interfascicular nerve repair
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content. Let me compile the answer.
Interfascicular Nerve Repair
Interfascicular (grouped fascicular) nerve repair is one of three main techniques for direct peripheral nerve suture, alongside epineurial repair and individual fascicular repair. It sits between these two in terms of dissection extent and precision.
Techniques of Direct Nerve Suture
| Technique | Description |
|---|---|
| Epineurial repair | Sutures placed in the outer epineurium only; most commonly used |
| Group fascicular (interfascicular) repair | Internal epineurium between fascicle groups is sutured; fascicle groups are matched separately |
| Individual fascicular repair | Each fascicle is sutured separately; rarely used due to surgical trauma |
Epineurial repair is the standard. External epineurial vessels are used as landmarks to align fascicles. Only 2–3 interrupted sutures are typically needed to coapt the nerve ends; the needle must pass only through epineurium without catching fascicles (which would cause intraneural neuroma formation).
Interfascicular / group fascicular repair involves separating fascicle groups and suturing the internal epineurium (perineurium) between them with fine interrupted nylon sutures, then completing an epineural repair. It is applied selectively:
- In large nerves where fascicle groups are easily identifiable
- At distal sites where fascicles are diverging into terminal branches, enabling group repair with minimal dissection
- Some studies report more intraneural scarring with this technique, so most peripheral nerve surgeons still prefer epineurial repair
"Group fascicular repair involves separating fascicles in a nerve and suturing matching structures separately. There is potential for more surgical trauma from this type of repair. A recent literature review found no evidence that one suturing technique is superior to another." — Rockwood and Green's Fractures in Adults, 10th ed.
Fascicular Alignment and Topography
Accurate alignment is critical regardless of technique. Strategies include:
- Epineurial vessel orientation — surface vessels serve as rotational markers
- Fascicular pattern matching — cross-sectional topography at the cut ends
- Intraoperative stimulation — identifies motor vs. sensory fascicles within the first few days after injury (while distal axons still conduct)
- Nerve action potential recording — useful at 2–3 months to detect regeneration across a lesion in continuity
- Histochemical stains — cholinesterase (motor) vs. carbonic anhydrase (sensory) differentiation
Proximal nerve trunks have extensive intermingling (plexus formation) of sensory/motor fibers; more distal injuries show better segregation into functional groups — this is why distal repairs have better outcomes than proximal ones.
Technical Principles of Suture Repair
- Suture material: Non-absorbable monofilament nylon
- Suture gauge: 6/0 for large nerves (sciatic), 8/0 for median/ulnar, 9/0–10/0 for digital nerves
- No tension: Tension causes ischemia and fibrosis at the repair site; it is the single most important predictor of poor outcome
- No fascicle overlap: Fascicles must be gently coapted, not overlapping
- Fibrin glue may augment the repair
- Approximate mesoneurial attachments to take tension off the nerve suture line
Managing Nerve Gaps
When direct end-to-end repair without tension is not possible:
| Strategy | Applicable gap |
|---|---|
| Nerve mobilization | Small gaps |
| Transposition (e.g., anterior ulnar) | Moderate gaps at elbow |
| Joint flexion + immobilization → gradual extension at 3 weeks | ≤2–3 cm |
| Bone shortening (e.g., comminuted humerus fracture) | Variable |
| Nerve graft | Larger gaps, delayed repairs, retracted stumps |
| Nerve conduits | Short gaps <3 cm, small sensory nerves (e.g., digital) |
| Nerve transfer | Avulsions, proximal injuries |
Surgical Timing — the "3+1 Rule"
| Timing | Window | Indication |
|---|---|---|
| Early | ≤3 days | Sharp transection (neurotmesis), acute compression from hematoma/bone |
| Subacute | ~3 weeks | Blunt/ragged transection |
| Delayed | 3–6 months | Blunt closed injury not recovering; neuroma-in-continuity with negative NAP |
| Late | >1 year | Tendon/muscle transfers if nerve repair window has passed |
Key Principle
Nerve regeneration occurs at ~1 mm/day. For proximal injuries, end-organ (muscle) viability may be lost before reinnervation arrives — if reinnervation is expected to take >12–18 months, tendon transfer or amputation may provide better functional outcomes than nerve repair.
