Malarial fever
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
malaria life cycle Plasmodium fever paroxysm
I have comprehensive information from multiple authoritative textbooks. Now I'll compose a thorough, well-organized answer.
Malarial Fever
Overview
Malaria is a protozoan disease transmitted by the bite of infected female Anopheles mosquitoes and is the most important parasitic disease of humans. In 2022, an estimated 249 million cases occurred in 85 endemic countries, causing ~608,000 deaths (~1,660 per day). The hallmark clinical feature is fever, which William Osler called humanity's greatest enemy.
— Harrison's Principles of Internal Medicine 22E, p. 1804
Causative Organisms
Six Plasmodium species infect humans:
| Species | Fever Pattern | Periodicity | Notes |
|---|---|---|---|
| P. falciparum | Quotidian → tertian | 36–48 h | Malignant tertian; most deaths; no persistent liver stage |
| P. vivax | Benign tertian | 48 h | Relapses (hypnozoites); widest distribution |
| P. ovale | Benign tertian | 48 h | Relapses; primarily tropical Africa |
| P. malariae | Quartan | 72 h | Long incubation (18–40 days); "quartan malaria" |
| P. knowlesi | Quotidian | 24 h | Severe disease in ~7%; Southeast Asia |
| P. simium / P. cynomolgi | — | — | Zoonotic, rare |
— Medical Microbiology 9e; Harrison's 22E
Life Cycle & Pathogenesis of Fever
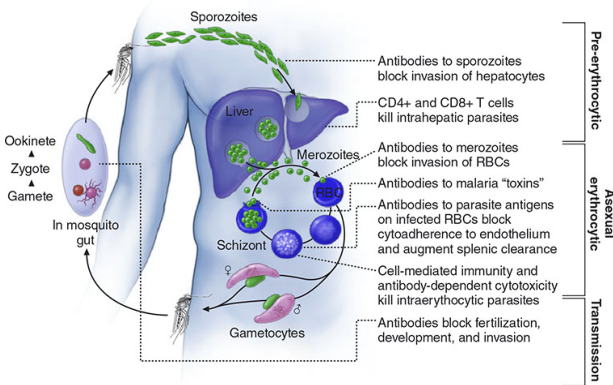
- Sporozoites inoculated by mosquito → travel to liver within minutes
- Intrahepatic schizogony: one sporozoite produces 10,000–30,000 merozoites
- Erythrocytic cycle: merozoites invade RBCs → trophozoite → schizont → rupture, releasing new merozoites
- Fever is triggered at schizont rupture: parasitized RBCs lyse synchronously, releasing hemozoin (malarial pigment), GPI anchors, and parasite debris → stimulates monocytes/macrophages → massive TNF-α and pro-inflammatory cytokine release → fever
- Temperatures ≥40°C damage mature parasites, further synchronizing the cycle → produces the classic regular periodic fever pattern
P. vivax and P. ovale have hypnozoites (dormant liver forms) → cause relapses weeks to months later.
— Harrison's 22E, p. 1805–1806; Goldman-Cecil Medicine
The Malarial Paroxysm
The classic malarial paroxysm has three stages:
- Cold stage (15–60 min) — intense chills, shaking rigors, patient feels cold despite rising temperature
- Hot stage (2–6 h) — high fever 39–41°C, headache, nausea, vomiting, dry skin
- Wet stage (defervescence) — profuse sweating, temperature falls, patient feels exhausted but transiently better
"The hallmark of malaria is fever, often with a nonspecific influenza-like prodrome including headache and fatigue followed by a classic malarial paroxysm including chills, high fever, and then sweats."
— Goldman-Cecil Medicine, p. 965
The classic periodic pattern (every 48 h for P. vivax/ovale, every 72 h for P. malariae) is now rarely seen as patients receive early treatment. P. falciparum initially produces daily (quotidian) fever before progressing to 36–48 h periodicity.
Clinical Features
Uncomplicated Malaria
- Fever (may be continuous early on; cyclical later)
- Headache, myalgia, arthralgia
- Nausea, vomiting, diarrhea
- Splenomegaly, hepatomegaly, anemia
- Jaundice (hemolysis)
Incubation periods: P. falciparum ~7–14 days; P. vivax/ovale ~8–14 days (up to months with hypnozoite reactivation); P. malariae 18–40 days.
