Can you teach me ssri…aa in everything, history, psychopharmacology,psychodynamics,kinetics, indications, guidelines, titration, switching, side effects, recent advances, contraindications, toxicity, discontinuation,dosage,formulation,evidence based trials,…and teach each drug individually too
Reading File
Finding Sources
Finding Sources
Searching PubMed
"selective serotonin reuptake inhibitors"[MeSH Terms] AND "antidepressive agents"[MeSH Terms]
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Searching the Web
SSRI antidepressants 2024 2025 clinical guidelines prescribing evidence-based advances
Selective Serotonin Reuptake Inhibitors (SSRIs) — Complete Teaching Guide
PART 1 — HISTORY
The story of SSRIs begins in the late 1960s when Arvid Carlsson (later Nobel laureate) proposed that selectively blocking serotonin reuptake might produce antidepressant effects without the dangerous cardiac and anticholinergic side effects of tricyclics (TCAs). Eli Lilly's chemists developed fluoxetine in 1974; it was approved by the FDA in December 1987 and marketed as Prozac. Its commercial success was unprecedented — it became one of the best-selling drugs in history and permanently shifted psychiatric prescribing.
Timeline of SSRI approvals (US):
| Drug | Brand | FDA Approval |
|---|---|---|
| Fluoxetine | Prozac | 1987 |
| Sertraline | Zoloft | 1991 |
| Paroxetine | Paxil | 1992 |
| Fluvoxamine | Luvox | 1994 |
| Citalopram | Celexa | 1998 |
| Escitalopram | Lexapro | 2002 |
The era of SSRIs essentially displaced MAOIs and TCAs from first-line status due to their far superior safety profile, simple once-daily dosing, and broad efficacy spectrum.
PART 2 — PSYCHOPHARMACOLOGY & MECHANISM OF ACTION
The Core Mechanism: SERT Inhibition
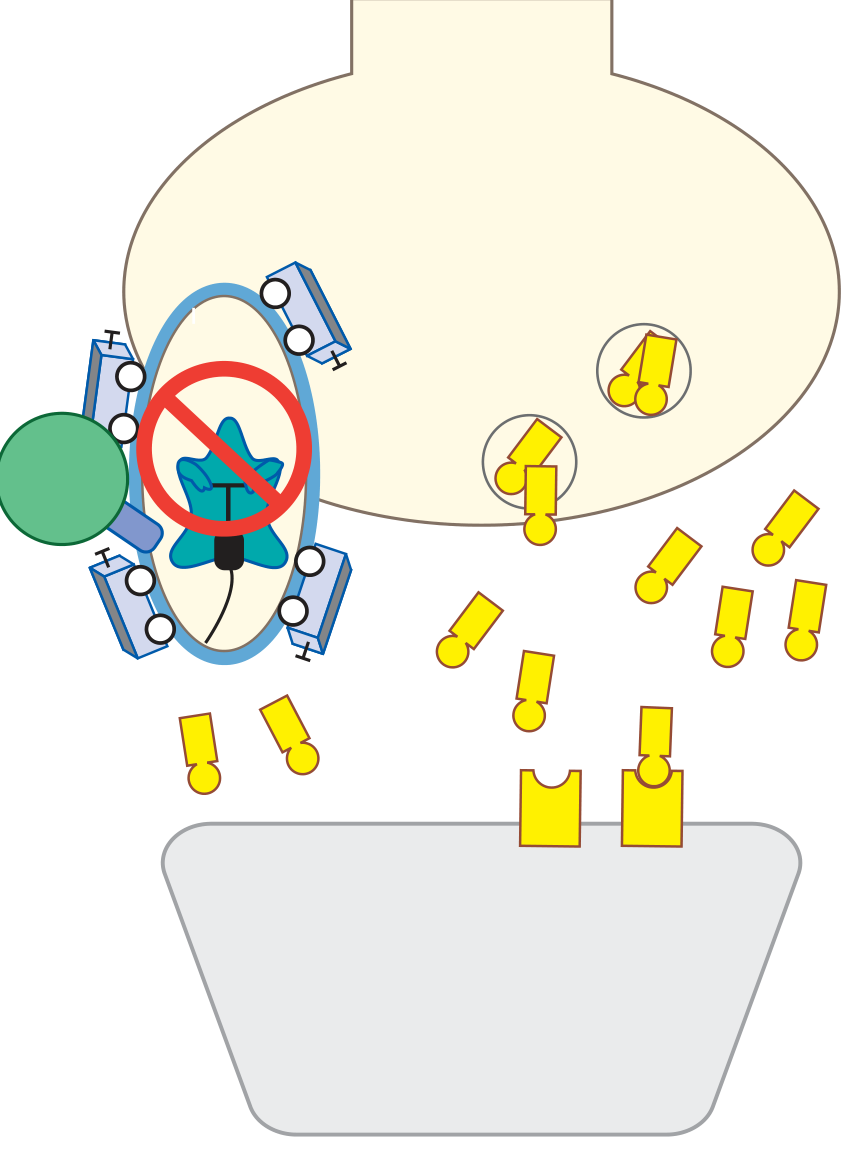
All six SSRIs share one essential action: potent, selective blockade of the serotonin transporter (SERT). SERT normally removes serotonin (5-HT) from the synapse back into the presynaptic neuron. By blocking it, SSRIs increase synaptic 5-HT availability.