Sources: Current Surgical Therapy 14e, p. 984 | Rockwood and Green's Fractures in Adults 10th ed., p. 778–779 | Sabiston Textbook of Surgery
what are the other nerve repair techniques entire notes on nerve repair a to z
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Peripheral Nerve Repair — Complete Notes A to Z
1. ANATOMY OF A PERIPHERAL NERVE
The hierarchical structure of a peripheral nerve (from inside out):
| Layer | Description |
|---|---|
| Axon | Basic functional unit; conducts electrical impulses |
| Endoneurium | Connective tissue surrounding individual axons |
| Fascicle | Bundle of axons + endoneurium |
| Perineurium | Encapsulates each fascicle; forms the blood-nerve barrier |
| Interfascicular epineurium | Connective tissue between fascicles |
| External epineurium | Outer sheath surrounding all fascicles |
| Mesoneurium | Outermost layer; allows nerve gliding; carries segmental blood supply |
Fascicular topography — proximal nerve trunks show extensive motor-sensory intermingling (plexus formation). More distally, fascicles segregate into distinct motor and sensory groups. This is why:
- Distal repairs → less fascicular mismatch → better outcomes
- Proximal facial nerve repair → high synkinesis risk due to poor topographic segregation
2. CLASSIFICATION OF NERVE INJURIES
Seddon Classification (3 grades)
| Grade | Name | Description | Recovery |
|---|---|---|---|
| I | Neurapraxia | Focal conduction block only; axons, endoneurium, perineurium, epineurium ALL intact; typically from stretch/compression | Complete; excellent prognosis |
| II | Axonotmesis | Axonal disruption + Wallerian degeneration; endoneurial tubes intact | Incomplete; unpredictable |
| III | Neurotmesis | Complete nerve disruption including epineurium; proximal neuroma + distal glioma form | Worst; requires surgery |
Sunderland Classification (5 grades, extended to 6 by Mackinnon & Dellon)
| Grade | Structures Disrupted | Equivalent |
|---|---|---|
| I | None (conduction block only) | Neurapraxia |
| II | Axon only | Axonotmesis |
| III | Axon + endoneurium | Axonotmesis |
| IV | Axon + endoneurium + perineurium | Axonotmesis → neuroma-in-continuity |
| V | All layers including epineurium | Neurotmesis |
| VI (Mackinnon) | Mixed injury (combination of I–V in same nerve) | — |
Key: Grades II–V all produce Wallerian degeneration distally. Grade I does NOT.
3. PATHOPHYSIOLOGY
Wallerian Degeneration (distal to injury)
- Begins within hours; axon and myelin fragment and degenerate
- Schwann cells phagocytose myelin debris
- Schwann cells then proliferate and change to a pro-regenerative phenotype — upregulating neurotrophins, forming bands of Büngner (longitudinally aligned cellular scaffolds for axon regrowth)
- Distal axon remains electrically excitable for 2–4 days after injury → neurophysiology unreliable until 3–6 weeks post-injury
Proximal (Retrograde) Changes
- Chromatolysis: cell body swells, Nissl substance disperses, nucleus peripheralizes
- Structural protein synthesis increases (pro-regenerative)
- ~30% of primary sensory neurons die after unrepaired distal transection
- After proximal injuries (e.g., brachial plexus postganglionic): greater motor neuron and sensory neuron death
- After preganglionic injury: up to 80% sensory + 50% motor neuron death
CNS Changes
- Sensory/motor homuncular maps reorganize within hours
- Denervation pain from spontaneous discharge of injured nociceptive afferents
- Root avulsion → crushing, unrelenting pain (worst prognosis)
Regeneration
- Proximal axon sprouts multiple regenerating sprouts
- Sprouts grow at ~1 mm/day (approximately 1 inch/month)
- Schwann cells guide axons along bands of Büngner
- Neurotrophic factor (BDNF, NGF) expression by denervated Schwann cells declines over time → window for repair
- Regenerated axons have smaller diameter than original; remyelination occurs but myelin sheath is thinner
4. TIMING OF REPAIR — THE "3+1 RULE"
| Timing | Window | Injury Type | Classification |
|---|---|---|---|
| Early | ≤3 days | Sharp laceration/transection; acute nerve compression from hematoma/bone; acute neurologic worsening | Neurotmesis |
| Subacute | ~3 weeks | Blunt or ragged transection (chainsaw, propeller) | Neurotmesis |
| Delayed | 3–6 months | Blunt closed injury not recovering; neuroma-in-continuity with negative NAP | Axonotmesis/Neurotmesis |
| Late | >1 year | Tendon/muscle transfers; bony procedures | — |
General principle: Best results achieved within 10–14 days of injury. "Time is muscle."
