Tetrology of Fallot
Reading File
Finding Sources
Searching PubMed
"Tetralogy of Fallot"[MeSH Terms]
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Reading File
Reading File
Here is a comprehensive overview of Tetralogy of Fallot (ToF).
Tetralogy of Fallot
Definition & Epidemiology
Tetralogy of Fallot is the most common cyanotic congenital heart disease, accounting for ~5% of all congenital cardiac malformations, with an incidence of approximately 420 per million live births.
The Four Cardinal Defects
All four features arise from a single embryologic defect: anterosuperior displacement of the infundibular (outlet) septum, causing unequal division of the truncus arteriosus.
| # | Defect | Details |
|---|---|---|
| 1 | Right Ventricular Outflow Tract (RVOT) Obstruction | Usually subpulmonic (infundibular) stenosis; may be valvular stenosis or complete pulmonary atresia |
| 2 | Large, Unrestrictive VSD | Near the membranous septum; allows bidirectional shunting |
| 3 | Overriding Aorta | Aortic valve straddles the VSD, receiving blood from both ventricles |
| 4 | Right Ventricular Hypertrophy | Secondary to high pressure load from RVOT obstruction |
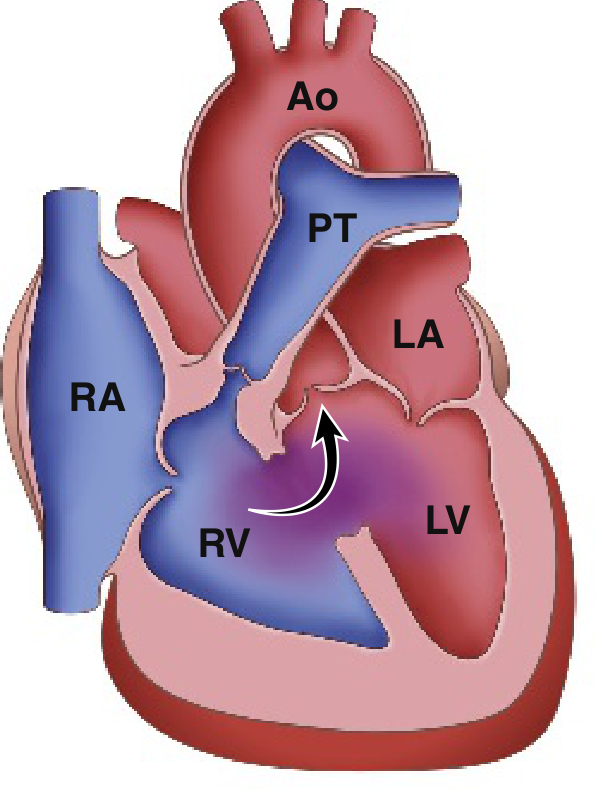
Classic ToF: right-to-left shunting across the VSD. Ao = Aorta, PT = Pulmonary Trunk, RA/RV/LA/LV = cardiac chambers. (Robbins & Kumar Basic Pathology)
Pathophysiology
The physiologic result is decreased pulmonary blood flow + right-to-left shunting of deoxygenated blood across the VSD into the aorta.
