TB Meningitis
Reading File
Finding Sources
Finding Sources
Searching Images
tuberculous meningitis CSF brain pathology
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Here is a comprehensive overview of Tuberculous (TB) Meningitis:
Tuberculous Meningitis
Epidemiology & Risk Factors
TB meningitis incidence closely parallels systemic tuberculosis. In developed countries, prevalence decreased sharply post-WWII but rose again after 1985, largely due to HIV. Among patients with full-blown HIV, the incidence of tuberculosis is nearly 500 times that of the general population. TB may be the first clinical manifestation of HIV infection. In sub-Saharan Africa, TB is 25 times more frequent than in the United States. High-risk populations include patients with HIV, alcoholics, immigrants from sub-Saharan Africa, Asia, India, and the former Soviet Union.
— Adams and Victor's Principles of Neurology, 12th Edition
Pathogenesis
The causative organism is almost always Mycobacterium tuberculosis; rare causes include M. bovis, M. avium, M. kansasii, and M. fortuitum (the last especially after neurosurgery or trauma). Pathogenesis occurs in two stages:
- Hematogenous seeding of the meninges and subpial brain regions → formation of tubercles (granulomas)
- Rupture of one or more tubercles → discharge of bacteria into the subarachnoid space → inflammatory meningitis
This distinguishes TB meningitis from conventional bacterial meningitis, where hematogenous implantation directly into the meninges is more typical.
— Adams and Victor's Principles of Neurology, 12th Edition
Pathology
- Small white tubercles scattered over the base of the cerebral hemispheres (basal predominance is hallmark)
- A thick gelatinous exudate accumulates at the base, obliterating the pontine and interpeduncular cisterns, extending to the medulla, floor of the third ventricle, optic chiasm, and undersurfaces of the temporal lobes
- Microscopically: meningeal tubercles show central caseation surrounded by epithelioid cells, giant cells, lymphocytes, plasma cells, and connective tissue
- The process extends beyond the subarachnoid space to invade underlying brain — making it a true meningoencephalitis
- Complications arising from the exudate:
- Cranial nerve palsies (traversing nerves caught in exudate)
- Vasculitis and arterial occlusion → infarction
- Hydrocephalus (obstructive, from blockage of basal cisterns; up to 25% need neurosurgical shunting)
- Arachnoid fibrosis, especially at the base of the brain (Robbins Pathology)
Gadolinium-enhanced MRI showing enhancement of basal meninges, multiple abscesses, and hydrocephalus:
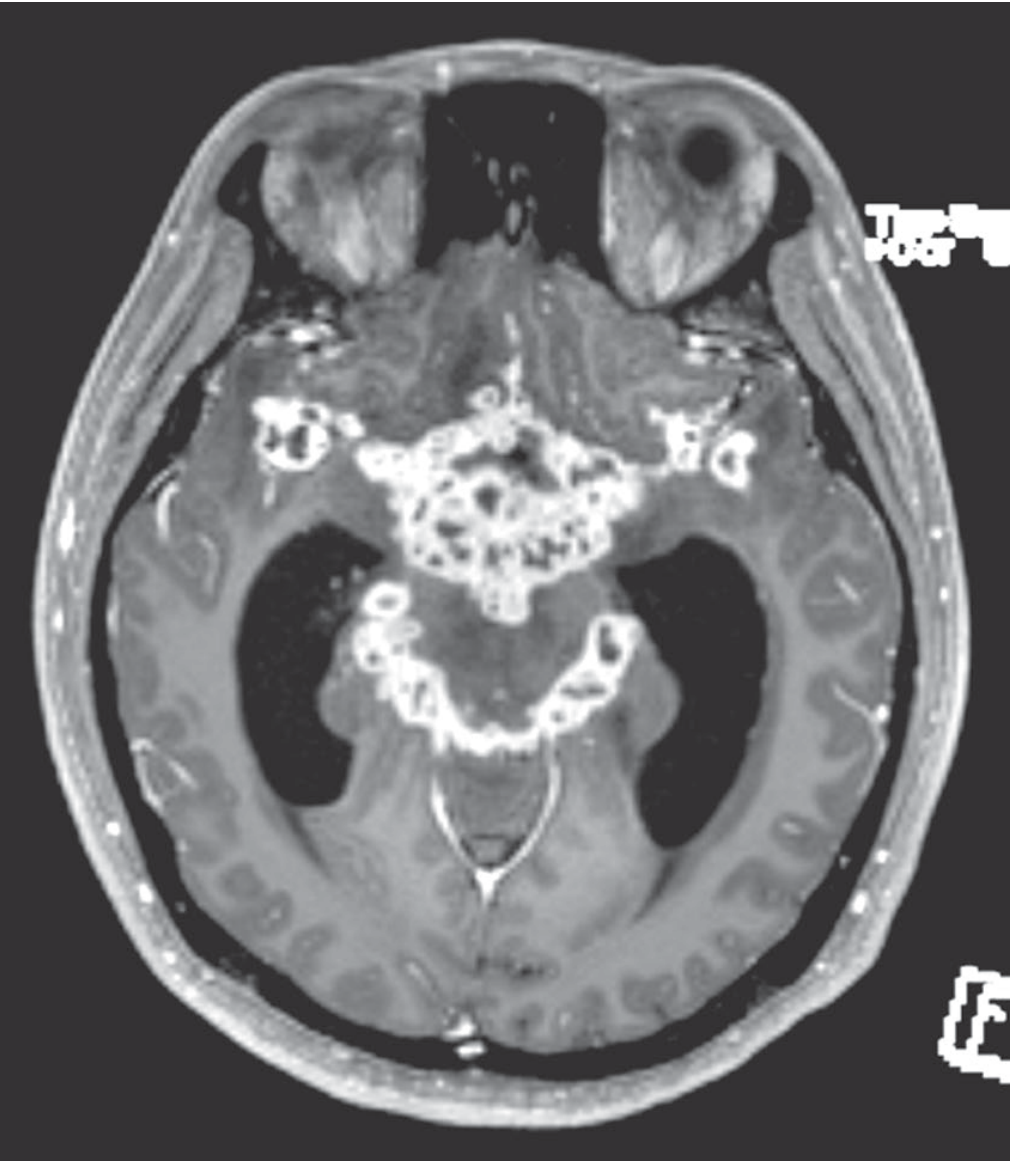
Figure: Gadolinium-enhanced MRI demonstrating intense basal meningeal enhancement and multiple small abscesses — the radiological hallmark of TB meningitis.
