Aortic regurgitation
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of aortic regurgitation (AR) synthesized from multiple authoritative sources.
Aortic Regurgitation
Definition
AR results from inadequate closure of the aortic valve cusps, allowing diastolic backflow from the aorta into the left ventricle. It may be caused by disease of the aortic valve cusps (primary AR), dilation or distortion of the aortic root/ascending aorta (secondary AR), or both (mixed AR).
Etiology
Valvular (leaflet) causes:
- Bicuspid aortic valve (most common congenital cause)
- Infective endocarditis
- Rheumatic heart disease
- Dopamine/serotonin receptor agonists (e.g., ergot derivatives)
Aortic root causes:
- Marfan syndrome and other connective tissue diseases
- Hypertension-induced annuloaortic ectasia
- Aortic dissection
- Syphilis
- Ankylosing spondylitis / psoriatic arthritis
- Aortic aneurysm (ascending aorta dilation → ring dilation → cusp malcoaptation)
Acute AR is typically caused by infective endocarditis or aortic dissection.
Pathophysiology
AR imposes a volume overload on the LV: during diastole, the regurgitant volume returns through the incompetent valve in addition to normal left atrial inflow. The LV must pump a much larger total stroke volume.
Chronic AR — compensated phase:
- Eccentric hypertrophy (LV cavity enlarges) accommodates the volume overload at lower filling pressure
- The large stroke volume elevates systolic pressure; the afterload excess produces a modest concentric component as well — distinguishing it from mitral regurgitation
- Pulse pressure widens; aortic diastolic pressure falls
- LV stroke volume may reach twice normal before decompensation
Chronic AR — decompensated phase:
- LV dilation exceeds adaptive capacity → EF falls
- LA pressure rises → pulmonary congestion and edema (once mean LA pressure exceeds ~25–40 mmHg)
Coronary ischemia mechanism: The fall in aortic diastolic pressure combined with the rise in LV diastolic pressure narrows the diastolic driving gradient for coronary flow → subendocardial ischemia, which can occur without obstructive CAD.
Acute AR (e.g., endocarditis):
- The LV is unprepared — no time for compensatory eccentric hypertrophy
- Sudden severe volume overload → precipitous fall in forward output + dramatic rise in LV filling pressure
- Reflex vasoconstriction raises afterload, worsening cardiac output
- Rapid decompensation and cardiogenic shock can develop quickly
Clinical Manifestations
Chronic AR — symptoms (often absent for years):
- Dyspnea on exertion, orthopnea, fatigue (left-sided HF)
- Angina (from reduced coronary reserve; may be accompanied by flushing)
- Carotid pulsation awareness; unpleasant heartbeat awareness
Prognosis: Once significant HF symptoms develop, expected survival without valve replacement is approximately 2–5 years. LV dilation, reduced EF, and moderate-or-greater pulmonary hypertension all predict higher mortality.
Physical Examination — Chronic AR
The hyperdynamic, volume-overloaded LV generates a constellation of findings:
| Sign | Description |
|---|---|
| Corrigan pulse | Sharp upstroke and rapid collapse of the carotid pulse ("water-hammer") |
| de Musset sign | Head bobbing with each heartbeat |
| Duroziez sign | Systolic + diastolic bruits on femoral artery compression |
| Quincke pulse | Systolic flushing / diastolic blanching of the nail bed |
| Hill sign | Femoral systolic BP ≥40 mmHg higher than brachial — the most reliable sign of severity |
| Displaced apex | Hyperactive impulse displaced downward and to the left |
Murmur: High-pitched blowing diastolic murmur at the lower left sternal border (if valvular origin) or upper right sternal border (if aortic root origin); heard best sitting upright and leaning forward. In mild AR, it is brief and early diastolic; in severe AR, it persists throughout diastole.
Austin Flint murmur: A mid-to-late diastolic rumble at the apex in severe AR — the regurgitant jet impinges on the anterior mitral leaflet, mimicking mitral stenosis.
