Teach me ketone bodies I am a nigerian 200lvl MBBS student
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Reading File
I now have comprehensive, high-quality content from authoritative sources. Let me compose the full teaching note.
🧪 Ketone Bodies — Complete 200L MBBS Guide
Sources: Basic Medical Biochemistry (6e), Lippincott Illustrated Reviews: Biochemistry (8e), Harper's Illustrated Biochemistry (32e)
1. What Are Ketone Bodies?
Ketone bodies are water-soluble, lipid-derived fuels produced by the liver from acetyl-CoA when carbohydrate availability is low (fasting, starvation, diabetes). There are three of them:
| Ketone Body | Also Called | Notes |
|---|---|---|
| Acetoacetate | Acetoacetic acid | Primary product; the "parent" ketone body |
| β-Hydroxybutyrate | 3-Hydroxybutyrate | Most abundant in blood (≈75%); technically not a "ketone" — it's a hydroxy acid |
| Acetone | — | Volatile; expired in breath; not metabolically useful |
Exam tip: β-Hydroxybutyrate is NOT a true ketone (no carbonyl group), but it is clinically grouped with the others.
2. When Are Ketone Bodies Made? (Physiological Triggers)
Ketogenesis is triggered whenever there is ↑ fatty acid oxidation + ↓ glucose availability:
- Starvation/prolonged fasting — liver glycogen depleted, insulin falls
- Uncontrolled Type 1 Diabetes Mellitus — absolute insulin deficiency
- High-fat, low-carbohydrate (ketogenic) diet
