Optical diagnoseis methods in medicine
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Searching Images
optical diagnosis endoscopy narrow band imaging chromoendoscopy OCT
Reading File
Loading Image
Now I have comprehensive material. Let me compile the full answer.
Optical Diagnosis Methods in Medicine
Optical diagnosis refers to a family of techniques that use light-based technologies to characterize tissue in real time, enabling clinicians to make or support a diagnosis without — or in addition to — conventional tissue biopsy. The field is most advanced in gastroenterology/endoscopy, ophthalmology, and laboratory medicine, though its principles extend across multiple specialties.
1. Chromoendoscopy (Dye-Based)
Chromoendoscopy uses dye agents sprayed onto the mucosa to enhance surface contrast and reveal architecture invisible to white-light endoscopy.
| Dye | Mechanism | Clinical use |
|---|---|---|
| Indigo carmine | Pools between mucosal grooves; 3D surface contrast | Polyp characterization, flat lesions |
| Methylene blue | Absorbed by normal epithelium; not absorbed by dysplasia | Barrett esophagus, gastric/duodenal neoplasia |
| Lugol solution | Stains glycogen in normal squamous epithelium; cancer = "Lugol-void lesions" | Early esophageal squamous cancer |
| Acetic acid | Denatures cytoplasmic proteins → "acetowhitening" | Intestinal metaplasia, cervical lesions |
Contrast chromoendoscopy is most powerful when combined with magnification endoscopy to evaluate pit patterns and mucosal architecture. — Yamada's Textbook of Gastroenterology, 7th ed.
2. Digital / Virtual Chromoendoscopy
These systems use electronic image processing rather than physical dyes, making them faster and easier to apply.
- Narrow-Band Imaging (NBI) — Olympus. Optical interference filters select 415 nm (blue) and 540 nm (green) light, both absorbed by haemoglobin. Blue light penetrates only the superficial capillary network; green light reaches deeper submucosal vessels. Result: dramatic enhancement of vascular patterns and mucosal surface detail.
- FICE (Fujifilm Intelligent Color Enhancement) — Electronically narrows white-light bandwidth post-acquisition to enhance pit patterns and microvasculature.
- i-SCAN — Pentax. Three algorithms: surface enhancement (SE), contrast enhancement (CE), and tone enhancement (TE). Post-acquisition software reconstructs images for enhanced mucosal contrast.
- Blue Laser Imaging (BLI) / Linked Color Imaging (LCI) — Fujifilm multi-light endoscopy platform; different modes optimized for vascular vs. colour contrast.
"Multiple meta-analyses comparing NBI and white light endoscopy have failed to show a difference in polyp detection rate or ADRs [adenoma detection rates]" — illustrating that enhanced imaging excels at lesion characterization rather than detection alone. — Clinical Gastrointestinal Endoscopy, Expert Consult, 3rd ed.
3. Magnification Endoscopy
High-resolution "zoom" endoscopes can magnify the mucosal surface up to ×100–150. Combined with NBI or chromoendoscopy, magnification endoscopy enables:
- Assessment of pit patterns (Kudo classification) to predict histology
- Evaluation of microvascular architecture (JNET classification) to differentiate adenoma grade
- Staging of early gastric cancer by mucosal morphology
- Detection of villous atrophy in coeliac disease
- Identification of dysplasia in Barrett esophagus
4. Optical Biopsy Techniques
The term "optical biopsy" describes technologies enabling real-time, in vivo tissue characterization at near-histological resolution — previously possible only with pathology.
a) Optical Coherence Tomography (OCT)
- Uses near-infrared light interference to produce cross-sectional images of tissue microstructure (analogous to ultrasound but using light).
- Imaging depth: 2–5 mm (into the submucosa).
- In Volumetric Laser Endomicroscopy (VLE): Fourier-domain OCT scans the full esophageal circumference, enabling assessment of Barrett esophagus at subcrypt level.