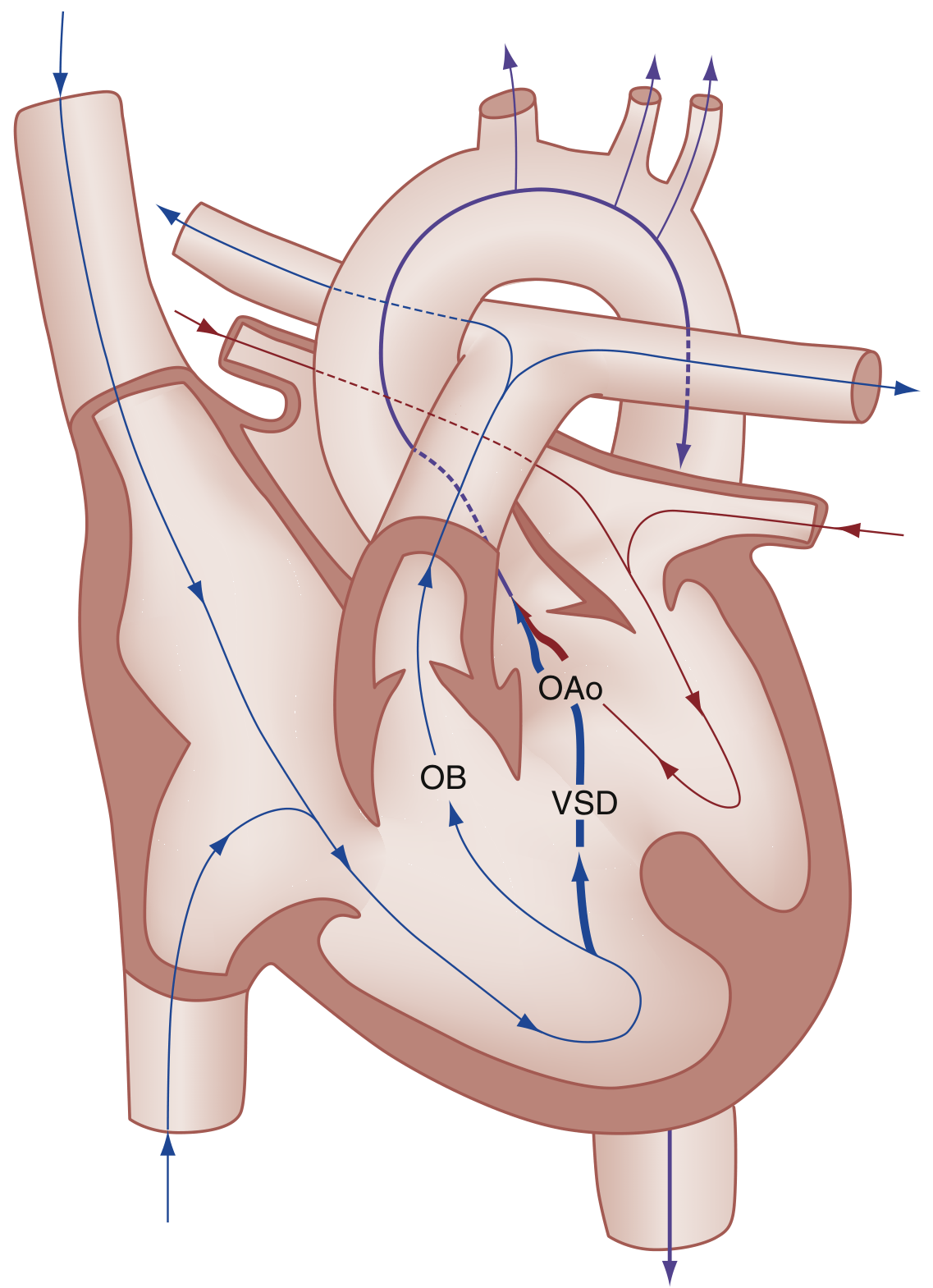
Hemodynamics of ToF: severity of shunt depends on degree of RVOT obstruction, VSD size, and systemic vascular resistance (SVR). (Rosen's Emergency Medicine)
- Severity is proportional to the degree of RVOT obstruction
- Mild obstruction → left-to-right shunt only → "Pink Tet" (acyanotic)
- Severe obstruction → early profound cyanosis
- In pulmonary atresia: pulmonary flow entirely via PDA or bronchial collaterals
Morphology
- Boot-shaped heart (coeur en sabot) — from RV hypertrophy elevating the cardiac apex
- Proximal aorta dilated; pulmonary trunk hypoplastic
- RV wall markedly hypertrophied (may exceed LV thickness)
- Left-sided chambers are normal in size
Clinical Features
| Feature | Details |
|---|---|
| Cyanosis | Worsens with crying, feeding, exertion; may be absent at birth |
| Systolic ejection murmur | Left sternal border (from RVOT obstruction, NOT the VSD) |
| Clubbing | Fingers and toes; from chronic hypoxemia |
| Polycythemia | Compensatory response to chronic hypoxia |
| Squatting | Older children squat to increase SVR and reduce R→L shunt |
| Chest X-ray | Boot-shaped heart, decreased pulmonary vascular markings, possible right-sided aortic arch (~25%) |
| ECG | RV hypertrophy, right axis deviation |
Tet Spells (Hypercyanotic/Hypoxic Spells)
Peak incidence: 2–4 months of age
Trigger: Anything that suddenly ↓SVR (crying, defecation, fever) or causes tachycardia/hypovolemia
Vicious cycle:
↓SVR → ↑R→L shunt → ↓PaO₂, ↑PCO₂, ↓pH → hyperpnea → ↑venous return to RV → more shunting → worsening hypoxia
Clinical signs: Hyperpnea, prolonged crying, deepening cyanosis, decreased murmur intensity, possible limpness, seizures, stroke, or death.
