HIFU in detail , give me each and every detail. And definition, principle ,mechanism , working, instrumentation and all you want to add
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
Here is a comprehensive, textbook-sourced breakdown of High-Intensity Focused Ultrasound (HIFU):
High-Intensity Focused Ultrasound (HIFU) — Complete Overview
1. Definition
HIFU is a completely non-invasive, thermal-based ablative technique that uses transcutaneously or transrectally focused ultrasonic waves to cause rapid, precise destruction of target tissue through coagulative necrosis, while leaving adjacent structures largely unharmed.
"HIFU is a unique thermal ablation technology in that it can be administered in an entirely noninvasive, extracorporeal fashion minimizing or eliminating the risk for tumor seeding, hemorrhage, or urinary extravasation." — Campbell Walsh Wein Urology
2. Historical Background
- The concept dates to Wood et al. in the 1920s, where HIFU was first proposed as an ablative technique.
- Research stagnated for decades due to lack of adequate imaging technologies.
- With the development of ultrasonography, CT, and MRI, accurate localization and real-time monitoring became possible, reviving clinical interest.
- Over the past 20 years, commercially available, precise HIFU delivery devices have been developed, particularly for urology.
— Hinman's Atlas of Urologic Surgery
3. Physical Principles
Ultrasound Fundamentals
- Ultrasound is an inaudible, high-frequency pressure wave emitted by a transducer.
- Diagnostic ultrasound uses 1–20 MHz frequencies.
- HIFU uses 0.8–4 MHz frequencies, but at energy levels approximately 10,000× greater than diagnostic ultrasound.
Focusing Principle
- When ultrasound waves are focused with an appropriately shaped transducer, energy is concentrated at a focal point.
- The intensity at the focal point can exceed 3,500 W/cm³, while surrounding tissue absorbs minimal energy.
- This achieves cell death at the focus while adjacent tissue is spared.
— Campbell Walsh Wein Urology
4. Mechanisms of Tissue Destruction
HIFU destroys tissue through two primary mechanisms:
A. Thermal (Heating) Mechanism
- Ultrasound waves are absorbed by tissue, causing molecular agitation.
- Resulting friction generates heat, rapidly raising tissue temperature to 80–100°C within seconds.
- This causes:
- Protein denaturation (melting of the lipid bilayer of the cellular membrane)
- Denaturation of lipoprotein structures in organelle membranes
- Coagulative necrosis — the predominant histologic finding
- Temperature elevation of 50–95°C in 1–2 seconds is sufficient for irreversible cell death.
- Heating is favored at higher ultrasound frequencies.
B. Cavitation (Mechanical) Mechanism
- At intensities >3,500 W/cm³, microbubbles form within the tissue (cavitation).
- These bubbles undergo rapid collapse, generating extremely high local temperatures and a mechanically disrupting "shock wave" effect — similar to extracorporeal shock wave lithotripsy (ESWL).
- Cavitation is favored at lower ultrasound frequencies.
- This adds further mechanical destruction on top of thermal injury.
— Hinman's Atlas of Urologic Surgery; Campbell Walsh Wein Urology; Dermatology 2-Volume Set
5. Instrumentation
Core Component: The Transducer
- The piezoelectric transducer generates ultrasound waves.
- It is specially curved/shaped so that waves converge at a defined focal point — delivering maximum energy at the target depth.
- The same transducer may serve dual purpose: treatment delivery + real-time imaging.
Key Design Parameters
| Parameter | Detail |
|---|---|
| Frequency | 0.8–4 MHz |
| Intensity at focus | >3,500 W/cm³ possible |
| Tissue temperature | Up to 80–100°C |
| Ablation zone size | 0.8 × 0.2 × 0.2 mm³ per pulse (small, precise) |
| Lesion size (clinical) | 10–26 mm depending on device |
Monitoring Systems
Two modalities are used to guide and monitor HIFU:
- Diagnostic Ultrasound (most common):
- Mounted with the treatment probe for real-time imaging
- Monitors gray-scale hyperechoic changes (hyperechoic = microbubble/steam formation from heated tissue)
- Portable and cost-effective; however, bubbles cause imaging artifacts
- MRI Guidance (MR-HIFU / MRgFUS):
- Uses MR thermometry for real-time temperature mapping in the target and surrounding structures (urethra, sphincter)
- Higher cost and less accessible but more precise for deep targets
Rectal Cooling System
- Devices used for prostate treatment include a cool water circulating system within the endorectal probe to prevent rectal wall thermal injury.
- Real-time rectal wall temperature monitoring and patient movement detection are built-in safety features.