— Adams and Victor's Principles of Neurology, 12th Edition
Clinical Features
Onset is subacute — evolving over 1–2 weeks (sometimes longer), in contrast to acute bacterial meningitis. In children it can be even more insidious.
| Feature | Details |
|---|---|
| Fever | Low-grade; present |
| Headache | >50% of cases |
| Stiff neck / meningismus | ~75% of cases; may be absent in infants |
| Confusion / lethargy | Common |
| Cranial nerve palsies | 20% at presentation — oculomotor (III) most common, also facial (VII), deafness |
| Papilledema | Present in raised ICP |
| Seizures | More prominent in children |
| Focal deficits | From infarction (vasculitis) or focal tuberculoma |
| Hyponatremia | From SIADH or adrenal TB (Addisonian state) |
In ~2/3 of patients, active TB is found elsewhere — usually lungs, occasionally small bowel, bone, kidney, or ear.
If untreated — uniformly fatal. In late-presenting comatose patients, mortality approaches 50%.
— Adams and Victor's Principles of Neurology, 12th Edition; Tintinalli's Emergency Medicine
CSF Findings
| Parameter | Typical Finding |
|---|---|
| Opening pressure | Elevated |
| Appearance | Clear / xanthochromic / viscous |
| WBC | 50–500 cells/mm³; lymphocytic predominance (early: mixed PMN + lymphocytes) |
| Protein | 100–200 mg/dL (can be much higher with spinal block) |
| Glucose | Reduced (<40 mg/dL); CSF:serum glucose ratio <0.5 |
| Chloride | Low (historically noted) |
Diagnostic tests on CSF:
- Acid-fast smear (Ziehl-Neelsen): Sensitivity only 10–50%; increased by large volumes and multiple LPs with concentration techniques
- Culture: Gold standard but slow (3–4 weeks); rapid liquid culture <1 week with newer techniques
- PCR (nucleic acid amplification): Sensitivity ~80%; ~10% false-positive rate; multiplex PCR is more sensitive
- ADA (adenosine deaminase): Significantly elevated in TB meningitis vs. other meningitides
- Dot-ELISA (CSF antigens/antibodies): Positive in ~86% of suspected cases
- Interferon-gamma release assay (IGRA): ~90% specific for active TB infection when used in blood; also used in CSF
— Henry's Clinical Diagnosis and Management by Laboratory Methods; Adams and Victor's Principles of Neurology
Imaging
MRI is preferred:
- Basal meningeal enhancement on gadolinium T1 (classic finding)
- Hydrocephalus (communicating > obstructive)
- Tuberculomas — ring-enhancing lesions with central caseation
- Infarcts from vasculitis (basal ganglia, internal capsule)
- CT normal in ~30% of mild disease
Multi-sequence MRI showing the classic triad of TB meningitis — basal enhancement, hydrocephalus, and parenchymal signal changes:
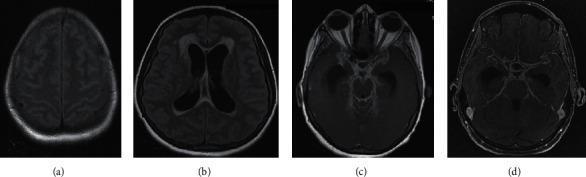
(a) FLAIR: subtle convexity signal; (b) FLAIR: hydrocephalus with periventricular edema; (c–d) Post-contrast T1: intense diffuse leptomeningeal enhancement at the basal cisterns.
Treatment
First-Line Regimen (4-drug induction)
| Drug | Adult Dose | Key Adverse Effects |
|---|---|---|
| Isoniazid (INH) | 5 mg/kg/day (max 300 mg) | Peripheral neuropathy, hepatitis |
| Rifampin (RMP) | 10 mg/kg/day (max 600 mg) | Hepatitis, drug interactions, orange secretions |
| Pyrazinamide (PZA) | 20–35 mg/kg/day | Hepatitis, hyperuricemia, rash |
| Ethambutol (EMB) | 15 mg/kg/day | Optic neuropathy (monitor visual acuity and color vision) |
Duration:
- Induction phase: All 4 drugs for 2 months
- Continuation phase: INH + RMP for remaining 7–10 months (total 9–12 months)
- Alternative regimen: INH + PZA + high-dose RMP + moxifloxacin
Key points:
- Give pyridoxine 50 mg/day with INH to prevent neuropathy (especially in alcoholics, pregnant women)
- All drugs penetrate the blood-brain barrier; INH and PZA have the best CNS penetration
- In multidrug-resistant (MDR-TB): add ethionamide (ETA) as 5th drug; doses 15–25 mg/kg/day in divided doses
- Drug-resistant organisms increasing — especially from certain geographic regions (high INH ± EMB resistance)
- Directly observed therapy (DOTS) for at least 2 months is now routine
Adjunctive Corticosteroids
Dexamethasone is recommended for all patients, particularly those with altered consciousness, raised ICP, or threatened subarachnoid block:
- 0.4 mg/kg/day IV for 1 week, then tapered over 3–6 weeks
- A randomized trial (Thwaites et al., Vietnam) showed dexamethasone reduced mortality from 41% to 32%, though without significant effect on residual disability
- Only used in conjunction with antituberculous drugs
Neurosurgical Management
- Ventriculoperitoneal shunting required in ~25% of patients for hydrocephalus
- Tuberculomas unresponsive to medical therapy may require surgical excision
