I now have all the material needed for a comprehensive answer. Let me compose the full detailed notes.
MULTIPLE SCLEROSIS — PART 1: CLINICAL TYPES & McDONALD'S CRITERIA
(Harrison's 22e · Bradley & Daroff's Neurology · Davidson's perspective)
⚡ QUICK EXAM FRAMEWORK (What Examiners Want)
MS = Chronic inflammatory demyelinating disease of CNS characterized by lesions disseminated in SPACE and TIME.
The two pillars of diagnosis: Dissemination in Space (DIS) + Dissemination in Time (DIT) → McDonald's criteria operationalize these.
SECTION 1: CLINICAL TYPES / PHENOTYPES OF MULTIPLE SCLEROSIS
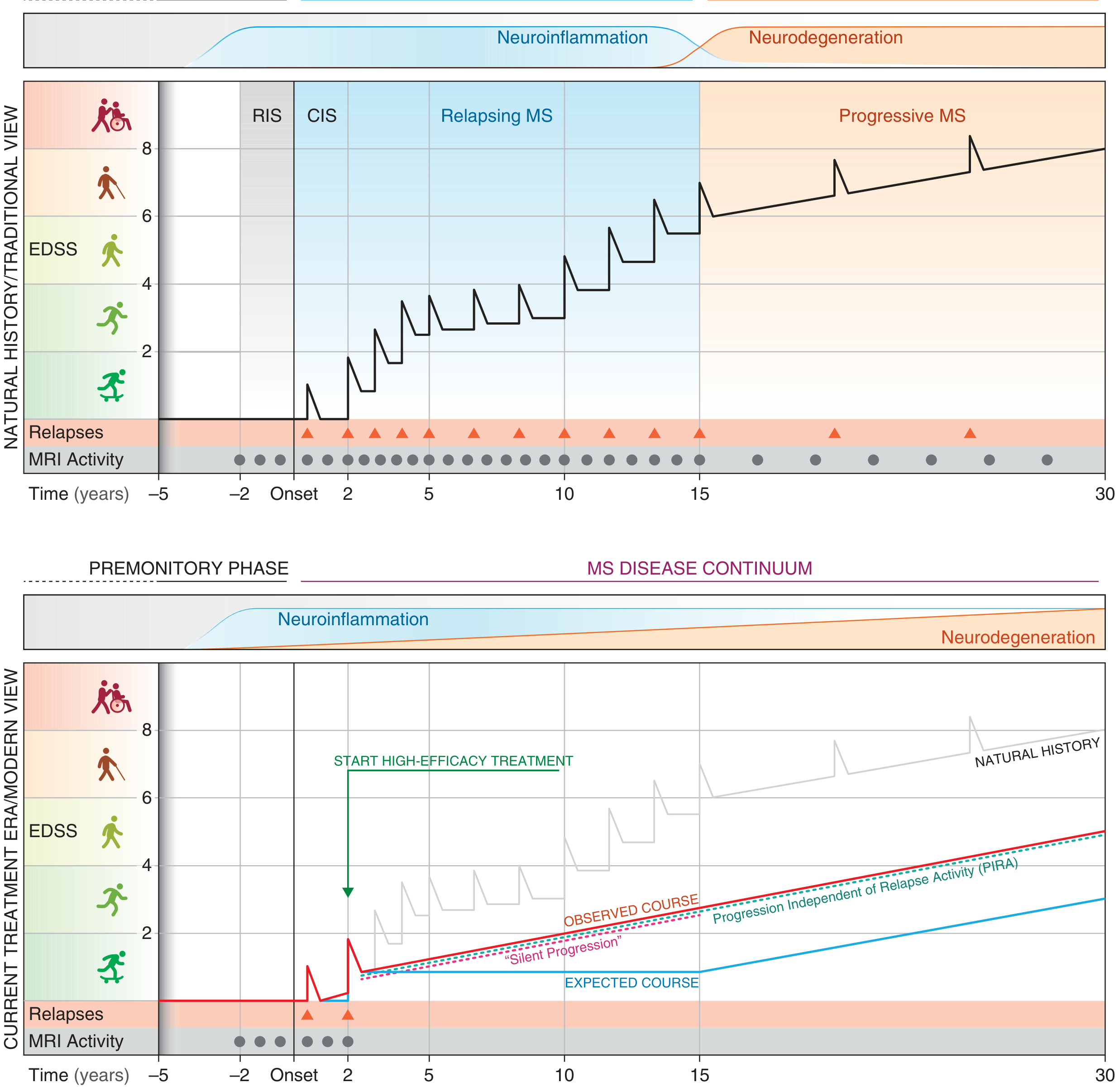
1.1 The Modern Unitary View (Harrison's 22e, 2025 — Key Conceptual Shift)
Before diving into subtypes, understand the paradigm shift that Harrison's 22e emphasizes and which examiners love asking about:
Traditional (old) view: MS had three distinct forms. Relapses caused by inflammation → progression caused by neurodegeneration. These were considered separate processes.
