Topics for independent work: 1. Cardiogenic shock: definition, mechanism of development, pathophysiology, signs, diagnostics. 2. Sudden cardiac death: definition, mechanism of development, pathophysiology, signs, diagnostics. 3. CRP/ AED algorithms 8 pages on this topic
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have comprehensive material from Harrison's, Rosen's, Tintinalli's, and Robbins to write a thorough 8-page document. Let me compose it now.
Cardiogenic Shock, Sudden Cardiac Death & CPR/AED Algorithms
Independent Work — Emergency Cardiology
PART 1: CARDIOGENIC SHOCK
1.1 Definition
Cardiogenic shock (CS) is a state of low cardiac output resulting in life-threatening end-organ hypoperfusion and hypoxia that cannot be attributed to hypovolemia or distributive causes. Clinically, it is defined by:
- Persistent systolic BP < 90 mmHg, or mean arterial pressure (MAP) < 60–65 mmHg, unresponsive to volume replacement
- Requirement of vasopressors to maintain adequate BP
- Clinical features of peripheral hypoperfusion: cold clammy extremities, oliguria, altered mental status
- Elevated arterial lactate > 2 mmol/L as an objective marker of hypoperfusion
In-hospital mortality ranges from 40–60%, depending on shock severity and underlying etiology.
— Harrison's Principles of Internal Medicine 22E, p. 2368
1.2 Etiology and Incidence
The most common cause is acute myocardial infarction (MI) with left ventricular (LV) dysfunction, accounting for approximately 80% of CS complicating MI. The incidence of CS complicating acute MI has declined to 5–10% due to widespread early mechanical reperfusion therapy. CS is more common in ST-elevation MI (STEMI) than non-STEMI.
Other causes of CS include:
| Category | Specific Causes |
|---|---|
| Mechanical MI complications | Acute severe mitral regurgitation (MR), ventricular septal rupture (VSR), free wall rupture/tamponade |
| Right ventricular failure | RV infarction, acute/chronic cor pulmonale |
| Non-ischemic myocardial | Myocarditis, dilated cardiomyopathy, Takotsubo syndrome |
