Patient ventilator asynchrony
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
I now have comprehensive content from two authoritative respiratory textbooks. Let me synthesize a thorough answer.
Patient-Ventilator Asynchrony
Patient-ventilator asynchrony (also called dyssynchrony) is a mismatch between the patient's neural respiratory drive and the mechanical breath delivered by the ventilator — specifically, disagreement in timing (trigger, flow delivery, or cycling) or magnitude of support. It is common, under-recognized, and clinically significant.
Definition
"Patient-ventilator dyssynchrony occurs when the trigger, flow, and/or cycling of a mechanical breath is not in time agreement with the initiation and termination of a patient's neural inspiration, respectively, or if the magnitude of mechanical assist does not respond to the patient's respiratory demand." — Fishman's Pulmonary Diseases and Disorders
In the context of NIV specifically, asynchrony is defined as a mismatch between the patient's neural inspiratory time and the ventilator insufflation time.
Prevalence and Clinical Impact
- Frequent asynchronies (>10% of respiratory efforts) are present in 43% of patients undergoing NIV in observational studies.
- Consequences include:
- Respiratory muscle overload — increased work of breathing
- Compromised ventilation — inadequate CO₂ clearance or oxygenation
- Sleep disruption — fragmented sleep architecture
- Patient discomfort — leading to poor tolerance and NIV failure
- In invasive MV: risk of ventilator-induced lung injury (VILI) from breath stacking and large tidal volumes
Types of Asynchrony
1. Trigger Asynchrony
Mismatch between the patient's inspiratory effort and when the ventilator triggers a breath.
| Subtype | Description | Cause |
|---|---|---|
| Ineffective effort | Patient makes an effort but no breath is delivered | High auto-PEEP (e.g., COPD), weak muscles (NMD), insensitive trigger |
| Double-triggering | Ventilator delivers two breaths for one patient effort | Short inspiratory time relative to neural Ti |
| Auto-triggering | Ventilator delivers a breath without patient effort | Expiratory leaks causing false flow signals, circuit water, cardiac oscillations |
Management:
- Minimize mask leaks (most common cause in NIV)
- Minimize upper airway resistance (e.g., treat OSA, glottic closure)
- Adjust trigger sensitivity: increase to High for weak efforts; decrease to Low for auto-triggering
- For double-triggering: increase pressure support and/or decrease cycle sensitivity; extend Ti in PC mode
2. Cycling Asynchrony
Ventilator cycling (end-inspiration) does not coincide with end of patient's neural inspiration.
| Subtype | Description | Cause |
|---|---|---|
| Premature cycling (early) | Ventilator ends before patient finishes inspiring | Short Ti setting, mask leaks reducing flow quickly |
| Delayed/late cycling | Ventilator continues after patient wants to exhale → active exhalation against pressure | Long Ti setting, large leaks keeping flow above cycling threshold |
Delayed cycling (prolonged inspiration) is the most common asynchrony during NIV, driven by inspiratory leaks — because the delivered flow remains above the cycling criterion, the ventilator continues to insufflate.
Management:
- Minimize mask leaks first
- Review Ti (or Ti max) settings
- Adjust cycle sensitivity to Low (lower % of peak flow threshold) for premature cycling
- Consider switching to pressure-control (PC) mode
3. Flow Asynchrony
The flow delivered does not match the patient's demand — either too slow (flow starvation, causing a scooped waveform on the pressure trace) or excessive (discomfort).
