Parkinson disease
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Here is a comprehensive overview of Parkinson Disease (PD) drawn from multiple authoritative medical textbooks.
Parkinson Disease
Definition & Epidemiology
PD is the second most common progressive neurodegenerative disorder after Alzheimer disease. It affects approximately 1% of people over age 60 and 4–5% of those over 85. The disease is uncommon before age 40, and incidence is higher in men than women. Up to 15% of patients have a first- or second-degree relative with PD, suggesting genetic predisposition.
Pathophysiology
PD results from loss of dopaminergic neurons in the substantia nigra pars compacta, which project to the striatum (nigrostriatal pathway) and are critical for motor control. The hallmark pathologic finding is the Lewy body — an eosinophilic, round intracytoplasmic inclusion composed of α-synuclein, neurofilaments, and ubiquitin.
Circuit-level mechanism:
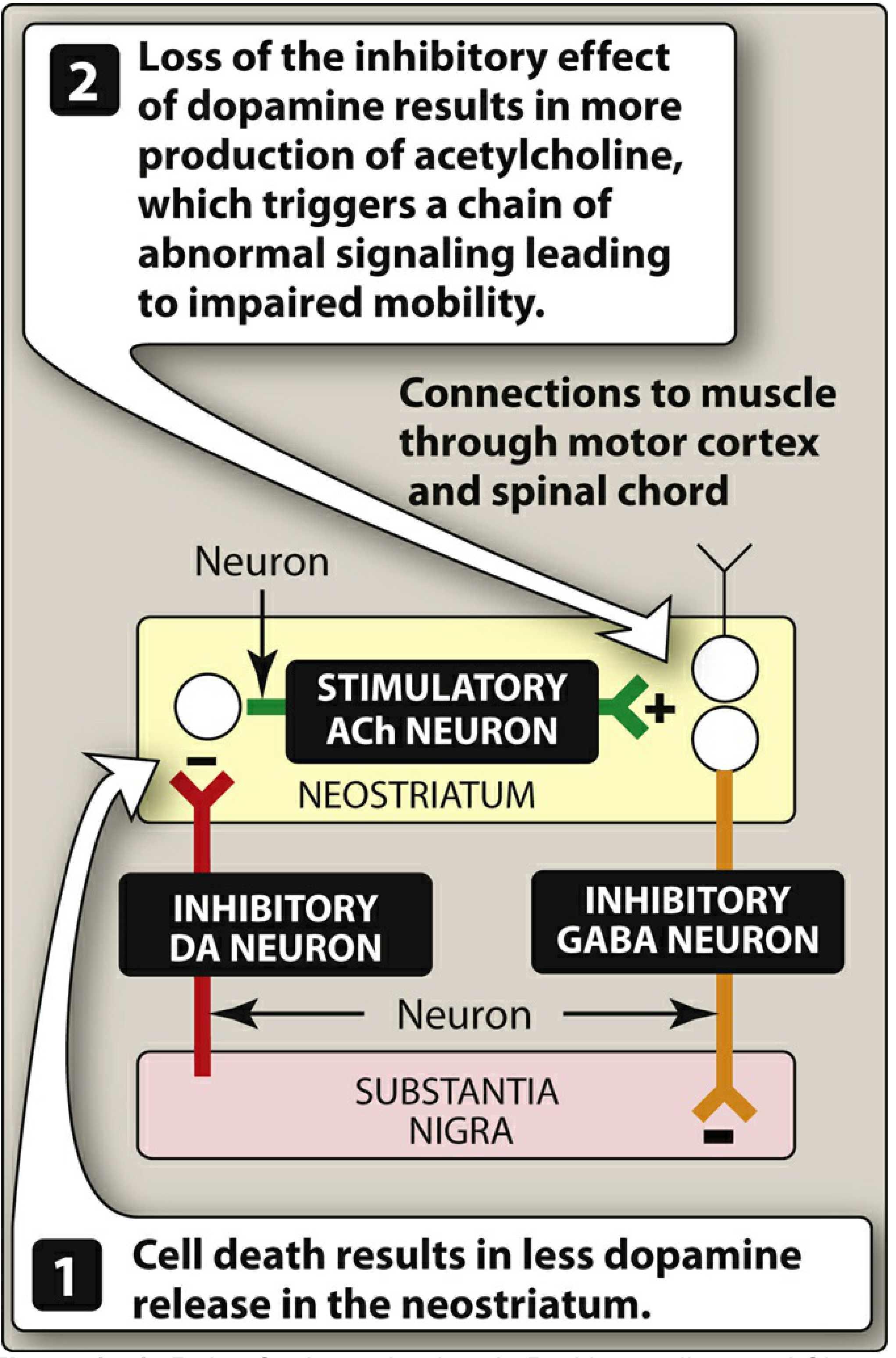
Normally, substantia nigra neurons release inhibitory dopamine onto neostriatal cholinergic neurons. In PD:
- Death of nigral cells → less dopamine release in the neostriatum
- Loss of dopaminergic inhibition → relative excess of excitatory acetylcholine
- Abnormal motor signaling → impaired voluntary movement
Molecular pathogenesis:
- α-synuclein misfolds and aggregates intraneuronally
- Defective autophagy and lysosomal degradation (Parkin, LRRK2, glucocerebrosidase mutations)
- Mitochondrial dysfunction
- Gross pathology: pallor of the substantia nigra and locus ceruleus
Clinical Features
The classic triad (usually asymmetric at onset):
| Feature | Description |
|---|---|
| Tremor | "Pill-rolling" resting tremor; presenting symptom in ~70%; virtually pathognomonic when asymmetric |
| Rigidity | Often with cogwheeling (ratchety passive resistance) |
| Bradykinesia | Slowness of movement; patient may describe it as "weakness" |
Other motor features: masked (expressionless) facies, micrographia, narrow shuffling gait, postural instability (late finding).