6. Commercial HIFU Devices
1. Sonablate (SonaCare Inc., USA)
- Mobile, minimally invasive endorectal probe device
- Treats prostates up to 40 mL
- Lesion size: 10–12 mm diameter
- Features real-time imaging, mpMRI fusion capability
- Color-coded temperature monitoring of treatment zone and adjacent structures (neurovascular bundles, rectal wall)
- Robotic transducer for whole-gland targeting
- Patient position: extended lithotomy
2. Ablatherm (EDAP TMS, France)
- Semi-automated device with endorectal probe
- Consists of: treatment module + control module + endorectal probe (with both treatment and imaging transducers)
- Lesion size: 19–26 mm via predefined power protocols
- Real-time intraoperative imaging with adjustable energy delivery
- Rectal cooling + movement detection safety systems
- Patient position: right lateral decubitus
— Hinman's Atlas of Urologic Surgery
3. MRgFUS / TULSA Systems
- MRI-guided focused ultrasound (MRgFUS): In-bore MRI guidance with thermometry
- Transurethral Ultrasound Ablation (TULSA): Miniaturized probe inserted transurethrally — especially useful for anterior prostate lesions that are harder to reach transrectally
- Spatial control of ablation within 1.3 mm on MRI thermometry
— Campbell Walsh Wein Urology
7. Working / Procedure
Pre-procedure
- Bowel preparation (full bowel prep the night before, or enema 1 hour prior)
- Antimicrobial prophylaxis: IV broad-spectrum antibiotics (metronidazole, cefuroxime, gentamicin)
- mpMRI for lesion characterization and treatment planning
- Patient consent with discussion of risks
- Anticoagulant/antiplatelet management
Anesthesia
- General or spinal anesthesia required
- If spinal: heavy sedation to prevent movement
- Paralysis + ventilation if prostate movement from breathing is detected
- ⚠️ Nitrous oxide is CONTRAINDICATED — causes microbubble formation in the prostate, producing artifactual changes that impair safe energy delivery
— Hinman's Atlas of Urologic Surgery
Intraoperative Steps
- Patient positioned (lithotomy or lateral decubitus)
- Urethral catheter inserted for planning
- Optional pre-procedure mini-TURP (4–6 weeks prior) to reduce prostate height and stricture risk
- Digital rectal exam to confirm empty rectum
- Endorectal HIFU probe introduced with lubricating jelly
- Prostate mapped ultrasonographically; target treatment area defined
- If mpMRI fusion used — calibrated at this step
- Rectal cooling system activated
- Energy delivered under real-time monitoring
- Operator monitors for hyperechoic changes (steam/microbubbles = effective treatment)
- Treatment paused if rectal wall or other structures are at risk
Post-procedure
- Usually day-case procedure (discharged same day)
- Urethral catheter left in place 5–10 days
- Oral antibiotics for 7 days (fluoroquinolones or beta-lactams)
- Alpha-blockers for up to 14 days
- Mild pain — simple oral analgesics usually sufficient
- PSA monitoring (may take months to reach nadir)
- Follow-up: 3-monthly × 1 year → 6-monthly × 1 year → annually
- mpMRI at 12 months; biopsy if recurrence suspected
— Hinman's Atlas of Urologic Surgery
8. Ablation Zone and Treatment Planning
Because each pulse creates a very small ablation zone (~0.8 × 0.2 × 0.2 mm³), multiple overlapping sonications are required to treat a whole lesion. The transducer is sequentially refocused to "paint" the target volume. This makes treatment times lengthy — reported mean duration of ~5.5 hours (range 1.5–9 hours) for renal HIFU; prostate treatment is generally shorter.
Ablation Strategies (Prostate):
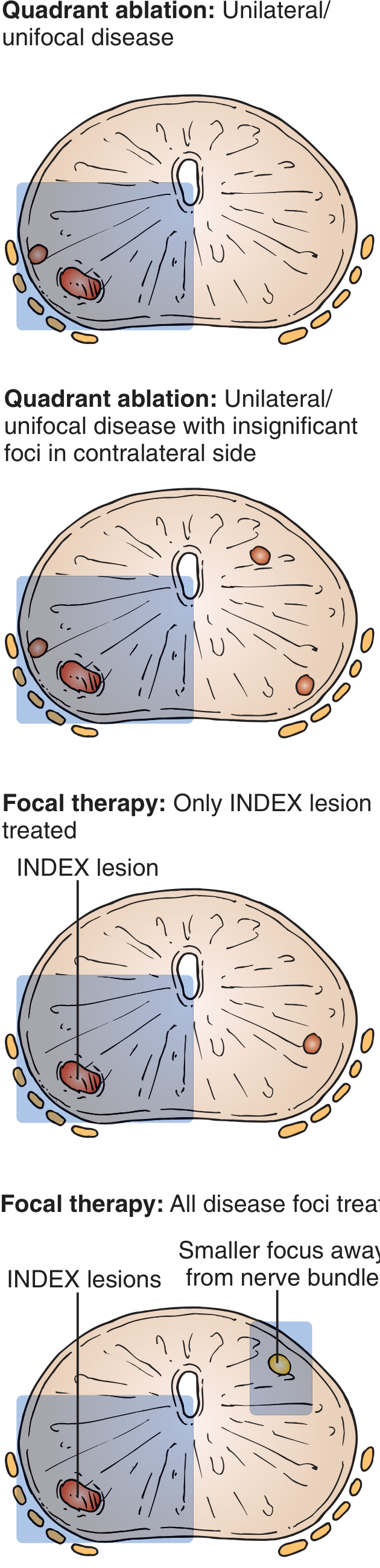
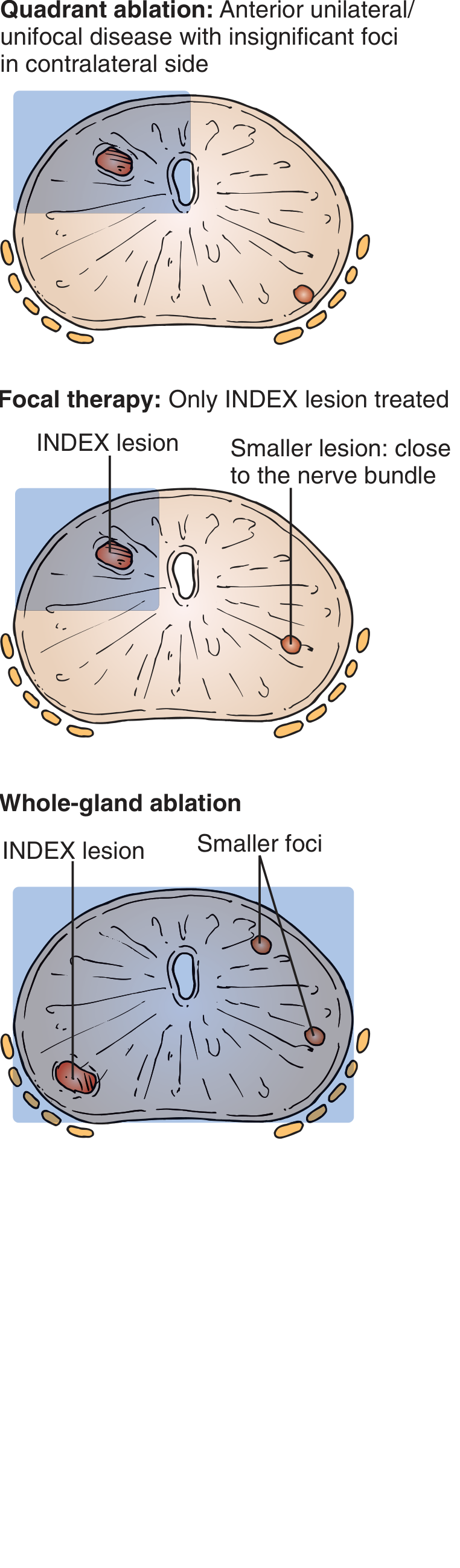
| Strategy | Indication |
|---|---|
| Focal therapy | Single index lesion ≥ Gleason 3+4, volume >0.2 mL |
| Hemiablation / Quadrant ablation | Unilateral/unifocal disease |