| Obstructive | Pericardial tamponade, massive pulmonary embolism |
| Arrhythmic | Malignant tachyarrhythmia, complete heart block |
| Valvular | Critical aortic stenosis, acute severe aortic or mitral regurgitation |
| Toxic/metabolic | Severe acidosis, severe hypoxemia, cardiotoxic drugs |
Risk factors in MI patients: older age, anterior MI location, prior MI, diabetes mellitus, multivessel coronary artery disease.
— Harrison's 22E, p. 2369; Tintinalli's Emergency Medicine, p. 39
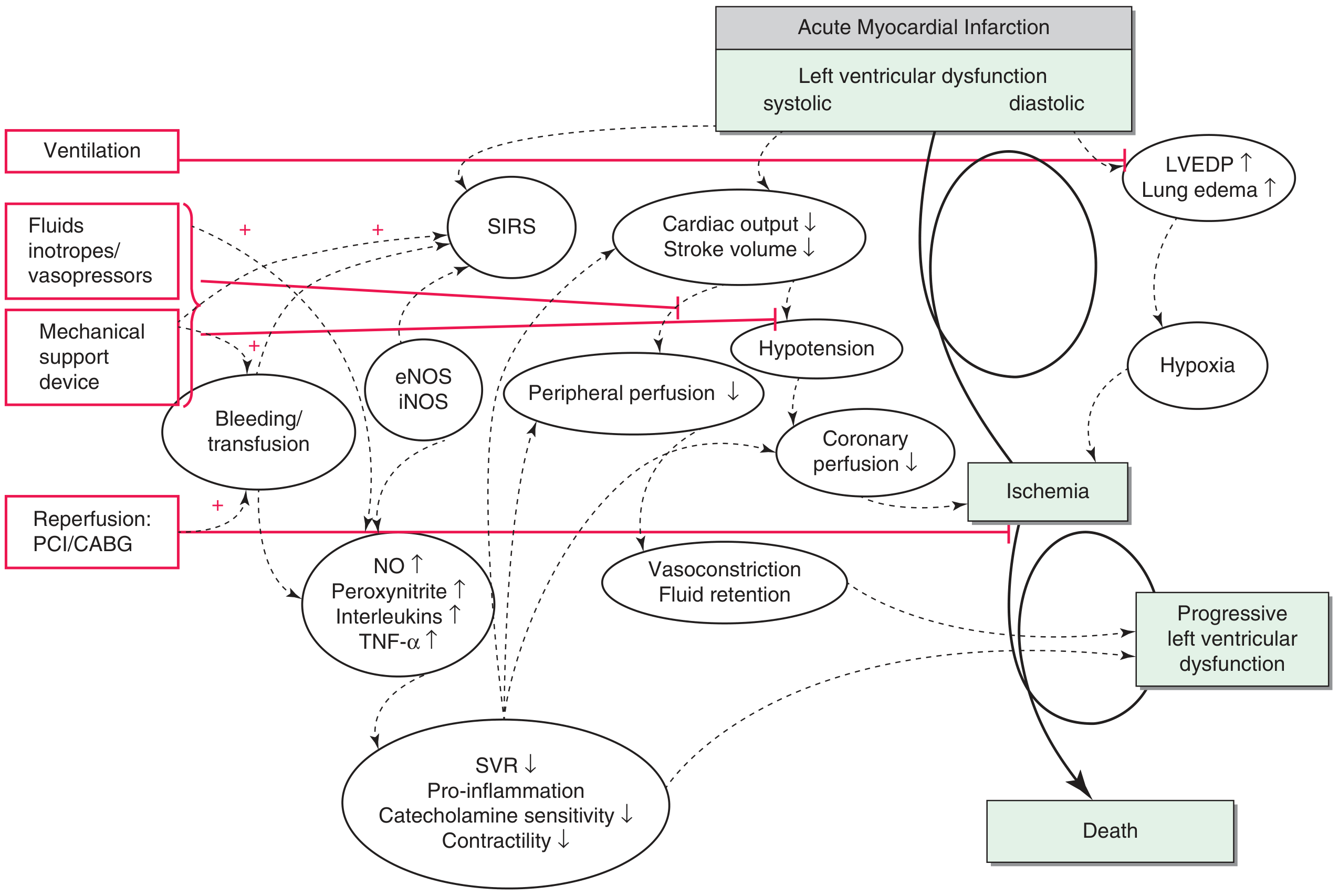
1.3 Pathophysiology
The pathophysiology of CS involves a self-perpetuating spiral of deterioration:
The Classic Spiral:
- Myocardial ischemia/necrosis → profound depression of myocardial contractility
- Reduced cardiac output (CO) → decreased systemic BP
- Decreased coronary perfusion pressure → worsening myocardial ischemia
- Further reduction of contractility → further reduction of CO (the "vicious cycle")
Expanded Pathophysiology (Modern Understanding):
In addition to the classic spiral, a systemic inflammatory response plays a critical role:
- Ischemic injury triggers release of cytokines (TNF-α, interleukins) and activation of inducible nitric oxide synthase (iNOS)
- Excessive nitric oxide (NO) and peroxynitrite cause myocardial depression and vasodilation — paradoxically, systemic vascular resistance (SVR) may be inappropriately low rather than elevated
- This creates a mixed picture: pump failure combined with a distributive component (SIRS)
Hemodynamic Consequences:
- ↓ Stroke volume → ↓ Cardiac Index (CI < 2.2 L/min/m²)
- Compensatory neurohormonal activation → tachycardia, vasoconstriction, fluid retention
- ↑ LV end-diastolic pressure (LVEDP) → pulmonary congestion / pulmonary edema
- ↑ Pulmonary capillary wedge pressure (PCWP > 18 mmHg)
- ↓ Mixed venous O₂ saturation (SvO₂) reflecting increased O₂ extraction
Mechanical Complications (e.g., acute MR, VSR) cause acute volume overload superimposed on myocardial dysfunction, accelerating hemodynamic deterioration.