- Prolonged intense exercise
- Excessive alcohol intake (generates excess NADH, blocks gluconeogenesis)
Hormonal signal: ↓ Insulin + ↑ Glucagon/Epinephrine → ↑ Lipolysis → ↑ Free fatty acids → ↑ Hepatic ketogenesis
3. Ketogenesis — Step-by-Step Synthesis (in Liver Mitochondria)
Location: Mitochondrial matrix of hepatocytes (liver cells only)
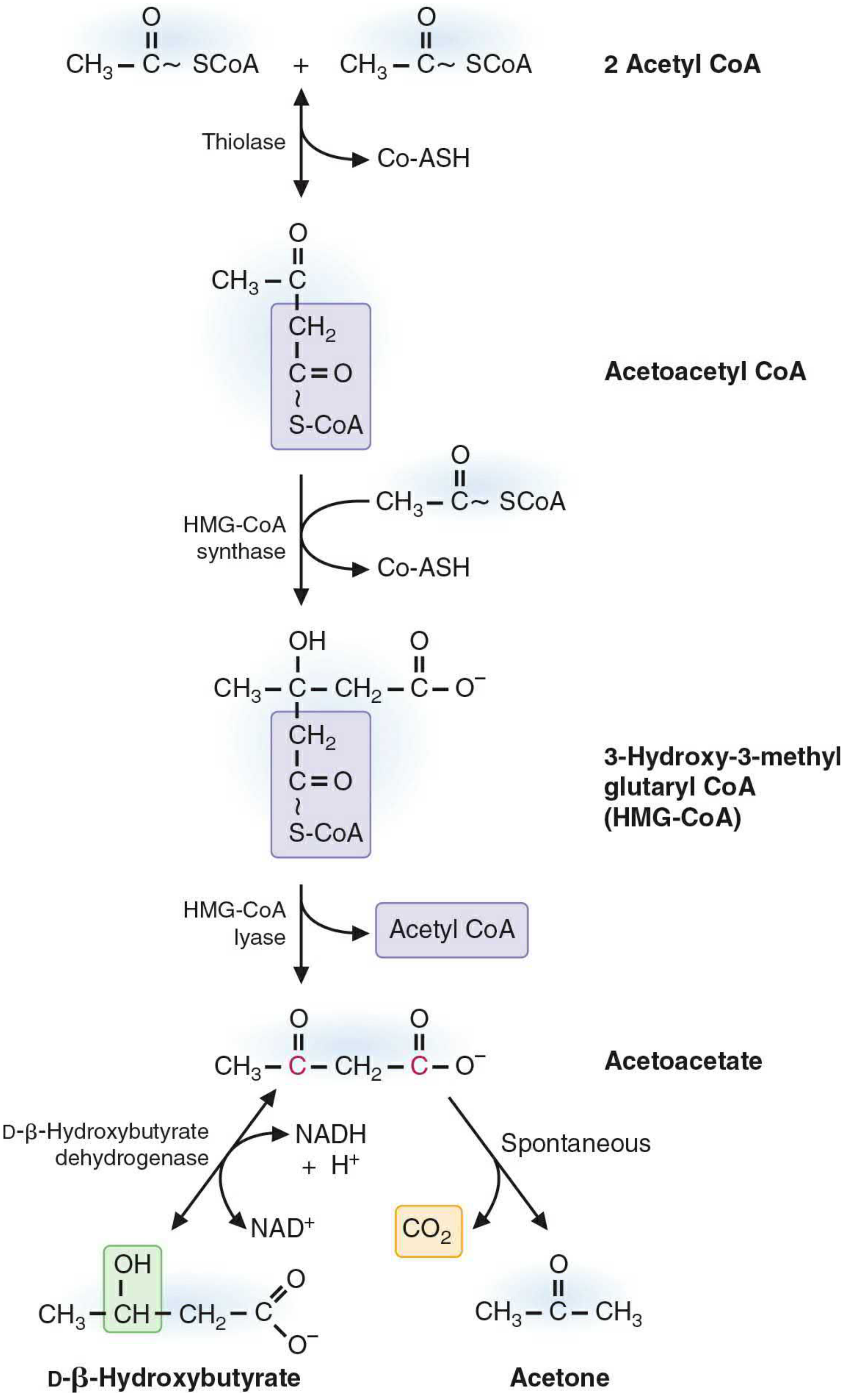
The pathway (4 steps):
Step 1 — Thiolase (reversible)
2 Acetyl-CoA → Acetoacetyl-CoA + CoA-SH
(This is the same thiolase as β-oxidation, running in reverse. High acetyl-CoA drives it forward here.)
Step 2 — HMG-CoA Synthase (rate-limiting step)
Acetoacetyl-CoA + Acetyl-CoA → HMG-CoA + CoA-SH
HMG-CoA = 3-Hydroxy-3-Methylglutaryl-CoA. This enzyme is the committed, regulated step.
Step 3 — HMG-CoA Lyase
HMG-CoA → Acetoacetate + Acetyl-CoA
Acetoacetate is the first free ketone body.
Step 4 — Two fates of Acetoacetate:
| Reaction | Product | Enzyme/Type | Notes |
|---|---|---|---|
| Reduction (NADH → NAD⁺) | β-Hydroxybutyrate | β-Hydroxybutyrate dehydrogenase | Main blood form; ratio β-HB:AcAc ≈ 3:1 normally |
| Spontaneous decarboxylation | Acetone + CO₂ | Non-enzymatic | Exhaled; causes "fruity breath" |
Key memory trick: "The HMG-CoA in ketogenesis is mitochondrial; the HMG-CoA in cholesterol synthesis is cytosolic. Same compound, different compartment, different enzyme sets."