Severe / Complicated Malaria (P. falciparum)
Caused by cytoadherence — parasitized RBCs express PfEMP-1 knobs that adhere to capillary endothelium → sequestration → organ ischemia.
| Complication | Features |
|---|---|
| Cerebral malaria | Impaired consciousness, seizures, coma; ~20% mortality |
| Severe anemia | Hb <7 g/dL; hemolysis + dyserythropoiesis |
| Blackwater fever | Massive intravascular hemolysis → hemoglobinuria (dark "blackwater" urine), renal failure |
| Acute pulmonary edema / ARDS | Non-cardiogenic; high mortality |
| Acute kidney injury | Acute tubular necrosis |
| Hypoglycemia | Parasite glucose consumption + quinine-stimulated insulin |
| Lactic acidosis | Impaired microcirculation |
| Algid malaria | Septic shock picture |
— Rosen's Emergency Medicine; Harrison's 22E
Protective Host Genetics
The geographic distribution of malaria co-evolved with several protective human polymorphisms:
- Sickle cell trait (HbA/S) — 6-fold reduction in severe falciparum malaria mortality; reduced PfEMP-1 surface expression
- HbC, HbE — partial protection
- G6PD deficiency — protective especially against P. vivax
- α-thalassemia — protects against severe disease after early frequent infections
- Duffy antigen negativity — near-complete protection against P. vivax (requires Duffy antigen for RBC invasion)
Diagnosis
| Method | Notes |
|---|---|
| Thick & thin blood films (Giemsa/Wright stain) | Gold standard; identify species, quantify parasitemia; repeat every 12–24 h if negative |
| Rapid Diagnostic Tests (RDTs) | Antigen detection (HRP-2 for P. falciparum, pLDH); field-deployable adjunct |
| PCR | Most sensitive; species confirmation |
| Serology | Not useful for acute diagnosis |
Key microscopic findings:
- P. falciparum: multiple ring forms per RBC, banana-shaped gametocytes, no enlarged RBCs
- P. vivax/ovale: enlarged RBCs, Schüffner's dots, amoeboid trophozoites
- P. malariae: band-form trophozoites, "rosette" schizonts
Treatment
Treatment depends on species, severity, and local resistance patterns:
Uncomplicated Malaria
| Species | First-line Treatment |
|---|---|
| P. falciparum (chloroquine-sensitive) | Chloroquine |
| P. falciparum (resistant) | Artemisinin-based combination therapy (ACT): artemether-lumefantrine, artesunate-amodiaquine |
| P. vivax / ovale | Chloroquine + primaquine (to eliminate hypnozoites; check G6PD first) |
| P. malariae | Chloroquine |
| P. knowlesi | ACT or chloroquine |
Severe Malaria
- IV artesunate — drug of choice for severe falciparum malaria worldwide
- IV quinine + doxycycline if artesunate unavailable
- Supportive: antipyretics, IV fluids, blood transfusion, mechanical ventilation as needed
Chemoprophylaxis
Options for travelers to endemic areas:
- Chloroquine (chloroquine-sensitive areas only)
- Atovaquone-proguanil (Malarone) — well tolerated, short pre/post travel course
- Doxycycline — daily; also covers other infections
- Mefloquine — weekly; neuropsychiatric side effects possible
- Primaquine — for P. vivax-endemic areas; check G6PD first
Combined with: insect repellents (DEET), permethrin-treated nets, protective clothing.
Epidemiology Note
P. falciparum almost exclusively in sub-Saharan Africa and tropics. P. vivax has the widest distribution (tropics, subtropics, temperate regions). Returning travelers, immigrants, and recipients of blood transfusions or organ transplants can present in non-endemic settings ("airport malaria" has also been described).
— Rosen's Emergency Medicine; Medical Microbiology 9e
Sources: Harrison's Principles of Internal Medicine 22E (2025) | Goldman-Cecil Medicine | Rosen's Emergency Medicine | Medical Microbiology 9e (Murray)
IV artesunate
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
I have comprehensive pharmacological data from Goodman & Gilman. Now I have everything needed to give a thorough answer on IV artesunate.
IV Artesunate
Overview
Artesunate is a water-soluble hemisuccinate ester of dihydroartemisinin (DHA), derived from artemisinin — a sesquiterpene lactone endoperoxide originally isolated from the Chinese herb Artemisia annua (sweet wormwood). It is the drug of choice for severe falciparum malaria worldwide, having replaced IV quinine.