— Harrison's 22E, p. 2368–2370
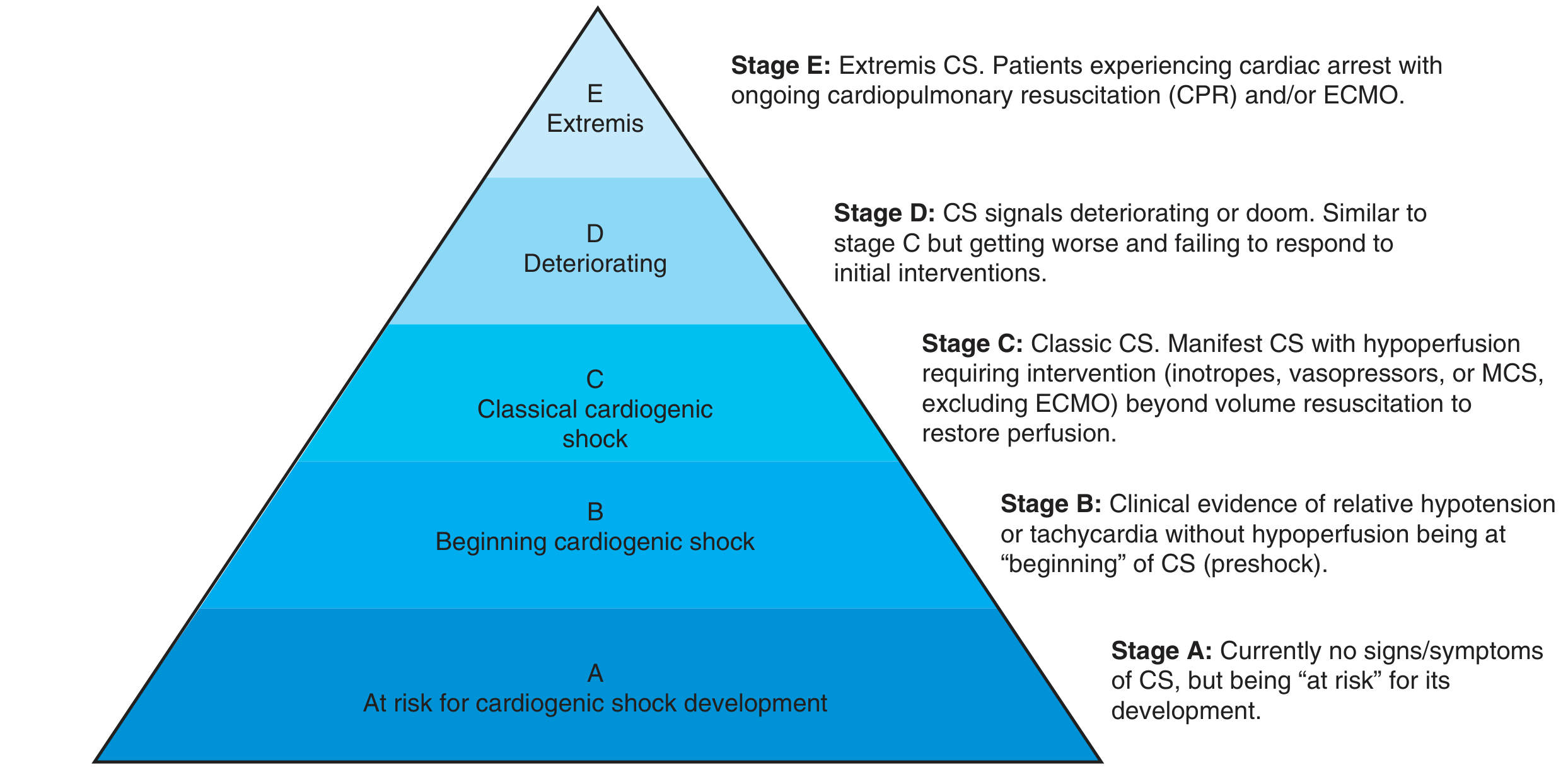
1.4 SCAI Classification (Severity Staging)
The Society for Cardiovascular Angiography and Interventions (SCAI) introduced a five-stage classification in 2019 (updated with validation studies):
| Stage | Name | Description |
|---|---|---|
| A | At Risk | No signs/symptoms of CS; risk factors present (e.g., large MI) |
| B | Beginning (Preshock) | Relative hypotension or tachycardia without hypoperfusion; requires early monitoring and intervention |
| C | Classic CS | Hypotension + hypoperfusion; requires vasopressors/inotropes |
| D | Deteriorating | Failure of initial CS therapy; escalating support required |
| E | Extremis | Cardiac arrest, ongoing CPR and/or ECMO; consideration of futility |
Stage E patients may need palliative care discussions. The updated model also incorporates a three-axis framework: shock severity + phenotype/etiology + risk modifiers (including cardiac arrest).
