tell me about liver cirrhosis in brief
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Reading File
I now have comprehensive content from Robbins Pathology, Goldman-Cecil Medicine, and Guyton & Hall. I have enough to provide a thorough, well-cited answer.
Liver Cirrhosis
Definition
Cirrhosis is the diffuse transformation of the liver into regenerative parenchymal nodules surrounded by fibrous bands. It represents the end stage of any chronic liver disease and is not a single specific entity — its morphology, pathophysiology, and prognosis vary by underlying cause.
— Robbins & Kumar Basic Pathology, p. 598
Causes
Most common worldwide:
- Chronic Hepatitis B
- Chronic Hepatitis C
- Non-alcoholic fatty liver disease (NAFLD/NASH)
- Alcohol-related liver disease
Less common (<2% of cases):
- Primary biliary cirrhosis / Primary sclerosing cholangitis
- Autoimmune hepatitis
- Metabolic disorders: Hemochromatosis, Wilson disease, α1-antitrypsin deficiency, glycogen storage diseases
- Biliary obstruction / biliary atresia / cystic fibrosis
- Hepatic venous outflow obstruction (Budd-Chiari)
- Cryptogenic cirrhosis — when no cause is identified
— Goldman-Cecil Medicine, p. 1614–1615
Pathogenesis
The central cellular event is activation of hepatic stellate cells (Ito cells / perisinusoidal cells):
- Normally quiescent, they reside in the Space of Disse and store retinoids (vitamin A)
- On injury, they lose vitamin A, proliferate, develop rough endoplasmic reticulum, and secrete extracellular matrix (collagen types I and III, sulfated proteoglycans, glycoproteins)
- They transform into contractile myofibroblasts, driving fibrosis
- Fibrous septa link portal tracts to each other and to central veins, distorting hepatic architecture
- Surviving hepatocytes undergo regeneration, forming nodules, but these lack normal vascular organization
Importantly, fibrosis can regress if the underlying injury is removed (e.g., cured hepatitis C, treated autoimmune hepatitis) — scars compact, thin, and fragment, allowing nodule coalescence.
— Goldman-Cecil Medicine, p. 1614; Robbins & Kumar, p. 598
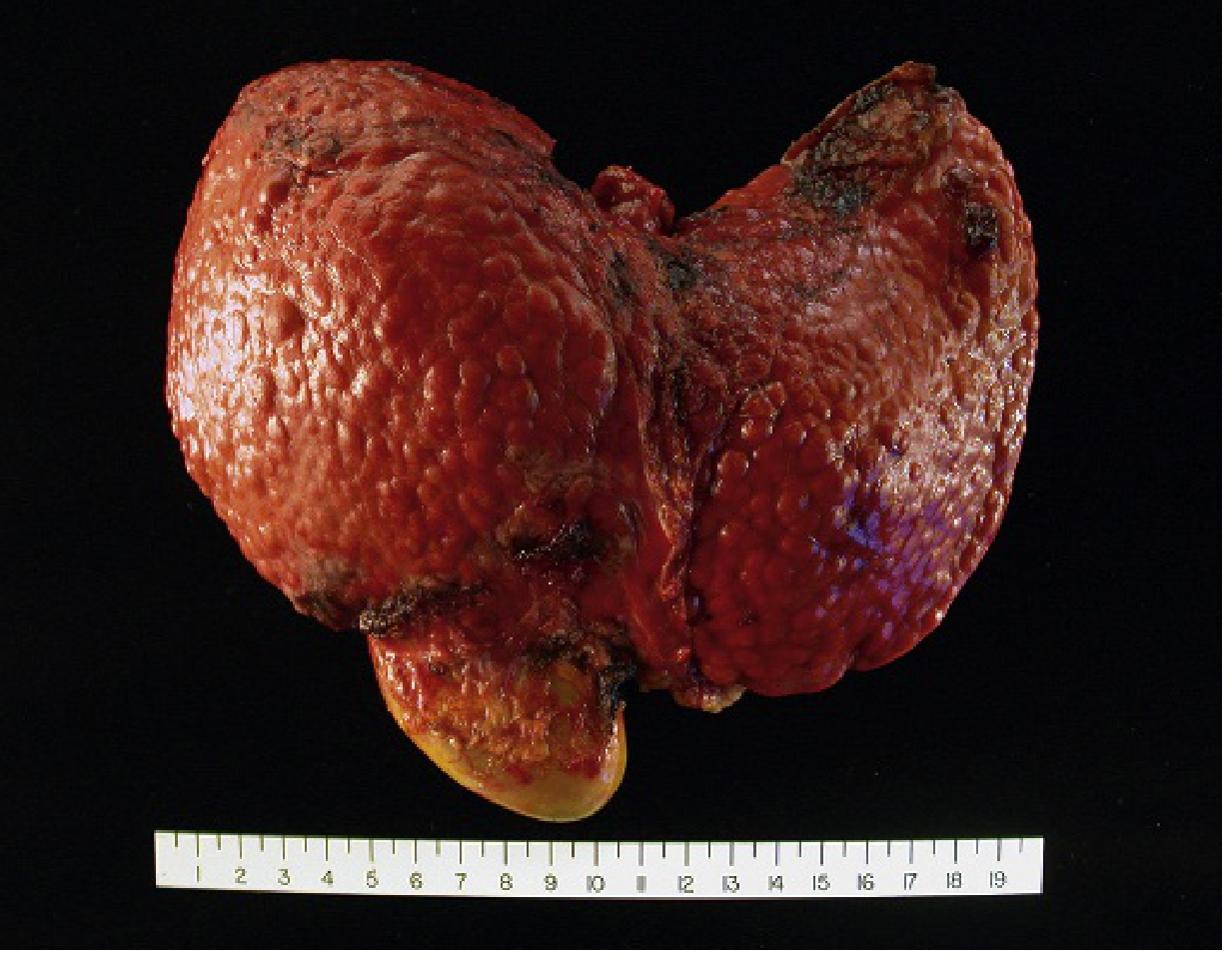
Morphology
- Gross: Nodular surface, orange-tawny color, irregular texture (vs. smooth normal liver)
- Microscopic: Disorganized architecture with regenerative nodules surrounded by fibrous tissue; prominent ductular reactions (stem cell-derived duct-like structures increase with disease progression)
- Nodule size and scar pattern vary by etiology
Staging: Compensated vs. Decompensated
| Stage | Definition |
|---|---|
| Compensated | Cirrhosis present but no major complications; may be asymptomatic (~40% of patients) |
| Decompensated | Development of ascites, variceal hemorrhage, hepatic encephalopathy, or jaundice |
Decompensation is driven by portal hypertension (and the hyperdynamic circulatory state) and/or liver insufficiency. Each stage has different predominant pathophysiologic mechanisms and management strategies.
— Goldman-Cecil Medicine, p. 1614
Clinical Features
Asymptomatic / nonspecific early:
- Anorexia, weight loss, weakness, fatigue