The Five-Step Mechanism of Action (Stahl)
The full antidepressant effect is not simply SERT blockade — it unfolds in stages:
-
Acute (hours): SSRI blocks SERT → serotonin accumulates at axon terminals AND at somatodendritic areas near the cell body. The cell body area has 5-HT1A autoreceptors that act as a brake — high local 5-HT activates them and inhibits neuronal firing.
-
Early (days): Because 5-HT1A autoreceptors are being continuously stimulated by elevated 5-HT, they begin to desensitize (downregulate). The brakes start releasing.
-
Intermediate (1–2 weeks): As somatodendritic 5-HT1A autoreceptors downregulate, the inhibition on neuronal firing lifts → impulse flow resumes normally.
-
Delayed (2–4 weeks): With normal impulse flow restored, serotonin is now released at axon terminals → synaptic 5-HT rises at target regions. This is when therapeutic effects begin. The delay explains why SSRIs don't work immediately.
-
Later (4–8 weeks): Postsynaptic 5-HT receptors downregulate as well → tolerance to side effects develops, and sustained clinical response is established.
Psychodynamic Considerations
From a psychodynamic lens, depression involves disrupted object relations, unresolved grief, internalized anger, and loss of self-object representations. SSRIs do not "cure" these dynamics but they lower the biological floor — reducing vegetative symptoms (anhedonia, fatigue, insomnia) enough that patients can engage meaningfully in psychotherapy. The biopsychosocial model frames SSRIs as one component: they create neurobiological conditions permissive for psychological change. Combined pharmacotherapy + psychotherapy (especially CBT) consistently outperforms either alone in moderate-severe depression.
PART 3 — PHARMACOKINETICS
Comparison Table
| Drug | Half-life | Active metabolite | CYP inhibition | Protein binding | Bioavailability |
|---|---|---|---|---|---|
| Fluoxetine | 2–3 days | Norfluoxetine (t½ ~2 wks) | 2D6 (potent), 3A4, 2C9 | 94% | 72% |
| Sertraline | 26 hrs | Desmethylsertraline (weak) | 2D6 (moderate), 3A4 | 98% | Variable |
| Paroxetine | 20 hrs | None (major) | 2D6 (potent), 3A4 | 95% | ~50% |
| Fluvoxamine | 15–20 hrs | None | 1A2 (potent), 2C9, 3A4 | 80% | 53% |
| Citalopram | 35 hrs | Desmethylcitalopram (weak) | Minimal | 80% | 80% |
| Escitalopram | 27–32 hrs | None significant | Minimal (2D6 weak) | 56% | 80% |
Key kinetic pearls:
- Fluoxetine has the longest combined half-life (~7 days including norfluoxetine). This means: (a) no significant discontinuation syndrome, (b) no need for taper on stopping, (c) requires 5-week washout before starting an MAOI.
- Paroxetine has the shortest half-life and is an autoinhibitor of its own metabolism (CYP2D6) — even small dose reductions cause disproportionately steep plasma level drops, causing the worst discontinuation syndrome of all SSRIs.
- Escitalopram has minimal CYP interactions — best choice when polypharmacy is a concern (Harrison's).
- All SSRIs achieve steady-state in ~5 half-lives. For fluoxetine this takes ~5 weeks.
- All are extensively protein-bound — less affected by renal disease; dose-adjust in severe hepatic impairment.
- Food does not significantly affect absorption of any SSRI.
CYP Interactions — Clinical Implications
| CYP | Inhibitor SSRI | Substrates at risk |
|---|---|---|
| 2D6 | Fluoxetine, paroxetine (potent); sertraline (moderate) | TCAs, antipsychotics, codeine→morphine conversion, tamoxifen |
| 1A2 | Fluvoxamine (potent) | Clozapine, olanzapine, theophylline, caffeine |
| 3A4 | Fluvoxamine, fluoxetine | Carbamazepine, digoxin, benzodiazepines |
| Minimal | Citalopram, escitalopram | Safest choice in complex regimens |
Tamoxifen note (2025 update): CYP2D6 inhibitors (fluoxetine, paroxetine) theoretically reduce conversion of tamoxifen to endoxifen. However, a 2022 systematic review of ~100,000 breast cancer patients found no adverse oncologic outcomes from concurrent use; updated guidelines recommend not switching antidepressants solely for this reason.
