Diaphragmatic hernia for pediatrics surgery everything
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
I now have comprehensive, well-sourced content. Here is the full synthesis:
Congenital Diaphragmatic Hernia (CDH) — Pediatric Surgery
1. Definition & Overview
Congenital diaphragmatic hernia (CDH) is a structural defect occurring during embryonic life where abdominal organs herniate into the thorax through a diaphragmatic defect, leading to pulmonary hypoplasia and persistent pulmonary hypertension (PPH). It is not a single clinical entity — outcomes are accordingly diverse.
- Incidence: 1:2,000–5,000 births (some sources report 1:10,000–4:10,000)
- Survival: 65%–90% at major centers with modern management
- Isolated CDH: ~60% of cases; when associated with other anomalies, mortality >85%
— Sabiston Textbook of Surgery, p. 2675; Fanaroff & Martin's Neonatal-Perinatal Medicine
2. Embryology & Types
The diaphragm derives from:
- Septum transversum
- Pleuroperitoneal folds
- Components of the abdominal wall
- Dorsal mesentery
Fusion begins at 3–4 weeks gestation and is typically complete by 9 weeks. Incomplete fusion leads to:
| Type | Location | Frequency |
|---|---|---|
| Bochdalek (posterolateral) | Left (85%), Right (13%), Bilateral (2%) | 70%–75% |
| Morgagni (anterior/retrosternal) | Central/anterior | 23%–28% |
| Central | Central tendon | 2%–7% |
Bochdalek hernias are by far the most clinically significant in the neonatal period.
— Sabiston Textbook of Surgery, p. 2675
3. Pathophysiology
Once abdominal contents herniate into the thorax:
- Pulmonary hypoplasia — compression of the developing ipsilateral lung → fewer bronchial branches, reduced alveolar surface area; contralateral lung also affected
- Abnormal pulmonary vasculature — increased arteriolar smooth muscle thickness; hypersensitive to vasoactive stimuli
- Persistent pulmonary hypertension (PPH) — right-to-left shunting across PDA/PFO → severe hypoxemia
- Mediastinal shift — compresses the contralateral lung
The severity of pulmonary hypoplasia and PPH is the primary determinant of morbidity and mortality.
4. Prenatal Diagnosis & Prognostication
Diagnosis
- Routine ultrasound detects ~2/3 of cases prenatally, as early as 15 weeks gestation
- Sonographic findings at 22–24 weeks:
- Left-sided CDH: rightward cardiac/mediastinal shift, stomach/intestinal loops in chest, polyhydramnios