Prerequisites before repair:
- Thorough wound debridement
- Fracture stabilization
- Vascular repair first
- Tendon repair
- Adequate soft tissue coverage (vascularized skin; flap if needed)
5. PREOPERATIVE ASSESSMENT
Clinical
- Document motor and sensory deficits at presentation
- Assess zone of injury (sharp → small zone; crush/blast → wide zone)
- Mechanism: open (laceration) vs. closed (stretch, compression, avulsion)
- Gunshot wounds behave as closed injuries (blast effect, not transection, usually)
Electrodiagnostic Studies
- NCS/EMG: Not reliable until 3–6 weeks post-injury (distal axons still conduct early)
- At 3–6 weeks: fibrillation potentials → confirm denervation
- Nerve Action Potential (NAP) recording at surgery:
- Positive NAP in a nerve in continuity → axons have regenerated across lesion → neurolysis only
- Negative NAP → resect neuroma + repair/graft
6. INTRAOPERATIVE NEUROPHYSIOLOGY
- Avoid neuromuscular blocking agents in anesthesia
- Bipolar stimulating electrode placed proximal to lesion; recording electrode distal
- Early exploration (within 2–4 days): stimulate distal stump to define functional fascicular topography (motor vs. sensory)
- Delayed exploration (2–3 months): NAP recording most useful for nerve in continuity
- Brachial plexus: somatosensory evoked potentials (SEPs) confirm root–cord continuity, excluding preganglionic injury
- Tourniquet limits nerve conductivity after ~30 min — deflate before neurophysiology if critical
7. SURGICAL EXPOSURE
- Nerves are longitudinal structures → extensive exposure required above and below injury zone
- If delayed >2 weeks: identify nerve in unscarred tissue proximally and distally first, then dissect into zone of injury
- Tourniquet used if injury is sufficiently distal (bloodless field)
8. NERVE REPAIR TECHNIQUES
A. Neurolysis
External neurolysis: Release of nerve from external compression or scar (e.g., fracture callus, carpal tunnel). Epineurium left intact. Highly effective for compression injuries.
