Intrparenchymal hemorrhage
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Finding Sources
Searching Images
intraparenchymal hemorrhage CT scan brain hypertensive
Reading File
I have extensive content from multiple authoritative textbooks. Now I'll compile a comprehensive, well-organized answer.
Intraparenchymal (Intracerebral) Hemorrhage
Definition
Intraparenchymal hemorrhage (IPH) is bleeding directly within the brain parenchyma — the cerebral hemispheres, brainstem, cerebellum, or spinal cord. It is distinct from epidural, subdural, and subarachnoid hemorrhage (see table below). It should also not be confused with hemorrhagic transformation of an ischemic infarct, which is secondary bleeding into already-infarcted tissue.
Epidemiology
- Peak incidence: middle to late adult life (~60 years)
- Accounts for ~15% of deaths among individuals with chronic hypertension
- Hypertension is the most common cause, responsible for >50% of clinically significant hemorrhages
- — Robbins, Cotran & Kumar Pathologic Basis of Disease, p. 1156
Etiology & Pathogenesis
| Cause | Key Features |
|---|---|
| Hypertension | Most common; deep ganglionic pattern; lipohyalinosis + Charcot-Bouchard microaneurysms in lenticulostriate arteries |
| Cerebral amyloid angiopathy (CAA) | Most common cause of lobar hemorrhage; Aβ deposited in vessel walls; associated with Alzheimer disease |
| Vascular malformations | AVM, cavernous malformations |
| Coagulopathy / anticoagulation | Warfarin; NOACs have lower ICH risk vs. warfarin |
| Tumor | Primary (GBM) or metastatic (melanoma, choriocarcinoma, renal cell) |
| Trauma | Contusions, shearing forces; coup and contrecoup injury |
| Vasculitis / mycotic aneurysm | Endocarditis setting |
| Hemorrhagic conversion | Prior ischemic infarct |
Hypertensive mechanism: Chronic hypertension causes:
- Accelerated atherosclerosis in larger arteries
- Hyaline arteriolosclerosis in small arteries → rigid, thickened walls vulnerable to rupture
- Microaneurysms of Charcot-Bouchard in penetrating arteries (most prominent in basal ganglia and subcortical white matter)
— Neuroanatomy through Clinical Cases, p. 180; Robbins p. 1156
Location Patterns