- Pott disease (spinal TB) with cord compression: surgical decompression after initial chemotherapy
— Adams and Victor's Principles of Neurology, 12th Edition; ROSEN's Emergency Medicine; Tintinalli's Emergency Medicine
Prognosis & Sequelae
- Overall mortality: ~10% in treated patients; significantly higher in HIV patients (~21%)
- Comatose patients at diagnosis: mortality approaches 50%
- 20–30% of survivors have neurologic sequelae:
- Cognitive impairment / intellectual decline
- Recurrent seizures
- Visual and oculomotor disorders
- Deafness
- Hemiparesis
- Psychiatric disturbances
- Early diagnosis is the single most important determinant of favorable outcome
— Adams and Victor's Principles of Neurology, 12th Edition
Summary: TB Meningitis vs. Bacterial Meningitis
| Feature | TB Meningitis | Pyogenic (Bacterial) Meningitis |
|---|---|---|
| Onset | Subacute (days–weeks) | Acute (hours) |
| CSF WBC | 50–500 (lymphocytes) | 1000–10,000+ (neutrophils) |
| CSF glucose | Mildly-moderately low | Very low |
| CSF protein | 100–200 mg/dL | Elevated (may be lower) |
| Basal predominance | Yes (hallmark) | Less characteristic |
| Cranial nerve palsies | Common (20%) | Less common |
| Hydrocephalus | Common complication | Less frequent |
| Treatment duration | 9–12 months | 10–21 days |
Clinical features of TB Meningitis in detail
Finding Sources
Finding Sources
Searching Images
TB meningitis clinical signs cranial nerve palsy hydrocephalus
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
Here is a detailed account of the clinical features of TB Meningitis, synthesized from multiple authoritative sources:
Clinical Features of Tuberculous (TB) Meningitis
1. Overview: The Subacute Nature
TB meningitis is distinguished from other forms of meningitis by its subacute to chronic onset. Symptoms evolve over 1–4 weeks (average ~12 days; range 5–30 days) — far more insidiously than pyogenic bacterial meningitis (hours) but overlapping with cryptococcal meningitis.
"TB meningitis, in contrast to other types of meningitis, can have a subacute or even chronic presentation, with symptoms developing months after onset of infection." — ROSEN's Emergency Medicine
If left untreated, death follows in a median of 19 days from symptom onset. — Murray & Nadel's Textbook of Respiratory Medicine
2. Prodromal / Constitutional Phase (Days 1–14)
Before overt meningeal signs appear, patients typically pass through a non-specific prodrome, especially prominent in TB meningitis compared to other meningitides:
| Symptom | Detail |
|---|---|
| Low-grade fever | Persistent; rarely high-spiking |
| Malaise & fatigue | Generalised, progressive |
| Night sweats | Characteristic of TB |
| Weight loss & anorexia | Especially in prolonged cases |
| Headache | Present in >50% of cases — initially mild, becomes severe |
| Lethargy / apathy | Early behavioural change |
| Nausea & vomiting | Common early symptom |
| Irritability | Particularly in children |
In children and infants, apathy, hyperirritability, vomiting, and seizures are the dominant early symptoms. The prodrome in children can be prolonged and easily misattributed to viral illness. — Adams and Victor's Principles of Neurology
3. Meningeal Phase — Classic Signs
As the basal exudate accumulates and the subarachnoid space is inflamed, frank meningeal signs emerge:
Headache
- The most prevalent symptom (>50% of cases)
- Becomes severe, persistent, and progressive
- Reflects raised intracranial pressure (ICP) and meningeal irritation
Neck Stiffness (Nuchal Rigidity)
- Present in approximately 75% of cases
- Reflects irritation of spinal meninges
- May be absent in infants, the very elderly, and immunosuppressed patients — an important diagnostic pitfall
Kernig's Sign
- With the patient supine and hip flexed at 90°, extension of the knee is resisted due to pain/spasm
- Reflects meningeal and nerve root irritation
Brudzinski's Sign
- Passive flexion of the neck causes involuntary flexion of both knees
- Also reflects meningeal irritation
"Characteristically, these symptoms evolve much less rapidly in tuberculous than in bacterial meningitis, usually over a period of a week or two, sometimes longer." — Adams and Victor's Principles of Neurology
4. Cranial Nerve Involvement (Very Characteristic of TBM)
Cranial nerve palsies occur in approximately 20% of patients at the time of presentation — far more commonly than in other bacterial meningitides. This is because the thick, gelatinous basal exudate envelops and compresses cranial nerves as they traverse the subarachnoid space at the base of the brain.