"Artesunate is the drug of choice when available. Data from large studies in Southeast Asia showed a 35% lower mortality rate than with quinine, and very large studies in Africa showed a 22.5% reduction in mortality rate compared with quinine."
— Harrison's Principles of Internal Medicine 22E, p. 1806
Mechanism of Action
Artesunate (and all artemisinins) are activated by intraparasitic heme, which is produced in the acidic digestive vacuole of Plasmodium as it digests hemoglobin. This interaction cleaves the endoperoxide bridge of the artemisinin molecule, generating free radicals that:
- Promiscuously alkylate a wide variety of parasite proteins
- Oxidize and damage macromolecules within the parasite
- May also form heme–artemisinin adducts that are directly toxic
This mechanism is unique among antimalarials — it requires a live, actively metabolizing parasite (containing heme) to be activated, making selectivity inherent.
Key effects:
- Active against asexual erythrocytic stages (rings through schizonts) — the stages responsible for clinical disease
- Also active against gametocytes → reduces onward transmission
- Each 48-hour erythrocytic cycle treated results in a ~4 log₁₀ (10,000-fold) reduction in parasite burden
- 3–4 treatment cycles (6–8 days) are required for complete parasite clearance
— Goodman & Gilman's Pharmacological Basis of Therapeutics, p. 1313
Pharmacokinetics (ADME)
| Parameter | Detail |
|---|---|
| Formulations | IV (artesunate only), IM (artesunate or artemether), oral (DHA, artesunate, artemether), rectal (artesunate) |
| Bioavailability (oral) | ≤30% |
| Peak serum levels | Rapid after IV/IM; 2–6 h after IM artemether |
| Plasma protein binding | Modest for artesunate and artemether |
| Metabolism | Artesunate is rapidly hydrolyzed (both in gut and plasma) to the active metabolite dihydroartemisinin (DHA) |
| Half-life | Very short — artesunate: <1 h; DHA: ~1–2 h |
| Elimination | DHA is glucuronidated; short t½ necessitates combination therapy to prevent recrudescence |
| Self-induction | Artemisinins induce their own hepatic metabolism (CYP enzymes) → plasma levels may decrease with repeated dosing |
Because of the very short half-life, artesunate monotherapy does not prevent recrudescence — it must always be followed by or combined with a longer-acting partner drug (ACT).
— Goodman & Gilman's, p. 1313–1315; Katzung's Basic & Clinical Pharmacology 16E
Dosing Protocol — IV Artesunate for Severe Malaria
Per WHO / Harrison's 22E:
| Patient | Dose |
|---|---|
| Adults and children ≥20 kg | 2.4 mg/kg IV at 0, 12, and 24 h, then once daily |
| Children <20 kg | 3 mg/kg per dose (higher dose per kg for better exposure) |
Route: IV injection (can also be given IM if IV access unavailable).
Switch to oral ACT as soon as the patient can tolerate oral medications, completing a full 3-day course.
In artemisinin-resistant areas: administer IV artesunate plus IV quinine together at standard doses (combination approach as a precaution).
Indications
- Severe / complicated P. falciparum malaria (primary indication)
- P. knowlesi severe malaria
- Severe P. vivax malaria
- Any patient unable to take oral medications regardless of severity
Pre-referral use: Rectal artesunate (not IV) can be given at community level to severely ill children in rural areas before transfer to a facility — shown to reduce mortality.
Advantages Over Quinine
| Feature | IV Artesunate | IV Quinine |
|---|---|---|
| Mortality reduction | ~35% lower (SE Asia), ~22.5% lower (Africa) | Reference standard |
| Parasite clearance rate | Much faster | Slower |
| Gametocyte clearance | Yes → reduces transmission | No |
| Hypoglycemia risk | Minimal | Significant (stimulates insulin) |
| Cardiac toxicity (QTc prolongation) | Minimal | Significant |
| Cinchonism (tinnitus, deafness) | Absent | Common |
| Ease of administration | Simple IV injection | Requires careful infusion rate control |
Adverse Effects
Artesunate is generally well tolerated, especially relative to quinine:
- Post-artesunate delayed haemolysis (PADH): A clinically important adverse effect — occurs 1–3 weeks after treatment; haemolysis of previously parasitized RBCs as they are re-released into circulation after splenic "pitting." Monitor Hb for 4 weeks post-treatment in non-immune patients (travelers).
- Transient neutropenia
- Mild transaminase elevation
- Neurotoxicity: Reported in animal studies at high doses; not established in clinical use at therapeutic doses.