Management of Tet Spells (Rosen's Box 165.8)
- Knee-to-chest position (or squatting) — increases SVR, reduces R→L shunt
- Supplemental oxygen (limited effect alone)
- Morphine 0.1–0.2 mg/kg IV/IM (sedation, ↓catecholamines) — note risk of histamine-mediated vasodilation
- Fentanyl 1 µg/kg IV/IM or 1.5–2 µg/kg intranasal (preferred over morphine)
- Midazolam 0.2–0.3 mg/kg intranasal
- Ketamine 1–2 mg/kg IV or 3–5 mg/kg IM — excellent (↑SVR + sedation)
- NaHCO₃ 1 mEq/kg IV — if metabolic acidosis (pH <7.4)
- Phenylephrine 0.01–0.02 mg/kg IV — α-agonist to ↑SVR
- Propranolol 0.1–0.2 mg/kg IV — may reduce infundibular spasm
- IV fluids — to ↑preload and pulmonary flow
Associated Defects
- Right-sided aortic arch (~25%)
- ASD
- Anomalous coronary artery origin (left coronary from right coronary artery in ~5% — critical surgical relevance)
- Pulmonary atresia with VSD = extreme form of ToF
Surgical Management
Palliative (staged)
- Modified Blalock-Taussig (BT) shunt: subclavian artery → ipsilateral pulmonary artery anastomosis
- Used when: severe cyanosis, very narrow RVOT, hypoplastic central pulmonary arteries
Definitive Repair
- VSD closure + RVOT relief (infundibular resection ± transannular patch across pulmonary valve annulus)
- Preferred: early single-stage repair in infancy
- Surgical mortality: ~5–10% in uncomplicated cases within first 2 years
- PGE₁ (prostaglandin E₁) infusion may be needed pre-operatively to maintain PDA patency in severely cyanotic neonates
Post-repair Residua (almost universal)
- Pulmonary regurgitation (PR) — most common long-term problem
- RV dilatation, RVOT aneurysm
- Residual RVOT obstruction or VSD
- Aortic root dilatation
- Right bundle branch block (from right ventriculotomy)
- Complete heart block, ventricular arrhythmias
- CMR is gold standard for RV assessment post-repair; guides timing of pulmonary valve replacement
Pulmonary Valve Replacement
- Surgical redo vs. transcatheter pulmonary valve replacement (TPVR) — both are options; recent 2025 systematic review (PMID 40122137) compared outcomes
- Timing of intervention guided by RV volumes/function on CMR
Recent Evidence
- Primary vs. staged repair in symptomatic neonates (Meta-analysis, Cardiol Young 2024; PMID 39444206): systematic review comparing outcomes of primary vs. staged surgical approach in symptomatic neonates.
- Surgical vs. transcatheter pulmonary valve redo (Expert Rev Cardiovasc Ther 2025; PMID 40122137): systematic review of redo pulmonary valve options in repaired ToF patients.