| Whole-gland ablation | Multifocal disease |
— Hinman's Atlas of Urologic Surgery
9. Clinical Applications
| Organ/System | Application |
|---|---|
| Prostate | Localized prostate cancer (primary & salvage therapy); BPH (symptomatic, refractory to medical therapy) |
| Kidney | Renal cell carcinoma (small tumors, investigational for renal HIFU) |
| Breast | Breast cancer ablation (MRI-guided); fibroadenoma reduction |
| Bone | Osteoid osteoma ablation (MRI-guided) |
| Thyroid | Toxic adenoma (render euthyroid — select institutions) |
| Liver/Pancreas | Tumor ablation (under investigation) |
| Brain | MR-guided FUS for essential tremor, Parkinson's disease (transcranial) |
| Aesthetics | Subcutaneous fat reduction (LipoSonix™) — FDA-cleared for waist circumference reduction; brow lifting |
| Uterine fibroids | MRgFUS ablation |
— Multiple sources: Dermatology 2-Volume Set; Grainger & Allison's Radiology; Current Surgical Therapy 14e; Sabiston Textbook of Surgery
10. Indications (Prostate Cancer — Most Established)
- Clinically localized prostate cancer
- Gleason score ≥ 3+4 (clinically significant disease)
- Tumor volume > 0.2 mL, visible on mpMRI and concordant with histology
- Patients with serious comorbidities precluding surgery or radiation
- Prior therapies excluding major surgery or RT
- Can be considered for salvage after radiotherapy failure
— Hinman's Atlas of Urologic Surgery; Miller's Anesthesia
11. Contraindications / Limitations
- Prostate size > 40 mL (Sonablate limit) — may require pre-TURP downsizing
- Anterior lesions — more difficult to treat with transrectal HIFU (energy dissipation over intervening tissue; edema displaces targets)
- Calcifications in the treatment zone interfere with energy delivery
- Inadequate imaging window (bowel gas, obesity)
- Nitrous oxide anesthesia (absolute contraindication intraoperatively)
- Respiratory movement — limits extracorporeal HIFU for kidney (acoustic shadowing, refraction)
12. Advantages
| Advantage | Detail |
|---|---|
| Non-invasive | No skin incision, no probe penetration of organ |
| No ionizing radiation | Unlike X-ray-based therapies |
| No tumor seeding | No needle tract |
| No hemorrhage risk | Purely extracorporeal/endoluminal |
| Organ preservation | Targeted; spares surrounding tissue |
| Repeatable | Can re-treat recurrent lesions |
| Cosmetic | No scarring (breast/aesthetic applications) |
| Outpatient | Day-case procedure |
13. Complications
| Complication | Notes |
|---|---|
| Urinary retention | Common; catheter left 5–10 days |
| Urinary tract infection | Managed with prophylactic antibiotics |
| Urethral stricture | Reduced by pre-TURP or suprapubic catheter |
| Recto-urethral fistula | Serious; prevented by rectal cooling |
| Erectile dysfunction | Less common than with surgery/cryotherapy; ~60–78% potency preservation with focal therapy |
| Urinary incontinence | Less than radical prostatectomy |
| Incomplete ablation | Especially for anterior gland or large prostates |
| Pain / discomfort | Especially in aesthetic applications (HIFU lipolysis) |
| Edema, ecchymosis, numbness | Aesthetic applications |
| Skin burns | Inadequate probe contact; aesthetic use |
| Pectoralis major injury | Breast HIFU |
— Current Surgical Therapy 14e; Hinman's Atlas; Dermatology 2-Volume Set
14. Monitoring Modalities Summary
| Modality | Advantages | Limitations |
|---|---|---|
| Ultrasound | Real-time, portable, inexpensive, combined with treatment probe | Artifacts from microbubbles; poor image quality |
| MRI (MRgFUS) | Real-time thermometry, precise anatomy, monitors urethra/sphincter | Expensive, less accessible, requires MR-compatible equipment |
15. Emerging Developments
- TULSA (Transurethral Ultrasound Ablation): Miniaturized intraluminal probe enabling precise anterior prostate treatment under in-bore MRI guidance — spatial control within 1.3 mm