- First-trimester pregnancy: Use with caution — limited data, but WHO recommends artesunate even in the first trimester for severe malaria (benefit outweighs risk).
Resistance
Partial artemisinin resistance — marked by delayed parasite clearance (slow clearance phenotype) — has emerged in the Greater Mekong Subregion (Cambodia, Myanmar, Thailand, Vietnam, Laos) and increasingly in sub-Saharan Africa, associated with mutations in the Kelch13 (K13) gene (P. falciparum). In confirmed resistance areas, IV artesunate + IV quinine combination is recommended.
Drug Stage Activity Summary
| Drug | Asexual Blood Stage | Gametocytes | Liver Stage (primary) | Hypnozoites |
|---|---|---|---|---|
| Artesunate | ✓ | ✓ | — | — |
| Chloroquine | ✓ | +/− | — | — |
| Primaquine | — | ✓ | ✓ | ✓ |
| Atovaquone/proguanil | ✓ | +/− | ✓ | — |
— Goodman & Gilman's, p. 1313
Sources: Harrison's Principles of Internal Medicine 22E (2025) | Goodman & Gilman's Pharmacological Basis of Therapeutics | Katzung's Basic & Clinical Pharmacology 16E | Goldman-Cecil Medicine
Primaquine
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Reading File
I now have comprehensive content from Goodman & Gilman, Katzung, Lippincott, and Goldman-Cecil. I have everything needed for a thorough answer.
Primaquine
Overview
Primaquine is an 8-aminoquinoline — the prototypical drug of its class — and holds a unique position among antimalarials because it is the only widely available drug that eradicates hypnozoites (dormant liver forms), making it essential for the radical cure of relapsing malaria (P. vivax and P. ovale). Its critical limitation is the risk of haemolytic anaemia in G6PD-deficient patients.
"Primaquine, in contrast to other antimalarials, acts on exerythrocytic tissue stages of Plasmodium spp. in the liver to prevent and cure relapsing malaria. Patients should be screened for G6PD deficiency prior to therapy."
— Goodman & Gilman's Pharmacological Basis of Therapeutics
Chemical Class
- 8-aminoquinoline (distinguished from 4-aminoquinolines like chloroquine)
- Structurally related to tafenoquine (its long-acting successor)
Mechanism of Action
Not fully elucidated. The metabolites of primaquine (not the parent drug) are the active species — they act as oxidants that severely disrupt plasmodial mitochondrial metabolism. Specifically:
- Oxidative disruption of the electron transport chain in the parasite's mitochondria
- Metabolites oxidize glutathione (GSH → GSSG), depleting the parasite's and, critically, the RBC's antioxidant defence
- This same oxidative mechanism underlies both schizonticidal activity and the haemolytic toxicity in G6PD-deficient patients
The drug is not effective against asexual erythrocytic stages and therefore cannot be used as monotherapy for treatment of acute malaria.
— Lippincott Illustrated Reviews Pharmacology; Goodman & Gilman's
Spectrum of Activity — Stage Specificity
| Parasite Stage | Primaquine Active? |
|---|---|
| Sporozoites | — |
| Primary liver stages (hepatic schizonts) | ✓ |
| Hypnozoites (P. vivax, P. ovale) | ✓ (unique action) |
| Asexual erythrocytic (blood) stages | Weak / minimal |
| Gametocytes (all species) | ✓ → interrupts transmission |
This stage profile makes primaquine complementary to blood schizonticides (chloroquine, ACTs) — the two are used together.
Pharmacokinetics
| Parameter | Primaquine | Tafenoquine (comparison) |
|---|---|---|
| Route | Oral only | Oral only |
| Absorption | Near 100%; rapid | Slow; peaks at 12–15 h; food ↑ exposure |
| Peak plasma level | 1–2 h | 12–15 h |
| Half-life | 3–8 h (short) | ~2 weeks (very long) |
| Distribution | Wide tissue distribution; low RBC/tissue binding | Wide |
| Metabolism | Rapid oxidation; primarily deaminated metabolites | Slow |
| Excretion | Minimal renal excretion | Slow urinary excretion |
The short half-life of primaquine necessitates daily dosing for 14 days. Tafenoquine's long half-life allows a single dose for radical cure.