Acute AR: Signs are subtle — no compensatory eccentric hypertrophy, no wide pulse pressure. Look for a short diastolic murmur and soft/absent S1 (due to premature mitral closure from high LV diastolic pressure — mitral valve preclosure).
Echocardiogram
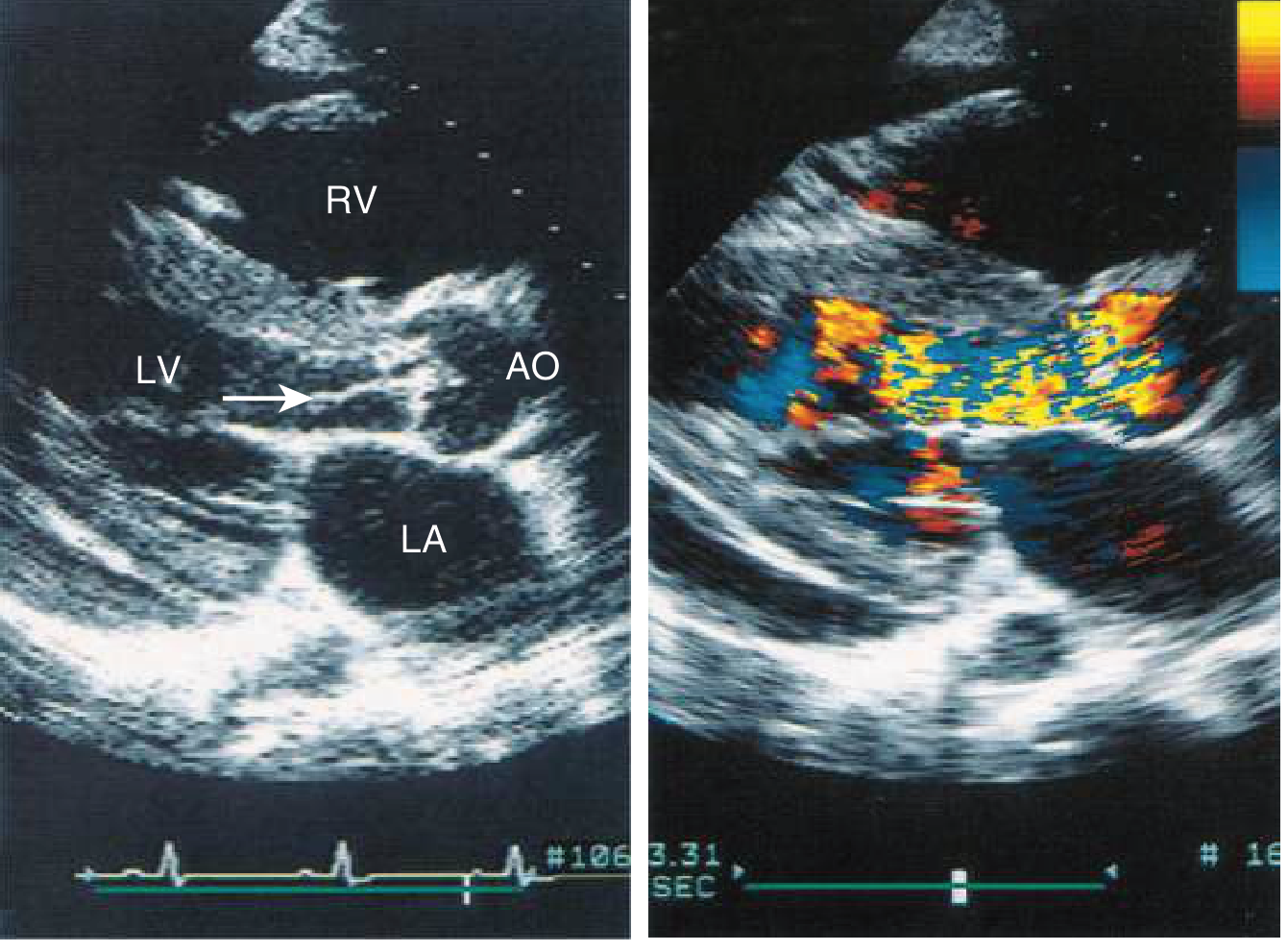
Echocardiogram showing a vegetation prolapsing into the LVOT from the aortic valve leaflet in diastole (left panel), and color-flow Doppler demonstrating turbulent regurgitant flow filling the LV tract during diastole (right panel). — Goldman-Cecil Medicine
TTE is the primary modality. Key echocardiographic parameters:
| Parameter | Severe AR Threshold |
|---|---|
| Regurgitant volume | ≥60 mL |
| EROA (proximal isovelocity surface area) | ≥0.30 cm² |
| Regurgitant fraction | >50% |
| Vena contracta | Largest diameter of regurgitant jet |
| Pressure half-time | <200 ms (vs. >500 ms for mild) |
| Holodiastolic flow reversal (descending aorta) | Peak velocity >20 cm/s |
LV dimensions, LVEF, and aortic root diameter are also measured. LV end-systolic dimension >4.5 cm or LVEF <50% triggers surgical indication even without symptoms.