4. Ketolysis — How Peripheral Tissues Use Ketone Bodies
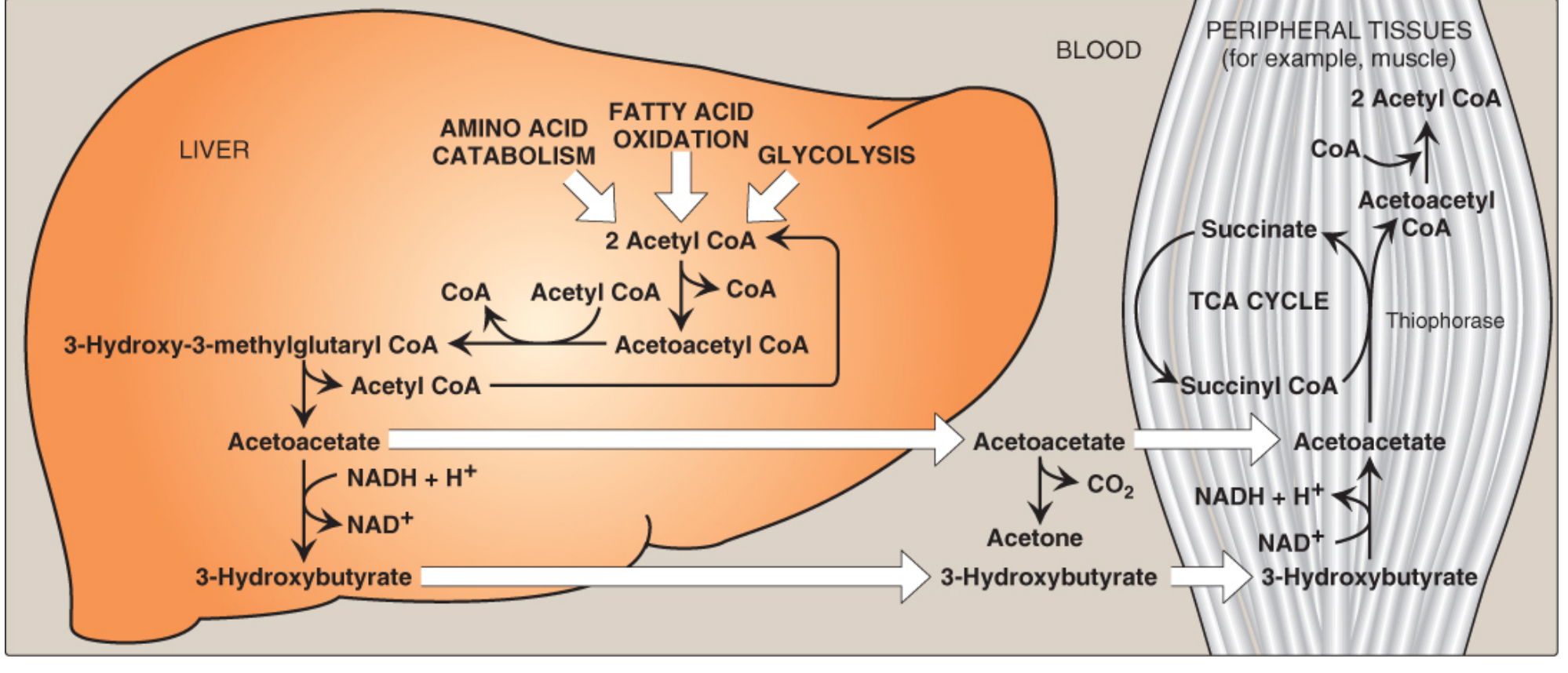
Which tissues use ketone bodies?
- ✅ Skeletal muscle, cardiac muscle
- ✅ Brain (especially during prolonged fasting — up to 75% of brain's fuel after 3+ days)
- ✅ Kidney cortex, intestinal mucosa
- ❌ Liver — cannot use ketone bodies (lacks thiophorase)
- ❌ Red blood cells — no mitochondria
The Ketolysis Pathway (in peripheral tissue mitochondria):
- β-Hydroxybutyrate → Acetoacetate (by β-hydroxybutyrate dehydrogenase; generates NADH)
- Acetoacetate + Succinyl-CoA → Acetoacetyl-CoA + Succinate
- Enzyme: Succinyl-CoA:acetoacetate CoA transferase (Thiophorase)
- This enzyme is absent in liver → liver cannot use its own ketone bodies
- Acetoacetyl-CoA → 2 × Acetyl-CoA (by thiolase)
- Acetyl-CoA enters the TCA cycle → ATP generation
Why does the liver lack thiophorase? Evolutionary advantage: the liver is the ketone body EXPORTER, not a consumer. It spares ketones for the brain and muscle.
