Diaphragmatic hernia for paediatric surgery everything
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Reading File
I now have comprehensive content from all three textbooks. Let me compile the complete answer.
Congenital Diaphragmatic Hernia (CDH) — Paediatric Surgery
1. Definition & Overview
Congenital diaphragmatic hernia (CDH) is a spectrum of developmental anomalies marked by a defect in the diaphragm that allows abdominal contents to herniate into the thoracic cavity, disrupting lung and pulmonary vascular development. The spectrum ranges from a small posterior muscle-rim defect to complete diaphragmatic agenesis.
- Incidence: ~1:2,000–5,000 live births (some studies cite 1:10,000–4:10,000)
- Survival: 65%–90% at high-volume centres
- 85% left-sided (Bochdalek), 13% right-sided, <2% bilateral/agenesis
- 60% are isolated; if associated with other anomalies, mortality >85%
— Sabiston Textbook of Surgery, p. 2675; Creasy & Resnik's Maternal-Fetal Medicine, p. [CDH section]
2. Types of Diaphragmatic Hernia
| Type | Location | Features |
|---|---|---|
| Bochdalek hernia | Posterolateral (pleuroperitoneal canal) | Most common; 85% left-sided; presents in neonatal period |
| Morgagni hernia | Anterior (retrosternal / parasternal) | Right-sided more common; often asymptomatic; delayed diagnosis into childhood |
| Central/agenesis | Central tendon | Rare; bilateral possible |
| Hiatal hernia | Oesophageal hiatus | Sliding or paraesophageal |
3. Embryology & Pathophysiology
The diaphragm forms between weeks 4–12 of embryonic life from four components:
- Septum transversum
- Pleuroperitoneal folds
- Dorsal mesentery of oesophagus
- Lateral body wall
Failure of the pleuroperitoneal canal to close (usually the left side) creates the Bochdalek defect. Herniated abdominal viscera compress the developing lung, causing:
- Pulmonary hypoplasia — bilateral; fewer airway branches, reduced lung compliance, abnormal pulmonary vasculature
- Pulmonary hypertension — persistent fetal-type circulation; right-to-left shunting through PDA/PFO
- Mediastinal shift — with contralateral lung compression
Associated anomalies (in 40% of cases):
- Congenital heart disease (most common)
- Chromosomal anomalies (trisomies 13, 18, 21)
- Neural tube defects, urogenital anomalies
4. Prenatal Diagnosis & Assessment
Ultrasound Findings
- Abdominal organs (bowel loops, stomach, liver) visualised in the chest
- Cardiac deviation/mediastinal shift
- Absence of normal diaphragmatic contour
- Polyhydramnios (bowel obstruction)
- Peristalsis of bowel in thorax
Left CDH: rightward heart shift + echolucent stomach/intestine in thorax
Right CDH: harder to diagnose (liver similar to fetal lung echo); leftward heart shift; use Doppler of umbilical/hepatic veins + gallbladder position
Can be detected as early as 15 weeks gestation; 22–24 weeks standard.
Prognostic Tools
| Tool | Description | Interpretation |
|---|---|---|
| LHR (Lung-to-Head Ratio) | Contralateral lung area ÷ head circumference | <1.0 = poor prognosis; >1.4 = ~100% survival |
| O/E LHR | Observed ÷ expected LHR for gestational age | O/E <25% → survival <20% |
| Liver herniation | Liver "up" in chest | Independent predictor of worse outcome |
| Total fetal lung volume (MRI) | Best quantification of lung volume | O/E TFLV <25–35% = severe |
Diagnosis should prompt referral to a tertiary centre for advanced imaging, genetic testing (karyotype/microarray), fetal echocardiography, and multidisciplinary counselling.