Leading Causes
| Cause | Mechanism |
|---|---|
| Mask leaks | #1 cause in NIV — inspiratory leaks → prolonged inspiration; expiratory leaks → auto-triggering |
| Auto-PEEP / intrinsic PEEP | Raises triggering threshold → ineffective efforts (esp. COPD) |
| Inappropriate ventilator settings | Ti too short/long, trigger sensitivity off, PS level insufficient |
| Upper airway obstruction | OSA, glottic closure → resistance impairs triggering |
| Weak respiratory muscles | NMD patients — effort too small to trigger standard sensitivity |
Monitoring and Detection
- Flow and pressure waveform analysis at the bedside — key clinical tool
- Esophageal electromyography (EAdi): diaphragm electrical activity monitoring allows detection of asynchrony, assessment of respiratory muscle unloading, and direct triggering/cycling (NAVA mode)
- Polysomnography in home NIV patients — detects asynchrony during sleep
Management Summary (Troubleshooting Table)
| Asynchrony | First Action | Second Action | Third Action |
|---|---|---|---|
| Ineffective triggering | Minimize mask leak | Minimize UA resistance | Increase trigger sensitivity / switch to Auto-Trak |
| Double-triggering | Increase PS, decrease cycle sensitivity | Extend Ti in PC mode | — |
| Auto-triggering | Set trigger sensitivity to Low | Check for expiratory leaks | NIV-dedicated ventilator algorithms |
| Premature cycling | Minimize mask leak | Review Ti/Ti max | Set cycle sensitivity to Low |
| Late cycling | Minimize mask leak | Shorten Ti max | Set cycle sensitivity to High |
Special Considerations
NAVA (Neurally Adjusted Ventilatory Assist): Triggers, limits, and cycles the ventilator directly from diaphragm electrical activity — theoretically eliminates most asynchrony and is not affected by leaks.
NIV-specific modes: Modern ICU ventilators and dedicated NIV devices include leak-compensation algorithms that markedly reduce both prolonged inspiration (from inspiratory leaks) and auto-triggering (from expiratory leaks) compared to older standard ICU ventilators.
ARDS patients: Neuromuscular blocking agents may be beneficial when patient-ventilator asynchrony leads to breath stacking or very large inspiratory efforts that risk VILI.
Sources: Fishman's Pulmonary Diseases and Disorders (Patient-Ventilator Dyssynchrony, Table 148-4); Murray & Nadel's Textbook of Respiratory Medicine (Asynchrony under BPAP, NIV chapter)
Weaning from mechanical ventilation
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
I now have everything needed for a comprehensive, well-structured answer.
Weaning from Mechanical Ventilation
"Weaning" refers to all methods of discontinuing mechanical ventilation (MV) — from gradual reduction in support to abrupt removal. A more modern framing calls it liberation: an active, daily process of assessing readiness rather than a slow, passive taper. Approaching it as a prolonged wean unnecessarily extends MV time by up to 40%.
1. When to Consider Weaning: Readiness Criteria
Before initiating a spontaneous breathing trial (SBT), assess all of the following daily:
| Domain | Criterion |
|---|---|
| Underlying disease | Primary cause of respiratory failure has improved |
| Neurological | Patient is awake, alert, minimal or no sedation |
| Oxygenation | FiO₂ ≤ 0.5; PEEP < 8 cmH₂O; SaO₂ > 88% |
| Hemodynamics | Stable — no vasopressors or low-dose only |
| Airway | Manageable secretions; adequate cough |
2. Weaning Predictor Tests
These are screening tools — not confirmatory. Their role is to flag patients who are ready before the clinician would otherwise think to trial them, offsetting cognitive errors in bedside decision-making.
Rapid Shallow Breathing Index (RSBI / f/Vt ratio — the Tobin Index)
- Formula: Respiratory frequency (f) ÷ tidal volume in litres (Vt)
- Interpretation: f/Vt < 105 breaths/min/L predicts weaning success
- Sensitivity ≥ 0.90 in multiple studies — excellent as a screening test
- Must be measured during unassisted spontaneous breathing (not on PS or CPAP, which falsely lowers the ratio)
- The higher the f/Vt, the worse the prognosis: patients who fail weaning immediately develop rapid, shallow breathing (↑ RR + ↓ Vt) upon ventilator disconnection
Other indices (less relied upon than SBT outcome)
- Minute ventilation (Ve)
- Negative inspiratory force (NIF / MIP) — reflects respiratory muscle strength
- Vital capacity
- P0.1 (airway occlusion pressure) — reflects neural drive
Relying on these physiologic variables rather than the outcome of an SBT leads to unnecessary delays in extubation. — Harrison's Principles of Internal Medicine
3. Spontaneous Breathing Trial (SBT)
The SBT is the cornerstone of the weaning process. Positive-pressure support is reduced to a minimum and the patient breathes near-spontaneously for 30–120 minutes.