PART 4 — INDICATIONS
FDA-Approved Indications (US) by Drug
| Indication | Flu | Ser | Par | Flv | Cit | Esx |
|---|---|---|---|---|---|---|
| Major Depressive Disorder | ✓ | ✓ | ✓ | — | ✓ | ✓ |
| OCD | ✓ | ✓ | ✓ | ✓ | — | — |
| Panic Disorder | ✓ | ✓ | ✓ | — | — | — |
| Social Anxiety Disorder | — | ✓ | ✓ | ✓ | — | — |
| GAD | — | — | ✓ | — | — | ✓ |
| PTSD | — | ✓ | ✓ | — | — | — |
| PMDD | ✓ | ✓ | ✓ | ✓ | — | — |
| Bulimia Nervosa | ✓ | — | — | — | — | — |
| BDD | — | — | ✓ | — | — | — |
Flu=fluoxetine, Ser=sertraline, Par=paroxetine, Flv=fluvoxamine, Cit=citalopram, Esx=escitalopram
Off-Label Uses (well-supported)
- Generalized anxiety disorder (all SSRIs)
- Body dysmorphic disorder
- Trichotillomania & skin-picking disorder
- Premenstrual dysphoric disorder (low-dose intermittent)
- Fibromyalgia (fluoxetine)
- Premature ejaculation (paroxetine — evidence-based off-label)
- Irritable bowel syndrome
- Hot flashes (menopause-related)
- Autism-associated repetitive behaviors
PART 5 — CLINICAL GUIDELINES & TREATMENT ALGORITHM
First-Line Selection (2024–2025 Guidelines)
Per recent clinical practice guidelines and Maudsley 15th ed.:
- Start with escitalopram or sertraline — best efficacy-tolerability balance, fewest drug interactions, most evidence.
- Alternatives: fluoxetine (if activating properties needed, eating disorders, pregnancy [most data]), paroxetine (if sedation needed, though avoid in elderly), citalopram (avoid >40 mg due to QTc).
- Paroxetine is least preferred for: elderly patients, pregnancy (neonatal withdrawal), patients likely to need future dose adjustments.
Step-Care Algorithm
Step 1: Start SSRI (escitalopram/sertraline preferred)
↓ 4–8 weeks at therapeutic dose
Step 2: Inadequate response?
→ Optimize dose (go to maximum tolerated)
→ 4 more weeks
↓
Step 3: Still inadequate?
→ Switch: SSRI → SNRI (or different SSRI)
→ Or Augment: lithium, aripiprazole, quetiapine, T3
↓
Step 4: Treatment-resistant (≥2 adequate trials failed)
→ ECT, ketamine/esketamine, TMS
→ Consider MAOIs (specialist)
PART 6 — DOSING & FORMULATIONS
Standard Doses
| Drug | Starting dose | Usual therapeutic | Maximum | Available forms |
|---|---|---|---|---|
| Fluoxetine | 10–20 mg/day | 20–40 mg | 80 mg (OCD/bulimia) | Capsule, tablet, liquid; weekly delayed-release capsule (Prozac Weekly, 90 mg) |
| Sertraline | 25–50 mg/day | 50–200 mg | 200 mg | Tablet, oral concentrate |
| Paroxetine IR | 10–20 mg/day | 20–40 mg | 60 mg | Tablet, liquid |
| Paroxetine CR | 12.5–25 mg/day | 25–50 mg | 62.5 mg | Extended-release tablet |
| Fluvoxamine | 50 mg/day | 100–300 mg | 300 mg | Tablet; CR capsule |
| Citalopram | 10–20 mg/day | 20–40 mg | 40 mg (QTc limit) | Tablet, liquid |
| Escitalopram | 5–10 mg/day | 10–20 mg | 20 mg | Tablet, liquid |
Titration Principles
- Start low, go slow — especially in anxiety disorders (initial activation effect can worsen anxiety)
- Increase dose after 2–4 weeks if tolerated but subtherapeutic response
- Allow 4–8 weeks at therapeutic dose before declaring a trial failed
- In elderly: start at half the adult starting dose; titrate more slowly
- In children/adolescents: fluoxetine has the most RCT evidence; start at 10 mg
PART 7 — THE SIX SSRIs INDIVIDUALLY
1. FLUOXETINE (Prozac)
Unique pharmacology: SERT + 5-HT2C antagonism + weak NET inhibition at high doses
The 5-HT2C effect: Blocking 5-HT2C receptors disinhibits release of norepinephrine and dopamine in the prefrontal cortex. This makes fluoxetine the most activating SSRI.