| Cranial Nerve | Deficit | Mechanism |
|---|---|---|
| CN III (Oculomotor) | Ptosis, mydriasis, "down and out" eye | Most common; compression in interpeduncular cistern |
| CN IV (Trochlear) | Vertical diplopia | Less common |
| CN VI (Abducens) | Lateral gaze palsy (failure of abduction), diplopia | Non-localising sign of raised ICP; also direct compression |
| CN VII (Facial) | Facial weakness (peripheral pattern) | Basal exudate involvement |
| CN VIII (Vestibulocochlear) | Deafness, vertigo | Exudate along VIII nerve |
| CN II (Optic) | Visual loss, papilledema | From raised ICP or direct optic nerve inflammation |
CN VI (abducens) palsy — failure of lateral eye movement, a common cranial nerve sign in TB meningitis:

"Cranial nerves are often involved by the inflammatory exudate as they traverse the subarachnoid space, indeed, far more often than with typical bacterial meningitis." — Adams and Victor's Principles of Neurology
5. Raised Intracranial Pressure (ICP)
Raised ICP is a major clinical feature and results from:
- Communicating hydrocephalus — basal exudate blocks CSF resorption at the arachnoid villi
- Obstructive hydrocephalus — ependymitis blocking the aqueduct or 4th ventricle
- Cerebral oedema
- Mass effect from tuberculoma(s)
Clinical signs of raised ICP:
- Severe, progressive headache (worse in morning, on Valsalva)
- Papilledema (visible on fundoscopy)
- Vomiting (without nausea = "projectile")
- Cushing's triad in severe cases (hypertension, bradycardia, irregular respirations) — late and ominous
- Altered consciousness (see next section)
- CN VI palsy (false localising sign — traction on long course of nerve)
- Up to 25% of patients ultimately require ventriculoperitoneal shunting for hydrocephalus — ROSEN's Emergency Medicine; Tintinalli's
6. Altered Consciousness & Neuropsychiatric Features
Cognitive and consciousness changes are a hallmark of TB meningitis and reflect both meningoencephalitic invasion and raised ICP:
| Stage | Manifestation |
|---|---|
| Early | Mild confusion, inattention, personality change |
| Intermediate | Progressive disorientation, lethargy, memory disturbance |
| Late/Untreated | Stupor → coma |
- Psychiatric disturbances may be the presenting feature, occasionally leading to initial misdiagnosis as a psychiatric disorder
- Mental confusion is listed as a cardinal symptom in multiple textbooks alongside headache and vomiting
- In untreated disease, the course is: "confusion and progressively deepening stupor and coma, coupled with cranial nerve palsies, pupillary abnormalities, focal neurologic deficits, raised ICP, and decerebrate postures; usually fatal within 4–8 weeks" — Adams and Victor's Principles of Neurology
7. Focal Neurological Deficits (Vascular Complications)
A distinctive and serious complication is vasculitis of basal cerebral arteries, leading to arterial occlusion and infarction:
- The basal exudate envelops and invades vessel walls → obliterative endarteritis (granulomatous infiltration with marked intimal thickening)
- Vessels most commonly affected: perforating branches of the middle cerebral artery and circle of Willis → basal ganglia, internal capsule
- Aneurysm formation, thrombosis, and focal haemorrhagic infarction can result
Focal deficits arising from vascular involvement:
- Hemiplegia / hemiparesis
- Aphasia
- Hemianopia
- Basal ganglia syndromes (chorea, tremor, rigidity)
- Acute stroke-like onset — can be the initial presentation of unrecognised TB meningitis
"Tuberculous cerebral involvement is most marked at the base of the brain, and vasculitis of local arteries and veins may lead to aneurysm formation, thrombosis, and focal haemorrhagic infarction. The vessels to the basal ganglia are usually involved." — ROSEN's Emergency Medicine
FLAIR MRI showing characteristic basal cistern hyperintense exudates (yellow arrows) and leptomeningeal inflammation on the ventral brainstem (red arrows):
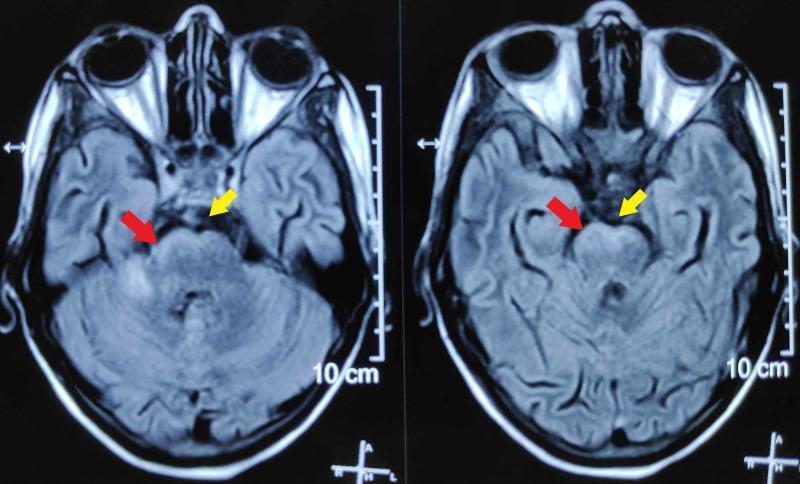
8. Seizures
- Can occur at any stage; more common in children
- May be focal or generalised
- Causes include:
- Cortical irritation from meningeal inflammation
- Cerebral infarction
- Hyponatraemia (a common metabolic complication — see below)
- Tuberculoma with cortical involvement
- Recurrent seizures are one of the significant long-term sequelae in survivors
9. Metabolic & Systemic Features
Hyponatraemia
- Common and clinically important
- Mechanisms:
- SIADH (Syndrome of Inappropriate ADH secretion) from hypothalamic involvement
- Addisonian state — if TB affects the adrenal glands
- Can precipitate or worsen seizures, confusion, and cerebral oedema
Hypothermia
- Occasionally reported at presentation (particularly in severe/hypothalamic disease)
10. Spinal Cord & Nerve Root Features
When the meningeal exudate extends to surround the spinal cord, a distinct syndrome can emerge (tuberculous myeloradiculitis):
- Radiculopathy — nerve root pain, sensory loss in dermatomal distribution
- Posterior column signs — impaired vibration and proprioception
- Lateral column signs — spasticity, upgoing plantars (myelopathy)
- Spinal block — when exudate obstructs CSF flow around the cord (protein can become extremely elevated)
- Cauda equina syndrome — if lumbosacral roots are involved
11. Features in Specific Populations
In Children and Infants:
- Prodrome is longer and more non-specific
- Neck stiffness may be absent — fontanelle bulging in infants
- Apathy, irritability, vomiting, seizures predominate
- Incidence up to 10% of all TB cases (vs ~1% in adults)
In HIV/Immunocompromised Patients:
- Course may be accelerated with more rapid deterioration
- Classic meningeal signs less reliable
- More frequent dissemination to extrapulmonary sites
- Higher mortality (~50% in treated HIV-positive patients with TBM)
- Less host inflammatory reaction pathologically
- May have atypical presentations with more focus on systemic symptoms
12. Concurrent Systemic TB
