Bone marrow cinical features
Reading File
Finding Sources
Finding Sources
Asking for Preferences
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
I now have comprehensive content to synthesize a thorough answer. Here is a full overview of Bone Marrow: Clinical Features.
Bone Marrow — Clinical Features
Overview of Bone Marrow Failure Syndromes
Bone marrow failure diseases share a common endpoint: deficient hematopoiesis leading to low blood counts. The major syndromes are:
| Syndrome | Dominant Feature |
|---|---|
| Aplastic anemia | Pancytopenia + hypocellular marrow |
| Myelodysplastic syndrome (MDS) | Dysplastic hematopoiesis ± cytopenias |
| Pure red cell aplasia (PRCA) | Isolated red cell precursor loss |
| Single-lineage cytopenias | Thrombocytopenia, neutropenia alone |
These must be distinguished from peripheral destruction of cells (hemolytic anemia, ITP, immune leukopenias), where the marrow itself is intact.
Aplastic Anemia
Definition
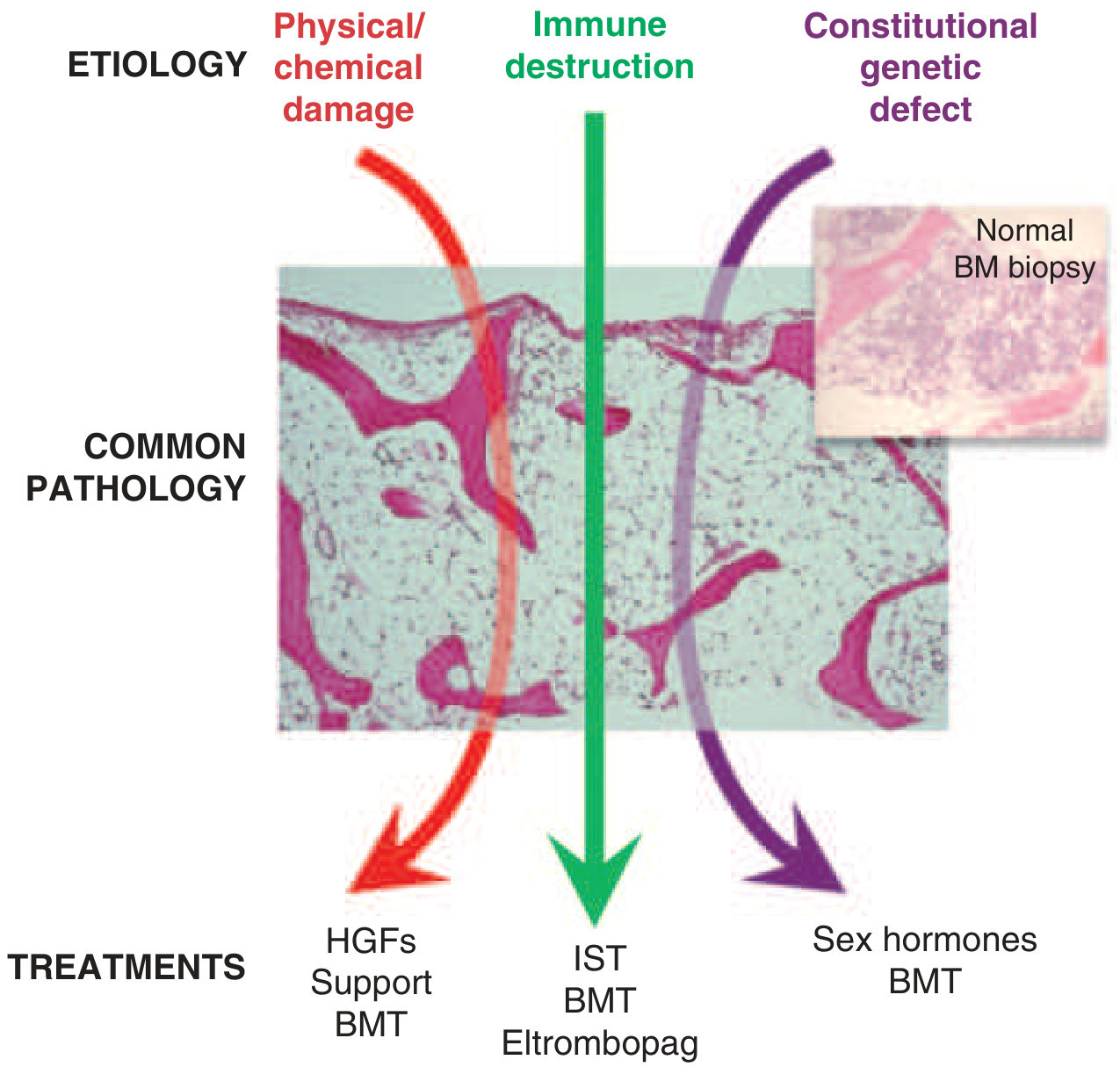
Aplastic anemia is pancytopenia with bone marrow hypocellularity. The empty marrow results from:
- Physical/chemical damage (benzene, radiation, cytotoxic chemotherapy)
- Immune-mediated destruction (most common acquired form — cytotoxic T cells targeting hematopoietic stem cells)
- Constitutional/genetic defects (Fanconi anemia, dyskeratosis congenita, telomere biology disorders)
Epidemiology
- Bimodal age distribution: children/young adults + second peak >60 years
- Rare in West: <2 cases/million/year; higher in Asia: 5–6 cases/million/year
- Males and females equally affected
Clinical Manifestations (Symptoms of Pancytopenia)
From anemia (low RBCs):
- Fatigue, weakness, pallor, headaches, exertional dyspnea
From thrombocytopenia (low platelets):
- Petechiae of skin and mucous membranes
- Epistaxis (nosebleeds)
- Gum/mucosal bleeding
From neutropenia (low WBCs):
- Fever
- Recurrent or severe bacterial/fungal infections
Some patients are identified incidentally on routine laboratory testing before symptoms develop.