— Harrison's 22E, p. 2368
1.5 Clinical Signs
Symptoms:
- Dyspnea, orthopnea
- Chest pain (if underlying MI)
- Profound weakness, fatigue
- Anxiety, confusion, altered consciousness
Physical Examination Findings:
| System | Findings |
|---|---|
| General | Pale, diaphoretic, apprehensive |
| Mental status | Confusion, agitation, somnolence (cerebral hypoperfusion) |
| Cardiovascular | Weak, rapid pulse (occasionally severe bradycardia with heart block); weak apical impulse; soft S1; S3 gallop; systolic murmur (MR or VSR if mechanical complication) |
| Blood pressure | Systolic < 90 mmHg (or requiring vasopressors) |
| Jugular veins | JVD (elevated in LV failure and RV failure) |
| Lungs | Bibasal crackles (LV failure); tachypnea |
| Skin/extremities | Cold, clammy, mottled skin; prolonged capillary refill; peripheral cyanosis |
| Urine output | Oliguria or anuria (< 0.5 mL/kg/h) |
— Harrison's 22E, p. 2369; Tintinalli's, p. 39
1.6 Diagnostics
Electrocardiogram (ECG):
- ST-elevation or depression (ischemia/infarction)
- New LBBB, Q waves
- Arrhythmias (VT, AF, complete heart block)
Laboratory Findings:
| Test | Expected Finding |
|---|---|
| Arterial blood gas (ABG) | Metabolic acidosis; low PaO₂ |
| Lactate | > 2 mmol/L (SCAI criterion); > 8 mmol/L = Stage E |
| Creatinine / BUN | Elevated (renal hypoperfusion) |
| Hepatic transaminases | Elevated (~20% of patients; marker of high mortality) |
| Troponin / CK-MB | Elevated (myocardial necrosis) |
| BNP / NT-proBNP | Markedly elevated (LV dysfunction) |
| WBC | Elevated (inflammatory response) |
| CRP | Elevated |
| Blood glucose | Hyperglycemia common; hypoglycemia must be avoided |
Chest X-ray:
- Cardiomegaly
- Pulmonary vascular congestion / pulmonary edema (butterfly pattern)
- Pleural effusions
Echocardiography (ECHO):
- First-line imaging tool — identifies cause, severity, and type of CS
- Assesses: LV/RV function and size; wall motion abnormalities; pericardial effusion/tamponade; valvular pathology (MR, AI); VSR
- Distinguishes LV, RV, or biventricular failure
Invasive Hemodynamic Monitoring (Pulmonary Artery Catheter — PAC):
Used when diagnosis is uncertain or to guide therapy:
- Cardiac Index (CI): < 2.2 L/min/m² (< 1.8 L/min/m² in severe CS)
- PCWP: > 18 mmHg (elevated in LV failure)
- SVR: typically elevated (occasionally low due to SIRS)
- SvO₂: decreased
Coronary Angiography:
- Immediately indicated in all CS complicating MI for reperfusion assessment
- Also consider in resuscitated cardiac arrest survivors without ST-elevation (~70% have significant CAD)
— Harrison's 22E, p. 2369–2371
PART 2: SUDDEN CARDIAC DEATH
2.1 Definition
Sudden Cardiac Death (SCD) is defined as unexpected death from cardiac causes occurring either:
- Within 1 hour of symptom onset, or
- Within 24 hours of being last seen well (if unwitnessed)
If the victim is successfully resuscitated, the event is called Sudden Cardiac Arrest (SCA) — an important clinical distinction, as SCA is potentially survivable.