Internal neurolysis: Splitting of epineurium + separating fascicles from intraneural scar. More controversial — potential for additional trauma to fascicles. Indicated when:
- Nerve in continuity with severe intraneural scarring
- Double-bulb swelling (Sunderland IV) to inspect fascicle integrity
- If fascicles are intact across lesion → injury ≤ Grade III → recovery possible without grafting
B. Direct Nerve Suture (Primary/Secondary Repair)
Three techniques — no technique proven superior in RCTs:
1. Epineurial Repair (MOST COMMON)
- Sutures placed in the outer epineurium only
- External epineurial blood vessels used as rotational alignment landmarks
- 2–3 interrupted sutures typically sufficient
- Needle must pass only through epineurium — NOT catching fascicles (causes intraneural neuroma)
- Fascicles gently coapted, not overlapping
- Fibrin glue may augment repair
2. Group Fascicular (Interfascicular) Repair
- Fascicle groups are identified and internal epineurium is sutured between groups
- Matching structures sutured separately with fine interrupted nylon
- Then standard epineurial repair added
- Useful in large nerves where fascicle groups are easily identifiable; also at distal sites where fascicles are diverging into terminal branches
- Risk: more intraneural scarring than epineurial repair
- Most peripheral nerve surgeons still prefer epineurial repair
3. Individual Fascicular Repair
- Each fascicle sutured separately with perineurial sutures
- Rarely used — highest potential for surgical trauma and intraneural scarring
- Considered only when individual fascicles are distinctly separable (very distal repairs)
Suture Material and Gauge
| Nerve | Suture |
|---|---|
| Sciatic (large) | 6/0 monofilament nylon |
| Median / Ulnar | 8/0 |
| Digital nerves | 9/0 – 10/0 |
Monofilament non-absorbable nylon is standard. Repair does not need to be "watertight" — just cover fascicles.
Fascicular Alignment Strategies
- Epineurial vessel orientation (primary landmark)
- Cross-sectional fascicular pattern matching
- Intraoperative electrical stimulation (motor vs. sensory fascicle identification)
- Histochemical stains: cholinesterase (motor), carbonic anhydrase (sensory)
- NAP recording
C. Managing Nerve Gaps (for tension-free repair)
| Technique | Applicable Gap |
|---|---|
| Nerve mobilization | Small gaps |
| Nerve transposition (e.g., anterior ulnar at elbow) | Moderate gaps |
| Joint flexion + immobilization → gradual extension at 3 weeks | ≤2–3 cm |
| Bone shortening at fracture site | Variable (humerus tolerates several cm; forearm less so) |
| Nerve graft | Larger gaps where tension-free direct repair impossible |
| Nerve conduit | <3 cm gaps, small sensory nerves |
| Nerve transfer | Avulsion injuries, proximal injuries |
Critical rule: Tension on repair causes ischemia + fibrosis at coaptation site → poor outcome. The repair must be tension-free within a clean, well-vascularized bed.
D. Nerve Grafting
Indication: Nerve gap that cannot be bridged without tension after mobilization and transposition.
Technique:
- Trim nerve stumps serially until healthy fascicles are visible
- Grafts placed 10% longer than the gap (slack to prevent tension)
- Where possible, connect equivalent fascicular bundles with individual graft strands
- Fascicular bundles transected at different levels to stagger suture lines
- 1–2 sutures (8/0 or 9/0) per strand through epineurium of graft + epineurium of nerve (or perineurium of fascicular bundle)
- Side-to-side sutures away from coaptation for added strength
- Fibrin glue over ends only (not across repair)
Donor Nerve Sources (autografts):
| Nerve | Yield | Notes |
|---|---|---|
| Sural nerve (most common) | Up to 40–45 cm per leg; both legs can be harvested | Purely sensory; small area of dorsolateral foot lost |
| Medial antebrachial cutaneous | — | Upper limb |
| Lateral antebrachial cutaneous | — | Upper limb |
| Superficial radial nerve | — | Attractive if already denervated by proximal radial injury |
| Superficial peroneal nerve | — | Lower limb |
| Distal posterior interosseous nerve | — | Small, wrist level |
| Great auricular nerve | — | Head/neck |
| Cervical plexus nerves | — | Head/neck |
Sural nerve harvest technique: Incision behind lateral malleolus → identify nerve → gentle traction to locate proximally → mid-calf incision → popliteal fossa incision → divide proximally, deliver distal to proximal, divide at ankle. Proximal stump placed deep to bury neuroma.
Outcome of autograft: Sensory recovery better than motor (fascicular mismatch in mixed nerves).