Energy yield:
- 1 mol acetoacetate → ~19 ATP (costs 1 GTP equivalent for thiophorase reaction)
- 1 mol β-hydroxybutyrate → ~21.5 ATP (extra NADH from dehydrogenase step)
5. Regulation of Ketogenesis
Three main control points:
| Level | Mechanism |
|---|---|
| Hormone | ↓ Insulin + ↑ Glucagon → activates HSL in adipocytes → ↑ free fatty acid supply to liver |
| Malonyl-CoA | Insulin → ↑ malonyl-CoA → inhibits CPT-1 (blocks fatty acid entry into mitochondria → blocks β-oxidation → blocks ketogenesis); fasting does the opposite |
| HMG-CoA synthase | Succinylation (inactivation) when acetyl-CoA supply is low; activated during fasting |
Key concept: Malonyl-CoA is the metabolic switch. High carbohydrate → high malonyl-CoA → CPT-1 inhibited → no fatty acid oxidation → no ketogenesis. Fasting/insulin deficiency → low malonyl-CoA → CPT-1 active → fatty acids flood the mitochondria → ketogenesis ON.
6. Diabetic Ketoacidosis (DKA) — The Critical Clinical Application
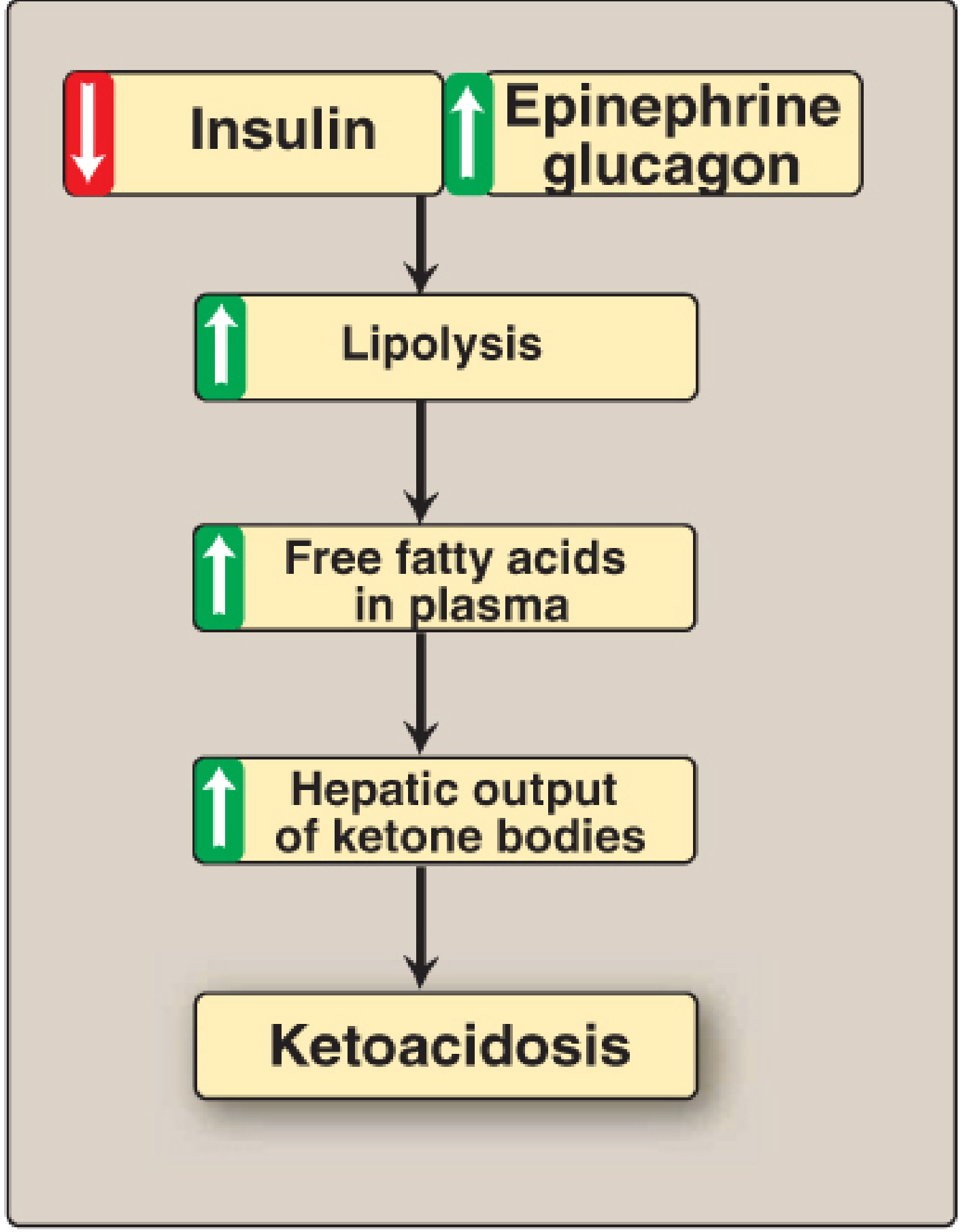
Trigger: Uncontrolled Type 1 Diabetes (absolute insulin deficiency)
Cascade:
↓ Insulin + ↑ Glucagon/Epinephrine → ↑ Lipolysis (adipose) → ↑ Free fatty acids in plasma → ↑ Hepatic ketogenesis → Ketone body accumulation in blood
What causes the acidosis?
- Each ketone body has a carboxyl group with pKa ≈ 4
- It dissociates in blood: AcAc⁻ + H⁺ → pH ↓
- Compounded by dehydration (osmotic diuresis from glycosuria + ketonuria → concentrated H⁺ in smaller plasma volume)
Blood values in DKA:
- Blood ketones: up to 90 mg/dL (normal: < 3 mg/dL)