Other Investigations
- ECG: Almost always shows LV hypertrophy (nonspecific)
- Chest X-ray: Cardiomegaly; uncoiling and enlargement of the aortic root
- Cardiac MRI: Useful when echo windows are poor; quantifies regurgitant fraction accurately
- CT: Aortic root/ascending aorta anatomy, annular sizing if TAVR considered
Natural History
- Chronic severe AR can remain asymptomatic for ≥10 years
- Once symptoms develop: ~5-year survival without surgery (shorter in older adults: ~2 years)
- Rate of progression to symptoms or LV dysfunction: ~4–6%/year in asymptomatic severe AR
- Higher risk in those with: LV dilatation, reduced EF, moderate-severe pulmonary hypertension
Management
Medical Therapy
- No proven disease-modifying therapy exists for chronic AR
- Vasodilators (e.g., nifedipine, ACE inhibitors) were once used to reduce afterload and delay surgery, but have not been shown to alter the natural history; they may have a role in hypertensive patients or those not yet candidates for surgery
- Diuretics for symptomatic relief of congestion
- Treatment of underlying causes (e.g., BP control, antibiotics for endocarditis)
Surgical Management — Indications for Aortic Valve Replacement (AVR)
AVR is the only treatment proven to reduce mortality. Indications:
| Indication | Details |
|---|---|
| Symptomatic severe AR | Class I — regardless of LVEF |
| Asymptomatic severe AR + LVEF <50% | Class I |
| Asymptomatic severe AR + LV end-systolic dimension >4.5 cm | Class I |
| Undergoing cardiac surgery for another indication | Add AVR if severe AR |
- Operative mortality increases with LV dysfunction: <5% with normal LVEF vs. ~14% if LVEF <35%
- Older/high-risk patients: TAVR for AR is emerging; small series show feasibility, though AR was historically a contraindication to TAVR (no annular calcification for anchor)
Acute Severe AR
- Medical stabilization: IV vasodilators (nitroprusside) to reduce afterload, inotropes if hypotensive; intra-aortic balloon pump is contraindicated (worsens regurgitation)
- Urgent/emergent surgical AVR is required; delay is associated with rapid deterioration
Key Distinctions: Chronic vs. Acute AR
| Feature | Chronic AR | Acute AR |
|---|---|---|
| Onset | Gradual | Abrupt |
| LV compensation | Eccentric hypertrophy | None |
| Pulse pressure | Widened | Normal or narrow |
| Signs | Many (Hill, Corrigan, Duroziez, etc.) | Minimal/absent |
| Murmur | Long diastolic blowing | Short, soft |
| S1 | Normal | Soft/absent (mitral preclosure) |
| Management | Elective surgery when indicated | Emergent surgery |
Sources: Fuster and Hurst's The Heart, 15th ed. | Goldman-Cecil Medicine | Braunwald's Heart Disease | Guyton and Hall Textbook of Medical Physiology | Braunwald's (Geriatric Cardiology chapter)
This is a shared conversation. Sign in to Orris to start your own chat.