Causes (Acquired)
- Idiopathic (majority)
- Medications (e.g., chloramphenicol, NSAIDs, antiepileptics)
- Viral infections: EBV, CMV, hepatitis A/B/C
- Radiation
- Autoimmune disease
- Pregnancy-associated
- Paroxysmal nocturnal hemoglobinuria (PNH)
Pathobiology
- Autoimmune attack by cytotoxic T cells → apoptosis of HSCs
- Usually normal cytogenetics (abnormal karyotype suggests MDS)
- ~60–70% have acquired clonal mutations on next-generation sequencing at diagnosis — these clones persist and can cause relapse or malignant transformation
Severity Classification
| Category | Criteria |
|---|---|
| Severe | Marrow cellularity <25% + ≥2 of: neutrophils <500/µL, platelets <20,000/µL, reticulocytes <60,000/µL |
| Very severe | Same as severe but neutrophils <200/µL |
| Non-severe | Cytopenias not meeting severe criteria |
Inherited Bone Marrow Failure Syndromes (IBMFS)
These are germline genetic defects with characteristic syndromic features:
| Syndrome | Gene Defect | Key Features |
|---|---|---|
| Fanconi anemia | DNA repair genes (FANC family) | Short stature, radial ray defects, café-au-lait spots, high cancer risk |
| Dyskeratosis congenita | Telomerase genes (TERT, TERC, DKC1) | Nail dystrophy, oral leukoplakia, skin pigmentation, pulmonary fibrosis |
| Diamond-Blackfan anemia | Ribosomal protein genes | Isolated red cell aplasia, congenital anomalies |
| Shwachman-Diamond | SBDS | Neutropenia + exocrine pancreatic insufficiency |
| GATA2 deficiency | GATA2 | Immunodeficiency, lymphedema, MDS/AML in adults |
Bone Marrow Examination Findings
Hypocellular Marrow (Aplastic Anemia)
- Marrow replaced by fat cells
- Markedly reduced hematopoietic precursors across all lineages (trilineage hypoplasia)
- Biopsy preferred over aspirate for cellularity assessment
Hypercellular/Dysplastic Marrow (MDS)
- Increased cellularity with dysplastic morphology (ring sideroblasts, hypolobated neutrophils, micromegakaryocytes)
- Variable cytopenias despite hypercellular marrow — "ineffective hematopoiesis"
Key Clinical Relationships
- Aplastic anemia ↔ PNH ↔ MDS: These three conditions are closely related; one can evolve into another. PNH clones are detected in ~50% of aplastic anemia patients.
- Separation between hypoplastic MDS and aplastic anemia can be diagnostically difficult and requires correlation of morphology, cytogenetics, and molecular studies.
- Many syndromes share immune-mediated marrow destruction and genomic instability, driving higher rates of malignant transformation (e.g., AML).
Treatment Principles
| Etiology | Treatment |
|---|---|
| Immune-mediated (acquired) | Immunosuppressive therapy (IST: ATG + cyclosporine), eltrombopag, allogeneic BMT |
| Physical/chemical damage | Hematopoietic growth factors (HGFs), supportive care, BMT |
| Constitutional/genetic | Sex hormones (androgens), BMT |
— Harrison's Principles of Internal Medicine 22E, pp. 852–856; Goldman-Cecil Medicine, Chapter 151
Pancytopenai
Finding Sources
Finding Sources
Reading File
Reading File
I now have comprehensive content from Harrison's and Textbook of Family Medicine. Here is a complete synthesis:
Pancytopenia — Clinical Features
Definition
Pancytopenia is the simultaneous reduction in all three peripheral blood cell lines:
- Red blood cells (anemia)
- White blood cells / neutrophils (leukopenia/neutropenia)
- Platelets (thrombocytopenia)
It is not a diagnosis itself, but a laboratory finding requiring identification of the underlying cause.
