Current guidelines for cervical cancer screening
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
I now have sufficient information to provide a comprehensive, well-cited response on cervical cancer screening guidelines.
Cervical Cancer Screening Guidelines
When to Start Screening
Screening begins at age 21, regardless of the age of sexual debut. This reflects the very low prevalence of invasive cervical cancer in adolescents, the slow multiyear course of cervical carcinogenesis, and the real (if small) risk of preterm birth associated with excisional procedures performed unnecessarily in young women. — Berek & Novak's Gynecology
Screening Intervals by Age Group
| Age Group | Recommended Test | Interval |
|---|---|---|
| 21–29 years | Pap smear (cytology) alone | Every 3 years |
| 30–65 years | Pap smear alone | Every 3 years |
| 30–65 years | Pap + HPV co-testing | Every 5 years (preferred by USPSTF 2017) |
| 30–65 years | HPV test alone (primary HPV screening) | Every 5 years (USPSTF 2017) |
HPV DNA testing is not recommended under age 30 because of the high prevalence of transient HPV infection in younger women, making the test poorly specific in that age group. — Robbins, Cotran & Kumar Pathologic Basis of Disease
Special Populations (More Frequent Screening)
The following groups require more intensive surveillance:
- HIV-positive women: Pap twice in the first year, then annually
- Immunosuppressed individuals (e.g., transplant recipients, long-term corticosteroids)
- DES (diethylstilbestrol) daughters
- History of CIN 2+ or cervical cancer: Annual screening for 20 years
— Berek & Novak's Gynecology
When to Stop Screening
- Ages 65–70: Discontinuation is reasonable if there is an adequate prior screening history (typically 3 consecutive negative Pap smears or 2 consecutive negative co-tests within the prior 10 years, with the most recent within 5 years)
- Annual reassessment of risk factors is still appropriate after stopping