Hypertensive (Deep / "Ganglionic") Hemorrhage — by frequency:
- Putamen (50–60%)
- Thalamus
- Cerebellar hemispheres
- Pons
- Other brainstem
Lobar Hemorrhage (CAA, older patients):
- Involves subcortical white matter of cerebral lobes
- Older, less often male, less hypertensive than ganglionic hemorrhage
- May extend into subarachnoid space
Traumatic:
- Contusions at crests of gyri (temporal and frontal poles > occipital poles)
- Coup and contrecoup pattern
- Shearing forces → petechial or confluent white matter hemorrhage
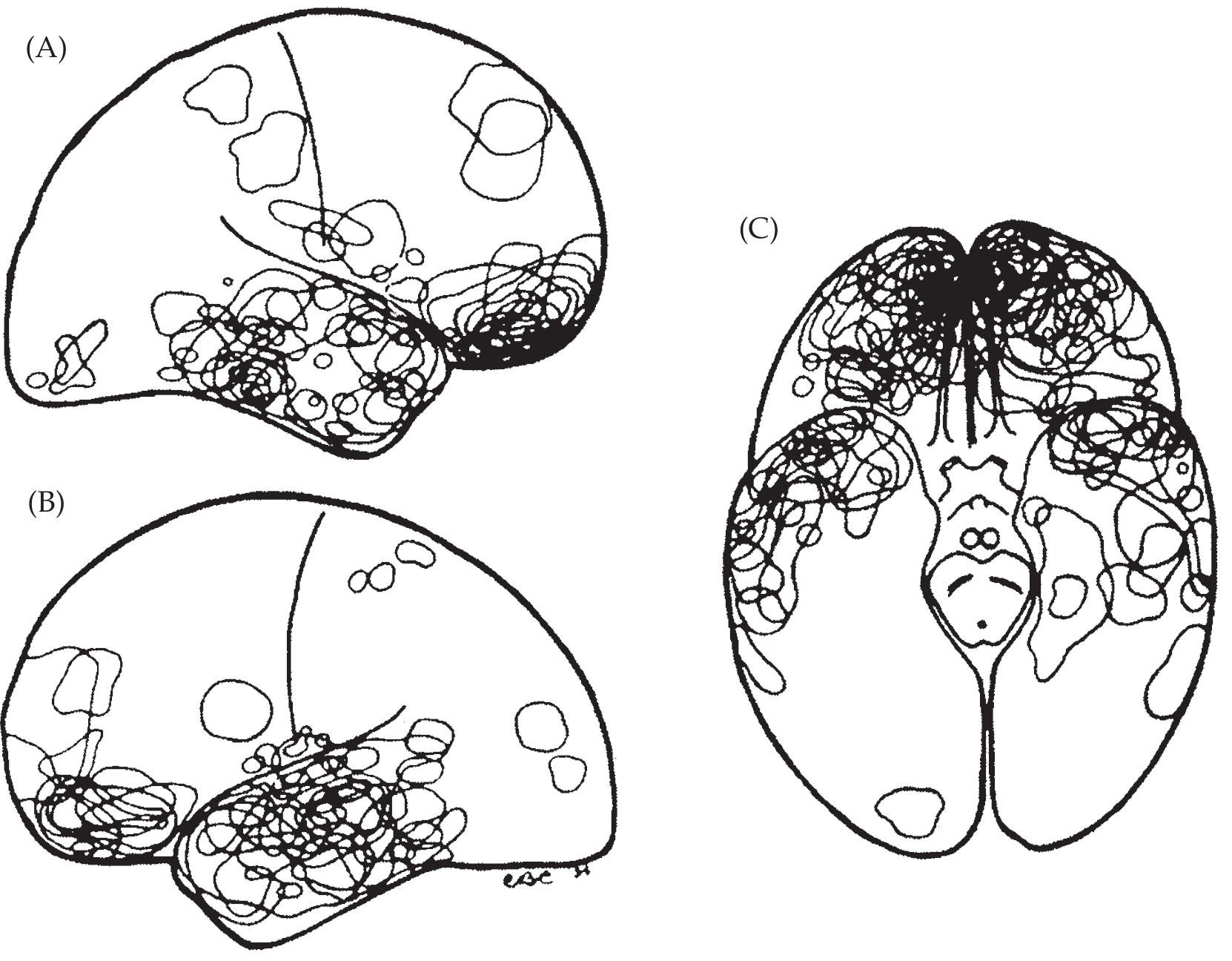
Common sites of cerebral contusion — right hemisphere (A), left hemisphere (B), inferior view (C). — Neuroanatomy through Clinical Cases
Compartments of Intracranial Hemorrhage
| Compartment | Common Etiology | Key Feature |
|---|---|---|
| Epidural | Trauma | Skull fracture; lucid interval; rapid evolution |
| Subdural | Trauma | May follow minor trauma; slow evolution |
| Subarachnoid | AVM, aneurysm | Thunderclap headache; vasospasm |
| Intraparenchymal | HTN, CAA, trauma, tumor | Depends on location more than etiology |
— Robbins, p. 1156
Clinical Presentation
Onset: Sudden neurologic deficit. Symptoms reflect location more than etiology.
Supratentorial:
- Lobar: Severe headache in ~90%; focal deficits in ~90%; decreased consciousness in ~50%; coma in ~20%; seizures common (EEG useful)
- Striatocapsular / putaminal: Contralateral hemiparesis; eye deviation toward the lesion; consciousness varies by subtype — massive lesions → coma (fatality ~50%)
- Thalamic: Stupor or coma in ~20%; ocular signs — skew deviation (lower eye ipsilateral), wrong-way gaze, loss of vertical gaze, miotic pupils ("peering at the tip of the nose" = nearly pathognomonic); sensory loss; ~25% mortality
Infratentorial:
- Pontine: Quadriplegia; pinpoint pupils; hyperthermia; coma — high mortality
- Cerebellar: Sudden headache, vomiting, ataxia, inability to stand; may have ipsilateral gaze palsy or facial palsy
Hematoma expansion: Unlike aneurysmal SAH, re-bleeding risk is low, but the hematoma often continues to expand for several hours, causing progressive neurologic deterioration. Surrounding edema peaks at ~3 days.