- Transcranial MRgFUS: Focused ultrasound through the skull for neurologic disorders (essential tremor, Parkinson's)
- MRI-guided breast HIFU: Complete ablation achieved in ~46% of patients in systematic review; ongoing refinement
- Extracorporeal renal HIFU: Currently investigational; hampered by respiratory motion and incomplete ablation rates
Summary Table
| Feature | Detail |
|---|---|
| Technology type | Non-invasive thermal ablation |
| Energy source | Focused ultrasound waves (0.8–4 MHz) |
| Mechanism | Thermal coagulation + cavitation |
| Target temperature | 55–100°C at focal point |
| Histologic result | Coagulative necrosis |
| Guidance | Ultrasound or MRI |
| Primary clinical use | Prostate cancer |
| Other uses | Breast, kidney, bone, thyroid, brain, fibroids, aesthetics |
| Anesthesia | General or spinal (NO nitrous oxide) |
| Key devices | Sonablate, Ablatherm, MRgFUS, TULSA |
Sources: Hinman's Atlas of Urologic Surgery; Campbell Walsh Wein Urology 3-Volume Set; Miller's Anesthesia 10e; Current Surgical Therapy 14e; Grainger & Allison's Diagnostic Radiology; Dermatology 2-Volume Set 5e; Sabiston Textbook of Surgery; Robbins & Kumar Basic Pathology
HIFU in detail , give me each and every detail. And definition, principle ,mechanism , working, instrumentation and all you want to add
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Finding Sources
Reading File
Reading File
Now I have comprehensive data from multiple authoritative textbooks. Here is the complete, fully sourced response:
High-Intensity Focused Ultrasound (HIFU) — Complete Reference
1. Definition
HIFU is a completely non-invasive, thermal-based ablative technique that uses precisely focused high-intensity ultrasonic waves — delivered transcutaneously or through an intracavitary probe — to cause rapid, targeted destruction of tissue via coagulative necrosis, while leaving adjacent structures largely unharmed.
"HIFU is a unique thermal ablation technology in that it can be administered in an entirely noninvasive, extracorporeal fashion, minimizing or eliminating the risk for tumor seeding, hemorrhage, or urinary extravasation." — Campbell Walsh Wein Urology
2. Historical Background
| Era | Development |
|---|---|
| 1920s | Wood et al. first proposed focused ultrasound as an ablative technique |
| 1940s–1980s | Limited progress — lacked adequate imaging for precise targeting |
| Early 1990s | Madersbacher et al. reported the first clinical use of HIFU in symptomatic BPH; further studies explored histologic changes |
| 1987 | Burgess, Iwamoto, and Coleman reported histologic tissue changes after HIFU exposure |
| 2000s onward | Commercially available devices developed; routine use established for prostate cancer |
| 2011 | FDA cleared HIFU (LipoSonix™) for non-invasive waist circumference reduction |
"In the early 1990s a number of authors investigated the therapeutic uses of high-intensity focused ultrasound (HIFU). Madersbacher et al. were the first to report the safety and efficacy of HIFU in patients with symptomatic BPH." — Campbell Walsh Wein Urology
3. Physical Principles
Ultrasound as a Wave
Ultrasound is an inaudible, oscillating pressure (stress) wave that propagates at the speed of sound through tissue with minimal scattering. This minimal scattering is what allows ultrasound waves to be focused deeply within soft tissues, unlike light or radio waves.
Frequency Comparison
| Type | Frequency Range | Energy Level |
|---|---|---|
| Diagnostic ultrasound | 1–20 MHz | Baseline |
| HIFU therapeutic | 0.8–4 MHz | ~10,000× greater than diagnostic |
Lower HIFU frequencies penetrate deeper; higher frequencies favor thermal heating. The critical point is that HIFU delivers an enormous concentration of energy exclusively at the focal point.