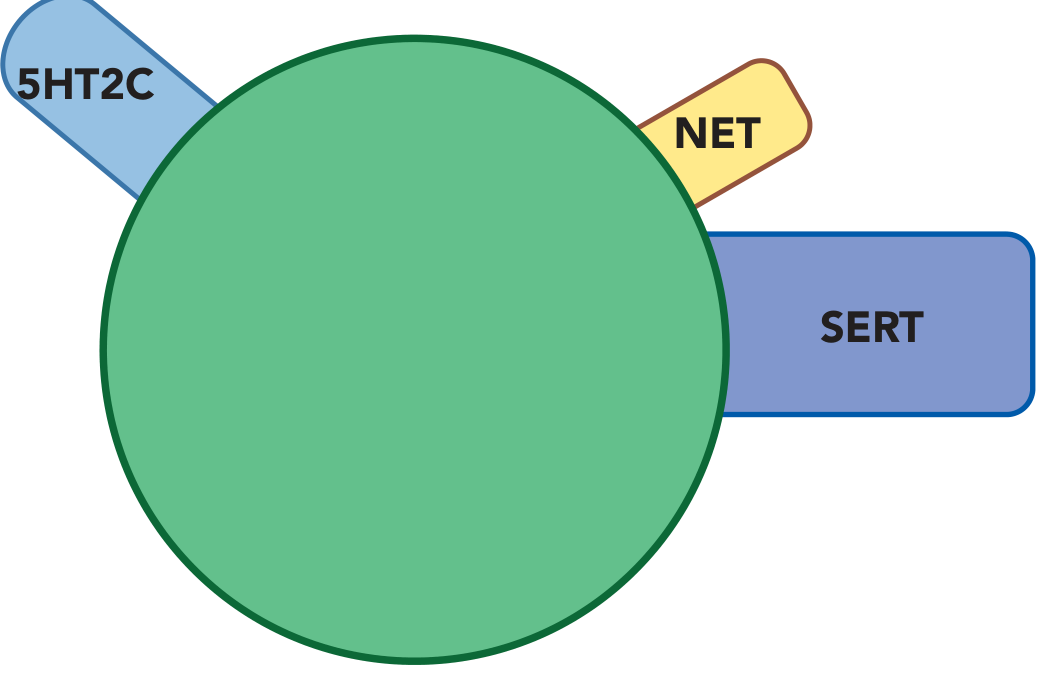
Clinical profile:
- Most activating → best for depression with hypersomnia, psychomotor retardation, fatigue, apathy
- May worsen agitation and insomnia in anxious patients
- Only SSRI approved for bulimia nervosa (higher dose, 60 mg)
- Approved for depression + bipolar when combined with olanzapine (Symbyax)
Kinetics: Half-life 2–3 days; active metabolite norfluoxetine half-life ~2 weeks → slowest elimination of all SSRIs → minimal discontinuation syndrome → 5-week MAOI washout
Formulations: Capsule 10/20/40 mg; liquid 20 mg/5 mL; Prozac Weekly 90 mg delayed-release capsule (for adherence)
Key uses: MDD, OCD, bulimia, PMDD, bipolar depression (with olanzapine), panic disorder
Doses: MDD: 20 mg/day → up to 80 mg; Bulimia: 60 mg/day; OCD: 20–80 mg
2. SERTRALINE (Zoloft)
Unique pharmacology: SERT + dopamine reuptake inhibition (DAT) at high doses + sigma-1 receptor binding
Clinical profile:
- Best overall efficacy-tolerability balance — most broadly prescribed SSRI worldwide
- Lowest risk of drug-drug interactions of the commonly used SSRIs (Harrison's)
- GI side effects (diarrhea) slightly more common than others
- DAT inhibition may improve motivation and drive at higher doses
- Sigma-1 binding: potential anxiolytic properties, mood stabilization
Kinetics: Half-life 26 hrs; active metabolite desmethylsertraline is weak; moderate CYP2D6 inhibition; 98% protein bound
Formulations: Tablets 25/50/100 mg; oral concentrate 20 mg/mL
Key uses: MDD, OCD, panic disorder, PTSD, PMDD, social anxiety, SAD
Doses: MDD: 50–200 mg/day; OCD: 50–200 mg; PTSD: 50–200 mg
Pearl: First-line in pregnancy due to extensive safety data and low neonatal withdrawal risk. First-line for cardiac patients (SADHART trial — safe post-MI).
3. PAROXETINE (Paxil, Paxil CR)
Unique pharmacology: SERT + muscarinic M1 antagonism + NET inhibition + NOS inhibition

Clinical profile:
- Most anticholinergic SSRI → dry mouth, constipation, urinary hesitancy, blurred vision, sedation
- NET inhibition → slightly more noradrenergic than other SSRIs → possibly more robust for anxiety
- NOS inhibition → contributes to sexual dysfunction and premature ejaculation treatment
- Most sedating SSRI → useful for patients with insomnia-predominant depression
- Most problematic for discontinuation syndrome (shortest half-life + autoinhibition of CYP2D6)
Kinetics: Half-life 20 hrs; autoinhibits its own CYP2D6 metabolism → non-linear kinetics; small dose reductions cause steep plasma drops
Formulations: IR tablets (10/20/30/40 mg), CR tablets (12.5/25/37.5 mg), liquid
Key uses: MDD, GAD, OCD, panic disorder, PTSD, PMDD, social anxiety, BDD
Avoid in: Elderly (anticholinergic burden), pregnancy (neonatal adaptation syndrome, possible septal cardiac defects — data controversial), patients who may need dose flexibility
Doses: MDD: 20–50 mg; GAD: 20–50 mg; CR formulation allows for better tolerability
4. FLUVOXAMINE (Luvox)
Unique pharmacology: SERT + sigma-1 receptor agonism + CYP1A2/1A3 inhibition

Clinical profile:
- Not FDA-approved for MDD in adults (approved for OCD and social anxiety)
- Sigma-1 receptor agonism: anxiolytic, antipsychotic-like, neuroprotective effects; may confer unique benefits in autism, OCD, schizophrenia
- OCD drug of first choice in children in some guidelines
- Most significant CYP interactions of all SSRIs (inhibits 1A2, 2C9, 3A4) → use caution with clozapine, olanzapine (can markedly raise levels), theophylline, warfarin
- Must be given twice daily (no once-daily formulation except CR capsule)
- COVID-19 note: Fluvoxamine gained attention in 2020–2022 (TOGETHER trial) for possible benefit in early COVID via sigma-1 agonism; results were modest and not definitive
Kinetics: Half-life 15–20 hrs; extensive 1A2 inhibitor; less protein-bound (80%)
Formulations: Tablets 25/50/100 mg (bid dosing); CR capsules 100/150 mg (once daily)
Key uses: OCD (adults and children), social anxiety disorder
Doses: OCD: 100–300 mg/day (divided); Social anxiety: 100–300 mg/day
5. CITALOPRAM (Celexa)
Unique pharmacology: Racemic mixture of S-citalopram (active) and R-citalopram (weakly active, possibly interfering); essentially pure SERT inhibitor; minimal other receptor binding