E. Nerve Allografts
Decellularized allografts (e.g., Avance, AxoGen):
- Cellular material removed; extracellular matrix scaffold preserved
- No living Schwann cells → effective length limited to ~3 cm
- No immunosuppression required
- Results: sensory recovery ~77% (useful S3 sensation); motor recovery poorer
- Increasingly used; more RCT data awaited
Fresh cadaveric nerve allografts:
- Theoretical advantage: living Schwann cells present
- Highly immunogenic; require immunosuppression → not routinely used clinically
F. Nerve Conduits (Tubes)
- Hollow tubes that bridge a nerve gap, channeling regenerating axons
- Materials: polyglycolic acid (PGA), collagen-based, poly-lactic-co-glycolic acid (PLGA)
- Indication: Short gaps (<3 cm), small-diameter nerves, usually sensory (e.g., digital nerves, gaps 8–20 mm)
- Limitations: Cannot support regeneration across >2–3 cm without Schwann cells
- Research ongoing: conduits seeded with cultured Schwann cells; not yet in clinical practice
- Freeze-thawed skeletal muscle autografts (basement membrane scaffold): useful for 1-cm defects but ineffective >5 cm
G. Nerve Transfers (Neurotization)
Principle: A healthy expendable donor nerve (or fascicle) is cut and coapted directly to a denervated recipient nerve, bypassing the injury site entirely.
Advantages over grafting:
- Shorter reinnervation distance → faster recovery
- Avoids long graft with multiple suture lines
- Can be performed even when proximal nerve root is avulsed (no proximal stump available)
Key principles:
- Donor nerve normal function will be permanently lost → select carefully
- Donor must be synergistic with recipient function (facilitates re-education)
- Direct suture preferred (no interpositional graft)
- One transfer = one function; multiple transfers needed for multiple movements
- Cannot achieve function independent of the donor nerve (co-contraction / coordination issues)
Common Upper Extremity Motor Nerve Transfers:
| Motor Deficit | Recipient Nerve | Donor Nerve |
|---|---|---|
| Elbow flexion | Biceps/brachialis branches of musculocutaneous nerve (MCN) | Ulnar fascicle to FCU OR Median fascicle to FDS/PL/FCR (Oberlin transfer) |
| Elbow flexion | MCN | Medial pectoral nerve branches OR Thoracodorsal nerve |