Manifestations of liver failure:
- Jaundice — chronic severe jaundice causes pruritus (from bile salt accumulation), which can be debilitating
- Coagulopathy — decreased synthesis of clotting factors
- Hepatic encephalopathy — altered mentation from nitrogenous waste accumulation
Portal hypertension complications:
- Ascites — raised portal pressure + reduced plasma oncotic pressure (from low albumin synthesis) causes fluid leakage into the peritoneal cavity; kidneys compensate by retaining salt and water
- Esophageal/gastric varices — portosystemic collaterals; rupture causes life-threatening hemorrhage
- Splenomegaly — splenic congestion → hypersplenism (thrombocytopenia, leukopenia)
- Caput medusae — dilated periumbilical veins
Other systemic effects:
- Spider angiomata, palmar erythema, gynecomastia (altered hormone metabolism)
- Hepatorenal syndrome — functional renal failure
- Hepatopulmonary syndrome / portopulmonary hypertension
- Increased risk of hepatocellular carcinoma (HCC)
Fluid/electrolyte physiology: Reduced plasma proteins → reduced oncotic pressure; portal obstruction → raised capillary pressure → ascites. The kidneys retain Na⁺ and water to restore volume; vascular capacity may actually increase due to portal vein distension, perpetuating fluid retention.
— Guyton and Hall Textbook of Medical Physiology; Robbins & Kumar, p. 598–599
Diagnosis
- LFTs: Elevated bilirubin, AST/ALT (may be normal in end-stage), low albumin, prolonged PT/INR
- CBC: Thrombocytopenia (hypersplenism), anemia
- Imaging (US/CT/MRI): Nodular liver surface, splenomegaly, ascites, collateral vessels
- Elastography (FibroScan): Non-invasive fibrosis assessment
- Liver biopsy: Gold standard for confirming cirrhosis and identifying etiology
- MELD score (Model for End-stage Liver Disease): Predicts 90-day mortality using bilirubin, INR, creatinine
Management Principles
- Treat the underlying cause — antiviral therapy for hepatitis B/C, alcohol abstinence, weight loss for NAFLD
- Prevent and manage complications:
- Ascites: Sodium restriction, diuretics (spironolactone ± furosemide), paracentesis for refractory cases
- Varices: Non-selective beta-blockers (propranolol/nadolol) for primary prophylaxis; endoscopic band ligation; TIPS (transjugular intrahepatic portosystemic shunt) for refractory bleeding
- Encephalopathy: Lactulose, rifaximin, dietary protein adjustment
- Spontaneous bacterial peritonitis (SBP): IV antibiotics (cefotaxime), long-term norfloxacin prophylaxis
- Liver transplantation — definitive treatment for decompensated cirrhosis; selection based on MELD score
Key Points Summary
| Feature | Detail |
|---|---|
| Core pathology | Nodules + fibrous bands throughout liver |
| Key cellular event | Hepatic stellate cell activation |
| Main causes | Alcohol, NAFLD, Hep B, Hep C |
| Decompensation triggers | Ascites, varices, encephalopathy, jaundice |
| Reversibility | Possible in early stages if cause removed |
| Complication to screen | HCC (6-monthly USS + AFP) |
This is a shared conversation. Sign in to Orris to start your own chat.