- Right-sided CDH: liver in right chest, leftward mediastinal shift, harder to diagnose (liver echogenicity similar to fetal lung)
- Liver herniation: difficult to distinguish sonographically; Doppler of umbilical vein/hepatic vessels helps
Differential Diagnosis (prenatal)
- CPAM, bronchogenic/enteric cysts, bronchopulmonary sequestration, bronchial atresia — no intra-abdominal organ displacement
Prognostic Markers
Lung-to-Head Ratio (LHR):
- Ratio of contralateral lung area to head circumference
- LHR < 1.0 → poor prognosis
- LHR > 1.4 → ~100% survival
Observed/Expected LHR (O/E LHR):
- Compares measured LHR to reference values for gestational age
- O/E LHR < 25% → survival < 20%
- More reliable than absolute LHR across gestational ages
Total Fetal Lung Volume (TFLV) by MRI:
- Used as supplement to ultrasound
- Predictive of mortality, ECMO use, and oxygen requirement at 30 days
Other poor prognostic factors:
- Liver herniation ("liver up")
- Right-sided CDH
- Associated cardiac or chromosomal anomalies
- Stomach herniation
— Sabiston Textbook of Surgery, p. 2715–2716; Fanaroff & Martin's Neonatal-Perinatal Medicine
5. Genetic Workup
- Genetic aberrations (trisomies, microdeletions, single-gene defects) occur in 6%–30% of CDH cases
- Array CGH / genome-wide comparative hybridization recommended
- ~30–40% of CDH cases have associated anomalies (congenital heart disease most common)
- Karyotyping + detailed fetal echo should be offered at diagnosis
6. Fetal Intervention — Fetal Endoscopic Tracheal Occlusion (FETO)
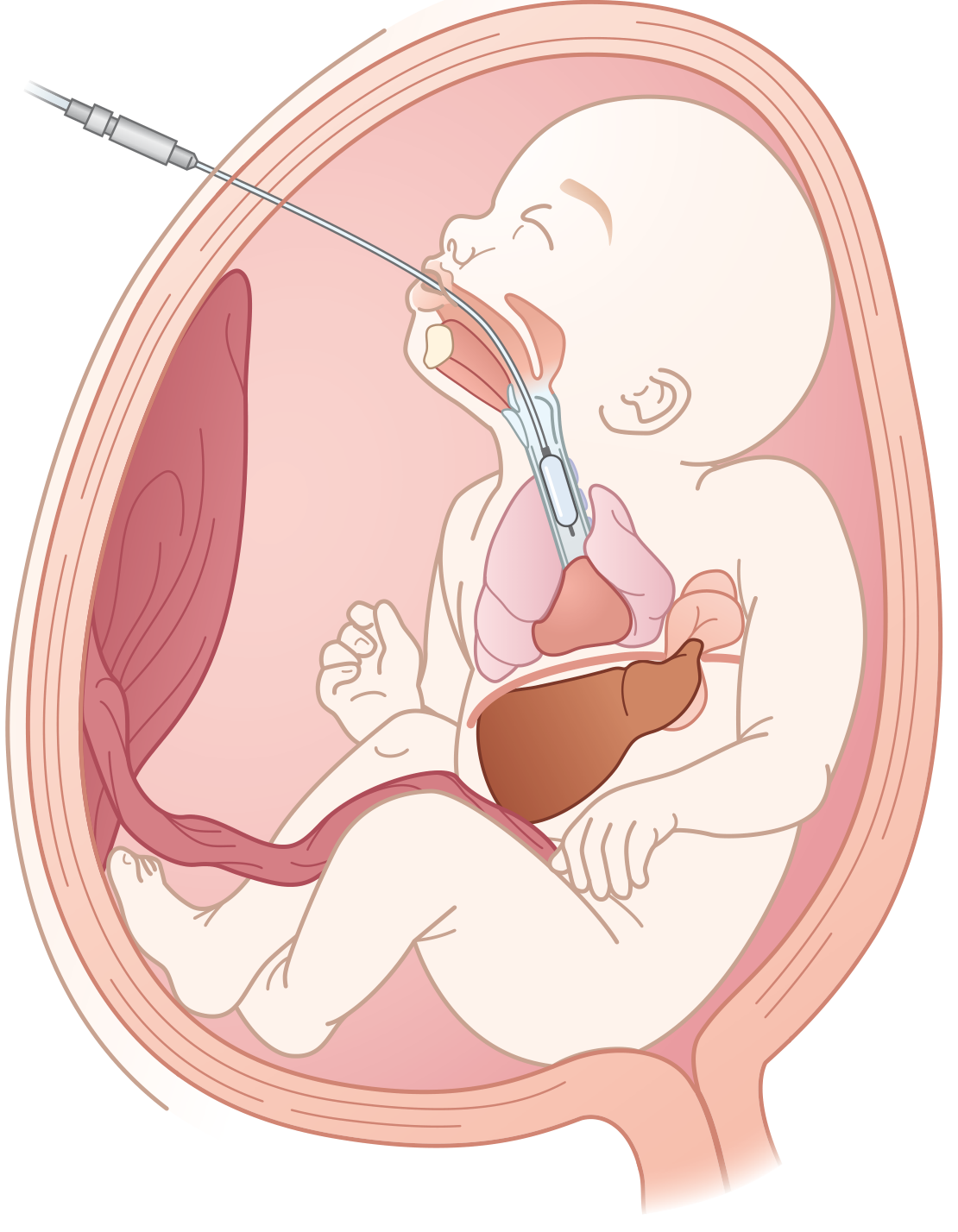
Rationale: Tracheal occlusion prevents egress of fetal lung fluid → fluid accumulation → lung growth and development
History:
- Open fetal CDH repair attempted in 1990s — no survival benefit over postnatal repair (even in severe cases) and increased prematurity risk
- Tracheal occlusion via metallic clip → tracheal stenosis, 15% survival
- Fetoscopic balloon (FETO) developed as minimally invasive alternative
The TOTAL Trials (landmark RCTs):
| Trial Arm | Criteria | Result |
|---|---|---|
| Severe TOTAL | O/E LHR <25% | 40% FETO survival vs. 15% expectant (stopped early for efficacy) |
| Moderate TOTAL | O/E LHR 25%–35% | No survival benefit vs. expectant management |
Current practice:
- FETO offered at select fetal centers for severe left-sided CDH (liver up + O/E LHR <25%)
- Balloon inserted fetoscopically at 27–29 weeks, removed at 34 weeks (or emergently if preterm labor)
- Main risk: preterm premature rupture of membranes (PPROM), preterm delivery
- Balloon must be removed before delivery to allow airway
— Sabiston Textbook of Surgery, p. 2716
7. Postnatal Clinical Presentation
Immediate neonatal presentation (Bochdalek):
- Respiratory distress at birth (tachypnea, grunting, cyanosis)
- Scaphoid abdomen (bowel in chest)
- Barrel-shaped chest
- Absent breath sounds on affected side (usually left)
- Bowel sounds auscultated in chest
- Heart sounds shifted to contralateral side
- NG tube coiling in chest on X-ray
Chest X-Ray findings:
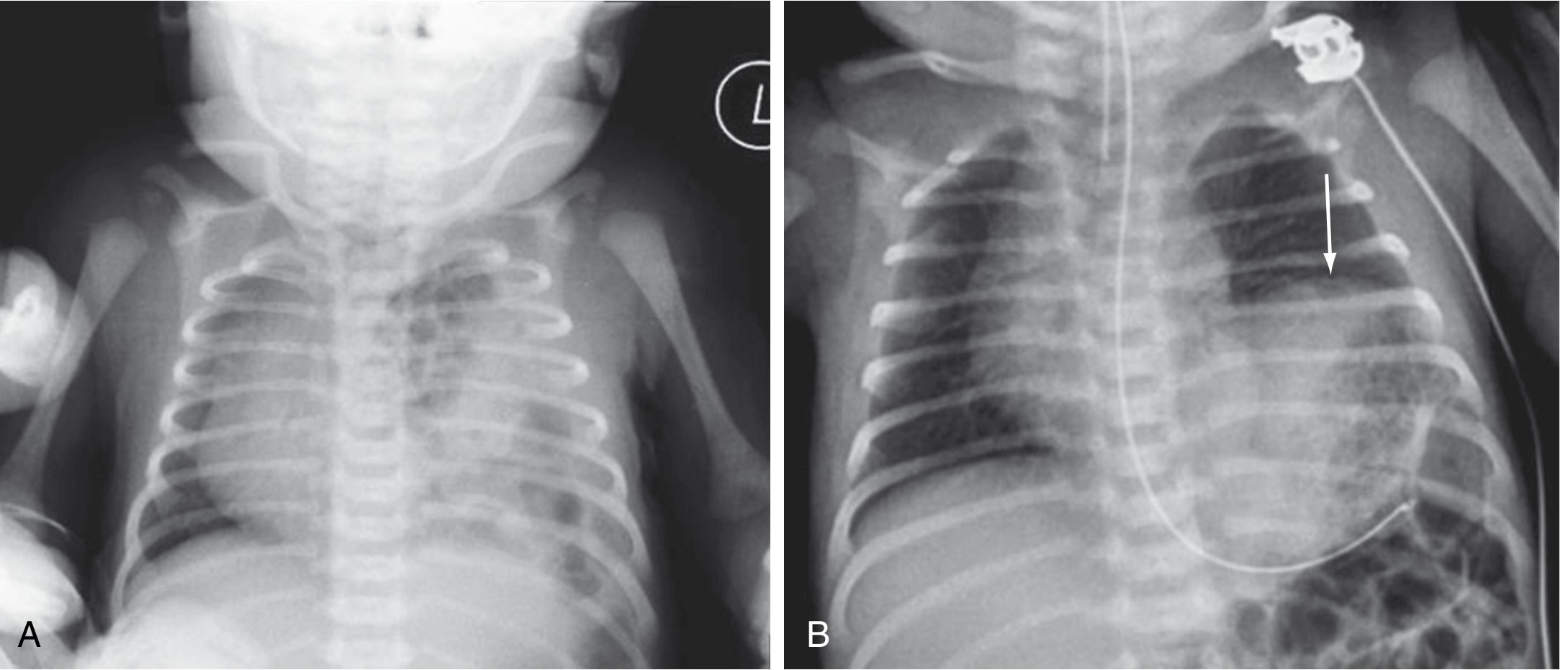
- Multiple gas-filled bowel loops in the hemithorax
- Mediastinal/cardiac shift to contralateral side
- Contralateral lung compression
- Paucity of bowel gas in abdomen
Delayed presentation (Morgagni / small Bochdalek):
- Can present weeks to years later with recurrent respiratory infections, GI symptoms, failure to thrive
8. Initial Stabilization & Resuscitation
Key principle: Stabilize FIRST, operate SECOND (surgery is not an emergency)
Airway & Ventilation
- Immediate intubation — avoid bag-mask ventilation (inflates herniated bowel → worsens lung compression)
- Gentle ventilation with low peak inspiratory pressures (PIP <25 cmH₂O) to prevent barotrauma to hypoplastic lungs
- Target: preductal SpO₂ 85%–95%, permissive hypercapnia (PaCO₂ 45–60 mmHg), preductal pH >7.25
- High-frequency oscillatory ventilation (HFOV) for refractory hypoxemia
Decompression
- Orogastric tube (Replogle) on suction — decompresses herniated bowel
Pulmonary Hypertension Management
- Inhaled nitric oxide (iNO): pulmonary vasodilator — first-line for PPH
- Sildenafil (PDE5 inhibitor): oral/IV pulmonary vasodilator
- Milrinone: inotrope + pulmonary vasodilator
- Avoid hypoxia, hypercarbia, acidosis, hypothermia — all worsen PPH
ECMO (Extracorporeal Membrane Oxygenation)
Indications when maximal medical therapy fails:
- Oxygenation index (OI) >40
- Unable to achieve preductal SpO₂ >85% despite iNO + maximal vent support
- VA-ECMO preferred (cardiac + respiratory support)
- VV-ECMO if cardiac function adequate