| Shoulder abduction | Axillary nerve | Radial nerve branch to triceps long head (Leechavengvong transfer) |
| Shoulder abduction | Suprascapular nerve | Spinal accessory nerve (CN XI) |
Sensory nerve transfers: e.g., ulnar nerve branch to 4th web space → transferred to median nerve branches (thumb/1st web space) for median nerve injury — restores some sensation, though quality is limited.
Brachial plexus avulsion: Nerve transfers are the cornerstone of reconstruction (no proximal stump for grafting). Extra-plexal donors used: phrenic nerve, intercostal nerves, contralateral C7, hypoglossal nerve.
H. Secondary (Delayed) Repair
- Performed after closed blunt injuries or failed primary repair
- Substantial exposure required (identify nerve in normal tissue before entering scar zone)
- Neuroma on proximal stump + swelling on distal stump → trim both to healthy fascicles
- Gap usually larger than anticipated after trimming
- Double-bulb swelling = Sunderland Grade IV (fascicles ruptured, only scarred epineurium in continuity) → resect and repair
I. Tendon and Muscle Transfers (Late Reconstruction)
Indicated when nerve repair is no longer viable (>12–18 months, end-organ denervation) or when recovery is inadequate:
Basic tenets:
- Donor must be expendable and of similar excursion/power
- One transfer → one function
- Synergistic transfers are easier to rehabilitate
- Straight line of pull optimal
- Expect one grade of motor strength loss after transfer
Common examples:
- High radial nerve palsy: Pronator teres → ECRB (wrist extension); FCU → EDC (finger extension); PL → EPL (thumb extension)
- Low median nerve palsy (opponensplasty): FDS ring finger / EIP / ADM / PL → APB
- Free functioning muscle transfer (e.g., gracilis): for late elbow flexion or finger flexion when no local donor available
9. OUTCOMES OF NERVE REPAIR
Factors Affecting Outcome
| Factor | Better Outcome | Worse Outcome |
|---|---|---|
| Age | Young (better regeneration, cortical plasticity, shorter limb) | Elderly |
| Level of injury | Distal (short distance to end organ) | Proximal |
| Nerve type | Pure sensory or pure motor | Mixed nerve (fascicular mismatch) |
| Specific nerve | Radial > Median > Ulnar; C5-C6 > C8-T1; Tibial > Peroneal | — |