- Extensively used in ophthalmology (retinal imaging, glaucoma, macular degeneration) and cardiology (coronary plaque characterization).
b) Confocal Laser Endomicroscopy (CLE)
- A miniaturized confocal microscope integrated into the endoscope tip or passed through its channel.
- Delivers ×1000 in vivo magnification of the mucosal surface after fluorescent contrast agent administration.
- Enables real-time histological assessment of crypt architecture, goblet cells, and vascular patterns.
- Applications: Barrett esophagus, IBD surveillance, colorectal neoplasia.
c) Autofluorescence Imaging (AFI)
- Tissues naturally emit fluorescence (autofluorescence) when excited by specific wavelengths.
- Neoplastic tissue has different fluorescence spectra than normal tissue (often appears dark purple vs. green normal mucosa) due to changes in NADH, porphyrins, and collagen cross-links.
- Used as a wide-field tool to flag suspicious areas for subsequent targeted biopsy or detailed optical examination.
d) Optical Spectroscopy (Raman / Reflectance)
- Raman spectroscopy uses a laser beam to measure molecular vibrational signatures of tissue — effectively a "chemical fingerprint."
- Can distinguish between normal, hyperplastic, and adenomatous tissue in real time.
- Surface-enhanced Raman scattering (SERS) nanoparticles can be used as targeted molecular contrast agents for endoscopy.
"These novel modalities, such as optical coherence tomography, confocal laser endomicroscopy, autofluorescence endoscopy, optical spectroscopy, and molecular imaging techniques, are still under investigation, and the hope is that these technologies will facilitate the detection of early dysplasia or cancer." — Yamada's Textbook of Gastroenterology, 7th ed.
5. Molecular Imaging
Fluorescently labelled probes targeting specific molecular markers (e.g., EGFR, CEA, lectins) can be administered topically or systemically. When excited during endoscopy, they highlight areas of high receptor expression — associated with dysplasia or cancer — enabling "targeted optical biopsy."
6. Artificial Intelligence–Augmented Optical Diagnosis
AI systems trained on large endoscopic image datasets can perform:
- Real-time polyp detection (computer-aided detection, CADe)
- Real-time polyp characterization (computer-aided diagnosis, CADx) — distinguishing adenomas from hyperplastic polyps, enabling the "resect-and-discard" or "leave-in-place" strategies
- Pattern recognition for early gastric cancer, Barrett esophagus, and IBD dysplasia
"After extensive validation, [AI systems] can accurately recognize patterns, facilitating optical diagnosis." — Yamada's Textbook of Gastroenterology, 7th ed.
7. OCT in Ophthalmology
OCT is the gold standard imaging modality in modern ophthalmology, providing non-invasive cross-sectional imaging of retinal layers with micrometre resolution.
- Diagnoses and monitors: diabetic macular oedema, age-related macular degeneration (AMD), glaucoma (retinal nerve fibre layer thickness), epiretinal membranes, vitreomacular traction
- OCT Angiography (OCTA): motion-contrast OCT that maps retinal and choroidal vasculature without dye injection — replacing fluorescein angiography in many settings.
