferred imaging pre-procedure | Multidetector CT angiography |
| Target vessels | Bronchial arteries ± non-bronchial systemic collaterals |
| Preferred embolic agent | PVA particles 300–500 µm |
| Immediate success rate | 70–99% |
| Recurrence rate | 10–57% |
| Most dreaded complication | Spinal cord ischemia (transverse myelitis) |
| Prevention of spinal ischemia | Superselective microcatheter placement beyond spinal artery origin |
| Post-embolization syndrome | Pleuritic pain, fever, dysphagia — resolves in 5–7 days |
| Surgery indication | Failure of embolization; aspergilloma |
Recent Evidence (2024–2025)
Two high-level systematic reviews confirm BAE superiority over conservative management (PMID 39215233 — BMC Pulmonary Medicine 2024) and a 2025 systematic review provides updated insights on outcomes and technical considerations (PMID 41286932 — BMC Pulmonary Medicine 2025). These reinforce BAE as the standard of care for massive hemoptysis.
Sources: Harrison's Principles of Internal Medicine 22E (2025); Murray & Nadel's Textbook of Respiratory Medicine; Schwartz's Principles of Surgery 11E; Tintinalli's Emergency Medicine; ICRI Guidelines on BAE (2020); Fan S et al., BMC Pulm Med 2024 (PMID 39215233); Ravetta P et al., BMC Pulm Med 2025 (PMID 41286932).
This is a shared conversation. Sign in to Orris to start your own chat.