SBT Methods
| Method | Description |
|---|---|
| T-piece trial | Patient disconnected from ventilator; breathes through humidified T-circuit with supplemental O₂. No ventilator support. |
| Low-level CPAP | CPAP 5 cmH₂O — maintains airway patency, compensates for ET tube resistance |
| Low-level PSV | PSV 5–7 cmH₂O — most common; compensates for ET tube and circuit resistance |
| SIMV reduction | Gradual stepwise reduction of mandatory breath rate (least effective method) |
In patients not anticipated to have weaning difficulty, a 30-minute SBT is as effective as a 2-hour trial.
Passing the SBT — All of the following:
| Parameter | Pass Criterion |
|---|---|
| Respiratory rate | < 35 breaths/min |
| SpO₂ | > 90% |
| Systolic BP | 90–180 mmHg |
| Heart rate | Stable, change < 20% |
| Clinical appearance | No marked anxiety, dyspnoea, diaphoresis, or use of accessory muscles |
Patients passing an SBT have a >70% chance of successful extubation.
Incorporating a daily readiness screen + SBT protocol leads to:
- 25% fewer ventilator days
- 10% decrease in ICU length of stay
4. Algorithm for Discontinuing Mechanical Ventilation
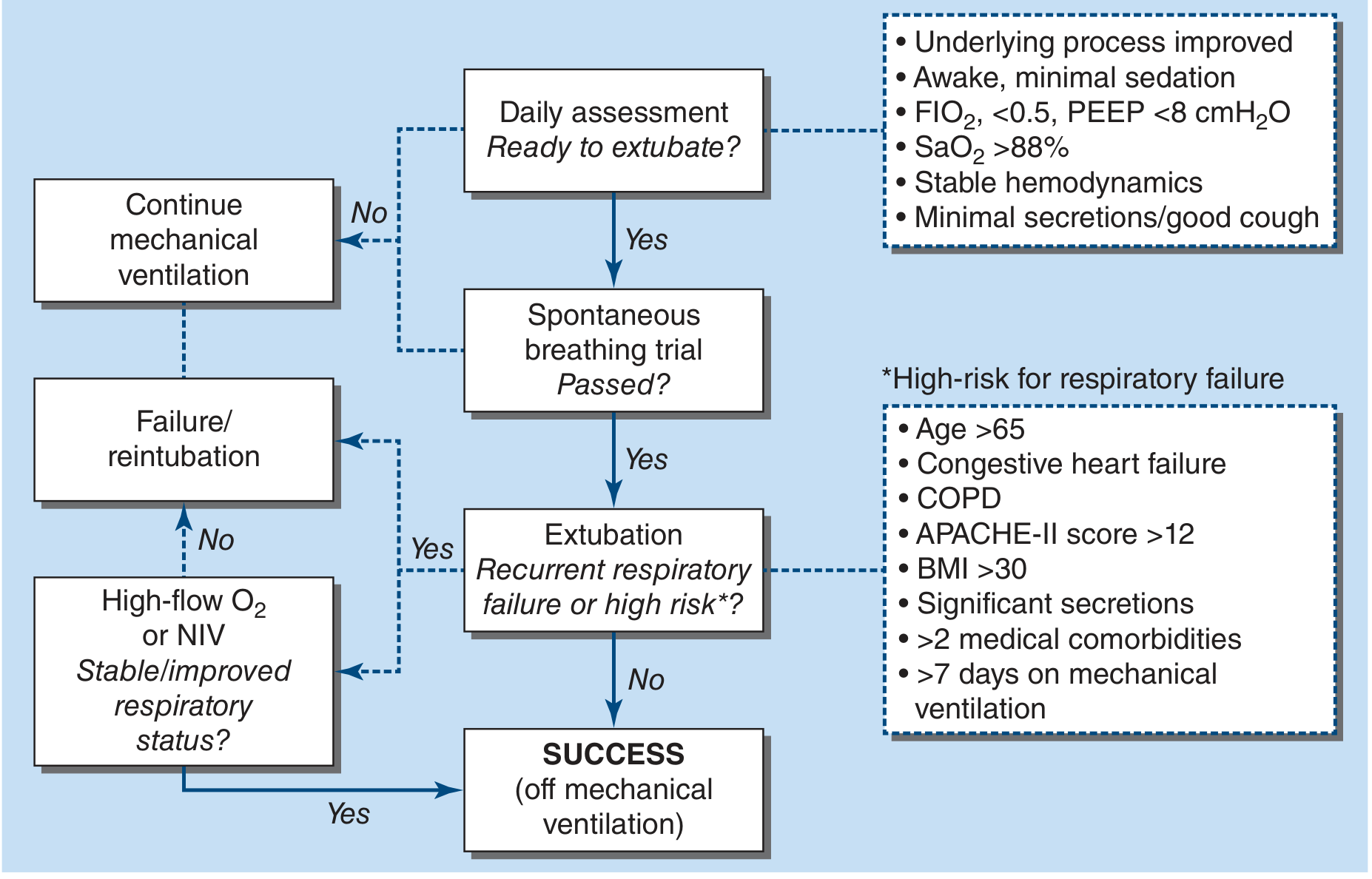
Algorithm for discontinuing mechanical ventilation. — Harrison's Principles of Internal Medicine, 22e
5. Causes of Weaning Failure
Up to 25% of patients experience respiratory distress requiring ventilator reinstitution after disconnection. The hallmarks of failed weaning are:
- Rapid, shallow breathing — immediately upon disconnection (↑ RR, ↓ Vt)
- Progressive respiratory effort over 30–60 min, reaching >4× normal by the end of a failed trial
Why mechanics worsen during a failed trial:
| Parameter | Change During Failed Trial |
|---|---|
| Respiratory resistance | Increases ~7× normal |
| Lung stiffness | Increases ~5× normal |
| Auto-PEEP (PEEPi) | More than doubles |
Importantly, respiratory mechanics before the trial are similar in patients who succeed and those who fail — the worsening is triggered by the act of spontaneous breathing itself (via unknown mechanisms), not pre-existing abnormality.
Cardiovascular stress of weaning failure:
- Extreme negative intrathoracic pressure swings → ↑ RV and LV afterload
- Pulmonary artery pressure ↑ 39%; systemic artery pressure ↑ 27%
- Patients who fail cannot increase O₂ delivery to meet demand → mixed venous O₂ saturation falls (occult cardiac dysfunction is unmasked)
Common causes of weaning failure:
- Unresolved underlying disease
- Respiratory muscle weakness / fatigue
- Excessive respiratory load (↑ resistance, ↓ compliance, auto-PEEP)