Visual Summary of Optical Endoscopy Technologies
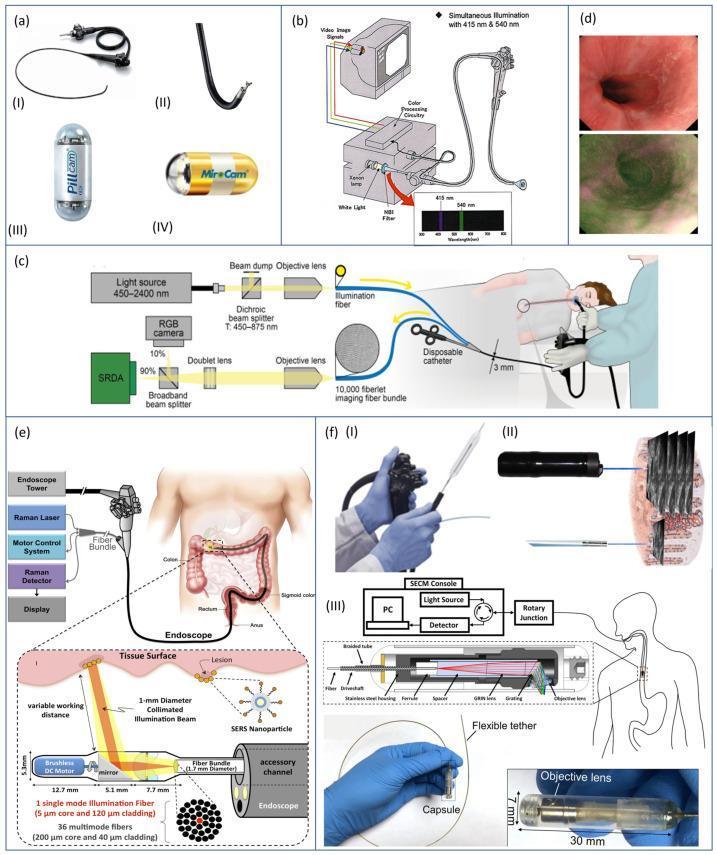
Composite diagram: (a) Standard vs. capsule endoscopes, (b) NBI light filtration at 415 & 540 nm, (c) Multi-spectral fiberscope, (d) AFI — neoplasia appears dark purple, (e) Raman spectroscopy with SERS nanoparticles, (f) OCT / CLE / tethered capsule endomicroscopy
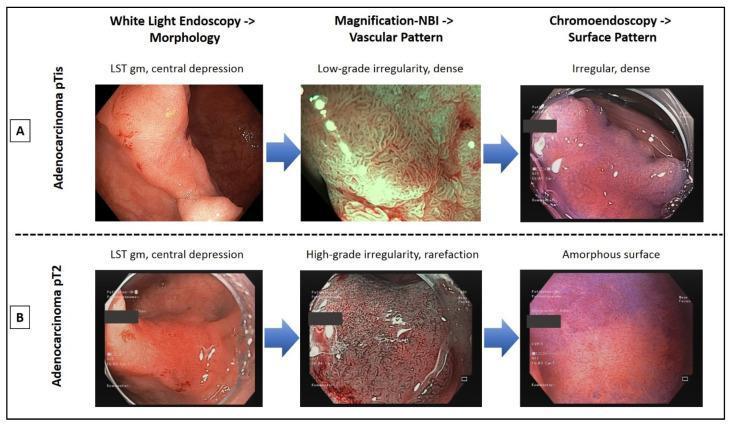
WLE vs. magnification NBI vs. chromoendoscopy: distinguishing carcinoma in situ (pTis, suitable for ESD) from invasive carcinoma (pT2, requiring surgery) based on vascular and pit-pattern analysis
Summary Table
| Method | Light used | Resolution | Depth | Primary role |
|---|---|---|---|---|
| White-light endoscopy | Broadband white | Macroscopic | Surface | Screening/general exam |
| NBI / virtual chromoendoscopy | 415 + 540 nm filtered | Enhanced surface/vascular | Mucosa | Lesion characterization |
| Dye chromoendoscopy | White + topical dye | Pit pattern detail | Surface | Dysplasia mapping |
| Magnification endoscopy | White/NBI | ×100–150 zoom | Surface | Architectural diagnosis |
| OCT / VLE | Near-infrared | ~10 µm | 2–5 mm | Subsurface layering |
| Confocal endomicroscopy | Laser (488 nm) | ~1 µm (histological) | 0–250 µm | Real-time histology |
| Autofluorescence | UV/blue excitation | Wide-field | Surface | Wide-field lesion flagging |
| Raman spectroscopy | Near-IR laser | Molecular | Surface | Tissue chemistry |
| Molecular imaging | Fluorescent probes | Targeted | Surface | Receptor-targeted detection |
| AI-CADe/CADx | Any modality | Algorithmic | — | Real-time decision support |
This is a shared conversation. Sign in to Orris to start your own chat.