In approximately two-thirds of patients, evidence of active TB is found elsewhere:
- Lungs (most common) — often miliary pattern on CXR
- Lymph nodes
- Small bowel, bone, kidney, ear (less common)
- Sputum cultures positive in 40–50% of TBM patients — making sputum culture a useful adjunct to diagnosis
13. Clinical Staging (British Medical Research Council — MRC Grading)
Widely used to grade severity and predict outcome:
| Grade | Consciousness | Focal Signs/Deficits |
|---|---|---|
| Grade I | GCS 15; fully conscious | No focal deficit |
| Grade II | GCS 11–14 (confusion) | Minor focal signs (CN palsy) |
| Grade III | GCS ≤10 (stupor/coma) | Dense hemiplegia or paraplegia |
- Grade III carries mortality approaching 50%
- Grade at presentation is the strongest predictor of outcome
Summary: Clinical Feature Spectrum at a Glance
PRODROME (1–2 wk) MENINGEAL PHASE ADVANCED / UNTREATED
───────────────────── ────────────────────────── ───────────────────────────
Low-grade fever Severe headache Stupor → Coma
Malaise, night sweats Neck stiffness (75%) Decerebrate posturing
Weight loss Kernig/Brudzinski signs Fixed dilated pupils
Mild headache Cranial nerve palsies (20%) Hemiplegia
Lethargy / irritability Papilledema Death (4–8 weeks)
Nausea, vomiting Hydrocephalus signs
Focal deficits (vasculitis)
Seizures
Hyponatraemia
— Adams and Victor's Principles of Neurology, 12th Edition; Murray & Nadel's Textbook of Respiratory Medicine; ROSEN's Emergency Medicine; Robbins Pathologic Basis of Disease; Goldman-Cecil Medicine
Aortic Regurgitation clinical features, diagnosis and treatment guidelines in detail
Reading File
Finding Sources
Finding Sources
Searching Images
aortic regurgitation echocardiogram murmur clinical signs
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
I now have comprehensive data. Let me compile the full detailed answer.
Aortic Regurgitation (AR): Clinical Features, Diagnosis & Treatment Guidelines
1. Definition & Aetiology
Aortic regurgitation (AR) — also called aortic insufficiency — is caused by either primary leaflet disease or aortic root/ascending aortic disease that prevents proper coaptation of the valve cusps in diastole, allowing retrograde blood flow from the aorta into the left ventricle.
Primary Valve (Leaflet) Causes:
| Cause | Notes |
|---|---|
| Bicuspid aortic valve (BAV) | Most common congenital cause; incomplete leaflet closure/prolapse; presents age 20–40 |
| Infective endocarditis | Leaflet destruction, perforation, or vegetation preventing coaptation — most common cause of acute AR |
| Rheumatic heart disease | Commissural fusion + fibrotic retraction → central defect; often combined AS + AR; mitral involvement common |
| Calcific aortic valve disease | Usually age >60; mild AR accompanying predominant AS |
| Aortic dissection | Acute: dissection tears into annulus disrupting leaflet support |
| Chest trauma | Valve disruption or aortic root tear |
| Myxomatous valve disease | Progressive prolapse |
| Congenital | VSD, subaortic stenosis, unicuspid/quadricuspid variants |
| Anorectic drugs, dopamine/serotonin agonists | Implicated in leaflet dysfunction |
| Systemic lupus, RA, Jaccoud arthropathy | Inflammatory leaflet involvement |
Aortic Root / Ascending Aorta Causes (now more common than primary valve disease for isolated AR requiring surgery):
| Cause | Notes |
|---|---|
| Marfan syndrome | Cystic medial necrosis → annuloaortic ectasia |
| Age-related degenerative aortic dilation | Most common root cause overall |
| Systemic hypertension | Annuloaortic ectasia |
| Aortic dissection | Acute AR from root involvement |
| Syphilitic aortitis | Now rare |
| Ankylosing spondylitis / psoriatic arthritis | Inflammatory aortopathy |
| Behçet syndrome, giant cell arteritis, relapsing polychondritis | Inflammatory aortopathies |
| Osteogenesis imperfecta | Connective tissue fragility |
| Post-TAVI paravalvular leak | Increasingly common iatrogenic cause |
— Goldman-Cecil Medicine; Braunwald's Heart Disease
2. Pathophysiology
Chronic AR — Volume Overload & Adaptive Remodelling
In contrast to mitral regurgitation, AR ejects the regurgitant volume against the full systemic afterload, imposing both volume and pressure overload on the LV simultaneously.
Adaptive (compensated) phase:
- LV accommodates the volume overload via eccentric hypertrophy (Laplace's law: increased wall thickness offsets the increased radius → normalised wall stress)
- This produces the largest LV end-diastolic volumes seen in any form of heart disease — cor bovinum
- Increased total stroke volume maintains forward output
- LV compliance increases → filling pressures remain near normal despite massive dilation
- The afterload excess from systolic hypertension also induces concentric hypertrophy (unlike mitral regurgitation)
- Patients remain asymptomatic for many years
Decompensated phase:
- Interstitial fibrosis accumulates → compliance falls → LVEDP rises
- EF begins to fall; LVESV rises
- Forward cardiac output falls — first on exertion, then at rest
- Left atrial, pulmonary, and right heart pressures rise → pulmonary congestion
Myocardial ischaemia mechanism:
- ↑ LV mass → ↑ myocardial O₂ demand
- ↓ Aortic diastolic pressure (diastolic "runoff" into LV) + ↑ LVEDP → ↓ coronary perfusion pressure
- Reduced coronary flow reserve → angina without significant coronary artery disease
Acute AR — No Time for Compensation
- The unprepared, normal-sized LV suddenly receives a large volume overload
- LV cannot dilate acutely → LVEDP rises dramatically
- Mitral valve closes prematurely (mitral "preclosure") to protect the pulmonary circulation
- Forward stroke volume falls → cardiogenic shock
- Reflex peripheral vasoconstriction (unlike chronic AR, where resistance is reduced)
- Rapidly fatal without urgent intervention
— Braunwald's Heart Disease; Goldman-Cecil Medicine
3. Clinical Features
3A. Chronic AR — Symptoms
Symptoms usually appear in the fourth or fifth decade, only after considerable cardiomegaly and myocardial dysfunction.