| Mechanism | Sharp laceration | Crush, avulsion, high-velocity gunshot |
| Timing | Early (<6 months) | Late (>1 year) |
| Type of repair | Neurolysis (positive NAP) > direct repair > graft > proximal nerve transfer | — |
| Wound bed | Clean, well-vascularized | Infected, scarred, irradiated |
Hierarchy of Expected Recovery (best to worst)
- Spontaneous recovery (no surgery needed)
- Neurolysis alone (positive intraoperative NAP)
- Primary direct end-to-end repair
- Nerve graft
- Nerve transfer (distal transfers better than proximal transfers with graft)
- Tendon/muscle transfers
Motor Strength Grading (MRC Scale — used for outcome reporting)
M0 → M5; M3 = movement against gravity (functional threshold)
Sensory Grading (modified Mackinnon-Dellon / BMRC)
S0 → S4; S3+ = two-point discrimination recovery (functional threshold)
10. SPECIAL SCENARIOS
Neuroma-in-Continuity
- Blunt/traction injury; nerve appears in continuity but may have internal disruption
- Intraoperative NAP:
- Positive NAP → axons regenerating → neurolysis only
- Negative NAP → resect neuroma + repair/graft
Brachial Plexus Injury
- Observe 3 months for spontaneous recovery before intervention
- Preganglionic signs: Horner syndrome (ptosis, miosis, anhidrosis — sympathetic chain), scapular winging (long thoracic nerve), elevated hemidiaphragm on CXR (phrenic nerve), preserved sensory NCS with absent motor (DRG intact)
- EMG: loss of innervation in paraspinal muscles = preganglionic
- Preganglionic → no proximal stump → nerve transfer mandatory
- Priority of reconstruction: elbow flexion > shoulder > hand
Nerve Repair Priorities (Upper Extremity)
- Minimum functional goals: stable shoulder, elbow flexion, median nerve sensibility, prehensile grip
Nerve Repair Priorities (Lower Extremity)
- Minimum: stable hip + knee, tibial nerve plantar sensation (allows ambulation)
11. KEY NUMBERS TO REMEMBER
| Parameter | Value |
|---|---|
| Axon regeneration rate | ~1 mm/day (~1 inch/month) |
| Distal axon conduction after injury | 2–4 days |
| Neurophysiology reliability post-injury | Unreliable for 3–6 weeks |
| Joint flexion technique: max gap for direct repair | ≤2–3 cm |
| Nerve conduit: max gap | <3 cm |
| Decellularized allograft: effective length | ~3 cm |