Modern (current) view: MS is a single disease (unitary disease concept) in which both inflammation and neurodegeneration are present simultaneously from the very beginning in most patients. This is supported by:
- Genetics — same HLA-DRB1*1501 risk gene across all phenotypes
- Epidemiology — similar geographic distribution
- Immunology — same autoimmune inflammatory process
- Pathology — demyelination and axonal loss occur in all forms
Exam trap: The classical subtypes are still clinically useful for assessment and management — so you must know both the old classification AND the new concept.
1.2 Classical Clinical Subtypes (2013 Lublin Revised Classification)
TYPE 1: RELAPSING-REMITTING MS (RRMS)
| Feature | Detail |
|---|
| Frequency | ~85–90% of all MS cases at onset |
| Alternate name | Bout-onset MS |
| Hallmark | Discrete attacks (relapses/exacerbations) of neurological dysfunction |
| Attack onset | Evolve over days to weeks (rarely hours) |
| Recovery | Often substantial or complete recovery over weeks to months early in disease; recovery becomes less complete as attacks continue |
| Between attacks | Patients were previously thought stable; now known that most have "silent" subclinical progression even when relapse-free |
| Sex ratio | Female:Male = 2–3:1 |
| Age of onset | Mean ~30 years, peak ~25 years |
| MRI activity | New MRI lesions occur 9–10 times more often than new clinical attacks |
Key modern concept — Silent Progression in RRMS: Even while on highly effective therapies that abolish relapses, patients show insidious worsening — this is called Progression Independent of Relapse Activity (PIRA).
Two mechanisms of disability accumulation in RRMS:
- RAW (Relapse-Associated Worsening): Disability from incomplete relapse recovery — once thought to be the primary driver
- PIRA (Progression Independent of Relapse Activity): Disability from ongoing neurodegeneration without clinical relapses — now recognized as the dominant driver in RRMS, SPMS, and PPMS
TYPE 2: SECONDARY PROGRESSIVE MS (SPMS)
| Feature | Detail |
|---|
| Origin | Always begins as RRMS — this is a definitional requirement |
| Transition | At some point, RRMS transitions to steady, progressive neurological deterioration |
| Relapses | Relapses may or may not continue (see active vs inactive SPMS) |
| Disability | Produces greater fixed neurological disability than RRMS |
| Practical definition | Patient with RRMS who has developed permanent walking disability NOT due exclusively to relapses |
| Objective measure | EDSS ≥4 + Functional Status Scale (FSS) motor score ≥2 supports SPMS diagnosis |
| Rate of conversion (pre-treatment era) | ~3% per year → majority of RRMS eventually became SPMS |
| Rate of conversion (treatment era) | <1% per year due to highly effective disease-modifying therapies |
TYPE 3: PRIMARY PROGRESSIVE MS (PPMS)
| Feature | Detail |
|---|
| Frequency | ~10–15% of all MS cases |
| Hallmark | Steady neurological decline from onset — NO initial relapsing phase |
| Sex ratio | More even (~1:1 female:male) — unlike RRMS |
| Age of onset | Later onset — mean ~40 years (vs ~30 in RRMS) |
| Disability | Develops faster relative to first symptom compared to RRMS |
| Relapses | Some PPMS patients DO experience relapses over course — shows overlap with RRMS |
| Common presentation | Progressive spastic paraparesis (spinal cord dominant) |
| MRI | Fewer gadolinium-enhancing lesions vs RRMS; more spinal cord atrophy |
Exam point: Despite clinical differences, PPMS represents the same underlying illness as RRMS/SPMS — genetics, pathology, and immunology are essentially identical.