- Urine ketones: up to 5,000 mg/24 hrs
- Arterial pH: < 7.3 (acidosis)
- Anion gap: elevated
Clinical signs:
- Kussmaul breathing (deep rapid respirations to blow off CO₂)
- Fruity/acetone breath (exhaled acetone)
- Nausea, vomiting, abdominal pain
- Polyuria, polydipsia, dehydration
- Altered consciousness (severe cases)
Diagnosis: Hyperglycemia + ketonuria (2+) + pH < 7.3
7. Other Causes of Ketoacidosis
| Condition | Mechanism |
|---|---|
| Prolonged fasting/starvation | ↓ Insulin, depleted glycogen |
| Alcoholic ketoacidosis | Excess NADH → blocks gluconeogenesis + TCA; acetyl-CoA diverted to ketogenesis |
| Ketogenic diet | Deliberate carbohydrate restriction |
8. Physiological Importance Beyond Starvation
-
Brain fuel during starvation: After 3+ days of fasting, the brain adapts to use ketone bodies as its primary fuel (up to 75%), reducing its glucose dependency and sparing muscle protein from being used for gluconeogenesis.
-
Ketogenic diet in epilepsy: A 3:1 lipid:carbohydrate diet reduces seizure frequency in children with refractory epilepsy — mechanism not fully known but may relate to altered brain excitability.
-
Pyruvate dehydrogenase deficiency: Ketone bodies provide alternative acetyl-CoA for energy and for acetylcholine synthesis in neurons when PDH is absent.
-
Ketogenic amino acids: Leucine, lysine, tryptophan, phenylalanine, tyrosine, and isoleucine are catabolized to acetyl-CoA/acetoacetyl-CoA → can feed ketogenesis. "Leu Lys Try Phe Tyr Ile" — these are purely or partly ketogenic.