- Cardiovascular dysfunction / occult heart failure
- Neurological impairment / impaired drive
- Metabolic derangements (hypophosphataemia, hypomagnesaemia → muscle weakness)
- Psychological dependence (in prolonged ventilation)
6. Risk Factors for Extubation Failure Despite Passing SBT
Even after a successful SBT, extubation may fail. High-risk features:
- Age > 65
- Congestive heart failure
- COPD
- APACHE-II score > 12
- BMI > 30
- Significant secretions
-
2 medical comorbidities
-
7 days on mechanical ventilation
In these patients, transition immediately post-extubation to high-flow nasal oxygen (HFNO) or non-invasive ventilation (NIV) significantly reduces reintubation rates.
7. Prolonged Weaning
Patients who fail ≥ 3 SBTs or require > 7 days of weaning after the first SBT are classified as difficult or prolonged weaning. Strategies include:
- Tracheostomy — reduces dead space, improves comfort, facilitates secretion clearance
- Progressive SBT duration increases — gradually extend unassisted breathing time each day
- Inspiratory muscle training
- Optimizing nutrition — avoid overfeeding (↑ CO₂ production worsens ventilatory demand)
- Management of comorbidities (heart failure, infection, thyroid, electrolytes)
Sources: Fishman's Pulmonary Diseases and Disorders (Weaning, Causes of Weaning Failure, Timing, Weaning Trials — Chapter 147); Harrison's Principles of Internal Medicine 22e (Liberation from Mechanical Ventilation, Fig. 313-4)
Ventilation in ARDS
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive, authoritative content from two major respiratory textbooks. I have enough to write a thorough, well-organized answer.
Ventilation in ARDS
Overview
Mechanical ventilation (MV) is the cornerstone of ARDS management — but MV itself can aggravate or even induce lung injury (ventilator-induced lung injury, VILI). The central goal of ventilatory management is therefore not to normalise blood gases, but to support gas exchange while minimising further lung damage — "lung-protective ventilation."
Pathophysiologic Basis: Why the ARDS Lung is Vulnerable
CT imaging transformed understanding of ARDS. Rather than diffuse homogeneous injury (as plain radiographs suggest), ARDS lungs show heterogeneous patchy injury — consolidated dependent regions interspersed with more normal-appearing lung. Positive pressure therefore preferentially inflates the "baby lung" (the smaller volume of recruitable normal lung), placing it at risk of overdistension even with seemingly normal tidal volumes.