5. Chest X-Ray (Classic Findings)
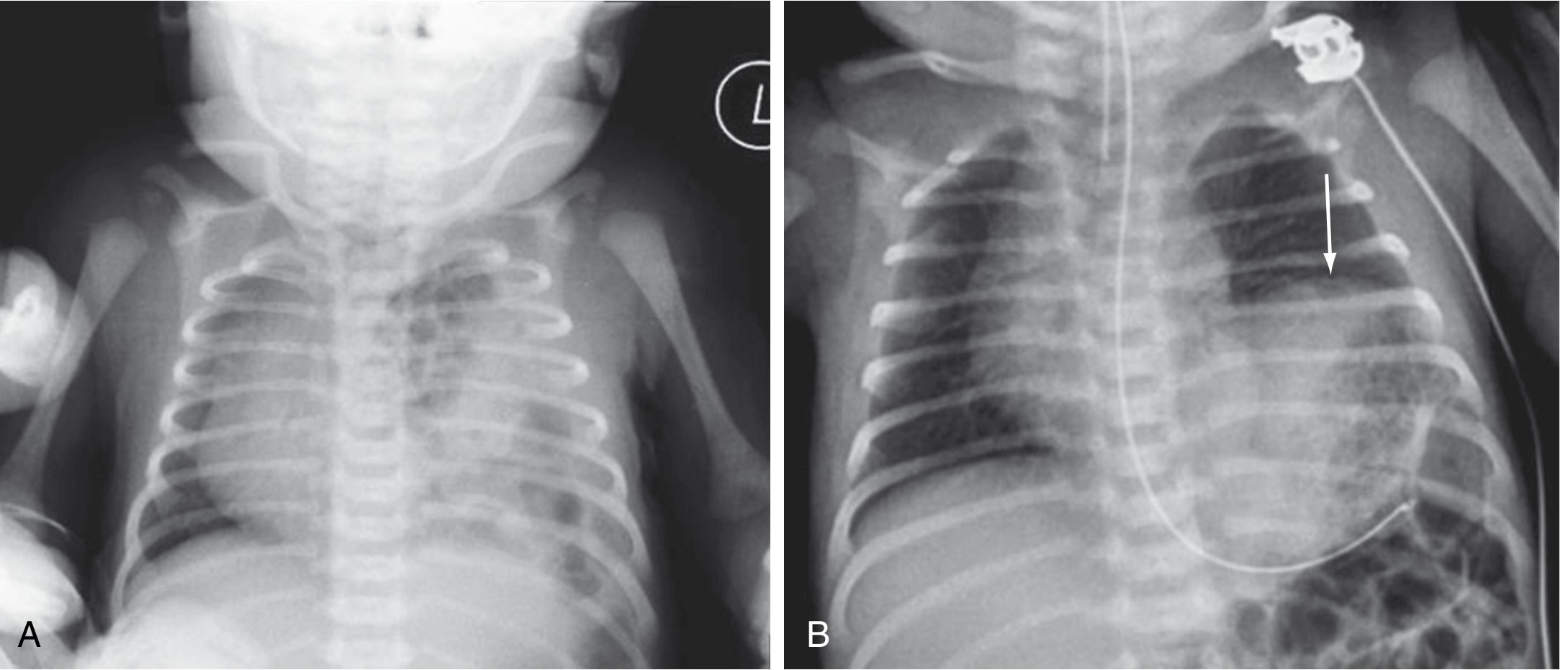
Figure: (A) CDH — multiple gas-filled bowel loops in left hemithorax; mediastinum shifted right. (B) Left diaphragmatic eventration — elevated hemidiaphragm (arrow) from phrenic nerve injury.
