All about orlistate medication
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of orlistat drawn from multiple authoritative medical textbooks.
Orlistat (Xenical / Alli)
1. Drug Class & Origin
Orlistat is the only agent in the lipase inhibitor class of antiobesity drugs. It is a synthetic hydrogenated derivative of lipostatin, a naturally occurring lipase inhibitor produced by the mold Streptomyces toxytricini. Chemically it is a pentanoic acid ester.
It was FDA-approved in 1999 for long-term use and became available over-the-counter (OTC) in 2007.
2. Mechanism of Action
Orlistat is a potent, slowly reversible inhibitor of:
- Gastric lipase
- Pancreatic lipase
- Carboxylester lipases
- Phospholipase A2
These enzymes are required for the hydrolysis of dietary fat (triglycerides) into fatty acids and monoacylglycerols that can be absorbed. By blocking them, dietary fat is not broken down and passes through the GI tract undigested.
Net effect: ~30% reduction in fat absorption, which is the primary mechanism of weight loss.
Because it acts locally in the GI tract, orlistat is minimally (<1%) absorbed systemically, meaning it has virtually no systemic pharmacological effects.
3. Indications
- Weight loss in obese/overweight patients
- Chronic weight maintenance
- Used as part of a reduced-calorie diet and exercise program
4. Dosing
| Formulation | Dose | Schedule |
|---|---|---|
| Prescription (Xenical) | 120 mg | Three times daily, with or up to 1 hour after each main meal containing fat |
| OTC (Alli) | 60 mg | Three times daily, same schedule |
Higher doses beyond 120 mg TID have not been shown to produce more weight loss.
5. Efficacy
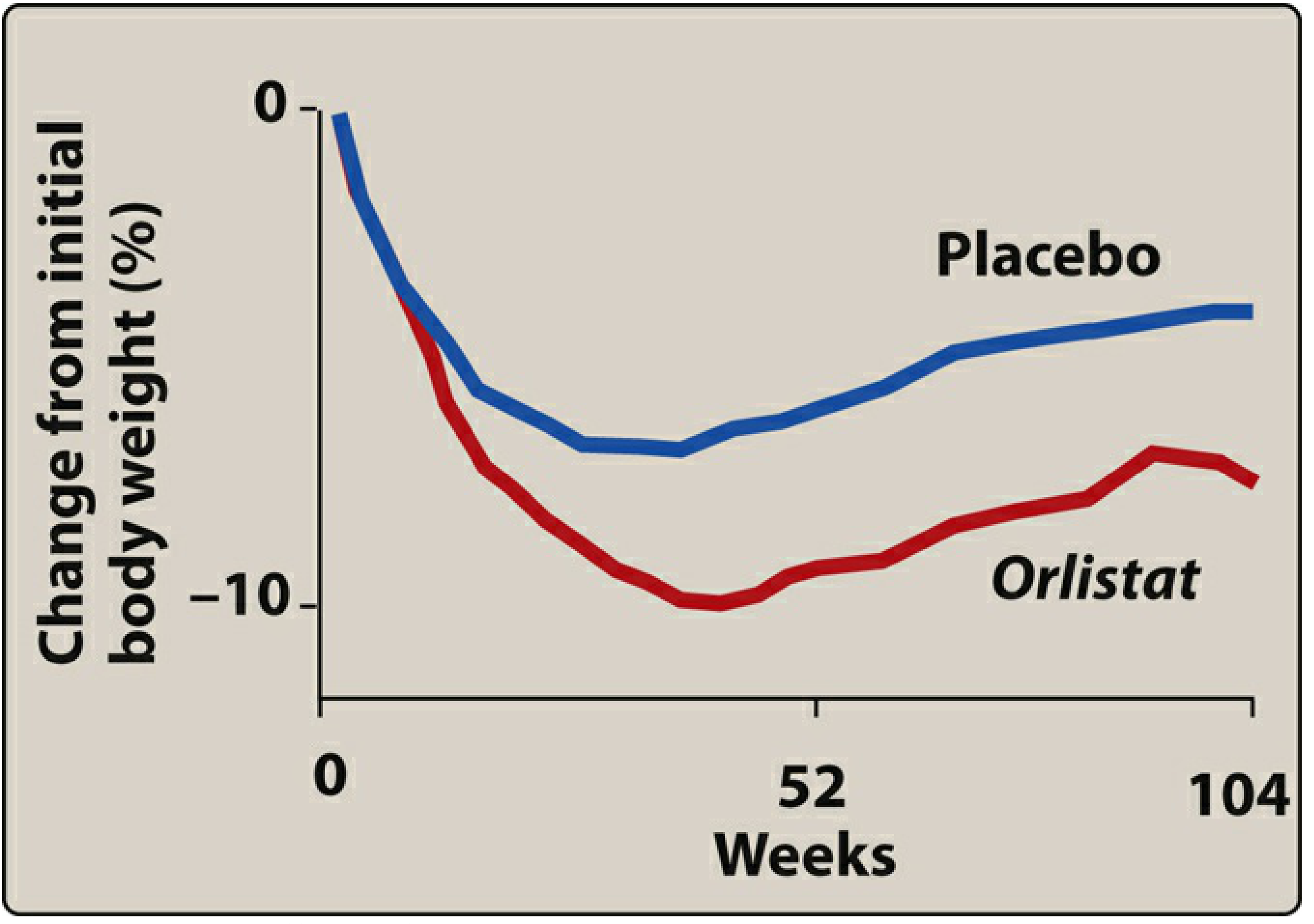
Figure: Orlistat (red) produces a sustained ~10% reduction in body weight at ~52 weeks compared to placebo (blue). Some weight regain occurs but orlistat maintains superior weight loss through 104 weeks.