Special Categories Under the 2013 Lublin Revised Classification
The 2013 revision added important activity and progression modifiers to the above subtypes — these appear in newer exam questions:
| Modifier | Definition | Applied To |
|---|
| Active | New/enlarging T2 lesions on MRI OR new relapses in previous year | RRMS, SPMS, PPMS |
| Not active | No new MRI lesions, no relapses | Any type |
| Worsening | Confirmed disability increase over time | Progressive forms |
| Not worsening | Stable disability | Progressive forms |
This gives combinations like:
- Active RRMS — relapsing (standard RRMS)
- Active SPMS — progressive + still having relapses or new MRI lesions
- Inactive SPMS — progressive disability without relapses/new MRI activity
- Active PPMS — same modifiers applied to primary progressive
Additional Phenotypes / Stages (Critical for Exam)
CIS — CLINICALLY ISOLATED SYNDROME
- First single clinical demyelinating episode lasting ≥24 hours at normal body temperature
- Does NOT yet meet criteria for MS (only ONE event so far)
- Can represent the very first attack of MS — ~50–85% convert to MS within years
- High-risk CIS → large T2 lesion burden on MRI → almost certain conversion to MS
- Low-risk CIS → normal MRI → much lower conversion risk
- Treatment implication: Starting DMT after high-risk CIS reduces conversion to CDMS (BENEFIT, CHAMPS, ETOMS trials)
RIS — RADIOLOGICALLY ISOLATED SYNDROME
- Incidental MRI lesions consistent with MS discovered on imaging done for another reason (e.g., headache workup)
- No clinical symptoms or attacks at time of discovery
- ~1/3 will develop a first clinical attack within 5 years
- Represents the presymptomatic phase of MS
RELAPSING MS (RMS)
- Umbrella term encompassing all relapsing patients: RRMS + SPMS with ongoing relapses
- Used in clinical trial eligibility and regulatory approvals for DMTs
ACTIVE PROGRESSIVE MS
- SPMS or PPMS patients who experience relapses OR have new MRI lesions
1.3 Disease Course — The Big Picture
Interpreting this diagram (Harrison's Figure 455-1):
- Top panel (Traditional/pre-treatment view): RIS → CIS → RRMS (disability accumulates from incomplete relapse recovery) → SPMS (when relapses stop but progression continues)
- Bottom panel (Modern/treatment era): With highly effective therapy, relapses are abolished. But "Silent Progression" (PIRA) remains. The disease continues as a continuum with both neuroinflammation AND neurodegeneration occurring together throughout
This diagram is extremely high-yield — knowing it differentiates a textbook student from a guess-paper student!
SECTION 2: McDONALD'S CRITERIA (2017 Revision)
2.1 Historical Evolution — Must Know for Short Notes/Viva
| Era | Criteria | Requirements |
|---|
| Pre-2001 | Poser criteria (1983) | 2 clinical attacks + examination evidence → Clinically Definite MS (CDMS) |
| 2001 | First McDonald criteria | Allowed MRI activity to substitute for second clinical attack |
| 2005 | Revised McDonald criteria | Simplified MRI rules |
| 2010 | Further revision | Made it easier to use MRI for DIS and DIT |
| 2017 (current) | Thompson et al., 2018, Lancet Neurology | Added cortical lesions to DIS; added OCBs to substitute for DIT in certain settings |
The core principle of McDonald criteria: Diagnosis can be made earlier and more reliably using MRI biomarkers rather than waiting for a second clinical attack. This enables earlier treatment.
2.2 The 2017 McDonald Criteria — Complete Breakdown
PREREQUISITE (Non-negotiable):
At least ONE clinical attack (CIS) attributable to CNS demyelination must be present before applying these criteria for RRMS.
A "clinical attack" = subjective report/objective finding of neurological symptom typical of MS, lasting ≥24 hours, at normal body temperature, without fever/infection.
PILLAR 1: DISSEMINATION IN SPACE (DIS)
DIS means lesions in ≥2 different anatomical locations in the CNS.
MRI DIS criteria (2017): ≥1 T2 lesion in at least 2 of 4 characteristic locations:
| Location | Notes |
|---|
| 1. Periventricular | Lesions adjacent to ventricles (classic Dawson's fingers) |
| 2. Cortical / Juxtacortical | NEW in 2017 — cortical lesions added; previously only juxtacortical (U-fibers) counted |
| 3. Infratentorial | Brainstem/cerebellum lesions |
| 4. Spinal cord | Cervical or thoracic cord lesions |
Key 2017 change: Cortical lesions now count as a distinct location AND can be used interchangeably with juxtacortical lesions.