- Contraindications: severe pulmonary hypoplasia incompatible with survival, irreversible non-pulmonary conditions, gestational age <34 weeks / weight <2 kg (relative)
- Timing of surgery: can repair on ECMO or after decannulation — center-dependent
Target "Honeymoon Period"
After initial stabilization, a "honeymoon period" of relative improvement may occur before PPH worsens. Repair is planned once:
- Pulmonary hypertension is controlled/stabilized
- Ventilator settings are improving or stable
- iNO weaning initiated
- Adequate urine output
9. Surgical Repair
Timing
- NOT emergent — delay until physiologically stable (typically 24–72 hrs or longer)
- If on ECMO: may repair on ECMO to allow lung recovery
Approach
Open Repair:
- Subcostal (transabdominal) incision — most common in neonates; allows reduction of all herniated contents, patch repair if needed
- Thoracotomy approach: used in some right-sided CDH or for thoracoscopic approach
Minimally Invasive (Thoracoscopic/Laparoscopic) Repair:
- Increasingly used in hemodynamically stable, non-ECMO patients
- Advantages: less pain, faster recovery, smaller scars
- Risks: hypercapnia/acidosis from CO₂ insufflation in neonates with already compromised lungs, higher recurrence rates with patch repairs
Steps of Repair
- Reduce herniated abdominal contents back into abdomen
- Identify diaphragmatic defect margins
- Primary repair (preferred) — interrupted non-absorbable sutures if edges can be approximated without tension
- Patch repair (for large defects without adequate tissue) — using:
- Gore-Tex (PTFE) — most common synthetic patch
- Biological/acellular dermal matrix
- Muscle flap (latissimus dorsi, abdominal wall)
- Chest tube placement: controversial — most centers do NOT routinely place chest tubes to avoid mediastinal shift; if placed, kept on water seal (not suction)
- Gastrostomy: may be placed for anticipated feeding difficulties
Primary vs. Patch Repair
| Primary Repair | Patch Repair | |
|---|---|---|
| Indication | Small–moderate defects | Large defects, absent diaphragm |
| Recurrence | Low (~5%) | Higher (up to 50% for large patches) |
| Preferred material | Suture | Gore-Tex / bioprosthetic |
10. Postoperative Management
- Continue PPH management — do NOT expect immediate improvement
- Pulmonary hypertension may worsen 24–48 hrs post-repair (common)
- Continue iNO, sildenafil, milrinone as needed