Clinical profile:
- Cleanest pharmacological profile of the original SSRIs — least receptor promiscuity
- Minimal CYP interactions → good for elderly and polypharmacy patients
- Critical safety issue: QTc prolongation
- FDA black box warning (2012): maximum dose 40 mg/day (20 mg/day in elderly, hepatic impairment, or with CYP2C19 inhibitors like omeprazole)
- Dose-dependent QTc prolongation → risk of torsades de pointes at higher doses
- Monitor ECG in high-risk patients
Kinetics: Half-life 35 hours (longest of commonly used SSRIs); minimal CYP inhibition; 80% bioavailability; 80% protein bound
Formulations: Tablets 10/20/40 mg; oral solution 10 mg/5 mL
Key uses: MDD, off-label anxiety, elderly depression
Doses: MDD: 20–40 mg/day; start at 10 mg in elderly/hepatic impairment
Pearl: R-citalopram has been described as a "hitch-hiker" that gets in the way of S-citalopram binding. Removing it (to get escitalopram) produced a more efficient, cleaner drug.
6. ESCITALOPRAM (Lexapro) — The Quintessential SSRI
Unique pharmacology: Pure S-enantiomer of citalopram — binds SERT at primary binding site AND at an allosteric site (unique), making it the most selective and potent SERT inhibitor available; essentially no other receptor binding
Clinical profile:
- Most selective SSRI — virtually no off-target effects
- Allosteric SERT binding: the S-enantiomer binds both the primary (orthosteric) and a secondary (allosteric) site on SERT — this may contribute to superior potency and possibly faster onset
- Minimal CYP inhibition → fewest drug interactions of all SSRIs (Harrison's)
- Best-tolerated SSRI in most comparative studies
- Consistently ranks #1 or #2 for efficacy in network meta-analyses (Cipriani 2018 Lancet)
- Effective across: MDD, GAD, social anxiety, panic disorder, OCD (off-label)
Kinetics: Half-life 27–32 hrs; 56% protein bound; minimal CYP inhibition; 80% bioavailability
Formulations: Tablets 5/10/20 mg; oral solution 5 mg/5 mL
Key uses: MDD (first-line), GAD (FDA-approved), off-label anxiety spectrum, elderly depression
Doses: MDD: 10–20 mg/day; start at 5 mg in elderly; maximum 20 mg/day
Pearl: In the elderly, escitalopram at 10–20 mg is considered by many guidelines as first-line due to minimal drug interactions, no anticholinergic effects, and good tolerability.
PART 8 — SIDE EFFECTS
Common Side Effects (All SSRIs)
GI: Nausea, diarrhea, dyspepsia (most common early; usually resolve in 1–2 weeks). Cause: excess 5-HT in gut (5-HT3/5-HT4 stimulation). Mitigation: Take with food, start low.
Sexual dysfunction: 30–60% of patients — decreased libido, delayed orgasm, anorgasmia, erectile dysfunction. Most underreported (must ask directly). Management:
- Dose reduction
- Drug holidays (2–3 times/month — not effective with fluoxetine due to long half-life)
- Switch to bupropion, mirtazapine, vortioxetine, or agomelatine (lower risk)
- Add sildenafil/tadalafil (evidence for men; limited for women)
- Add bupropion 100–150 mg/day (Maudsley)
- Amantadine 100 mg tid; buspirone 10 mg tid (Harrison's)
Sleep disturbance: Insomnia (especially fluoxetine/sertraline) or excessive sedation (paroxetine, fluvoxamine). Insomnia can be managed by morning dosing; sedation by bedtime dosing.
Activation/jitteriness syndrome: Anxiety, restlessness, insomnia in the first 1–2 weeks, especially in panic disorder. Use lowest starting doses; reassure; consider short-term benzodiazepine.
Weight gain: Modest but real, especially with paroxetine. Escitalopram and sertraline are relatively weight-neutral.
Headache: Common early; usually transient.
Sweating: Especially night sweats; can persist.
Hyponatremia (SIADH): All SSRIs cause SIADH — particularly in elderly, women, those on diuretics. A 2024 meta-analysis (Gheysens et al., Eur Psychiatry) found SSRIs carry higher hyponatremia risk than SNRIs or other antidepressants. Monitor sodium in high-risk patients within the first 4–6 weeks.
Bruising/bleeding: SSRIs reduce platelet aggregation (serotonin depletion in platelets). Increased GI bleeding risk — add PPI if concurrent NSAIDs.