Mechanisms of VILI
| Mechanism | Description |
|---|---|
| Volutrauma | Overdistension from large tidal volumes → increased alveolar-capillary permeability → pulmonary edema. The dominant determinant — high Vt causes injury regardless of whether airway pressure is high or low |
| Barotrauma | High airway pressures → pneumothorax, pneumomediastinum, subcutaneous emphysema |
| Atelectrauma | Repetitive cyclic opening and closing of collapsed terminal units → high shear stress at the interface of aerated and collapsed tissue |
| Biotrauma | Mechanical ventilation triggers local and systemic inflammation; high Vt + zero PEEP causes synergistic elevation of inflammatory cytokines in BAL and blood → multiorgan dysfunction |
Lung-Protective Ventilation: The ARDSNet Protocol
The landmark ARDSNet ARMA trial (861 patients) demonstrated that low tidal volume ventilation reduces mortality from 39.8% → 31.0% (P = 0.007) and increases ventilator-free days. This is now the standard of care.
Part I — Ventilator Setup
| Step | Detail |
|---|---|
| Mode | Assist-Control (volume-cycled) |
| Initial Vt | 8 mL/kg predicted body weight (PBW) — reduce by 1 mL/kg every ≤2 h |
| Target Vt | 6 mL/kg PBW (reduce to 4 mL/kg if Pplat > 30 cmH₂O) |
| Respiratory rate | Set to approximate baseline minute ventilation (max 35 bpm) |
| Inspiratory flow | >80 L/min; I:E ratio 1:1.0–1.3 |
Use predicted (ideal) body weight, not actual body weight — actual BW can be ~20% higher due to fat and oedema, leading to dangerous over-ventilation.
Part II — Oxygenation Goal: PaO₂ 55–80 mmHg or SpO₂ 88–95%
Use these preset FiO₂/PEEP combinations (titrate upward as needed):
| FiO₂ | 0.3 | 0.4 | 0.4 | 0.5 | 0.5 | 0.6 | 0.7 | 0.7 | 0.8 | 0.9 | 0.9 | 1.0 | 1.0 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PEEP | 5 | 5 | 8 | 8 | 10 | 10 | 10 | 12 | 14 | 14 | 16–18 | 20 | 22–24 |
Part III — Plateau Pressure Goal: ≤30 cmH₂O
- Check Pplat with a 0.5-second inspiratory pause
- If Pplat > 30 cmH₂O → reduce Vt by 1 mL/kg (minimum 4 mL/kg PBW)
- If Pplat < 25 cmH₂O and Vt < 6 mL/kg → increase Vt by 1 mL/kg
No plateau pressure is known to be truly "safe" — the data show a continuous relationship between Pplat and mortality with no safe threshold.
Part IV — pH Goal: 7.30–7.45
| pH | Action |
|---|---|
| < 7.30 | Increase RR (max 35); consider sodium bicarbonate infusion |
| > 7.45 | Decrease RR |
Permissive hypercapnia — accept elevated PaCO₂ to maintain low Vt. This is the physiologic cost of lung protection.
PEEP in ARDS
PEEP serves two purposes:
- Prevent atelectrauma — keeps terminal units open, avoiding cyclic collapse/reopening
- Improve oxygenation — recruits collapsed alveoli, reduces shunt
Evidence on High vs. Low PEEP
- ALVEOLI, LOVS, EXPRESS trials: Higher PEEP (combined with low Vt) did not reduce 60-day mortality vs. lower PEEP strategies
- Meta-analysis of the three trials: Higher PEEP improved survival in moderate-to-severe ARDS (PaO₂/FiO₂ < 200) but not in mild ARDS
- Higher PEEP improved secondary outcomes: reduced rescue therapy use (LOVS), shorter MV duration and less organ failure (EXPRESS)
The optimal PEEP level in ARDS remains unclear. Higher PEEP is safe and may benefit severe ARDS; PEEP increments should only be applied when they produce reliable alveolar recruitment. — Fishman's Pulmonary Diseases and Disorders
Recruitment Maneuvers
Transient increases in airway pressure (e.g., sustained inflation at 40 cmH₂O for 40 seconds, or stepwise PEEP increments) to open collapsed alveoli. Benefit is transient unless adequate PEEP is applied afterward to prevent derecruitment. Used as a rescue strategy — not routine.