Classic CXR findings:
- Multiple gas-filled bowel loops in hemithorax
- Mediastinal shift to contralateral side
- Scarcity of bowel gas in abdomen
- Nasogastric tube coiling in thorax (if stomach herniated)
- Absent left diaphragmatic shadow
6. Clinical Presentation
Neonatal (most common)
- Respiratory distress at birth: grunting, dyspnea, retractions, cyanosis
- Scaphoid abdomen (bowel contents in chest)
- Diminished/absent breath sounds on affected side
- Bowel sounds in chest
- Displaced heart tones
- Pre- and post-ductal SpO₂ difference (right-to-left shunting through PDA)
Delayed Presentation (10–20% after 24 hours; Morgagni more common)
- Feeding difficulties
- Recurrent chest infections / pneumonia
- Respiratory distress triggered by respiratory illness
- Occasionally, incidental finding on CXR
7. Fetal Intervention — FETO
Fetoscopic Endoluminal Tracheal Occlusion (FETO):
- Tracheal balloon placed fetoscopically at 27–29 weeks gestation
- Occludes trachea → retained lung fluid → lung growth stimulus
- Balloon retrieved at ~34 weeks (or emergently if preterm labour)
TOTAL Trials (RCTs):
- Severe CDH (O/E LHR <25%): FETO → 40% survival vs 15% expectant (trial stopped early for efficacy); benefit sustained at 6 months
- Moderate CDH (O/E LHR 25–34.9%): No survival benefit shown at 30–34 weeks
Complications: Preterm labour, pPROM, preterm birth, fetal demise, tracheal stenosis
FETO is limited to specialist fetal therapy centres.
8. Postnatal Stabilisation — "Gentle Ventilation" Protocol
CDH repair is NOT a surgical emergency. Priority is cardiorespiratory stabilisation before surgery.
Airway & Ventilation
- Avoid bag-mask ventilation (air swallowing worsens herniation)
- Early intubation and nasogastric tube insertion for GI decompression
- "Gentilation" / lung-protective strategy:
- Peak inspiratory pressure (PIP) ≤25 cmH₂O
- Permissive hypercapnia (PaCO₂ 45–65 mmHg, pH >7.2)
- Pre-ductal SpO₂ target: 85–95%
- Avoid high PEEP (worsens pulmonary hypertension)
- High-frequency oscillatory ventilation (HFOV) if conventional fails
Pulmonary Hypertension Management
| Agent | Mechanism | Notes |
|---|---|---|
| Inhaled Nitric Oxide (iNO) | Selective pulmonary vasodilator | First-line for iPPHN |
| Sildenafil | PDE-5 inhibitor | Oral/IV; combination with iNO |
| Milrinone | PDE-3 inhibitor (inodilator) | For RV dysfunction |
| PGE₁ / Epoprostenol | Prostanoids | Second-line |
| Bosentan | Endothelin receptor antagonist | Emerging role |
ECMO (Extracorporeal Membrane Oxygenation)
Indicated when:
- FiO₂ 1.0 + optimal ventilation → pre-ductal SpO₂ <85% or PaO₂ <40 mmHg
- OI (Oxygenation Index) >40
- Refractory pulmonary hypertension
VA-ECMO preferred for CDH (RV support). Repair can be performed on ECMO but carries higher bleeding risk.