— Neuroanatomy through Clinical Cases, p. 180; Plum & Posner, p. 272–274
Imaging
CT (non-contrast) — first-line:
- Acute blood appears hyperdense (bright white)
- Reveals mass effect, midline shift, intraventricular extension
- Hypodense rim = surrounding edema
MRI:
- More sensitive for small hemorrhages and subacute/chronic blood
- SWI (susceptibility-weighted imaging): detects hemosiderin → identifies microhemorrhages (chronic CAA)
- DWI/ADC: distinguishes from ischemic stroke
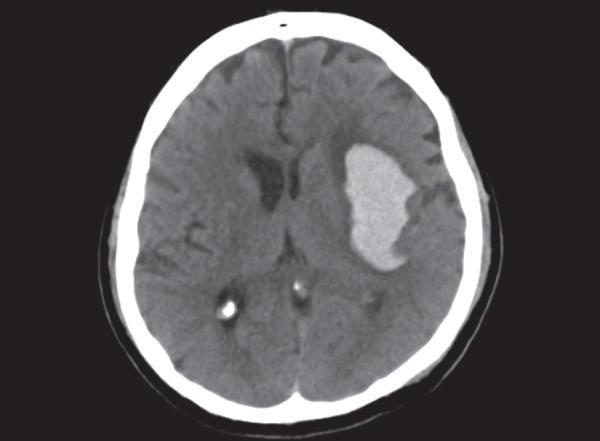
Non-contrast CT: large hyperdense hematoma in left basal ganglia (lenticulostriate territory) with rightward midline shift — classic hypertensive ICH
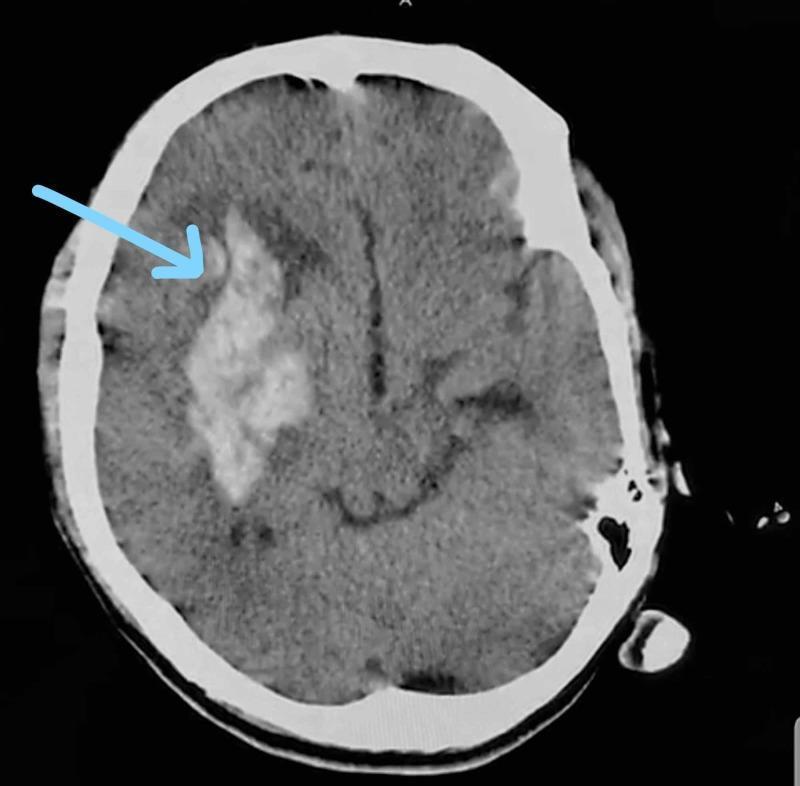
Right gangliocapsular ICH with intraventricular extension — sudden-onset contralateral hemiparesis
Pathology (Gross & Microscopic)
- Acute: Clotted blood displacing and compressing surrounding parenchyma; adjacent petechiae
- Subacute–Chronic: Hemoglobin degradation → hemosiderin-laden macrophages (brown discoloration); reactive astrocytosis; eventual cavitation with orange-brown scar
- In hypertension: vessels show hyaline arteriolosclerosis — thickened, rigid walls (Fig. 28.14B in Robbins)
- In CAA: amyloid deposits in walls of meningeal and cortical vessels; vessels fail to collapse on histologic sectioning
Management Overview
Acute Medical
- Airway / ventilation: Intubate if GCS ≤8 or airway at risk
- Blood pressure control: Target SBP <140 mmHg (AHA/ASA 2022 guideline for spontaneous ICH) — prevents hematoma expansion
- Reverse coagulopathy: Vitamin K + 4-factor PCC (warfarin); idarucizumab (dabigatran); andexanet alfa (factor Xa inhibitors)
- ICP management: HOB 30°, osmotherapy (mannitol, hypertonic saline), avoid hyperthermia/hypoglycemia
- Seizure prophylaxis: Treat clinical seizures; EEG monitoring for subclinical seizures
Surgical
Considered when:
- Cerebellar hemorrhage >3 cm with neurologic deterioration or brainstem compression → craniotomy/evacuation is strongly indicated
- Lobar hematoma within 1 cm of cortical surface in patients with deterioration
- Obstructive hydrocephalus → external ventricular drain (EVD)
- Minimally invasive surgery (MIPS): trials comparing MIPS to medical management for volumes 30–80 mL
- Deep ganglionic hemorrhages generally not improved by open surgery (STICH trials)
Prognosis Predictors
- Hematoma volume (strongest predictor)
- Glasgow Coma Scale at presentation
- Intraventricular extension (worsens prognosis)
- Infratentorial location (higher mortality)
- Anticoagulant use
— Plum & Posner, p. 271–274; Neuroanatomy through Clinical Cases, p. 180; Robbins p. 1156; ROSEN's Emergency Medicine
Key Distinguishing Points
| Feature | Hypertensive ICH | CAA-related ICH |
|---|---|---|
| Location | Deep (putamen, thalamus, pons) | Lobar (cortical/subcortical) |
| Age | Middle-aged | Elderly |
| Recurrence | Moderate | High (multifocal microhemorrhages on SWI) |
| Association | HTN history | Alzheimer disease |
| Vessel change | Hyaline arteriolosclerosis | Amyloid deposition |
This is a shared conversation. Sign in to Orris to start your own chat.