Approximately 325,000–450,000 individuals die from SCD annually in the United States; globally, 4–5 million deaths per year are attributed to SCD. The estimated overall out-of-hospital cardiac arrest survival rate is approximately 5.4% (EMS-treated cases ~10.8%).
— Robbins & Kumar Basic Pathology, p. 362; Tintinalli's Emergency Medicine, p. 93
2.2 Epidemiology
- SCD is most prevalent in individuals > 45–50 years old; 60% occur in males
- Circadian pattern: highest incidence in the first few hours after awakening from sleep (increased sympathetic activity)
- Most events occur in the home (~70%), though public cardiac arrests have better survival rates (higher likelihood of shockable rhythm, bystander CPR, and AED access)
- Socioeconomic gradient: SCD is 30–80% higher in the lowest vs. highest socioeconomic quartile
— Tintinalli's Emergency Medicine, p. 93
2.3 Etiology
Adults:
- Coronary artery disease (CAD): 65–80% of cases — the leading cause; SCD may be the first manifestation of IHD
- Cardiomyopathy (dilated, hypertrophic): 10–15%
- Other structural/electrical causes: 5–10%
Important autopsy findings in CAD-related SCD:
- Chronic severe atherosclerotic stenosis is found in most; acute plaque rupture in only 10–20% of cases
- 80–90% of resuscitated SCA victims show no enzymatic or ECG evidence of myocardial necrosis
- Healed remote MIs present in ~40%
- Subendocardial myocyte vacuolization (chronic severe ischemia) is common
Younger Patients (Non-atherosclerotic Causes):
- Hereditary channelopathies (Long QT syndrome, Brugada syndrome, CPVT)
- Hypertrophic cardiomyopathy (HCM) — most common cause of SCD in young athletes
- Dilated cardiomyopathy
- Congenital coronary artery abnormalities
- Myocarditis / sarcoidosis
- Mitral valve prolapse
- Pulmonary hypertension
- Pericardial tamponade, pulmonary embolism
- Drug-induced (cocaine, methamphetamine)
— Robbins & Kumar Basic Pathology, p. 362; Robbins, Cotran & Kumar Pathologic Basis of Disease; Tintinalli's, p. 93–94
2.4 Mechanisms and Pathophysiology
The mechanism of SCD is almost always a lethal arrhythmia:
Primary Mechanisms:
1. Ventricular Fibrillation (VF):
- Most common terminal arrhythmia in witnessed SCD (~70–80% of initial rhythms in public arrests)
- Triggered by electrical irritability of the myocardium (usually ischemia-mediated)
- Multiple re-entry circuits generated within the ventricular myocardium → ventricular tachycardia (VT) → degenerates to VF
- VF is present in only 23% of all cardiac arrests overall (many are unwitnessed and have degenerated to asystole by first EMS contact)
- Shockable rhythm: responds to defibrillation
2. Pulseless Ventricular Tachycardia (pVT):
- Organized ventricular rhythm at high rate without effective cardiac output
- Degenerates rapidly to VF
- Shockable rhythm
3. Asystole:
- Complete cessation of electrical activity
- Common in unwitnessed or prolonged arrests
- Non-shockable; prognosis very poor if unwitnessed
- Primary vs. secondary: primary = failure of conduction system; secondary = external factors (hypoxia, drugs)
4. Pulseless Electrical Activity (PEA):
- Organized electrical rhythm without detectable pulse
- Caused by profound reduction of CO due to myocardial depression or mechanical obstruction (tamponade, tension pneumothorax, PE, hypovolemia)
- Non-shockable; treatment targets reversible causes (the "Hs and Ts")
Electrophysiological Basis of Arrhythmia in SCD:
- Ischemic injury causes heterogeneity of conduction and refractory periods → substrate for re-entry