Serious/Rare Side Effects
Suicidality (FDA Black Box): Increased risk of suicidal ideation in children, adolescents, and young adults (up to age 24) — especially in the first few weeks of treatment. Monitor closely. This is a risk-benefit discussion, not an absolute contraindication.
Akathisia: Inner restlessness, resembles anxiety. Can be mistaken for worsening depression. Manage with propranolol, mirtazapine, or benzodiazepines.
Hypomania/mania activation: Can precipitate mania in undiagnosed bipolar disorder. Always screen for personal/family history of bipolar.
QTc prolongation: Most significant with citalopram; also escitalopram at high doses. Rare with others.
Extrapyramidal effects: Parkinsonism, dystonia (rare; more with paroxetine).
Post-SSRI Sexual Dysfunction (PSSD): Persistent sexual dysfunction continuing after SSRI discontinuation — not fully understood; possibly related to epigenetic changes, neurosteroid disruption. Low prevalence but real. No proven treatment; some case reports of recovery with time (Maudsley 15th ed.).
PART 9 — SEROTONIN SYNDROME
A pharmacodynamic toxidrome from excess serotonergic stimulation, typically from drug combinations.
Hunter Criteria for Diagnosis (requires ONE of):
- Spontaneous clonus
- Inducible clonus + agitation/diaphoresis
- Ocular clonus + agitation/diaphoresis
- Tremor + hyperreflexia
- Hypertonia + temperature >38°C + clonus
Classic Triad:
- Neuromuscular: clonus (pathognomonic), hyperreflexia, tremor, myoclonus, rigidity
- Autonomic: hyperthermia, tachycardia, diaphoresis, hypertension
- Cognitive: agitation, confusion
Precipitating Combinations:
- SSRI + MAOI (most dangerous — avoid; requires washout)
- SSRI + tramadol, fentanyl, meperidine
- SSRI + triptans (theoretical; clinical risk debated)
- SSRI + linezolid (weak MAOI)
- SSRI + St. John's Wort
- SSRI + dextromethorphan
- SSRI + lithium (at toxic lithium levels)
Management:
- Stop all serotonergic agents immediately
- Supportive care: IV fluids, cooling
- Benzodiazepines for agitation and seizures
- Cyproheptadine 12 mg PO then 2 mg every 2 hrs (5-HT2A antagonist — antidote)
- Severe cases: ICU, intubation, temperature control
- Do NOT use physical restraints (cause hyperthermia)
PART 10 — OVERDOSE & TOXICITY
SSRIs have a high therapeutic index — pure SSRI overdoses are rarely fatal (Kaplan & Sadock; Tintinalli).
Clinical Features of Overdose:
- Tachycardia, mild hypotension, lethargy (especially with co-ingestants)
- Citalopram/escitalopram: most dangerous in overdose → dose-dependent QTc prolongation, QRS widening, seizures, torsades de pointes (even at overdoses of 600–1000 mg)
- Serotonin syndrome occurs in ~10% of overdoses
Treatment (Tintinalli):
| Intervention | Indication |
|---|---|
| IV access + cardiac monitor + ECG | All intentional ingestions |
| Activated charcoal (single dose) | Within 1 hour of ingestion |
| Benzodiazepines | Seizures (first-line) |
| Phenobarbital | Refractory seizures |
| Sodium bicarbonate | Prolonged QRS |
| Magnesium sulfate | QTc prolongation, torsades |
| Cyproheptadine | Serotonin syndrome |
- Observe 6 hours minimum; admit if: persistent tachycardia, altered mental status, citalopram/escitalopram ingestion, cardiac conduction abnormalities, serotonin syndrome features
- Gastric lavage, ipecac, whole-bowel irrigation: NOT recommended
PART 11 — SWITCHING STRATEGIES
Why Switch?
- Inadequate response after adequate trial (4–8 weeks at therapeutic dose)
- Intolerable side effects
- Drug interaction burden
Switching Principles
1. Direct switch (most common):
Stop current SSRI, start new SSRI at a low dose the next day. For SSRI-to-SSRI switches, washout is rarely needed (similar mechanisms, no dangerous interactions). Cross-taper (overlap while tapering old drug) can reduce discontinuation symptoms.
Stop current SSRI, start new SSRI at a low dose the next day. For SSRI-to-SSRI switches, washout is rarely needed (similar mechanisms, no dangerous interactions). Cross-taper (overlap while tapering old drug) can reduce discontinuation symptoms.
2. Cross-taper:
- Gradually taper down the first SSRI while gradually titrating up the new one
- Preferred for: paroxetine (due to discontinuation risk), patients sensitive to discontinuation
3. SSRI to SNRI:
- Can usually do direct switch or cross-taper
- No washout required
4. SSRI to MAOI — CRITICAL washout rules:
| From | Washout before MAOI |
|---|---|
| Fluoxetine | 5 weeks (norfluoxetine half-life) |
| All other SSRIs | 2 weeks |
5. MAOI to SSRI:
- Always 2 weeks washout after stopping MAOI before starting any SSRI
Step-down titration for discontinuation (Maudsley Deprescribing Guidelines):
- Never abrupt discontinuation (except fluoxetine which can usually be stopped without taper)
- Paroxetine, venlafaxine: hyperbolic tapering — small percentage reductions over months
- Use liquid formulations or pill-cutting for micro-tapering
- Fluoxetine bridge: switch to fluoxetine (long half-life), stabilize, then taper fluoxetine slowly
PART 12 — DISCONTINUATION SYNDROME
Also called antidepressant discontinuation syndrome (avoid the term "withdrawal" in clinical settings to prevent stigma, though the mechanism is similar).