- Post-hysterectomy for benign indications: Screening can be discontinued if there is no prior history of high-grade CIN or cervical cancer — Berek & Novak's Gynecology
HPV Testing Details
| Test | FDA Approval |
|---|---|
| Hybrid Capture 2 | ASC-US triage, Co-test |
| Cobas HPV (L1 DNA, PCR) | ASC-US triage, Co-test, Primary HPV screening |
| APTIMA (E6/E7 mRNA) | ASC-US triage, Co-test, reflex typing for 16/18/45 |
| Cervista | ASC-US triage, Co-test |
— Berek & Novak's Gynecology, Table 16-2
Management of Abnormal Results
- ASC-US: Reflex HPV testing; if HPV positive → colposcopy
- LSIL: Colposcopic examination; confirmed LSIL can be managed conservatively (watchful waiting) or ablated (cryotherapy) depending on follow-up reliability
- HSIL: Cervical conization (superficial excision) is the standard treatment
- HPV positive, cytology normal (in women ≥30): Repeat cytology every 6–12 months — Robbins, Cotran & Kumar
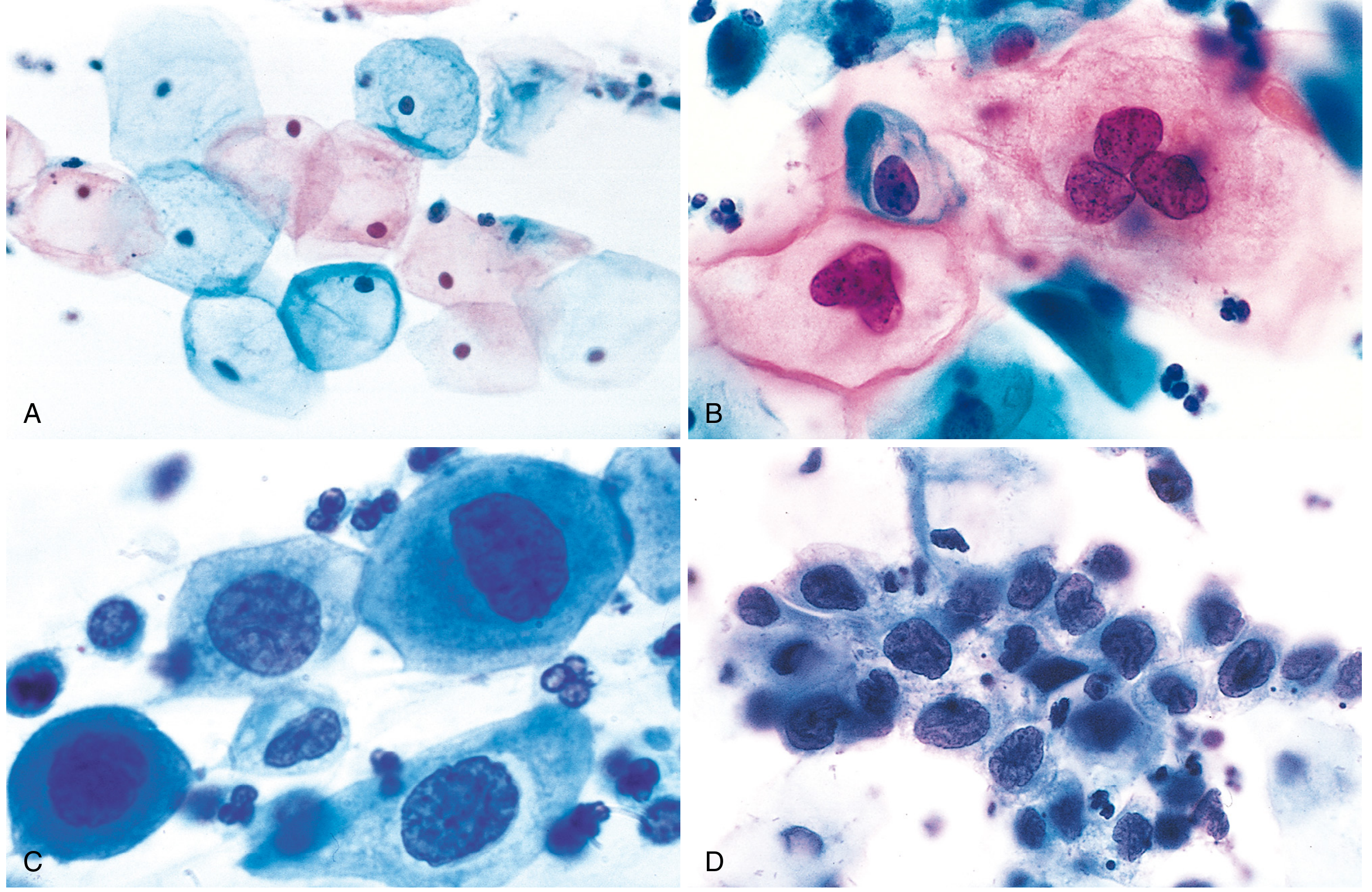
The Pap Smear — Cytologic Spectrum
The transformation zone is circumferentially scraped; cells are fixed and stained using the Papanicolaou method. The cytologic spectrum from normal to HSIL reflects a progressive reduction in cytoplasm and increase in the nucleus-to-cytoplasm ratio as differentiation is lost.
Prevention: HPV Vaccination
Vaccination against oncogenic (and low-risk) HPV types is recommended for:
- All children and adults ages 9–26 years
- Selected adults 27–45 years (shared decision-making based on risk)
— Robbins, Cotran & Kumar Pathologic Basis of Disease
Key takeaway: The USPSTF 2017 and ACS guidelines converge on starting at age 21, using cytology alone every 3 years for ages 21–29, and offering cytology + HPV co-testing or primary HPV testing every 5 years for ages 30–65. Routine annual Pap smears are no longer recommended for average-risk women. Always individualize based on risk factors and immunocompromise status.
This is a shared conversation. Sign in to Orris to start your own chat.