- Healed infarct scar provides anatomical substrate for re-entrant circuits
- Acute ischemia, catecholamine surges, electrolyte disturbances, and autonomic imbalance act as triggers
- In structurally normal hearts (channelopathies): mutations in ion channels (Na⁺, K⁺, Ca²⁺) cause aberrant depolarization/repolarization → spontaneous VF
— Robbins & Kumar Basic Pathology, p. 362; Tintinalli's Emergency Medicine, p. 94–97
2.5 Risk Factors / Predictors
| Risk Factor | Details |
|---|---|
| Reduced LV ejection fraction | EF ≤ 35% = best predictor; ICD indicated |
| Prior MI | Scar = re-entry substrate |
| CAD | 80% of SCD victims |
| Ventricular hypertrophy | Independent risk factor; increased mass in young athletes |
| Channelopathies | Long QT, Brugada, CPVT |
| Non-sustained VT on Holter | Marker of myocardial irritability |
| Family history of SCD | Channelopathies, HCM |
| Syncope of unexplained origin | May represent aborted SCA |
β-Blockers provide significant protection against SCD, particularly in post-MI patients with reduced ejection fraction.
2.6 Clinical Signs (Cardiac Arrest Presentation)
SCD by definition is fatal unless interrupted. The clinical presentation of cardiac arrest (SCA) includes:
- Sudden loss of consciousness (collapse)
- No pulse / no detectable blood pressure
- Apnea or agonal gasping
- Cyanosis developing rapidly
- Fixed, dilated pupils (if prolonged)
- ECG: VF, pVT, asystole, or PEA
Pre-arrest warning signs (in some cases):
- Chest pain, palpitations, dizziness, syncope
- However, in >50% of cases, SCD occurs without prior warning
2.7 Diagnostics
Diagnostics in the acute setting focus on rhythm identification and reversible cause detection:
ECG / Cardiac Monitor:
- Identify rhythm: VF, pVT, asystole, PEA
- Post-ROSC: look for ST elevation (STEMI → emergent cath), QTc prolongation, Brugada pattern, delta waves (WPW)
Point-of-Care Ultrasound (POCUS):
- Identifies tamponade, massive PE, severe wall motion abnormality, hypovolemia
- Differentiates true asystole from pseudo-PEA
Laboratory (post-ROSC):
- Troponin, CK-MB (myocardial necrosis)
- ABG (acidosis, hypoxia, hypercarbia)
- Electrolytes (K⁺, Mg²⁺, Ca²⁺ — reversible causes)
- Lactate (degree of hypoperfusion)
- Toxicology screen (drug-induced arrest)
- TSH (hypothyroidism)
Coronary Angiography:
- Immediately for post-arrest STEMI
- Considered in hemodynamically unstable post-arrest patients without ST-elevation
Electrophysiology Study (EPS):
- For risk stratification in survivors; identifies inducible arrhythmias
Implantable Loop Recorder / Holter Monitor:
- For unexplained syncope or suspected channelopathy
PART 3: CPR AND AED ALGORITHMS
3.1 Overview and the "Chain of Survival"
Effective resuscitation from SCA depends on a sequence of time-critical interventions — the Chain of Survival (AHA):
- Early recognition and activation of emergency medical services (EMS)
- Early high-quality CPR
- Rapid defibrillation (AED or manual defibrillator)
- Advanced cardiac life support (ACLS)
- Post-cardiac arrest care (integrated care system)
Survival from VF/pVT is inversely related to time between onset and defibrillation. Community strategies that combine early CPR + early AED deployment can double survival to hospital discharge compared to CPR-only approaches. — Tintinalli's, p. 98
3.2 Basic Life Support (BLS) — Adult CPR Algorithm
Upon finding an unresponsive adult:
Step 1 — Scene safety: Ensure the environment is safe.