FINISH Mnemonic:
- Flu-like symptoms (myalgias, sweating, nausea)
- Insomnia (vivid dreams, nightmares)
- Nausea/GI distress
- Imbalance/dizziness
- Sensory disturbances ("brain zaps" — electric shock sensations in head)
- Hyperactivation (anxiety, agitation, irritability)
Risk by Drug (Highest → Lowest):
- Paroxetine (shortest half-life + autoinhibition = steepest drop)
- Venlafaxine (short half-life, though technically SNRI)
- Sertraline, citalopram, escitalopram (moderate)
- Fluvoxamine
- Fluoxetine (virtually none — long half-life provides self-taper)
Management:
- Prevention: Always taper; educate patients
- Mild: Reassurance; usually resolves 1–2 weeks
- Moderate-severe: Restart drug → stabilize → slower taper
- Fluoxetine bridge: Switching to fluoxetine, stabilizing, then tapering leverages its self-tapering property
- Kaplan & Sadock: movement symptoms (tremor, akathisia, parkinsonism) on discontinuation — manage akathisia with propranolol, anticholinergics, or benzodiazepines
PART 13 — CONTRAINDICATIONS
Absolute:
- Concurrent MAOI use (fatal serotonin syndrome risk)
- Known hypersensitivity to the drug
Relative / Caution:
| Situation | Guidance |
|---|---|
| Bipolar disorder (unprotected) | Risk of manic switch — use mood stabilizer first |
| QTc >500 ms | Avoid citalopram/escitalopram |
| Seizure disorders | Generally safe but use cautiously |
| Severe hepatic impairment | Halve dose; fluoxetine safest (long half-life allows slow titration) |
| Elderly | Start low, watch for hyponatremia, falls |
| Pregnancy | Fluoxetine, sertraline safest (most data); paroxetine least preferred (cardiac anomaly signal) |
| Lactation | Sertraline: lowest breast milk transfer; paroxetine also low; fluoxetine: infant serum levels higher |
| Tamoxifen | Avoid fluoxetine and paroxetine (strong 2D6 inhibitors) — use escitalopram or sertraline instead |
| Bleeding risk / NSAIDs | Add PPI; monitor |
| Linezolid / methylene blue | Contraindicated (MAOI-like activity — serotonin syndrome risk) |
PART 14 — SPECIAL POPULATIONS
Children & Adolescents:
- Fluoxetine: only SSRI FDA-approved for depression in children ≥8 years
- Fluoxetine and escitalopram: approved for adolescent depression
- All SSRIs: FDA black box warning — increased suicidal ideation in under-25s; monitor weekly initially
- OCD in children: sertraline, fluoxetine, fluvoxamine (all FDA-approved)
- CBT must accompany pharmacotherapy; do not use medication as sole treatment
Elderly:
- Start at half the adult dose; titrate slowly
- Preferred: escitalopram, sertraline (fewest interactions, no anticholinergic)
- Avoid paroxetine (Beers criteria — anticholinergic)
- Monitor: sodium (SIADH), falls (dizziness), QTc (avoid citalopram >20 mg)
Pregnancy:
- SSRIs are the most studied antidepressants in pregnancy
- Neonatal Adaptation Syndrome: transient in newborns — jitteriness, respiratory distress, poor feeding (especially paroxetine, venlafaxine); resolves in days; not a reason to withhold treatment
- Persistent Pulmonary Hypertension of Newborn (PPHN): weak signal with late-pregnancy SSRI exposure; absolute risk very low
- 2025 umbrella review (Fabiano et al., Mol Psychiatry): overall, SSRIs do not show strong teratogenic signal; risk of untreated depression > risk of SSRI exposure
Cardiac Disease:
- SADHART trial (2002): Sertraline safe in post-MI patients with depression; does not worsen cardiac outcomes
- ENRICHD trial: CBT + sertraline improved depression post-MI
- Preferred: sertraline, escitalopram (minimal CYP, no QTc at therapeutic doses)
- Avoid: citalopram >40 mg; paroxetine (anticholinergic, tachycardia)
PART 15 — EVIDENCE-BASED TRIALS
Landmark Studies
STAR*D (Sequenced Treatment Alternatives to Relieve Depression), 2006:
- Largest real-world antidepressant trial (n=4,041); used citalopram as first step
- 28% remission at step 1 (citalopram)
- Only ~33% remission after 4 steps → established treatment-resistance as a major clinical challenge
- Key message: most patients need more than one treatment step
Cipriani et al. (Lancet 2018) — Network Meta-Analysis:
- 522 trials, 116,477 patients, 21 antidepressants
- Escitalopram and sertraline had best combination of efficacy and acceptability
- All SSRIs were more efficacious than placebo
- Escitalopram ranked highest for both efficacy and tolerability