Clinical Features (Symptoms by Cell Line)
Anemia (↓ RBCs)
- Fatigue, weakness, malaise
- Pallor (skin, conjunctiva, nail beds)
- Dyspnea on exertion
- Headaches, dizziness, tinnitus
- Palpitations, tachycardia
Thrombocytopenia (↓ Platelets)
- Petechiae (skin and mucous membranes)
- Purpura, ecchymoses
- Epistaxis (nosebleeds)
- Gingival / mucosal bleeding
- Menorrhagia
- Spontaneous bruising
Neutropenia (↓ WBCs)
- Recurrent or severe bacterial/fungal infections
- Fever (may be the presenting sign)
- Oral ulcers
- Perirectal infections
- Sepsis in severe cases
Differential Diagnosis by Bone Marrow Appearance
Pancytopenia with Hypocellular Bone Marrow
| Cause |
|---|
| Acquired (immune) aplastic anemia |
| Constitutional aplastic anemia (Fanconi anemia, telomere biology disorders) |
| Hypoplastic myelodysplastic syndrome (MDS) |
| Rare aleukemic leukemia |
| Some acute lymphoblastic leukemia (ALL) |
| Rare bone marrow lymphoma |
| Copper deficiency |
Pancytopenia with Cellular Bone Marrow
| Primary Bone Marrow Disease | Secondary / Systemic Causes |
|---|---|
| Myelodysplastic syndromes (MDS) | Systemic lupus erythematosus (SLE) |
| Paroxysmal nocturnal hemoglobinuria (PNH) | Hypersplenism |
| Myelofibrosis | Vitamin B12 / folate deficiency |
| Aleukemic leukemia | Copper deficiency |
| Myelophthisis | Alcohol |
| Bone marrow lymphoma | HIV infection |
| Hairy cell leukemia | Brucellosis, Sarcoidosis, TB |
| Leishmaniasis, Sepsis |
Hypocellular Marrow ± Pancytopenia (Other Causes)
- Q fever
- Legionnaires' disease
- Anorexia nervosa / starvation
- Mycobacterial infection
Common Specific Causes — Key Features
1. Aplastic Anemia
- Most common cause of pancytopenia with hypocellular marrow
- Immune-mediated destruction of hematopoietic stem cells by cytotoxic T cells
- Abrupt onset in a previously well young adult
- May be precipitated by seronegative hepatitis, EBV, CMV, or drugs
2. Megaloblastic Anemia (B12 / Folate Deficiency)
- Hypercellular marrow with ineffective erythropoiesis
- Peripheral blood: macrocytosis (MCV up to 125–130 fL)
- Elevated LDH and indirect bilirubin (intramedullary hemolysis)
- Neurological features with B12 deficiency (subacute combined degeneration)
- Responds rapidly to replacement (reticulocyte peak in 5–7 days)
3. Drug-Induced
- Chloramphenicol: bone marrow vacuolation + ringed sideroblasts; can be permanent
- Linezolid: similar but less severe
- Alcohol: reversible pancytopenia or selective thrombocytopenia via folate deficiency or direct toxicity
- NSAIDs (e.g., indomethacin): rare aplastic anemia
4. Myelodysplastic Syndrome (MDS)
- Dysplastic morphology in ≥1 cell line
- Hypercellular marrow despite peripheral cytopenias ("ineffective hematopoiesis")
- Risk of transformation to AML
5. Hypersplenism
- Splenic sequestration of all blood cell lines
- Splenomegaly on exam and imaging (spleen normally 9–10 cm)
- Marrow is normal or hypercellular
6. Hairy Cell Leukemia
- Classically causes pancytopenia with splenomegaly
- "Dry tap" on aspiration due to marrow fibrosis
- Hairy cells with tartrate-resistant acid phosphatase (TRAP) positivity
Diagnostic Approach
| Test | Purpose |
|---|---|
| CBC with differential + reticulocyte count | Confirm and characterize pancytopenia |
| Peripheral blood smear | Morphology — blasts, dysplasia, macro-ovalocytes |
| Bone marrow aspirate + biopsy | Essential — assess cellularity, morphology, infiltration |
| Flow cytometry | Leukemia/lymphoma immunophenotyping, PNH clone |
| Cytogenetics / FISH | MDS, leukemia |
| B12, folate, copper levels | Nutritional causes |
| LFTs, LDH, bilirubin | Hemolysis, liver disease |
| Abdominal ultrasound | Spleen size (most cost-effective for hypersplenism evaluation) |
| Viral serology | HIV, EBV, CMV, hepatitis |
| ANA, anti-dsDNA | Autoimmune (SLE) |
Bone marrow biopsy is far more frequently indicated in pancytopenia than in single-lineage cytopenia, especially when malignancy (MDS, AML, NHL) is suspected. Flow cytometry and chromosome analysis should be added.
— Harrison's Principles of Internal Medicine 22E, p. 851; Textbook of Family Medicine 9e; Goldman-Cecil Medicine
This is a shared conversation. Sign in to Orris to start your own chat.