From randomized, double-blind, placebo-controlled studies:
- 30–50% of patients achieve ≥5% decrease in body mass
- ~20% achieve ≥10% decrease in body mass
- Placebo-subtracted 1-year weight loss: 2.7–4.1%
- After stopping orlistat, up to one-third of patients regain the lost weight
Additional metabolic benefits include:
- Reduced blood pressure
- Reduced risk of developing type 2 diabetes
6. Adverse Effects
The clinical utility of orlistat is significantly limited by GI adverse effects, which result directly from unabsorbed fat in the colon:
| Effect | Notes |
|---|---|
| Oily/fatty stools (steatorrhea) | Most common |
| Flatulence with oily discharge | Very common |
| Fecal urgency / incontinence | Distressing for patients |
| Increased defecation frequency | Dose-related |
- Side effects are worst at initiation and tend to improve as patients adopt a lower-fat diet
- These effects can be minimized by consuming a low-fat diet and using cholestyramine
- Paradoxically, the GI effects have been used therapeutically to treat constipation caused by psychotropic drugs (e.g., TCAs, clozapine)
Serious but rare adverse effects:
- Severe liver injury (rare cases reported; FDA added a safety label update in 2010)
- Acute kidney injury (rate is higher among orlistat users than non-users)
7. Drug Interactions
| Drug | Interaction | Management |
|---|---|---|
| Cyclosporine | Orlistat reduces cyclosporine plasma levels | Do NOT co-administer; administer separately if needed |
| Levothyroxine | Impaired absorption of levothyroxine | Separate doses by at least 4 hours |
| Amiodarone | Impaired absorption | Monitor clinical response |
| Fat-soluble vitamins (A, D, E, K, β-carotene) | Reduced serum concentrations | Mandatory multivitamin supplementation |
| Warfarin | Potential for altered anticoagulation via vitamin K malabsorption | Monitor INR |
8. Fat-Soluble Vitamin Supplementation
Because orlistat inhibits fat absorption, it also reduces absorption of fat-soluble vitamins and nutrients. Patients must take a daily multivitamin containing vitamins A, D, E, K, and β-carotene — preferably at bedtime (separated from orlistat doses).
9. Contraindications
- Chronic malabsorption syndrome
- Cholestasis
- Reduced gallbladder function
- Hypersensitivity to orlistat
- Pregnancy
- Impaired liver function (use with caution)
- Impaired renal function (use with caution)
- Pancreatic disease (use with caution)
10. Special Populations
- Adolescents: Approved for use in adolescents; GI side effects (oily spotting, oily discharge) are the most common reason for early discontinuation in this age group
- OTC availability: Available as Alli (60 mg) since 2007 without a prescription
11. Place in Therapy (Context Among Antiobesity Drugs)
Orlistat was the only approved long-term antiobesity drug prior to 2012. It remains unique as the only peripherally acting (non-systemic) antiobesity medication. Compared to newer agents:
| Agent | Mechanism | 1-year Placebo-Subtracted Weight Loss |
|---|---|---|
| Orlistat | GI lipase inhibitor | ~2.7–4.1% |
| Phentermine/topiramate | Appetite suppression | Higher |
| Liraglutide (GLP-1 RA) | GLP-1 receptor agonist | ~3.4–6.1% |
| Semaglutide 2.4 mg | GLP-1 receptor agonist | ~6.2–14.8% |
| Tirzepatide 15 mg | GLP-1/GIP dual agonist | ~11.6–21.4% |
Orlistat produces more modest weight loss than newer agents but offers the advantage of no systemic exposure and a well-characterized safety profile over decades of use.
Sources: Lippincott Illustrated Reviews: Pharmacology | Harrison's Principles of Internal Medicine, 22nd Ed. | Kaplan & Sadock's Synopsis of Psychiatry | Sleisenger & Fordtran's Gastrointestinal and Liver Disease
This is a shared conversation. Sign in to Orris to start your own chat.