Non-motor features: dysphagia (objective prevalence up to 82%), aspiration (15–56% of patients), drooling, autonomic dysfunction, depression, hallucinations, and dementia in later stages. Death often results from aspiration pneumonia or trauma from falls.
Differential Diagnosis (Parkinsonism-Plus Syndromes)
Up to 20% of patients initially diagnosed with PD ultimately have an alternative diagnosis. Red flags suggesting a parkinsonism-plus syndrome:
- Hallucinations or early dementia
- Early postural instability or autonomic dysfunction
- Paralysis of vertical gaze
- Failure to respond to levodopa
- Involuntary movements other than tremor
Key mimics:
| Condition | Distinguishing Features |
|---|---|
| Progressive Supranuclear Palsy (PSP) | Vertical gaze palsy (especially downward), axial rigidity, poor levodopa response |
| Dementia with Lewy Bodies | Early cognitive impairment and hallucinations, rigidity > tremor |
| Multisystem Atrophy | Autonomic dysfunction (orthostatic hypotension), cerebellar signs, poor levodopa response |
| Vascular Parkinsonism | Step-wise progression, MRI multi-infarcts, lower-body predominance |
| Drug-induced Parkinsonism | History of dopamine blockers (neuroleptics, metoclopramide, amiodarone, valproic acid, lithium) — usually reversible |
Treatment
Strategy
Therapy aims to:
- Restore dopamine in the basal ganglia
- Antagonize excess cholinergic activity
Drugs offer symptomatic relief only — none currently slow neurodegeneration.
Levodopa + Carbidopa (First-line)
- Levodopa is the immediate precursor of dopamine and crosses the blood-brain barrier; dopamine itself does not
- Carbidopa inhibits peripheral DOPA decarboxylase → reduces peripheral conversion of levodopa to dopamine, cutting the required levodopa dose by 4–5× and reducing peripheral side effects (nausea, vomiting, arrhythmias)
- Effective for bradykinesia, rigidity, and tremor
- Substantially reduces symptoms in ~2/3 of patients for the first few years; decline in response typically begins in the 3rd–5th year
- "On-off" phenomenon: unpredictable motor fluctuations, not simply related to plasma levels
Adverse effects of levodopa:
- Peripheral: nausea/vomiting (CTZ stimulation), orthostatic hypotension, tachycardia, dyskinesias
- CNS: visual/auditory hallucinations, abnormal involuntary movements (dyskinesias) from dopamine overactivity
- Urine/saliva may darken (melanin from catecholamine oxidation)
Key interactions:
- Pyridoxine (B6) → increases peripheral levodopa breakdown → reduces efficacy
- Non-selective MAOIs → contraindicated (risk of hypertensive crisis)
MAO-B Inhibitors
| Drug | Notes |
|---|---|
| Selegiline | Selective MAO-B inhibitor; reduces dopamine metabolism; metabolized to amphetamine (risk of insomnia) |
| Rasagiline | 5× more potent than selegiline; not metabolized to amphetamine |
| Safinamide | MAO-B inhibitor; adjunct to levodopa-carbidopa |
All: avoid concurrent serotonergic agents (serotonin syndrome risk).
Dopamine Agonists
Used as monotherapy in early disease or adjuncts to levodopa. Help reduce motor fluctuations.
Anticholinergics
Reduce relative excess of ACh; useful particularly for tremor and rigidity.