— Katzung's Basic & Clinical Pharmacology 16E
Clinical Uses
1. Radical Cure of P. vivax and P. ovale Malaria
The primary indication. Standard regimen:
- Chloroquine first → eradicates erythrocytic forms (terminates the acute attack)
- Then primaquine 15 mg base/day × 14 days → eradicates liver hypnozoites and prevents relapse
Primaquine appears most effective when started before completion of chloroquine dosing — G6PD status must be checked first.
Some P. vivax strains (notably from Southeast Asia/Oceania) show relative resistance — higher doses (30 mg/day × 14 days) or repeated courses may be needed.
2. Terminal Prophylaxis (Post-exposure)
Given after return from an endemic area to prevent delayed relapse from dormant hypnozoites. Standard blood-stage chemoprophylaxis (chloroquine, mefloquine, etc.) does not cover hypnozoites.
3. Primary Chemoprophylaxis
- Primaquine 30 mg base (0.5 mg/kg) once daily — CDC-listed alternative prophylactic regimen
- Effective against both P. falciparum and P. vivax
- Targets primary liver stages of P. falciparum (no hypnozoites in P. falciparum)
- Must be taken daily; started 1–2 days before travel, continued 7 days after return
4. Gametocidal Action — Transmission Blocking
- Renders P. falciparum gametocytes non-infective to mosquitoes
- A single low dose (0.25 mg base/kg) added to falciparum treatment reduces onward transmission
- WHO recommends this in low-transmission settings; may be safe even without G6PD testing at low doses
5. Pneumocystis jirovecii Pneumonia (PCP)
- Clindamycin + primaquine is an alternative regimen for mild-to-moderate PCP
- Better tolerated than high-dose TMP-SMX or pentamidine; less data for severe disease
— Katzung 16E; Goldman-Cecil Medicine; Goodman & Gilman's
The Critical Adverse Effect: G6PD-Related Haemolysis
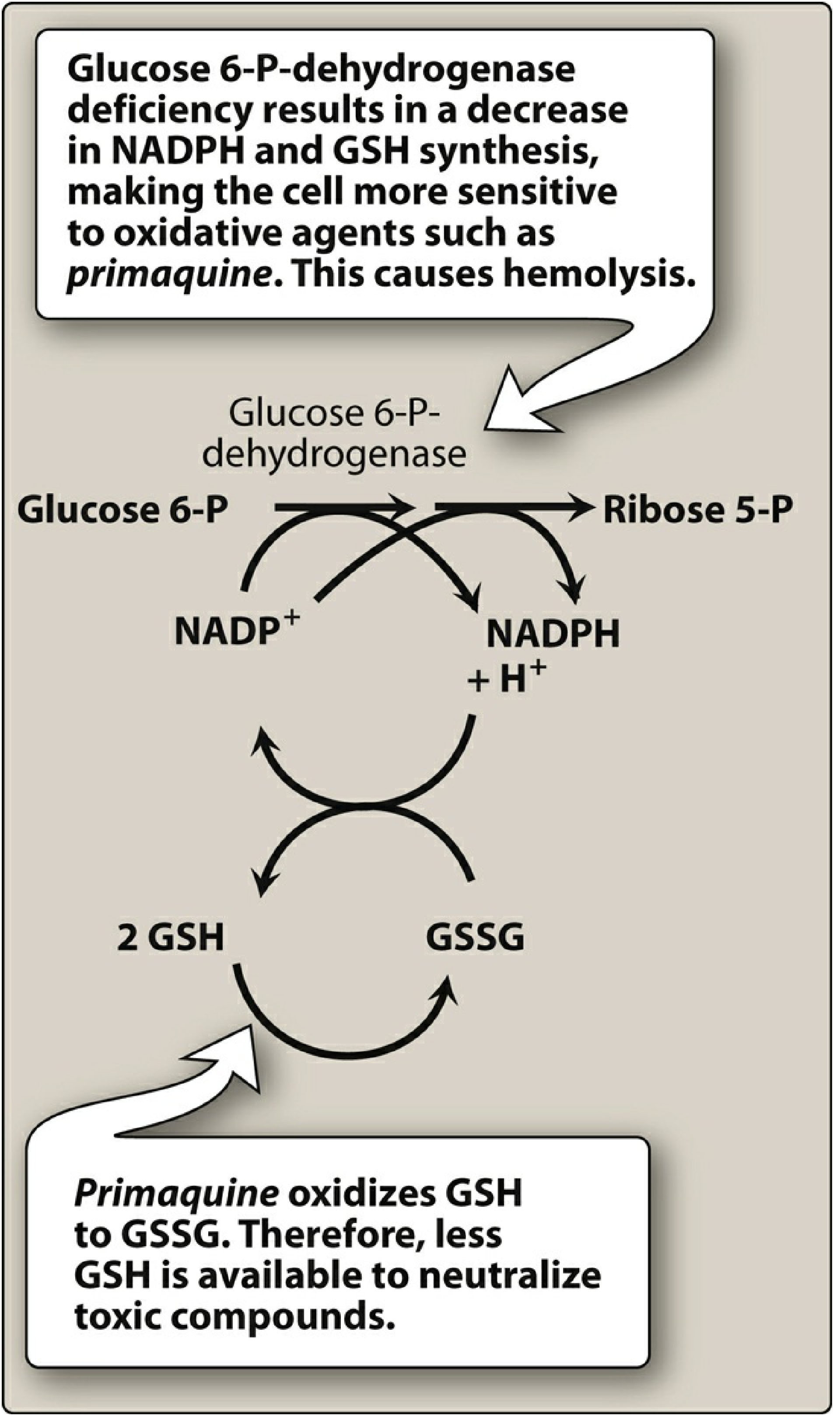
Mechanism:
- RBCs rely on the hexose monophosphate shunt (G6P → Ribose-5-P) to generate NADPH
- NADPH maintains glutathione (GSH) in its reduced, protective form
- Primaquine metabolites oxidize GSH → GSSG, generating reactive oxygen species
- In G6PD-deficient patients, NADPH cannot be regenerated → RBCs are overwhelmed by oxidative stress → Heinz body formation → intravascular haemolysis
Populations at risk: People of African, Mediterranean, Middle Eastern, and South/Southeast Asian ancestry (G6PD deficiency is X-linked; higher prevalence in malaria-endemic regions due to selective pressure).