Sources: Robbins & Kumar Basic Pathology | Rosen's Emergency Medicine | Grainger & Allison's Diagnostic Radiology | The Developing Human (Moore) | Braunwald's Heart Disease
How crying fever decreases SVR
Finding Sources
Finding Sources
Searching the Web
how does crying decrease systemic vascular resistance SVR physiology
Finding Sources
Finding Sources
Finding Sources
Searching the Web
fever decreases SVR mechanism prostaglandin nitric oxide vasodilation physiology
This is an important clarification — crying and fever are two separate triggers, each decreasing SVR through different mechanisms. The textbooks (and Merck Manual) actually categorize them differently.
How Crying and Fever Each Affect SVR
First — A Clarification on the Textbook Statement
Looking back at Rosen's and the Merck Manual carefully, the triggers for tet spells are listed as:
- ↓ SVR triggers: Playing, kicking legs on awakening, vigorous physical activity
- ↓ O₂ saturation triggers: Crying, defecating
- Other: Tachycardia, hypovolemia
Crying is not primarily a direct SVR-lowering stimulus — its main effect is different. Here's the full breakdown:
1. Crying → ↑ Intrathoracic Pressure → ↑ Venous Return → Worsens Shunting
Crying involves a Valsalva-like maneuver and forceful expiratory effort:
- The child strains and holds breath during a cry, raising intrathoracic pressure transiently
- This compresses pulmonary vasculature, increasing pulmonary vascular resistance (PVR) and reducing pulmonary blood flow
- Simultaneously, crying causes agitation and catecholamine release (sympathetic activation), which:
- ↑ Heart rate → shortens RV filling time
- Causes infundibular (RVOT) spasm via β-adrenergic stimulation of infundibular muscle → worsens RVOT obstruction
- The increased venous return overwhelms the obstructed RV, forcing more blood across the VSD into the aorta
So crying's primary mechanism is ↑ RVOT obstruction + ↑ PVR, not ↓SVR. The result is the same — more R→L shunting — but via a different pathway.
This is why morphine helps tet spells: it suppresses the catecholamine surge and reduces the cry-driven agitation, breaking the infundibular spasm cycle.
2. Fever → ↓ SVR via Peripheral Vasodilation
Fever decreases SVR through direct vascular mechanisms:
| Mediator | Mechanism |
|---|---|
| PGE₂ (prostaglandin E₂) | Released during fever via COX → arachidonic acid → PGH₂ → PGE₂; acts on vascular smooth muscle to cause vasodilation (same prostaglandins that cause the flushed, warm skin of fever) |
| Nitric Oxide (NO) | Cytokine-stimulated endothelial NO release → cGMP in smooth muscle → relaxation → vasodilation |
| Direct thermal effect | Heat itself causes smooth muscle relaxation in arterioles (metabolic vasodilation) |
| Increased metabolic demand | Fever raises tissue O₂ consumption → local metabolic byproducts (CO₂, lactate, adenosine) cause local arteriolar dilation → ↓ SVR |
This is why febrile children with ToF are at risk: the systemic vasodilation from fever lowers the "wall" of SVR that normally keeps blood flowing left-to-right (or balanced), tipping the balance toward R→L shunting across the VSD.
Summary: Pathways to R→L Shunting
CRYING FEVER
↓ ↓
Agitation + catecholamines PGE₂, NO, heat
↓ ↓
Infundibular spasm Peripheral vasodilation
↑ RVOT obstruction ↓ SVR
↓ ↓
↑ RV pressure Pressure gradient shifts
↓ ↓
└──────────────────────────────┘
↑ Right-to-Left shunting across VSD
↓
↑ Cyanosis (Tet Spell)
Both converge on the same endpoint but through mechanistically distinct pathways — which is why treatment targets both (knee-chest/phenylephrine for SVR; morphine/fentanyl for infundibular spasm/catecholamine surge).
This is a shared conversation. Sign in to Orris to start your own chat.