| Symptom | Details |
|---|---|
| Exertional dyspnoea | Most common first symptom; reflects reduced cardiac reserve |
| Orthopnoea / PND | As LVEDP rises; pulmonary congestion |
| Angina pectoris | Later course; nocturnal; accompanied by flushing and diaphoresis (heart rate slows in sleep → lower diastolic pressure → lower coronary perfusion) |
| Palpitations | Awareness of forceful heartbeat, especially lying on left side |
| Head pounding | With exertion or tachycardia |
| Carotid artery pain | Uncomfortable pulsation in neck |
| Premature ventricular contractions | Particularly distressing due to post-extrasystolic heave of the volume-loaded LV |
"Patients with severe AR often complain of an uncomfortable awareness of the heartbeat, especially in supine position lying on the left side, and thoracic discomfort caused by pounding of the heart against the chest wall." — Braunwald's Heart Disease
Natural history (untreated symptomatic AR):
- Death usually within 4 years after onset of angina
- Death within 2 years after onset of heart failure
- 4-year survival in NYHA class III–IV: only ~30%
3B. Chronic AR — Physical Signs
Chronic severe AR generates a hyperdynamic, high-stroke-volume circulation producing a remarkable constellation of eponymous signs:
Peripheral Vascular Signs (from wide pulse pressure):
| Sign | Description |
|---|---|
| Corrigan pulse (Water-hammer pulse) | Abrupt sharp upstroke + rapid collapse of carotid/radial pulse; best felt with arm elevated |
| de Musset sign | Head bobbing with each heartbeat |
| Duroziez sign | Systolic + diastolic femoral bruits on gentle compression of femoral artery with stethoscope |
| Quincke pulse | Capillary pulsation in nail bed (systolic plethora / diastolic blanching with gentle pressure) |
| Hill sign | Femoral systolic BP ≥40 mmHg higher than brachial BP — most reliable sign of severe AR |
| Müller sign | Visible systolic pulsation of uvula |
| Traube sign | "Pistol shot" sound over femoral artery |
| Bisferiens pulse | Double-peaked pulse (best felt in brachial and femoral arteries) |
| Wide pulse pressure | SBP elevated; DBP very low (sometimes <40 mmHg) |
Precordial Examination:
| Finding | Significance |
|---|---|
| Displaced, hyperdynamic apex | Downward and lateral displacement due to LV enlargement; forceful, heaving quality |
| Rocking/apical heave | Characteristic motion of enlarged volume-loaded LV |
| Diastolic thrill at left sternal border | In severe AR |
Auscultation:
| Sound | Details |
|---|---|
| Early diastolic blowing murmur | High-pitched; heard best along left sternal border (3rd/4th ICS) with patient sitting forward, breath held in expiration; decrescendo pattern; radiates to aortic area |
| Mild AR: short early diastolic murmur only | Gradient highest at onset of diastole |
| Severe AR: holodiastolic murmur | Persists throughout diastole |
| Austin Flint murmur | Mid-diastolic / late diastolic low-pitched apical rumble; mimics mitral stenosis; created by regurgitant jet impinging on anterior mitral leaflet causing it to vibrate; present in severe AR with normal mitral valve |
| S3 gallop | Correlates with increased LVEDV; may signal impaired LV function |