If ≥2 clinical attacks: DIS can be demonstrated clinically (different anatomical locations of the two attacks) without requiring MRI.
PILLAR 2: DISSEMINATION IN TIME (DIT)
DIT means evidence of disease activity at two different time points.
Three ways to satisfy DIT (2017):
| Method | Details |
|---|
| A. Simultaneous lesions on single MRI | Presence of both gadolinium-enhancing AND non-enhancing T2 lesions at any time on the same scan |
| B. New lesion on follow-up MRI | A new T2 or Gd-enhancing lesion on follow-up scan compared to baseline (regardless of when the baseline scan was done) |
| C. Second clinical attack | A second episode of neurological dysfunction consistent with MS (in different location from first = also satisfies DIS) |
Key 2017 change: The 2010 criteria required that symptomatic brainstem/spinal cord lesions NOT be used for DIS or DIT. The 2017 criteria removed this restriction — any typical MS location now counts.
THE GAME-CHANGING 2017 ADDITION: CSF Oligoclonal Bands (OCBs)
For patients with ONE clinical attack meeting DIS criteria:
→ Presence of CSF oligoclonal bands (OCBs) can substitute for DIT
This means: CIS + DIS on MRI + positive OCBs = Diagnosis of MS (without waiting for DIT)
Why was OCBs added?
- Multiple studies showed OCBs are an independent risk factor for further clinical activity
- OCBs found in >90% of confirmed MS patients
- Absence of OCBs does NOT exclude MS (10–20% of confirmed MS lack OCBs at any given time)
- The IgG index (CSF IgG/Albumin ÷ Serum IgG/Albumin) is elevated in >90% of MS patients
PPMS Diagnosis — Special Criteria
PPMS has its own McDonald criteria section (important for exams):
For diagnosis of PPMS, need ALL THREE:
- 1 year of progressive disability independent of relapse
- PLUS two of the following three MRI criteria:
- ≥1 DIS T2 lesion in the brain (periventricular, cortical, juxtacortical, or infratentorial)
- ≥2 T2 lesions in the spinal cord
- Positive CSF OCBs
Note: For PPMS, if CSF OCBs are absent, intrathecal IgG synthesis documentation can also support diagnosis.
2.3 McDonald Criteria Summary Table (2017) — Exam-Ready
| Clinical Presentation | Additional Data Needed for Diagnosis |
|---|
| ≥2 attacks + ≥2 objective clinical lesions | None — clinical diagnosis is sufficient |
| ≥2 attacks + 1 objective lesion | DIS by MRI (≥1 T2 lesion in ≥2 of 4 locations) OR wait for another attack in different CNS site |
| 1 attack + ≥2 objective lesions | DIT by MRI (simultaneous Gd+ & Gd– lesions, or new T2/Gd+ lesion on follow-up) OR second clinical attack; OR positive CSF OCBs |
| 1 attack + 1 objective lesion (CIS) | DIS by MRI AND DIT by MRI; OR DIS by MRI + positive CSF OCBs |
| Progressive neurological decline from onset (PPMS) | 1 year disability progression + 2 of 3: ≥1 brain T2 DIS lesion, ≥2 cord T2 lesions, positive CSF OCBs |
(Adapted from Thompson et al., 2018, Lancet Neurol 17:162 — Harrison's 22e Table 455-4)
2.4 Key MRI Concepts for McDonald Criteria
Dawson's Fingers:
- Lesions oriented perpendicular to the lateral ventricles on sagittal MRI
- Represent perivenular inflammation along medullary veins
- Located in periventricular white matter
- Classic and highly characteristic of MS
Gadolinium Enhancement:
- Gd crosses disrupted blood-brain barrier (BBB) at sites of acute inflammation
- Marks active/new inflammatory lesions
- Enhancement typically lasts <1 month
- Active Gd+ lesion = acute MS plaque