The Focusing Principle
When ultrasound waves are emitted from a curved (concave) transducer, they converge geometrically at a single focal point — like sunlight through a magnifying glass. Energy intensity is concentrated at the focus to levels exceeding 3,500 W/cm³, while tissues in the path to and beyond the focus absorb minimal energy and are spared.
"The energy levels delivered are about 10,000 times greater in a HIFU beam than a modern diagnostic ultrasound beam. This amount of energy can rapidly increase the temperature in the beam's focus to 80°C or higher, which is adequate for causing cell death. Outside of the focus, little damage occurs to tissue." — Hinman's Atlas of Urologic Surgery
4. Mechanisms of Tissue Destruction
HIFU destroys tissue through two fundamental mechanisms that often act simultaneously:
Mechanism 1 — Thermal Coagulation (Primary)
Sequence of events:
- Ultrasound waves travel through tissue and are absorbed by molecules
- Absorption causes molecular agitation — the molecules vibrate at ultrasonic frequencies
- The resulting intermolecular friction generates heat
- Tissue temperature rises to 55–100°C within 1–2 seconds
- This causes:
- Melting of the phospholipid bilayer of the cell membrane
- Denaturation of proteins (structural and enzymatic)
- Denaturation of lipoprotein structures in organelle membranes (mitochondria, ER, nucleus)
- Irreversible coagulative necrosis
Thermal threshold for cell death: ≥ 55°C. At 80–100°C, cell death is instantaneous.
"The ultrasound waves are absorbed by the tissue and agitate the molecules within. The subsequent friction generates heat and the temperature in the tissue affected can rise to 100°C in seconds. The heat denatures protein and lipoprotein structures in the cell membrane and the membranes of organelles contained within." — Hinman's Atlas of Urologic Surgery
"The focused ultrasound waves cause rapid (1–2 seconds) temperature elevation within the targeted tissue up to 50°C to 95°C. This causes melting of the lipid bilayer of the cellular membrane and protein denaturation, leading to tissue necrosis, whereas tissues outside the targeted area are spared." — Current Surgical Therapy 14e
Mechanism 2 — Acoustic Cavitation (Secondary)
Sequence of events:
- At very high intensities (> 3,500 W/cm³), dissolved gases in tissue nucleate and form microbubbles
- These bubbles undergo rapid oscillation (stable cavitation) and then violent collapse (inertial/transient cavitation)
- Bubble collapse generates:
- Extremely high local temperatures (thousands of degrees at the bubble wall)
- High-pressure shock waves — mechanically destructive, analogous to ESWL (extracorporeal shock wave lithotripsy)
- Free radical production — chemical damage to DNA and membranes
- This causes additional mechanical tissue disruption on top of thermal injury
"At sufficiently high intensities (>3500 W/cm³), cavitation and microbubble formation occur that yield extremely high temperatures and a mechanically disrupting 'shock wave' effect similar to that seen with extracorporeal shock wave lithotripsy." — Campbell Walsh Wein Urology
Note: The relative contribution of each mechanism depends on frequency:
- Thermal heating is favored at higher frequencies
- Cavitation is favored at lower frequencies
Three Mechanisms Summary (Dermatology perspective)
"Intensity is concentrated near the focal point, to the extent that tissue damage can occur by one or more of three mechanisms — heating, cavitation, or shock waves. Heating is favored at high ultrasound frequencies while cavitation (a rapidly expanding gas bubble) is favored at low ultrasound frequencies; shock waves are high-pressure, supersonic pressure waves that can be very destructive." — Dermatology 2-Volume Set 5e
5. Histologic Result
The end tissue result is coagulative necrosis — the same pattern seen with ischemic infarction:
- Cell outlines preserved but nuclear pyknosis/karyolysis
- Ghost cells with no viable nuclei
- Sharp demarcation between treated and untreated tissue
- Surrounding tissue intact (the hallmark of precise HIFU ablation)
6. Instrumentation
A. The Transducer — Core Component
The transducer is the heart of every HIFU system. It:
- Is made of piezoelectric crystal material (converts electrical energy → acoustic energy)
- Is curved/concave in shape so that emitted waves converge at a preset focal depth
- Operates at 0.8–4 MHz for HIFU therapy
- May be dual-function: treatment delivery + real-time imaging (combined in one probe)
Lesion geometry is determined by:
- Transducer design (curvature, aperture, focal length)
- "On" and "off" beam time (duty cycle)
- Power delivered (in watts)
- Tissue acoustic properties
B. Ablation Zone Dimensions
| Parameter | Value |
|---|---|
| Single-pulse ablation zone | ~0.8 × 0.2 × 0.2 mm³ (very small, precise) |
| Clinical lesion size (Sonablate) | 10–12 mm diameter |
| Clinical lesion size (Ablatherm) | 19–26 mm diameter |
| Temperature at focus | 55–100°C |
| Energy intensity at focus | Can exceed 3,500 W/cm³ |
Because each pulse ablates only a tiny volume, multiple overlapping sonications are needed — the transducer is robotically repositioned to sequentially "paint" the target volume.