Step 2 — Assess responsiveness: Tap shoulders, shout "Are you okay?"
Step 3 — Call for help: Activate EMS (call 911 or send someone); get an AED.
Step 4 — Check for breathing and pulse simultaneously (≤10 seconds):
- No normal breathing (absent or only gasping) AND no pulse → begin CPR
Step 5 — Begin CPR (C-A-B sequence):
- Compressions first (C before A-B per current AHA guidelines)
- Start 30 chest compressions before giving 2 breaths
Chest Compression Technique:
| Parameter | Standard |
|---|---|
| Rate | 100–120 compressions/minute |
| Depth | ≥ 2 inches (5 cm), ≤ 2.4 inches (6 cm) |
| Recoil | Allow full chest recoil after each compression |
| Interruptions | Minimize; pause < 10 seconds for rhythm check |
| Position | Heel of hands on lower half of sternum; arms straight |
| Compression fraction | > 60% of total CPR time |
Rescue Breaths (if trained):
- Ratio: 30:2 (30 compressions : 2 breaths)
- Each breath: 1 second; visible chest rise
- Tilt head – lift chin (jaw thrust if trauma suspected)
Compression-Only CPR is acceptable for untrained rescuers.
3.3 AED Algorithm
Automated External Defibrillator (AED) — use as soon as it arrives:
- Power ON the AED (open lid or press power button)
- Apply pads as directed (right clavicle/upper sternum; left lateral chest/apex)
- Analyze rhythm — ensure no one is touching the patient ("CLEAR!")
- If shock advised: deliver shock → immediately resume CPR (do not check pulse first)
- If no shock advised (PEA/asystole): resume CPR immediately; reanalyze every 2 minutes
- Continue until:
- Signs of life return (ROSC)
- ACLS providers take over
- Victim is declared deceased
Key AED Principle: Each minute of delay to defibrillation reduces survival by ~7–10%; prompt AED use is the single most important determinant of survival from witnessed VF arrest.
Public Access Defibrillation (PAD): Randomized trials demonstrate that laypersons trained with AEDs in public locations doubled survival compared to bystander CPR alone. — Tintinalli's, p. 98
3.4 ACLS Algorithms
Shockable Rhythms: VF / Pulseless VT
VF / pVT identified
↓
Shock (biphasic: 120–200 J; monophasic: 360 J)
↓
Immediately resume CPR × 2 minutes (no pulse check)
↓
Rhythm check
├── VF/pVT → 2nd Shock → CPR × 2 min
│ → IV/IO access → Epinephrine 1 mg IV q3–5 min
│ → 3rd Shock → CPR × 2 min
│ → Amiodarone 300 mg IV bolus (or Lidocaine 1–1.5 mg/kg)
│ → Continue: Shock → CPR × 2 min → Epinephrine → Amiodarone...