Severity varies by G6PD variant:
- African variant (A−): Milder haemolysis (only older RBCs affected; reticulocytes relatively protected)
- Mediterranean variant: Severe haemolysis; affects all RBC ages
Rule: Always test G6PD status before prescribing primaquine. If G6PD deficient:
- Mild deficiency: Weekly primaquine 45 mg × 8 weeks (alternative under supervision)
- Severe deficiency: Primaquine contraindicated → consider tafenoquine is also contraindicated → no radical cure option; accept risk of relapse with chloroquine as suppressive therapy
Other Adverse Effects
| Effect | Notes |
|---|---|
| GI disturbance | Nausea, abdominal cramps, especially with chloroquine co-administration; take with food |
| Methemoglobinaemia | At higher doses; metabolites oxidize Hb Fe²⁺ → Fe³⁺ |
| Leucopenia / agranulocytosis | Rare |
| Mild anaemia | Even without G6PD deficiency at high doses |
Contraindications
| Contraindication | Reason |
|---|---|
| G6PD deficiency (severe) | Acute haemolytic anaemia |
| Pregnancy | Risk of haemolysis in G6PD-deficient fetus (fetal G6PD status unknown) |
| Rheumatoid arthritis | Potential for exacerbation |
| Systemic lupus erythematosus (SLE) | Contraindicated |
| Neonates | Not established |
Primaquine vs. Tafenoquine
| Feature | Primaquine | Tafenoquine |
|---|---|---|
| Class | 8-aminoquinoline | 8-aminoquinoline |
| Half-life | 3–8 h | ~2 weeks |
| Radical cure dosing | 15 mg/day × 14 days | Single dose 300 mg |
| Prophylaxis dosing | 30 mg/day | 200 mg/day × 3 days loading, then weekly |
| G6PD testing required | Yes | Yes (even more critical — long t½ means prolonged haemolysis if deficient) |
| FDA approval | Yes | 2018 (prophylaxis + radical cure of P. vivax) |
Summary: Why Primaquine Is Irreplaceable
Primaquine is the only drug routinely available that:
- Eradicates hypnozoites → prevents relapse of P. vivax / P. ovale
- Kills gametocytes of all Plasmodium species → reduces community transmission
- Covers primary liver stages → useful for prophylaxis
Its use is inseparable from mandatory G6PD screening before every prescription.
Sources: Goodman & Gilman's Pharmacological Basis of Therapeutics | Katzung's Basic & Clinical Pharmacology 16E | Lippincott Illustrated Reviews Pharmacology | Goldman-Cecil Medicine
This is a shared conversation. Sign in to Orris to start your own chat.