| Systolic ejection murmur | Due to large forward stroke volume across aortic valve (not true stenosis) |
3C. Acute AR — Clinical Features
Acute AR (from endocarditis or dissection) presents very differently from chronic AR:
- Gravely ill appearance: tachycardia, severe peripheral vasoconstriction, cyanosis
- Pulmonary oedema / cardiogenic shock — the dominant presentation
- Subtle peripheral signs: Pulse pressure may be only mildly widened; the dramatic eponymous signs of chronic AR are absent (no time for compensatory changes)
- Soft or absent S1 — due to premature mitral valve closure (high LVEDP closes the mitral valve before systole; only tricuspid component of S1 heard)
- Short, low-pitched early diastolic murmur (shorter than chronic AR because LV/aortic pressures equalise quickly)
- Austin Flint murmur of brief duration
- Signs of the underlying cause (endocarditis vegetations, marfanoid habitus, aortic dissection features such as unequal pulses, tearing chest pain)
4. Diagnostic Workup
4A. ECG
- Non-specific but almost always shows left ventricular hypertrophy (LVH)
- Left axis deviation; voltage criteria for LVH (Sokolow-Lyon, Cornell)
- ST-T changes of LV strain in severe/chronic cases
4B. Chest X-Ray
- Cardiomegaly — enlarged cardiac silhouette (cor bovinum in severe chronic AR)
- Uncoiling and enlargement of the aortic root / ascending aorta
- Pulmonary venous congestion in decompensated AR or acute AR
4C. Echocardiography (Primary Diagnostic Tool)
Transthoracic Echo (TTE) is the cornerstone of evaluation:
Colour Doppler (PLAX view) in severe AR — turbulent mosaic retrograde jet filling the LVOT with vena contracta measurement of 7.0mm:
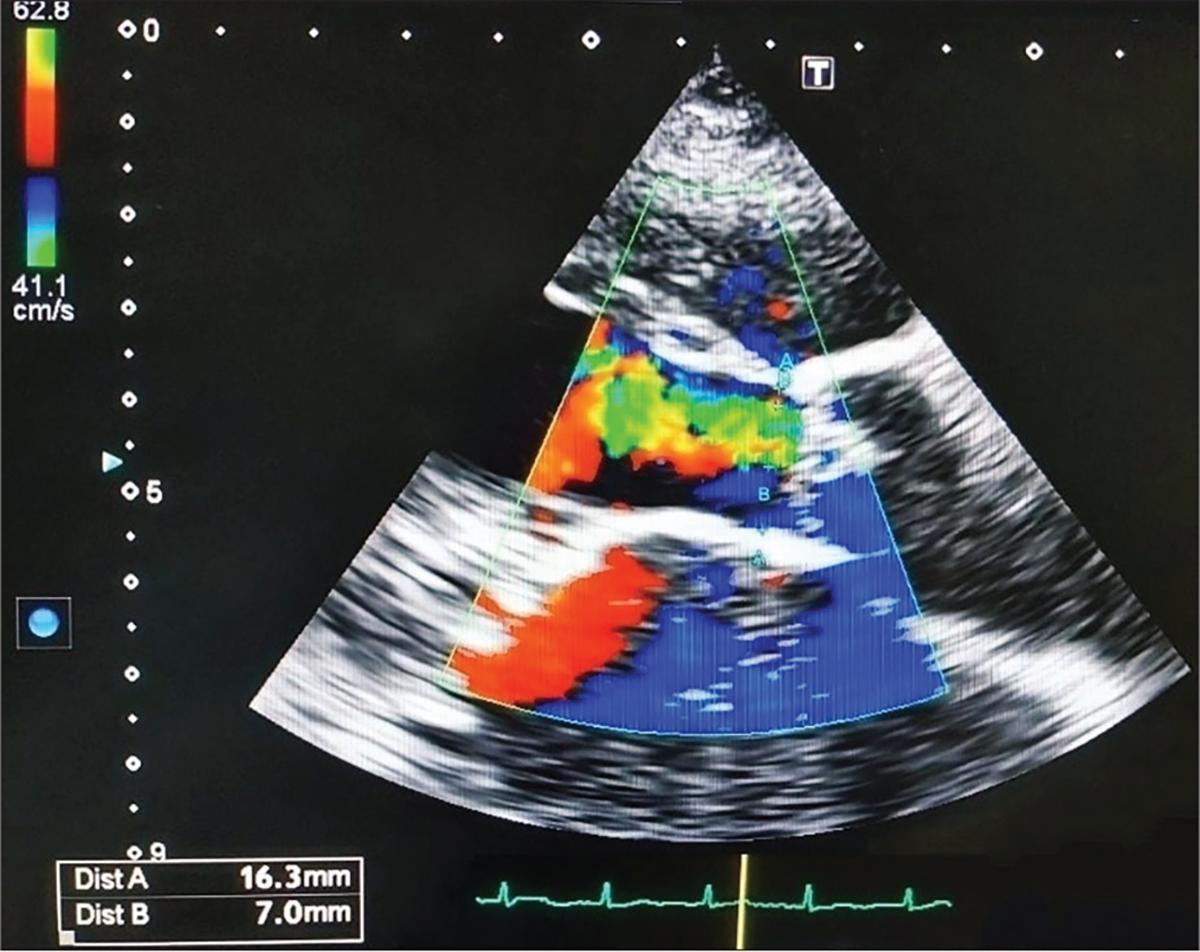
Echocardiographic assessment includes:
- Valve morphology: BAV, leaflet thickening, prolapse, flail, vegetation, perforation
- Aortic root size and shape: proximal ascending aorta may need CT/CMR if inadequate
- Colour Doppler: jet width in LVOT, jet area, vena contracta
- Continuous-wave Doppler: pressure half-time (PHT); steep deceleration slope = severe (acute) AR; diastolic half-time <200ms = severe
- Pulsed-wave Doppler: holodiastolic flow reversal in descending thoracic or abdominal aorta (abdominal reversal = severe AR)
- LV dimensions and function: LVEDD, LVESD, LVEF, LVESV — critical for surgical timing
- Quantitative measures: Regurgitant volume (RVol), Regurgitant fraction (RF), Effective Regurgitant Orifice Area (EROA)
Grading of AR Severity by Echocardiography (ACC/AHA Guidelines):
| Parameter | Mild | Moderate | Severe |
|---|---|---|---|
| Jet width (% of LVOT) | <25% | 25–64% | ≥65% |
| Vena contracta | <0.3 cm | 0.3–0.6 cm | >0.6 cm |
| Regurgitant volume | <30 mL/beat | 30–59 mL/beat | ≥60 mL/beat |
| Regurgitant fraction | <30% | 30–49% | ≥50% |
| EROA | <0.10 cm² | 0.10–0.29 cm² | ≥0.30 cm² |
| Holodiastolic aortic reversal | — | Partial | Holodiastolic (abdominal) |
Transesophageal Echo (TEE): provides better detail of aortic root, leaflet anatomy, vegetations, and is particularly useful pre-operatively.
3D Echocardiography / Contrast Echo: enhances accuracy of LV volume measurements.