| Sural nerve graft length | Up to 40–45 cm per leg |
| Motor end plate viability | 12–18 months after denervation |
| Best surgical timing | ≤10–14 days |
| Primary sensory neuron death (unrepaired distal transection) | ~30% |
| Sensory neuron death (preganglionic) | Up to 80% |
| Motor neuron death (preganglionic) | Up to 50% |
Sources: Current Surgical Therapy 14e (p. 980–987) | Rockwood and Green's Fractures in Adults 10th ed. (p. 767–781) | Miller's Review of Orthopaedics 9th ed. | Sabiston Textbook of Surgery
GIVE PICTURE FOR anatomyand wallerian degeneration
Loading Image
Loading Image
Anatomy of a Peripheral Nerve
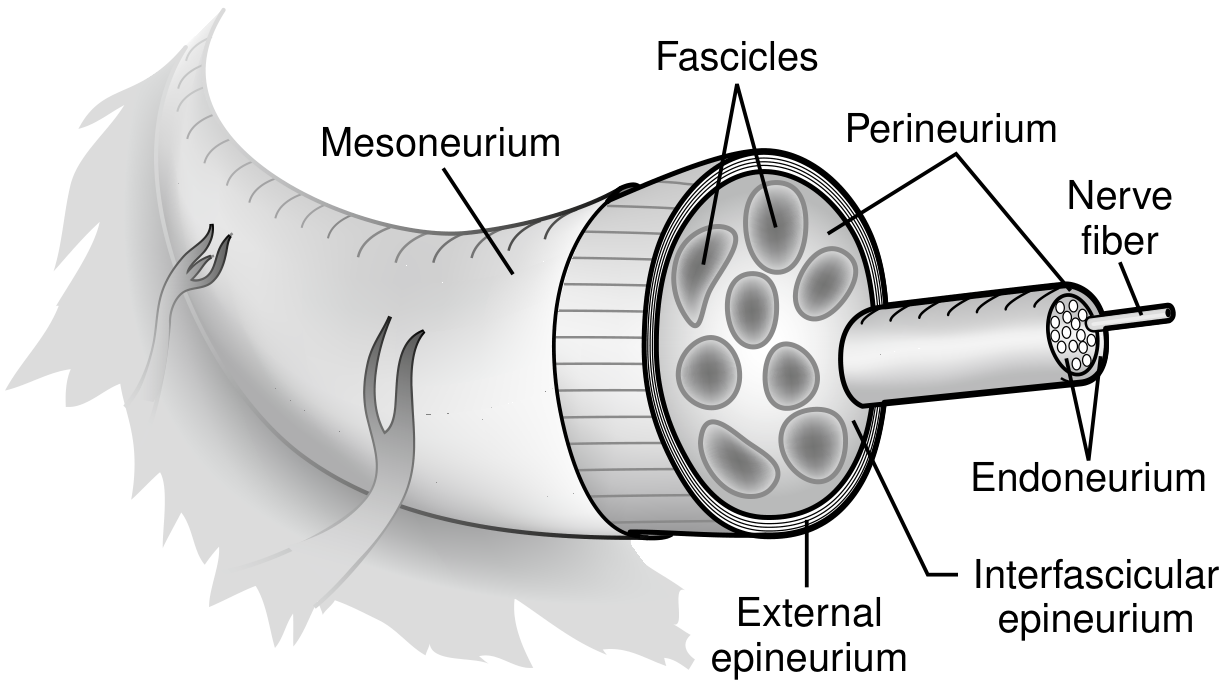
From Current Surgical Therapy 14e — showing Mesoneurium, External epineurium, Interfascicular epineurium, Perineurium, Fascicles, Endoneurium, and Nerve fiber, from outside in.
Wallerian Degeneration & Axonal Regeneration
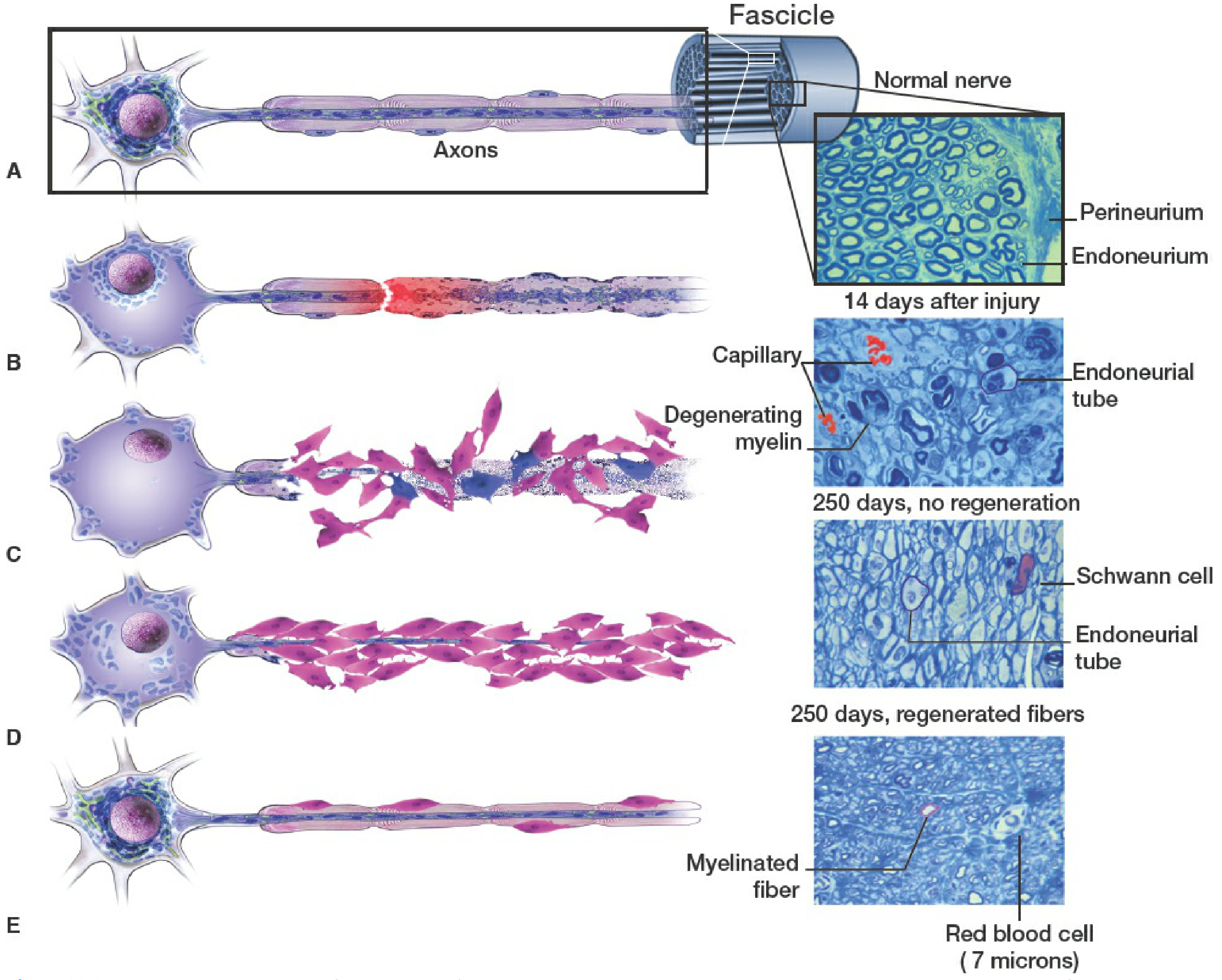
From Rockwood & Green's Fractures in Adults 10th ed. — Sequential stages after nerve transection:
| Stage | What Happens |
|---|---|
| A | Intact myelinated axon within a fascicle (normal nerve cross-section: perineurium + endoneurium visible) |
| B | Axon + myelin fragment and degenerate distal to the transection site (red = injury zone) |
| C | Schwann cells proliferate, phagocytose myelin debris — degenerating myelin fragments visible; at 14 days endoneurial tubes are emptied; at 250 days without regeneration only Schwann cells + empty endoneurial tubes remain |
| D | Regenerating axon sprouts grow distally from the proximal stump, guided by Schwann cells lining the endoneurial tubes (bands of Büngner) |
| E | Regenerated axon — smaller diameter than original, remyelinated; connects with target organ. At 250 days with regeneration: myelinated fibers visible in cross-section |
Key histological finding: The cross-sections show that with successful regeneration (250 days), myelinated fibers re-populate the endoneurial tubes, though thinner than normal. Without regeneration, only empty endoneurial tubes lined by Schwann cells remain — these tubes progressively shrink and fibrosis sets in, explaining why delayed repair has worse outcomes.
This is a shared conversation. Sign in to Orris to start your own chat.