9. "The Liver Paradox" — High-Yield Exam Point
| Feature | Liver | Peripheral Tissues |
|---|---|---|
| Synthesizes ketone bodies | ✅ YES | ❌ No |
| Uses ketone bodies | ❌ No (lacks thiophorase) | ✅ YES |
| Why? | Exports fuel for others | Consumer of exported fuel |
10. Quick Summary Table
| Feature | Detail |
|---|---|
| Site of synthesis | Liver mitochondria |
| Substrate | Acetyl-CoA (from β-oxidation of fatty acids) |
| Rate-limiting enzyme | HMG-CoA synthase (mitochondrial) |
| Three ketone bodies | Acetoacetate, β-Hydroxybutyrate, Acetone |
| Most abundant in blood | β-Hydroxybutyrate |
| Key enzyme for use | Thiophorase (absent in liver) |
| Normal blood level | < 3 mg/dL |
| Normal β-HB:AcAc ratio | ≈ 3:1 |
| DKA blood level | Up to 90 mg/dL |
| Tissues that CANNOT use | Liver, RBCs |
| Fruity breath | Acetone (exhaled) |
Key Mnemonics for the Nigerian Student
- "Hungry livers Make Huge Amounts" = HMG-CoA synthase is the hungry liver's key enzyme
- "The Liver Gives, Never Takes" = liver makes but cannot use ketone bodies
- "DKA: No Insulin = No off-switch" = insulin deficiency → malonyl-CoA falls → CPT-1 opens → FA floods in → ketosis goes unchecked
These concepts appear in MBBS Part 1 Biochemistry exams, and the clinical aspects (DKA) appear repeatedly in Internal Medicine, Endocrinology, and Emergency Medicine in later years. Nail this now and you'll always have the foundation.
— Basic Medical Biochemistry 6e, pp. 1097–1100 | Lippincott Illustrated Reviews: Biochemistry 8e, pp. 555–559
Does beta-oxidation occur in the liver?
Yes, beta-oxidation absolutely occurs in the liver — and it is actually the source of the acetyl-CoA used for ketogenesis, which we just covered.
Here is the full picture:
Beta-Oxidation in the Liver
Site: Mitochondrial matrix of hepatocytes (same as all other tissues)
The liver is one of the major sites of fatty acid oxidation. When fatty acids arrive at the liver (from adipose tissue during fasting/starvation), they undergo:
- Activation → Fatty acyl-CoA (in cytosol)
- Transport into mitochondria via CPT-1 / CPT-2 system (carnitine shuttle)
- Beta-oxidation → produces Acetyl-CoA + NADH + FADH₂
So What Happens to the Acetyl-CoA in the Liver?
This is the key distinction:
| Condition | Fate of Acetyl-CoA in Liver |
|---|---|
| Fed state | Enters TCA cycle → ATP; or used for fatty acid/cholesterol synthesis |
| Fasting/starvation | TCA cycle is saturated (oxaloacetate is pulled into gluconeogenesis) → acetyl-CoA diverted to ketogenesis |
This is exactly why fasting produces ketone bodies: beta-oxidation runs fast in the liver, but the TCA cycle cannot keep up because oxaloacetate (OAA) is being used for gluconeogenesis. With no OAA to condense with, acetyl-CoA has nowhere to go except into the ketone body pathway.
The "OAA Bottleneck" — Most Important Concept Here
Fatty acids → β-oxidation → Acetyl-CoA
↓
OAA needed to enter TCA
(OAA + Acetyl-CoA → Citrate)
↓
BUT in fasting: OAA → Gluconeogenesis
↓
Acetyl-CoA CANNOT enter TCA
↓
→ Ketogenesis instead
This is why the liver both performs beta-oxidation AND makes ketone bodies at the same time during fasting — they are sequential events, not alternatives.
Why the Liver Is Special Compared to Muscle
| Feature | Liver | Skeletal Muscle |
|---|---|---|
| Beta-oxidation | ✅ Yes | ✅ Yes |
| Ketone body synthesis | ✅ Yes (has HMG-CoA synthase) | ❌ No |
| Ketone body use | ❌ No (lacks thiophorase) | ✅ Yes |
| Gluconeogenesis | ✅ Yes (pulls OAA away from TCA) | ❌ No |
Bottom line: Beta-oxidation occurs in the liver, generates large amounts of acetyl-CoA during fasting, and because the TCA cycle is diverted toward gluconeogenesis, that acetyl-CoA is channelled into ketone body synthesis. The liver is the only organ that does all this — oxidise fatty acids and export the products as ketone bodies for the rest of the body to use.
This is a shared conversation. Sign in to Orris to start your own chat.