C. Imaging & Monitoring Systems
Two modalities guide HIFU delivery:
1. Ultrasound Guidance (Most Common)
- The imaging transducer is mounted alongside or within the treatment probe — enabling real-time simultaneous treatment + imaging
- Hyperechoic (bright) changes on gray-scale ultrasound = microbubble/steam formation from heated tissue — confirms effective treatment
- Portable, inexpensive, widely accessible
- Limitation: Bubbles cause imaging artifacts that can obscure visualization; poor image quality in difficult cases
2. MRI Guidance (MRgFUS — Gold Standard for Precision)
- Uses MR thermometry to produce real-time temperature maps of the target zone and surrounding structures (urethra, sphincter, rectal wall)
- Provides superior anatomic resolution for accurate targeting
- More expensive, requires MR-compatible HIFU equipment and dedicated MRI suite
- Used for brain, uterine fibroids, bone, and emerging prostate applications
D. Safety Systems (Prostate Devices)
- Rectal cooling system: Circulating cool water within the endorectal probe protects the rectal wall from thermal injury
- Real-time rectal wall temperature monitoring
- Patient movement detection (automatic treatment pause if movement detected)
- Color-coded temperature display (Sonablate) for neurovascular bundle and rectal wall
7. Commercial HIFU Devices
1. Sonablate (SonaCare Inc., Indiana, USA)
- Mobile, minimally invasive
- Endorectal probe (transrectal approach)
- Treats prostates up to 40 mL
- Lesion size: 10–12 mm per sonication
- Robotic transducer for precise targeting throughout the gland
- Real-time color-coded temperature monitoring of treatment zone and adjacent critical structures
- mpMRI fusion imaging capability
- Operator can adjust power in real time based on tissue response
- Patient position: extended lithotomy
2. Ablatherm (EDAP TMS, Lyon, France)
- Semi-automated system
- Components: treatment module + control module + endorectal probe (dual transducer: treatment + imaging)
- Lesion size: 19–26 mm via predefined power protocols
- Real-time intraoperative imaging with adjustable energy delivery
- Rectal cooling + movement detection + rectal wall monitoring
- Power is preset (fixed protocols)
- Patient position: right lateral decubitus
3. MRgFUS Systems (e.g., ExAblate — InSightec)
- In-bore MRI guidance with real-time thermometry
- Superior targeting accuracy — spatial control within 1.3 mm
- Applications: uterine fibroids, bone metastases, brain (transcranial), prostate
4. TULSA (Transurethral Ultrasound Ablation)
- Miniaturized HIFU probe inserted transurethrally (not transrectally)
- In-bore MRI guidance
- Particularly suited for anterior prostate lesions inaccessible to transrectal HIFU
- Multicenter phase I trial: spatial control within 1.3 mm, 3.3% grade 3 complication rate, no grade 4/5 complications
— Campbell Walsh Wein Urology
5. LipoSonix™ (Aesthetic HIFU)
- Focal depth: 1.3 cm into subcutaneous fat
- Temperature up to 70°C — thermally destroys adipocytes
- FDA-cleared (2011) for non-invasive waist circumference reduction
- Single treatment reduces waist circumference by 2–3 cm
— Dermatology 2-Volume Set 5e
8. Step-by-Step Working / Procedure (Prostate Cancer Model)
Pre-Procedure Assessment
- Complete staging workup: PSA, multiparametric MRI (mpMRI), targeted + systematic biopsy
- Gleason score and tumor volume confirmation
- Zonal risk assessment (which zones harbor disease)
- Anticoagulant/antiplatelet medication management
Bowel Preparation
- Full bowel preparation the evening before, OR
- Enema administered 1 hour prior to surgery
- Purpose: optimize perirectal ultrasonographic imaging quality
Antimicrobial Prophylaxis
- Intravenous broad-spectrum antibiotics: metronidazole + cefuroxime + gentamicin (gram-negative and anaerobic coverage)
- Surgery postponed if active urinary tract infection or bacteriuria present
- Postoperative oral antibiotics: ciprofloxacin × 7 days
Anesthesia
- General or spinal anesthesia required (any patient movement impairs precision)
- Spinal: heavy sedation mandatory
- If prostate movement from breathing is detected → neuromuscular paralysis + ventilation
- ⚠️ Nitrous oxide is ABSOLUTELY CONTRAINDICATED — causes microbubble formation throughout the prostate, producing widespread artifacts that make safe energy delivery impossible
— Hinman's Atlas of Urologic Surgery; Miller's Anesthesia 10e
Intraoperative Steps
| Step | Detail |
|---|---|
| 1 | Patient positioned: lithotomy (Sonablate) or right lateral decubitus (Ablatherm) |
| 2 | Warming blanket applied to prevent hypothermia |
| 3 | Urethral catheter inserted for planning |
| 4 | Optional pre-procedure mini-TURP (performed 4–6 weeks prior) to reduce prostate volume and minimize post-HIFU stricture risk |
| 5 | Digital rectal exam to confirm empty rectum |
| 6 | Endorectal HIFU probe introduced with lubricating jelly |
| 7 | Prostate mapped ultrasonographically; target treatment zone defined |
| 8 | If mpMRI fusion used: calibrated at this point |
| 9 | Rectal cooling system activated |
| 10 | Energy delivered; operator monitors for hyperechoic changes (steam = effective ablation) |