SADHART (2002): Sertraline safe and effective post-myocardial infarction
CANMAT Guidelines (2023–2024):
- SSRIs + SNRIs first-line for MDD, GAD, PTSD, OCD, panic disorder, social anxiety
- Augmentation with atypical antipsychotics (aripiprazole, quetiapine, brexpiprazole) for partial responders
- Level 1 evidence for escitalopram and sertraline in MDD
ISPOT-D & pharmacogenomics: Studies exploring CYP2D6 and CYP2C19 genotyping to guide SSRI selection — not yet standard of care but evidence accumulating
Recent (2023–2025) meta-analyses:
- Escitalopram vs. other antidepressants (Yin et al., BMC Psychiatry 2023, PMID 38001423): Escitalopram showed superior response and remission vs. several other antidepressants
- Panic disorder (Guaiana et al., Cochrane 2023, PMID 38014714): SSRIs and benzodiazepines both effective; SSRIs preferred long-term
- Hyponatremia risk (Gheysens et al., Eur Psychiatry 2024, PMID 38403888): SSRIs carry higher hyponatremia risk than other antidepressant classes
- SSRI safety in pregnancy (Fabiano et al., Mol Psychiatry 2025, PMID 39266712): low absolute teratogenic risk confirmed
PART 16 — RECENT ADVANCES (2023–2026)
-
Post-SSRI Sexual Dysfunction (PSSD): EMA and FDA have mandated label updates acknowledging PSSD as a real, potentially persistent adverse effect. Research ongoing into mechanisms (epigenetic? neurosteroid?).
-
Pharmacogenomics in SSRI prescribing: CYP2D6 and CYP2C19 genotyping is increasingly available; strong metabolizers may need higher doses; poor metabolizers risk toxicity. Not yet in routine guidelines but several countries are moving toward it.
-
Antidepressants and neuroplasticity: Beyond the monoamine hypothesis — antidepressants enhance BDNF signaling, promote neurogenesis in the hippocampus, and facilitate synaptic remodeling. This "neuroplasticity hypothesis" may explain why combining SSRIs with psychotherapy is synergistic.
-
Esketamine (Spravato) as augmentation / monotherapy: FDA approved esketamine as first monotherapy for treatment-resistant depression in January 2025 — not an SSRI, but defines the "after SSRI fails" landscape.
-
SSRI hyperbolic tapering (Maudsley Deprescribing Guidelines, 2022+): Evidence-based protocol for SSRI discontinuation using hyperbolic dose reductions (small % reductions of remaining dose) rather than linear steps — prevents protracted withdrawal. Widely adopted in UK.
-
Gepirone (Exxua) FDA approved 2023: A 5-HT1A partial agonist/5-HT2 antagonist antidepressant — new class but with serotonergic mechanism, positioned alongside SSRIs.
-
Digital phenotyping and prediction of SSRI response: Machine learning models using EEG biomarkers (e.g., frontal alpha asymmetry) and digital biomarkers to predict SSRI response before prescribing — early research stage.
QUICK REFERENCE SUMMARY TABLE
| Drug | Unique feature | Best for | Avoid in | Key interaction | Half-life |
|---|---|---|---|---|---|
| Fluoxetine | 5-HT2C antagonism, activating | Hypersomnia, bulimia, low adherence | Agitated/anxious patients | 5-wk MAOI washout | ~7 days (with norfluoxetine) |
| Sertraline | Fewest interactions, PTSD | Pregnancy, cardiac disease, PTSD | — | Minimal | 26 hrs |
| Paroxetine | Anticholinergic, sedating | Insomnia, PE treatment | Elderly, pregnancy, dose-flexible patients | 2D6 potent | 20 hrs (autoinhibitor) |
| Fluvoxamine | Sigma-1 agonism | OCD, autism, unique receptor profile | Polypharmacy (1A2) | 1A2: clozapine↑↑↑ | 15–20 hrs |
| Citalopram | Racemic, cleanest profile | Simple cases, elderly | QTc risk, >40 mg | Minimal | 35 hrs |
| Escitalopram | S-enantiomer, allosteric SERT | First-line, elderly, MDD+GAD | — | Minimal | 27–32 hrs |
Sources
- Stahl's Essential Psychopharmacology, Cambridge University Press
- Harrison's Principles of Internal Medicine, 22nd ed. (McGraw-Hill, 2025)
- Kaplan & Sadock's Comprehensive Textbook of Psychiatry, 10th ed.
- The Maudsley Prescribing Guidelines in Psychiatry, 15th ed.
- Tintinalli's Emergency Medicine, 9th ed.
- Cipriani et al., Lancet 2018 (network meta-analysis, 21 antidepressants)
- Yin et al., BMC Psychiatry 2023 [PMID 38001423]
- Guaiana et al., Cochrane 2023 [PMID 38014714]
- Gheysens et al., Eur Psychiatry 2024 [PMID 38403888]
- Fabiano et al., Mol Psychiatry 2025 [PMID 39266712]
Explain further
Asking for Preferences
This is a shared conversation. Sign in to Orris to start your own chat.