└── ROSC → Post-cardiac arrest care
Drug Therapy in Shockable Arrest:
- Epinephrine 1 mg IV/IO every 3–5 minutes (given after 2nd shock, then every other cycle) — improves coronary perfusion pressure; evidence for neurologically intact survival is limited but remains standard
- Amiodarone 300 mg IV bolus as first antiarrhythmic; second dose 150 mg if persistent VF/VT
- Lidocaine 1–1.5 mg/kg IV — alternative if amiodarone unavailable
Non-Shockable Rhythms: Asystole / PEA
Asystole / PEA identified
↓
CPR × 2 minutes (high-quality, continuous)
↓
IV/IO access → Epinephrine 1 mg IV q3–5 min (as soon as possible)
↓
Identify and treat REVERSIBLE CAUSES ("5 Hs and 5 Ts")
↓
Rhythm check every 2 minutes
├── Remains asystole/PEA → Continue CPR + Epi → Treat reversible causes
└── Shockable → Switch to VF/pVT algorithm
└── ROSC → Post-cardiac arrest care
Reversible Causes — The "5 Hs and 5 Ts":
| Hs | Ts |
|---|---|
| Hypovolemia | Tension pneumothorax |
| Hypoxia | Tamponade (cardiac) |
| Hydrogen ion (acidosis) | Toxins (drug overdose) |
| Hypo/Hyperkalemia | Thrombosis — pulmonary (PE) |
| Hypothermia | Thrombosis — coronary (MI/ACS) |
3.5 Post-Cardiac Arrest Care (Post-ROSC)
After Return of Spontaneous Circulation (ROSC):
- Airway: Secure advanced airway (endotracheal intubation); confirm placement
- Ventilation: Target SpO₂ 92–98%; avoid hyperoxia; target PaCO₂ 35–45 mmHg (avoid hyperventilation)
- Hemodynamics: Target MAP ≥ 65 mmHg; vasopressors (norepinephrine preferred) if hypotensive
- 12-lead ECG: Identify STEMI → emergent PCI
- Targeted Temperature Management (TTM): Maintain temperature 32–36°C for 24 hours in comatose survivors (neuroprotection)
- Coronary angiography: Emergent if STEMI; consider in unstable patients without ST elevation
- Treat precipitating cause: ACS, electrolyte disturbance, toxin, PE, etc.
- Neurological assessment: Glasgow Coma Scale, pupillary reflexes, CT brain if trauma/hemorrhage suspected
3.6 Special Considerations
Interposed Abdominal Compression CPR, Active Compression-Decompression CPR, Mechanical CPR devices (e.g., LUCAS, AutoPulse): can maintain adequate coronary perfusion pressure during prolonged resuscitation; used in select settings (cath lab CPR, prolonged transport).
ECMO-CPR (Extracorporeal CPR, eCPR): For highly selected refractory cardiac arrest patients — provides mechanical circulatory support to allow time for diagnosis and intervention (e.g., PCI for STEMI-related arrest). Survival to discharge rates of 30–50% reported in select centers.
Implantable Cardioverter-Defibrillator (ICD):
- Primary prevention: EF ≤ 35%, NYHA Class II–III on optimal medical therapy ≥ 3 months
- Secondary prevention: survivors of VF/VT arrest not from reversible cause
- ICDs "sense and electrically counteract episodes of ventricular fibrillation" — markedly improve prognosis in at-risk patients. — Robbins & Kumar Basic Pathology, p. 362
Summary Table
| Feature | Cardiogenic Shock | Sudden Cardiac Death |
|---|---|---|
| Definition | Low CO → end-organ hypoperfusion | Unexpected cardiac death within 1 h of symptoms |
| Leading cause | Acute MI (LV failure ~80%) | CAD (65–80%); channelopathies in young |
| Key mechanism | Myocardial contractility failure → ↓CO spiral | Lethal arrhythmia (VF, pVT, asystole, PEA) |
| Key signs | Hypotension, cold extremities, oliguria, S3 | Collapse, pulselessness, apnea |
| Key diagnostics | Echo, PAC, lactate, ABG, ECG, troponin | ECG rhythm, POCUS, post-ROSC labs |
| Mortality | 40–60% | ~90% out-of-hospital without prompt treatment |
| Key treatment | Reperfusion (PCI/CABG), vasopressors, MCS | Defibrillation, CPR, ACLS, ICD (secondary prevention) |
References:
- Harrison's Principles of Internal Medicine, 22nd Edition (2025) — Ch. 316
- Tintinalli's Emergency Medicine: A Comprehensive Study — Ch. 11, 12
- Robbins & Kumar Basic Pathology (10th Ed.) — Ch. 9
- Robbins, Cotran & Kumar Pathologic Basis of Disease — Ch. 9
This is a shared conversation. Sign in to Orris to start your own chat.