T2/FLAIR lesions:
- Remain as permanent hyperintense foci after inflammation resolves
- Represent the cumulative "burden of disease"
- Found in virtually all MS patients (>95%)
Black Holes (T1 hypointense lesions):
- ~1/3 of T2 lesions appear as T1 black holes
- Indicate irreversible demyelination and axonal loss
- Progress over time in chronic lesions
Central Vein Sign:
- A vein visible at the center of an MS lesion on susceptibility-weighted imaging
- Helps distinguish MS from other white matter diseases
- Now mentioned in Harrison's 22e as diagnostically useful
2.5 CSF in McDonald Criteria Context
| CSF Finding | Significance |
|---|
| Oligoclonal bands (OCBs) ≥2 | Present in >90% confirmed MS; absent in paired serum; substitute for DIT in 2017 criteria |
| IgG index elevated (>0.7) | Raised in >90% MS; reflects intrathecal IgG synthesis |
| IgG synthesis rate | Elevated; calculated from serum and CSF IgG and albumin |
| Mild pleocytosis (<50 cells/μL) | Present in ~25% MS, usually early RRMS |
| Predominantly lymphocytes/T cells | |
| CSF protein | Usually normal or mildly elevated |
| Red flags against MS in CSF | >75 cells/μL, PMN-predominant pleocytosis, protein >100 mg/dL → suspect alternative diagnosis |
2.6 Evoked Potentials (Supporting Evidence)
Not part of McDonald criteria but support DIS in atypical presentations:
| Test | Detects | MS finding |
|---|
| VEP (Visual Evoked Potential) | Optic nerve/visual pathway delay | Prolonged P100 latency — most useful; normal amplitude initially |
| SSEP (Somatosensory EP) | Dorsal column/posterior thalamic pathway | Prolonged latency |
| BAER (Brainstem Auditory EP) | Brainstem lesions | Prolonged latency |
Key point: EPs detect subclinical lesions at sites remote from the clinical lesion — thus providing objective evidence of spatial dissemination.
SECTION 3: RELATED CONCEPTS FOR COMPLETE UNDERSTANDING
3.1 Pre-MS Stages — The Disease Continuum
Harrison's 22e and the modern unifying view describe MS as a continuum:
Genetic susceptibility (HLA-DRB1*1501)
↓
Environmental triggers (EBV infection, low Vitamin D, smoking)
↓
RIS (Radiologically Isolated Syndrome) — subclinical MRI lesions, no symptoms
↓
CIS (Clinically Isolated Syndrome) — first clinical attack
↓
RRMS (Relapsing-Remitting MS) — relapses + remissions + silent progression
↓
SPMS (Secondary Progressive) — relapse-independent disability accumulation
PPMS represents a distinct clinical entry point to the same continuum.
3.2 EDSS — Expanded Disability Status Scale
Every MS type is monitored with EDSS (Kurtzke scale). High-yield for exams:
| EDSS Score | Functional Status |
|---|
| 0 | Normal neurological exam |
| 1–1.5 | Minimal signs, no disability |
| 2–2.5 | Minimal disability |
| 3–3.5 | Moderate disability, fully ambulatory |
| 4.0 | Ambulatory without aid for ~500m — threshold for progressive disease |
| 4.5 | Ambulatory ~300m |
| 5.0 | Ambulatory ~200m |
| 5.5 | Ambulatory ~100m |
| 6.0 | Unilateral assistance (cane/crutch) to walk 100m |
| 6.5 | Bilateral assistance to walk 20m |
| 7.0 | Wheelchair-bound, self-propelled |
| 7.5 | Requires aid for wheelchair transfer |
| 8.0 | Essentially restricted to chair/bed |
| 9.0 | Helpless bed patient |
| 10 | Death due to MS |
EDSS ≥4 + FSS motor score ≥2 = supports SPMS diagnosis
3.3 Practical Diagnostic Algorithm (How to Apply McDonald Criteria Clinically)
Step 1: Does the patient have a typical demyelinating attack?