Prone Positioning
One of the most evidence-supported rescue interventions in severe ARDS:
- Redistributes perfusion toward non-dependent (now ventral) lung regions, better matching ventilation to perfusion
- Reduces dorsal compression atelectasis
- PROSEVA trial: Prone positioning ≥16 hours/day in severe ARDS (PaO₂/FiO₂ < 150) reduced 28-day mortality from 32.8% → 16.0%
- Indicated in moderate-to-severe ARDS (PaO₂/FiO₂ < 150 on FiO₂ ≥ 0.6, PEEP ≥ 5, on lung-protective ventilation)
Rescue / Salvage Strategies
For patients with severe refractory hypoxaemia despite standard lung-protective ventilation and PEEP:
| Intervention | Mechanism | Notes |
|---|---|---|
| Prone positioning | V/Q matching, recruits dorsal lung | Strongest mortality evidence (PROSEVA) |
| Neuromuscular blockade | Eliminates patient-ventilator asynchrony, reduces VILI from effort-driven injury | Beneficial in severe ARDS (PaO₂/FiO₂ < 150); reduces breath stacking |
| Inhaled NO / prostacyclin | Selective pulmonary vasodilation → V/Q improvement | Improves oxygenation transiently; no mortality benefit; used as bridge |
| High-frequency oscillatory ventilation (HFOV) | Ultra-small Vt at very high frequency | No mortality benefit in RCTs (OSCILLATE, OSCAR); not routinely recommended |
| ECMO (VV-ECMO) | Extracorporeal gas exchange — permits "ultra-lung-rest" | For refractory ARDS failing all conventional measures; EOLIA trial |
| ECCO₂R | Extracorporeal CO₂ removal | Permits further reduction of Vt below 6 mL/kg |
| Corticosteroids | Anti-inflammatory | Limited evidence; may be used in fibroproliferative phase |
| PC-Inverse Ratio Ventilation (PC-IRV) | Prolongs inspiration (I:E > 1) → ↑ mean airway pressure → improved oxygenation | Risk of auto-PEEP, dynamic hyperinflation, haemodynamic compromise; salvage only |
| APRV (Airway Pressure Release Ventilation) | High baseline PEEP with brief release phases for CO₂ clearance; allows spontaneous breathing | Limited controlled evidence; promotes recruitment |
Modes Allowing Spontaneous Breathing
APRV and BIPAP allow spontaneous breathing at any phase of the ventilatory cycle:
- APRV: sustained high pressure (P-high) to recruit + brief release to a low pressure (P-low) for CO₂ clearance, without allowing derecruitment
- Theoretical benefits: reduced sedation, preserved diaphragm function, improved haemodynamics
- Controlled data are limited; risk of patient self-inflicted lung injury (P-SILI) in vigorous spontaneous breathers
Fluid Management
The FACTT trial (ARDSNet) demonstrated that a conservative fluid strategy (targeting lower CVP/PAOP) vs. liberal strategy reduced ventilator days and ICU length of stay without increasing non-pulmonary organ failure. Conservative fluid management is therefore preferred once haemodynamic stability is achieved.
Summary: Priorities in ARDS Ventilation
- Vt 6 mL/kg PBW — non-negotiable foundation
- Pplat ≤ 30 cmH₂O — reduce Vt further if needed
- Permissive hypercapnia — tolerate high PaCO₂ to protect the lung
- Adequate PEEP — prevent atelectrauma; higher PEEP in moderate-severe disease
- Prone positioning — for PaO₂/FiO₂ < 150 early and sustained
- Conservative fluids after resuscitation
- Rescue strategies — stepwise escalation for refractory hypoxaemia
Sources: Murray & Nadel's Textbook of Respiratory Medicine (VILI, Mechanical Ventilation in ARDS, Role of PEEP and Recruitment Maneuvers — Chapter 134); Fishman's Pulmonary Diseases and Disorders (ARDSNet ARMA Trial, NIH NHLBI Low Tidal Volume Strategy Table 141-9, Rescue Interventions Table 141-12 — Chapter 141)
This is a shared conversation. Sign in to Orris to start your own chat.