| 11 | Transducer repositioned robotically; overlapping sonications paint the full target volume |
| 12 | Treatment paused/stopped if rectal wall or critical structures at risk |
| 13 | Power adjusted in real time (Sonablate) or follows preset protocol (Ablatherm) |
| 14 | Urethral catheter left in situ at end of procedure |
Post-Procedure Care
| Item | Detail |
|---|---|
| Catheter | Left in place 5–10 days |
| Discharge | Usually same day (day-case procedure) |
| Antibiotics | Fluoroquinolones or beta-lactams × 7 days |
| Alpha-blockers | Up to 14 days post-procedure (facilitates voiding, reduces retention) |
| Analgesia | Simple oral analgesics (pain is usually mild) |
| Follow-up | 3-monthly × 1 year → 6-monthly × 1 year → annually thereafter |
| PSA | Monitored at each visit; can take several months to reach nadir — do not interpret early results in isolation |
| mpMRI | At 12 months post-procedure |
| Rebiopsy | If concern for incomplete treatment or recurrence |
9. Ablation Zone Strategies (Prostate)
Because each HIFU pulse creates a tiny (sub-millimeter) ablation, treatment strategy must be planned based on disease distribution:


| Strategy | Indication |
|---|---|
| Focal therapy (index lesion only) | Single clinically significant lesion ≥ Gleason 3+4, volume > 0.2 mL, visible on mpMRI |
| Quadrant / Hemiablation | Unilateral disease; may spare contralateral neurovascular bundle |
| Whole-gland ablation | Multifocal bilateral disease |
Lesion must be visible on mpMRI and concordant with histology before planning.
10. Indications
Prostate
- Localized prostate cancer (primary treatment):
- Clinically significant disease: Gleason ≥ 3+4, volume > 0.2 mL
- Focal, hemiablation, or whole-gland approach
- Patients with comorbidities precluding surgery or radiation
- Salvage HIFU (after radiotherapy failure):
- In an open-label trial (n=100): 81% of patients had negative biopsy at 12 months; 50/78 achieved PSA nadir ≤ 0.5 ng/mL
- BPH (benign prostatic hyperplasia) — symptomatic, refractory to medical therapy
- Emerging option for low-risk disease alongside cryotherapy (Schwartz's Principles of Surgery)
Other Organs
| Organ | Application |
|---|---|
| Kidney | Renal cell carcinoma — currently investigational; challenged by respiratory movement |
| Breast | Breast cancer ablation (MRI-guided); fibroadenoma volume reduction |
| Bone | Osteoid osteoma ablation (image-guided); osseous metastases palliation |
| Uterus | Fibroids (MRgFUS — well established) |
| Thyroid | Toxic adenoma — render euthyroid (select institutions) |
| Brain | Essential tremor, Parkinson's disease (transcranial MRgFUS) |
| Liver/Pancreas | Tumor ablation (under investigation) |
| Aesthetic | Subcutaneous fat reduction (LipoSonix™); brow lifting (Ulthera™) |
11. Contraindications / Limitations
| Factor | Explanation |
|---|---|
| Prostate > 40 mL | Exceeds Sonablate limit; requires pre-TURP downsizing |
| Anterior prostate lesions | Energy dissipates over intervening prostate tissue; progressive edema displaces anterior targets during treatment |
| Calcifications in treatment zone | Cause acoustic shadowing and scatter |
| Active UTI / bacteriuria | Must be treated before procedure |
| Nitrous oxide anesthesia | Absolute contraindication intraoperatively |
| Respiratory motion (renal HIFU) | Causes targeting errors; acoustic shadowing and refraction |
| Poor imaging window | Bowel gas, obesity, large prostate |
12. Complications
Prostate HIFU Complications (Hinman's Atlas)
| Complication | Rate |
|---|---|
| Debris, dysuria, LUTS | Common |
| Acute urinary retention | 1–2% |
| Urinary tract infection | 5–10% |
| Epididymo-orchitis | 5–10% |
| Hematuria / hematospermia | Self-limiting, may persist weeks |
| Retrograde ejaculation / anejaculation | ~50% |
| Urinary incontinence (pad-requiring) | 1–2% |
| Urethral stricture | Uncommon; reduced by pre-TURP |
| Rectourethral fistula | ~1 in 500 — most severe complication |
| Need for second treatment | ~1 in 5 patients |
Salvage HIFU (Post-Radiation) — Higher Complication Rates
From a 100-patient open-label trial:
- 80 patients: Grade 2 toxicity
- 20 patients: Grade 3 toxicity
- Complications included: 5 rectal fistulas, 3 cases osteitis pubis, 3 cases recalcitrant hematuria requiring intervention
— Campbell Walsh Wein Urology
Aesthetic HIFU (LipoSonix™)
- Post-procedural pain (moderate to severe in some)
- Numbness, edema, ecchymoses
Breast HIFU (Systematic Review, 167 patients)
- Pain: 40.1%
- Edema: 16.8%
- Skin burn: 4.2%
- Pectoralis major injury: 3.6%
— Current Surgical Therapy 14e
13. Comparison with Other Ablative Modalities
| Modality | Invasiveness | Mechanism | Local Recurrence Rate |
|---|---|---|---|
| HIFU | Non-invasive | Thermal + cavitation | 2.9% |
| Cryoablation | Percutaneous probes | Freeze-thaw | 1.4% |
| RFA | Percutaneous probe | Thermal (RF current) | 3.1% |
| Microwave ablation | Percutaneous probe | Thermal (microwave) | 0% (limited data) |
| Laser ablation | Percutaneous probe | Thermal (laser) | 10.7% |
— Current Surgical Therapy 14e
HIFU's unique advantage: It is the only thermal ablation technique that can be entirely extracorporeal (no skin puncture, no needle tract, no tumor seeding risk).