- Extubation when off or minimal respiratory support
- Early enteral feeds (via NG or gastrostomy) once stable
- Watch for recurrence (especially with patch repairs)
11. Complications & Long-Term Morbidity
| Complication | Details |
|---|---|
| Pulmonary hypertension | Persists or recurs; may need chronic sildenafil |
| Bronchopulmonary dysplasia (BPD) | From barotrauma, O₂ toxicity |
| Gastroesophageal reflux (GER) | Very common (up to 50–80%); may need fundoplication |
| Feeding difficulties | Oral aversion, aspiration; often need G-tube |
| Hernia recurrence | 5% primary repair; up to 50% large patch repairs |
| Chest wall deformity | Scoliosis, pectus excavatum |
| Neurodevelopmental delay | Especially post-ECMO (sensorineural hearing loss, cognitive delays) |
| Intestinal obstruction | Malrotation, adhesions |
| Failure to thrive | Multifactorial |
12. Morgagni Hernia
- Anterior (retrosternal) defect through the foramen of Morgagni
- Usually presents later (beyond infancy) — often incidental or with recurrent chest infections, GI symptoms
- Less physiologically severe than Bochdalek
- CXR: anterior mediastinal mass or air-fluid levels in right cardiophrenic angle
- Repair: laparoscopic approach preferred; generally excellent outcomes
- Primary suture repair usually sufficient
13. Diaphragmatic Eventration
- Not a true hernia — thinned, non-muscularized diaphragm that paradoxically elevates with respiration
- Cause: phrenic nerve injury (birth trauma, cardiac surgery), developmental failure of muscularization
- May mimic CDH on imaging
- Management: plication of the diaphragm (open or thoracoscopic) when symptomatic
14. Survival & Prognosis Summary
| Factor | Better Prognosis | Worse Prognosis |
|---|---|---|
| Side | Right? | Left more common, bilateral worst |
| Liver position | Liver down | Liver up ("liver herniation") |
| O/E LHR | >35–45% | <25% |
| Associated anomalies | None (isolated) | Cardiac defect, chromosomal |
| ECMO requirement | Not needed | Required |
| Defect size | Small, primary repair | Large, requires patch |
Overall survival at experienced centers: 65%–90% (down from <50% in older era).
Sources:
- Sabiston Textbook of Surgery, 21st ed., Ch. 117 & 118 (pp. 2675–2716)
- Fanaroff & Martin's Neonatal-Perinatal Medicine, Ch. 34
This is a shared conversation. Sign in to Orris to start your own chat.