4D. Cardiac MRI (CMR)
- Most accurate noninvasive technique for LVESV, LVEDV, and LV mass
- Accurate quantification of regurgitant volume and fraction from ascending aorta flow
- Preferred when echocardiographic evaluation is suboptimal
- Best for quantifying aortic root and ascending aortic dimensions (especially in BAV/Marfan)
4E. Cardiac Catheterisation / Angiography
- Rarely needed solely to diagnose AR; reserved for haemodynamic assessment when non-invasive data are discordant
- Aortography (contrast injection into aortic root at 55–60 mL, 20 mL/sec): graded I–IV
- Coronary angiography pre-operatively in patients with risk factors for CAD
5. Clinical Staging (ACC/AHA 2020 Guidelines)
| Stage | Definition | Key Features |
|---|---|---|
| A | At risk | BAV or other congenital anomaly; no significant AR |
| B | Progressive AR | Mild-to-moderate AR; normal LV size and function; asymptomatic |
| C1 | Asymptomatic severe AR | Severe AR; LVEF >55%; LVEDD ≤65 mm; no symptoms |
| C2 | Asymptomatic severe AR with LV dysfunction | Severe AR; LVEF ≤55% OR LVESD >50 mm (or indexed >25 mm/m²); no symptoms |
| D | Symptomatic severe AR | Severe AR + exertional dyspnoea, angina, or heart failure |
6. Treatment Guidelines (ACC/AHA 2020)
6A. Medical Management
Vasodilator therapy:
- ACE inhibitors / ARBs / dihydropyridine CCBs (e.g., nifedipine) — reduce afterload, potentially slow disease progression in patients with systemic hypertension
- When hypertension accompanies AR, treat according to standard hypertension guidelines
- Evidence for vasodilators in normotensive asymptomatic severe AR with normal LV function is less compelling and not routinely recommended as a strategy to delay surgery
- Beta-blockers: used cautiously (heart rate slowing increases regurgitant time per beat); may be useful in aortic root disease (Marfan/BAV) to reduce aortic wall stress
Endocarditis prophylaxis: No longer routinely recommended for isolated AR without prior endocarditis, except in highest-risk situations per current guidelines.
Serial monitoring (asymptomatic patients):
- Mild/moderate AR, normal LV: echocardiography every 1–2 years
- Asymptomatic severe AR, normal LV function: every 6–12 months
- Acute decompensation: urgent surgical evaluation
6B. Surgical Treatment — Indications (ACC/AHA 2020 Guidelines — Table 73.3, Braunwald's)
| Class | LOE | Recommendation |
|---|---|---|
| Class I (Indicated) | B-NR | 1. Symptomatic patients with severe AR (Stage D) — aortic valve surgery indicated regardless of LV systolic function |
| Class I | B-NR | 2. Asymptomatic severe AR with LV systolic dysfunction (LVEF ≤55%, Stage C2) — surgery indicated if no other cause for dysfunction |
| Class I | C-EO | 3. Severe AR (Stage C or D) undergoing cardiac surgery for other indications — concomitant aortic valve surgery indicated |
| Class IIa (Reasonable) | B-NR | 4. Asymptomatic severe AR with normal LVEF (>55%) but severely dilated LV: LVESD >50 mm or indexed LVESD >25 mm/m² (Stage C2) |
| Class IIa | C-EO | 5. Moderate AR undergoing cardiac/aortic surgery for other indications |
| Class IIb (May be considered) | B-NR | 6. Asymptomatic severe AR, normal LVEF (55–60% on serial studies), or progressive LV dilation (LVEDD >65 mm) in low surgical risk patients |
| Class III: Harm | B-NR | 7. Isolated severe AR candidates for SAVR should NOT undergo TAVI |
Key principle: Surgery should be performed before irreversible LV dysfunction develops. The LVEF threshold of 55% and LVESD thresholds are sentinel values because myocardial damage can occur even when EF appears preserved.
6C. Surgical Options
| Procedure | Indication |
|---|---|
| Surgical aortic valve replacement (SAVR) | Standard; mechanical vs. bioprosthetic based on age, anticoagulation preference |
| Aortic valve repair | Increasingly used for selected cases (leaflet prolapse, BAV with preserved leaflets) — avoids anticoagulation |
| Bentall procedure (composite root replacement — valve + aortic root) | When aortic root/ascending aorta is also dilated (Marfan, BAV with root dilation) |
| David procedure (valve-sparing root replacement) | For aortic root dilation with relatively normal valve leaflets |
| TAVI | Contraindicated for isolated severe AR as primary indication (Class III: Harm); may be considered in inoperable cases at very high surgical risk (off-label) |
6D. Acute Severe AR — Emergency Management
Acute AR is a surgical emergency:
- Stabilise haemodynamically: IV vasodilators (sodium nitroprusside) to reduce afterload and forward flow; avoid beta-blockers (tachycardia is compensatory)
- Intra-aortic balloon pump contraindicated (worsens regurgitation in diastole)
- Urgent surgical valve replacement — mortality approaches 75% with medical management alone in acute endocarditis/dissection-related AR
- Treat underlying cause (antibiotics for endocarditis; urgent aortic repair for dissection)
7. Prognosis
| Clinical State | Natural History |
|---|---|
| Asymptomatic mild/moderate AR | Excellent; low progression rate |
| Asymptomatic severe AR, preserved LV | Progression to symptoms or LV dysfunction ~4–6%/year |
| Symptomatic severe AR (untreated) | Death from angina ~4 years; from HF ~2 years |
| NYHA III–IV (untreated) | 4-year survival ~30% |
| After successful SAVR | Near-normal survival if operated before severe LV dysfunction |
| Post-op with LVEF <35% pre-op | Reduced benefit from surgery; irreversible myocardial damage |
"Data compiled in the presurgical era indicate that without surgical treatment, death usually occurred within 4 years after the development of angina pectoris and within 2 years after the onset of heart failure." — Braunwald's Heart Disease
8. Summary Algorithm
AORTIC REGURGITATION
│
┌────┴────┐
ACUTE CHRONIC
│ │
Emergency Stage A/B (mild-moderate)
Surgery → Serial echo, medical Rx BP
↑ │
Stabilise Stage C1 (asymptomatic severe, preserved LV)
Nitroprusside → Echo every 6-12 mo
NO IABP → Surgery if: LVEF decline to ≤55%
LVESD >50mm or indexed >25mm/m²
LVEDD >65mm (IIb)
│
Stage C2 (asymptomatic severe + LV dysfunction)
→ CLASS I SURGERY
│
Stage D (symptomatic severe)
→ CLASS I SURGERY (regardless of LVEF)
— Braunwald's Heart Disease; Goldman-Cecil Medicine; ACC/AHA 2020 Valvular Heart Disease Guidelines
This is a shared conversation. Sign in to Orris to start your own chat.