14. Advantages of HIFU
| Advantage | Detail |
|---|---|
| Truly non-invasive | No incision, no needle, no probe penetration of the organ |
| No ionizing radiation | Unlike X-ray therapies |
| No tumor seeding | No needle tract through tumor |
| No hemorrhage | No vascular disruption |
| Organ-sparing | Targeted destruction preserves surrounding tissue |
| Outpatient procedure | Day-case, rapid recovery |
| Repeatable | Can re-treat recurrent disease |
| No scarring | Important for breast and aesthetic applications |
| Real-time monitoring | Treatment effect visualized intraoperatively |
| Erectile function preservation | 60–78% with focal therapy (vs. ~15–20% with cryoablation) |
15. Erectile Function Outcomes (Prostate HIFU)
"Preservation of function with whole-gland high-intensity focused ultrasound (HIFU) is more common but may vary with the aggressiveness and oncologic efficacy of the procedure." — Campbell Walsh Wein Urology
- Focal/hemiablation HIFU: 60–78% recovery of erectile function
- Whole-gland HIFU: less favorable, but better than whole-gland cryoablation
- Re-treatment carries increased rates of erectile dysfunction and stricture
16. Oncologic Outcomes (Prostate)
Primary HIFU (Focal/Hemiablation)
- French hemiablation trial (n=111): Largest published series
- Infield positive biopsy rate: 15.5–34.6% (all cancer); 8.2–11.5% (clinically significant)
- Outfield positive biopsy: 7.7–36.7% (all cancer); up to 2% (clinically significant)
- Recurrence-free rate: 90% at 5 years, 38% at 10 years (early series, pre-MRI era)
Salvage HIFU (Post-Radiation)
- 81% negative biopsy at 12 months
- 64% (50/78) achieved PSA nadir ≤ 0.5 ng/mL with negative biopsy
17. Emerging Innovations
| Innovation | Detail |
|---|---|
| TULSA (Transurethral Ultrasound Ablation) | Miniaturized transurethral probe; MRI-guided; precision ≤1.3 mm; treats anterior prostate |
| Transcranial MRgFUS | Non-invasive neurosurgery; FDA-approved for essential tremor and Parkinson's |
| MRI-guided breast HIFU | Complete tumor ablation in 46.2%; ongoing refinement |
| Laparoscopic HIFU | Developed to overcome respiratory motion artifact for renal HIFU |
| AI-assisted treatment planning | Automatic target delineation from mpMRI |
| Immunological priming | Emerging evidence that HIFU-induced necrosis may stimulate anti-tumor immune response |
18. Summary Reference Table
| Feature | Detail |
|---|---|
| Full name | High-Intensity Focused Ultrasound |
| Technology class | Non-invasive thermal ablation |
| Energy source | Acoustic (ultrasound) waves |
| Frequency | 0.8–4 MHz |
| Energy at focus | ~10,000× greater than diagnostic US; >3,500 W/cm³ |
| Target temperature | 55–100°C at focal point |
| Time to ablation | 1–2 seconds per sonication |
| Mechanisms | Thermal coagulation + acoustic cavitation + shock waves |
| Histologic result | Coagulative necrosis |
| Ablation zone/pulse | ~0.8 × 0.2 × 0.2 mm³ |
| Guidance modalities | Ultrasound (real-time) or MRI (thermometry) |
| Primary use | Prostate cancer |
| Key devices | Sonablate, Ablatherm, MRgFUS (ExAblate), TULSA, LipoSonix™ |
| Anesthesia | General or spinal (NO nitrous oxide) |
| Re-treatment rate | ~1 in 5 patients |
| Most feared complication | Rectourethral fistula (~1 in 500) |
| Erectile preservation (focal) | 60–78% |
Sources: Hinman's Atlas of Urologic Surgery E-Book; Campbell Walsh Wein Urology 3-Volume Set; Miller's Anesthesia 10e; Current Surgical Therapy 14e; Dermatology 2-Volume Set 5e; Grainger & Allison's Diagnostic Radiology; Schwartz's Principles of Surgery 11e; Robbins & Kumar Basic Pathology
This is a shared conversation. Sign in to Orris to start your own chat.