- ≥24 hours duration, at normal temperature, no fever/infection
- Typical symptoms: optic neuritis, transverse myelitis, brainstem/cerebellar syndrome, hemispheric syndrome
Step 2: Count clinical attacks and objective lesions
Step 3: Apply McDonald table above to determine what additional evidence is needed
Step 4: Exclude alternative diagnoses (Red flags against MS):
- Fever, systemic illness during attack → suspect ADEM
- CSF protein >100 mg/dL, >75 WBCs, PMNs → not typical MS
- Symmetric lesions → metabolic/genetic disorder
- Basal ganglia involvement → ADEM, vasculitis, CADASIL
- Anti-AQP4 antibody positive → Neuromyelitis Optica (NMOSD)
- Anti-MOG antibody positive → MOGAD
- No OCBs with atypical course → reconsider diagnosis
3.4 Differential Diagnosis of MS (For McDonald Criteria — "Exclusion" Step)
The McDonald criteria explicitly require exclusion of more likely alternative diagnoses. Key differentials:
Inflammatory:
- Neuromyelitis optica spectrum disorder (NMOSD) — anti-AQP4 Ab+, area postrema syndrome, severe attacks
- MOG antibody-associated disease (MOGAD) — anti-MOG Ab+, ADEM phenotype
- ADEM (Acute Disseminated Encephalomyelitis) — monophasic, post-infectious/vaccine
- SLE, Sjögren's syndrome, Behçet's disease, vasculitis, sarcoidosis
Infectious:
- Neuroborreliosis (Lyme disease)
- HTLV-1/2 (tropical spastic paraparesis)
- HIV encephalopathy
- Progressive Multifocal Leukoencephalopathy (PML — JC virus)
- Neurosyphilis
Vascular:
- CADASIL (subcortical infarcts, family history, NOTCH3 mutation)
- Antiphospholipid antibody syndrome
- CNS vasculitis
- Spinal dural AV fistula
Metabolic/Nutritional:
- Vitamin B12 deficiency (subacute combined degeneration)
- Copper deficiency myelopathy
Genetic/Hereditary:
- Metachromatic leukodystrophy (adults)
- Adrenoleukodystrophy/adrenomyeloneuropathy
Structural:
- Arnold-Chiari malformation
- Cervical spondylotic myelopathy
3.5 High-Yield Exam Facts — "The Points That Win Marks"
🎯 These are the facts that separate toppers from average scorers:
- RRMS = 85–90%, PPMS = 10–15% — SPMS always starts as RRMS
- PPMS: more equal sex ratio (1:1), later onset (~40 yrs), more spinal cord dominant
- McDonald 2017 key changes: (a) Cortical lesions added as DIS location, (b) OCBs substitute for DIT in CIS+DIS, (c) Symptomatic brainstem/spinal cord lesions now CAN be used for DIS/DIT
- MRI lesions occur 9–10× more often than clinical relapses — most MS activity is "clinically silent"
- Dawson's fingers = periventricular lesions perpendicular to ventricles on sagittal MRI
- Gd enhancement persists <1 month — marks acute active lesion
- OCBs in >90% MS — but 10–20% can be negative; OCBs must be absent from paired serum
- EDSS 6.0 = unilateral walking assistance; EDSS 6.5 = bilateral assistance
- PIRA (Progression Independent of Relapse Activity) is now recognized as the dominant driver of disability even in RRMS
- CSF red flags (NOT typical MS): >75 cells/μL, protein >100 mg/dL, PMN predominance
- "Active" modifier in progressive MS = still having relapses or new MRI lesions
- CIS → high-risk if large T2 lesion burden on MRI; treat early to delay CDMS
- VEP prolonged P100 latency = optic nerve demyelination (most common EP abnormality in MS)
- For PPMS diagnosis: need 1 year progression + 2 of 3 MRI/CSF criteria
- Anti-AQP4 antibody = NMOSD (NOT MS); if positive, McDonald criteria do NOT apply
Sources Used
- Harrison's Principles of Internal Medicine, 22nd Edition (2025) — Chapter 455: Multiple Sclerosis
- Bradley and Daroff's Neurology in Clinical Practice — Chapter 80: Multiple Sclerosis (McDonald Criteria, MRI criteria, CSF)
- Thompson AJ et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17:162, 2018 (cited in Harrison's)
- Lublin FD et al. 2013 revised clinical course descriptions
📚 What's Coming Next
This was Part 1: Clinical Types + McDonald's Criteria. The full MS topic breakdown:
- Part 2: Pathogenesis, Epidemiology, Clinical Features (Symptoms & Signs), Investigations
- Part 3: Treatment (Acute attacks, DMTs, Symptomatic therapy), Prognosis, Special Situations (Pregnancy, Fatigue, Uhthoff's phenomenon)
Let me know when you're ready for Part 2!