COMT Inhibitors (e.g., entacapone, tolcapone)
Inhibit peripheral breakdown of levodopa → prolong its effect; adjuncts that reduce "wearing off."
Deep Brain Stimulation (DBS)
Electrodes implanted in the globus pallidus or subthalamic nucleus modulate basal ganglia circuitry, allowing significant reduction in levodopa dose in appropriate candidates.
Managing PD Psychosis
A therapeutic challenge: dopamine-enhancing treatments cause hallucinations, while dopamine blockers (antipsychotics) worsen motor symptoms.
- Most antipsychotics are contraindicated in PD
- Low-dose quetiapine or clozapine (weak D2 blockade) may be used cautiously
- Pimavanserin (5-HT2A inverse agonist/antagonist) — approved specifically for PD psychosis; no direct dopamine antagonism, thus low risk of worsening the movement disorder
Disease Course
PD typically progresses over 10–15 years, eventually producing severe motor slowing approaching near-immobility. Early initiation of treatment is recommended when symptoms cause functional impairment.
Sources: Textbook of Family Medicine 9e; Robbins & Kumar Basic Pathology; Lippincott Illustrated Reviews: Pharmacology; Bradley and Daroff's Neurology in Clinical Practice
pyramidal role
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of the Pyramidal (Corticospinal) System — its anatomy, physiology, and clinical significance.
The Pyramidal System
Terminology
The term "pyramidal tract" is a near-synonym for the corticospinal tract. The name comes from the triangular-shaped medullary pyramids on the ventral surface of the medulla oblongata, where these fibers collect before crossing. The terminology is somewhat imprecise, as the medullary pyramids also include some reticulospinal and other brainstem pathway fibers — but in clinical practice, "pyramidal" refers to the corticospinal system.
Origin: Motor Cortex
The corticospinal tract does not arise solely from the primary motor cortex. Its origins are:
| Source | Contribution |
|---|---|
| Primary motor cortex (Brodmann area 4, precentral gyrus) | ~30–50% |
| Premotor & supplementary motor areas (area 6) | ~30% |
| Somatosensory cortex (areas 3, 1, 2, 5, 7, posterior to central sulcus) | ~30–40% |
The cortical neurons contributing to this tract reside mainly in cortical layer 5. About 3% are giant Betz cells — the largest neurons in the human nervous system (~60 µm diameter), found exclusively in the primary motor cortex. Their axons conduct at ~70 m/s, the fastest of any corticospinal fibers. The total fiber count per tract exceeds 1 million.
Course of the Pyramidal Tract
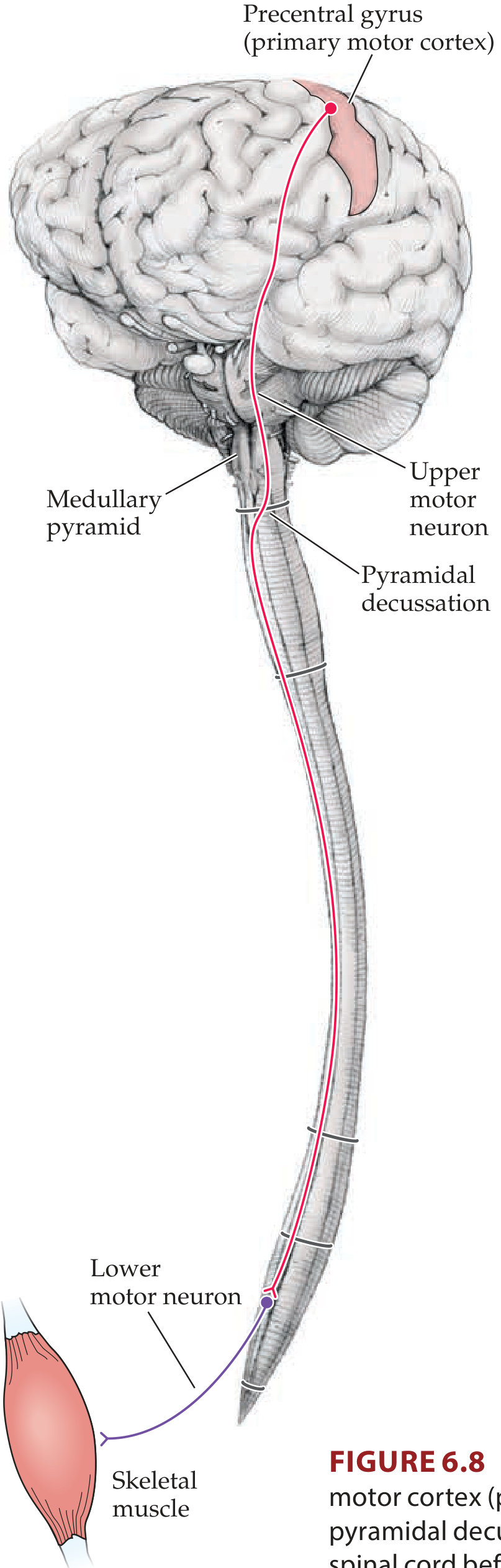
Step-by-step descent:
-
Corona radiata — axons from the motor cortex fan out into the cerebral white matter
-
Internal capsule (posterior limb) — fibers condense into a compact bundle; somatotopic organization is preserved:
- Face fibers → most anterior (genu)
- Arm fibers → mid-posterior limb
- Leg fibers → most posterior
- Lesions here often produce contralateral hemiplegia affecting face, arm, and leg together
-
Cerebral peduncles (basis pedunculi) — middle third carries corticospinal and corticobulbar fibers; face medial, leg lateral
-
Ventral pons — fibers scatter into fascicles as pontine nuclei and transverse fibers (destined for the cerebellum) interrupt them
-