9. Surgical Repair
Timing
- Defer 48–72 hours after birth to allow haemodynamic stabilisation and reduce pulmonary vascular lability
- On ECMO: timing controversial — some centres repair on ECMO; others wean first
- Morgagni hernias: can be semi-elective
Approach
Open repair (standard):
- Subcostal (transabdominal) incision — provides best access, allows bowel reduction and patch placement
- Thoracotomy — used for thoracoscopic approach or Morgagni hernia
Minimally invasive (thoracoscopic / laparoscopic):
- Increasingly used in stable infants >2.5 kg
- Thoracoscopic approach most common for Bochdalek
- Advantages: reduced trauma, faster recovery, better cosmesis
- Concerns: hypercarbia and acidosis from CO₂ insufflation; higher recurrence rates in some series
Key Surgical Steps
- Reduction of herniated viscera into abdomen
- Assessment of diaphragmatic defect size
- Primary repair — if rim of posterior muscle present; non-absorbable sutures
- Patch repair — for large/absent rim
- Synthetic: Gore-Tex (PTFE) — most common; risk of recurrence ~10–50% with large defects
- Biological: porcine small intestinal submucosa (SIS), acellular dermal matrix
- Muscle flaps: latissimus dorsi, serratus anterior — for recurrences or agenesis
- Avoid creating pneumothorax on ipsilateral side — do NOT drain unless tension
- Contralateral chest: assess for undrained pneumothorax
Primary vs Patch Repair
- Small defect → primary closure (best outcomes, lowest recurrence)
- Large/absent muscle rim → patch required (recurrence up to 50% with synthetic patch)
- Patch recurrence presents with intestinal obstruction months–years later → requires re-exploration
10. Postoperative Management
- Continue gentle ventilation; pulmonary hypertension may worsen post-op (inflammatory response)
- Maintain iNO if pre-operatively required
- Ipsilateral pleural drain is NOT routinely placed (lung re-expansion is gradual over days–weeks)
- Early enteral feeding when tolerated
- Monitor for intestinal obstruction (adhesions, recurrent hernia)
11. Complications & Long-Term Morbidity
| Complication | Notes |
|---|---|
| Recurrent hernia | 10–50%; higher with patch; may present with obstruction years later |
| Pulmonary hypertension | Persistent; requires ongoing sildenafil/bosentan in some |
| Bronchopulmonary dysplasia | Chronic lung disease; oxygen dependence |
| GERD / Oesophagitis | Very common (>50%); often requires fundoplication |
| Feeding difficulties | Dysphagia, poor weight gain; G-tube in ~30% |
| Neurodevelopmental delay | Especially in ECMO survivors |
| Chest wall deformities | Pectus excavatum/carinatum |
| Scoliosis | Long-term surveillance required |
| Hearing loss | Especially after aminoglycosides/ECMO |
| Intestinal obstruction | Adhesions or hernia recurrence |
12. Diaphragmatic Eventration
Distinct from CDH — the diaphragm is intact but thinned/weakened (usually from phrenic nerve injury or congenital muscular weakness), causing paradoxical movement.
- Acquired: phrenic nerve injury during cardiac/thoracic surgery
- Congenital: true aplasia of diaphragmatic muscle fibres
Management: Diaphragmatic plication (thoracoscopic or open) — indicated for symptomatic cases with paradoxical movement and respiratory compromise. Especially in neonates who cannot wean from ventilation.
13. Summary Flowchart
Prenatal US diagnosis
↓
Tertiary referral → Echo + MRI + Genetics
↓
Severe (O/E LHR <25%)?
YES → Consider FETO at 27–29 wks
NO → Expectant + planned delivery at tertiary centre
↓
Delivery → Immediate intubation + NGT
↓
Stabilise: Gentle ventilation, iNO, pulmonary vasodilators
↓
Refractory? → ECMO
↓
Stable 48–72 hrs → Surgical repair
(Primary closure if small defect; patch if large)
↓
Long-term follow-up: GI, pulmonary, neuro, hearing
Key Teaching Points for Exams:
- CDH is a physiological emergency, not a surgical emergency — stabilise before operating
- Left-sided (85%); Bochdalek (posterolateral) most common
- Scaphoid abdomen + respiratory distress at birth = CDH until proven otherwise
- LHR <1.0 = poor prognosis; FETO for severe CDH improves survival (TOTAL trial)
- Permissive hypercapnia + avoid high PIP/PEEP = "gentilation"
- iNO = first-line pulmonary vasodilator
- Patch repair needed for large defects; recurrence is a major long-term problem
- GERD and feeding difficulties are nearly universal survivors' morbidities
— Sabiston Textbook of Surgery, pp. 2675–2676; Creasy & Resnik's Maternal-Fetal Medicine (CDH chapter); Current Surgical Therapy 14e
This is a shared conversation. Sign in to Orris to start your own chat.