Medullary pyramids — fibers reconverge on the ventral medulla surface, forming the visible pyramids
-
Pyramidal decussation (cervicomedullary junction, foramen magnum level):
- ~85% of fibers cross → enter lateral white matter of spinal cord → lateral corticospinal tract
- ~15% remain ipsilateral → anterior (ventral) corticospinal tract (most eventually cross in the cervical/upper thoracic cord; control bilateral postural movements)
-
Spinal cord — fibers descend somatotopically in the lateral column and synapse onto:
- Interneurons in the intermediate gray (mainly)
- Directly onto anterior horn cells (lower motor neurons) — particularly in cervical and lumbosacral enlargements controlling the hands and feet
Function
The pyramidal system is the primary pathway for voluntary, skilled, fine motor control, especially of the distal extremities (hands and fingers). Key functions:
- Initiation and precise execution of voluntary movements
- Speed and fractionation of finger movements
- Modulation of spinal reflex circuits
- Some corticospinal axons (from somatosensory cortex) regulate afferent sensory input (gate proprioceptive and tactile signals entering the dorsal horn)
Additional Fiber Pathways from Motor Cortex
Beyond the main corticospinal tract, the motor cortex also sends fibers to:
- Caudate nucleus & putamen → basal ganglia circuits (postural control)
- Red nucleus (midbrain) → rubrospinal tract
- Reticular formation & vestibular nuclei → reticulospinal and vestibulospinal tracts
- Pontine nuclei → cerebellum (corticopontocerebellar pathway)
Upper vs. Lower Motor Neuron
The pyramidal system is synonymous with the upper motor neuron (UMN) system.
| Feature | UMN Lesion (Pyramidal) | LMN Lesion |
|---|---|---|
| Weakness | Yes (contralateral) | Yes (ipsilateral) |
| Tone | Increased (spasticity) | Decreased (flaccidity) |
| Reflexes | Hyperreflexia | Hyporeflexia / areflexia |
| Babinski sign | Positive (extensor plantar) | Absent |
| Atrophy | Minimal (disuse only) | Prominent (denervation) |
| Fasciculations | Absent | Present |
| Clonus | May be present | Absent |
Clinical significance of lesion location:
- Internal capsule (e.g., capsular stroke) → contralateral face + arm + leg hemiplegia
- Cerebral peduncle → contralateral hemiplegia ± ipsilateral CN III palsy (Weber syndrome)
- Ventral pons → contralateral hemiplegia ± ipsilateral CN VI palsy
- Lateral spinal cord → ipsilateral UMN weakness below the level (tract already crossed), combined with Brown-Séquard features
- Foramen magnum / upper cervical cord → may cause quadriparesis + lower cranial nerve signs
Why "Pyramidal" in Parkinson Disease Context?
In Parkinson disease, the extrapyramidal system (basal ganglia circuits) is primarily diseased — NOT the pyramidal tract. This is why PD produces:
- Hypokinesia/bradykinesia (reduced movement initiation)
- Rigidity (not spasticity)
- Resting tremor
- Normal or reduced reflexes (no Babinski sign)
This is the key distinguishing feature: pyramidal lesions → spasticity + hyperreflexia + Babinski; extrapyramidal (PD) → rigidity + bradykinesia, normal reflexes.
Sources: Neuroanatomy through Clinical Cases 3rd Ed.; Guyton and Hall Textbook of Medical Physiology; Bradley and Daroff's Neurology in Clinical Practice
This is a shared conversation. Sign in to Orris to start your own chat.