I am a medical student of 200lvl Nigeria teach me beta oxidation
Reading File
Finding Sources
Finding Sources
Searching Images
beta oxidation of fatty acids pathway diagram
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have everything I need for a comprehensive, well-illustrated lecture. Here is the full teaching on beta oxidation:
🔬 Beta Oxidation of Fatty Acids
A Complete Lecture for 200-Level Medical Students
1. What Is Beta Oxidation?
Beta (β) oxidation is the major pathway by which the body breaks down fatty acids to generate energy. The name comes from the fact that oxidation occurs at the β-carbon (carbon 3) of the fatty acyl chain. The end products — acetyl-CoA, NADH, and FADH₂ — feed directly into the TCA cycle and the electron transport chain to produce large amounts of ATP.
Key point: Beta oxidation is the reverse process of fatty acid synthesis — but it takes place in a completely different compartment (mitochondrial matrix vs. cytosol) and uses different enzymes.
2. Where Does It Occur?
- Primarily: Mitochondrial matrix
- Also: Peroxisomes (for very-long-chain fatty acids, C>22)
- Key tissues: Liver, heart muscle, skeletal muscle — especially during fasting, starvation, and prolonged exercise
3. Step 0: Activation of the Fatty Acid (Before Entry)
Before any oxidation can begin, the fatty acid must be activated in the cytosol to form acyl-CoA. This is the only step that requires ATP.
Reaction:
Fatty acid + CoA + ATP → Acyl-CoA + AMP + PPi
Enzyme: Acyl-CoA synthetase (thiokinase)
Location: Outer mitochondrial membrane / ER / peroxisomes
The PPi (pyrophosphate) is immediately hydrolyzed by pyrophosphatase, which drives the reaction to completion. This costs the equivalent of 2 ATP (ATP → AMP uses 2 high-energy phosphate bonds).
4. The Carnitine Shuttle — Entry into the Mitochondria
The inner mitochondrial membrane is impermeable to CoA and long-chain acyl-CoA. So long-chain fatty acids (>C12) must be carried across as acylcarnitine using the carnitine shuttle.
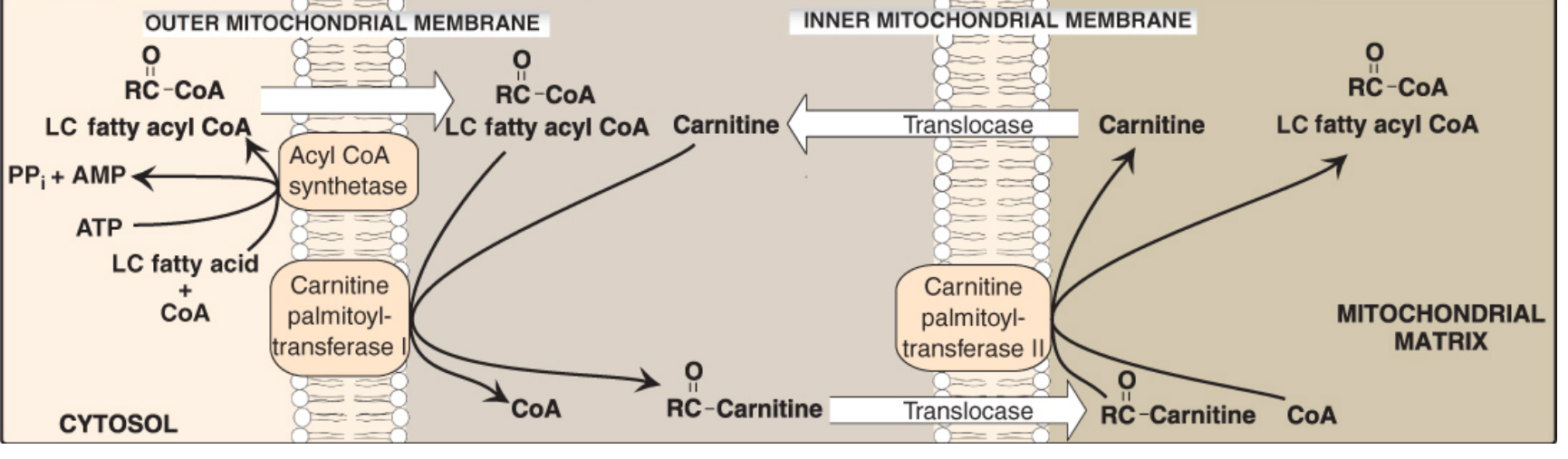
Three steps:
| Step | Enzyme | Action |
|---|---|---|
| 1 | CPT-I (outer membrane) | Transfers acyl group from CoA → carnitine to form acylcarnitine |
| 2 | Carnitine-acylcarnitine translocase | Moves acylcarnitine INTO the matrix (exchanges with free carnitine going out) |
| 3 | CPT-II (inner membrane, matrix side) | Transfers acyl group from carnitine → CoA, regenerating acyl-CoA inside matrix |
⚠️ Short- and medium-chain fatty acids (≤C12) can cross freely without carnitine and are activated by matrix enzymes directly.
Clinical note — CPT-I deficiency: Inability to oxidize long-chain fatty acids in the liver → impaired gluconeogenesis (energy-dependent) → severe hypoglycemia, coma, death during fasting.
Regulation: Malonyl-CoA (the first intermediate in fatty acid synthesis) inhibits CPT-I — this ensures that when you are synthesizing fatty acids (fed state), you are not simultaneously importing them for breakdown. It is a classic reciprocal regulatory mechanism.
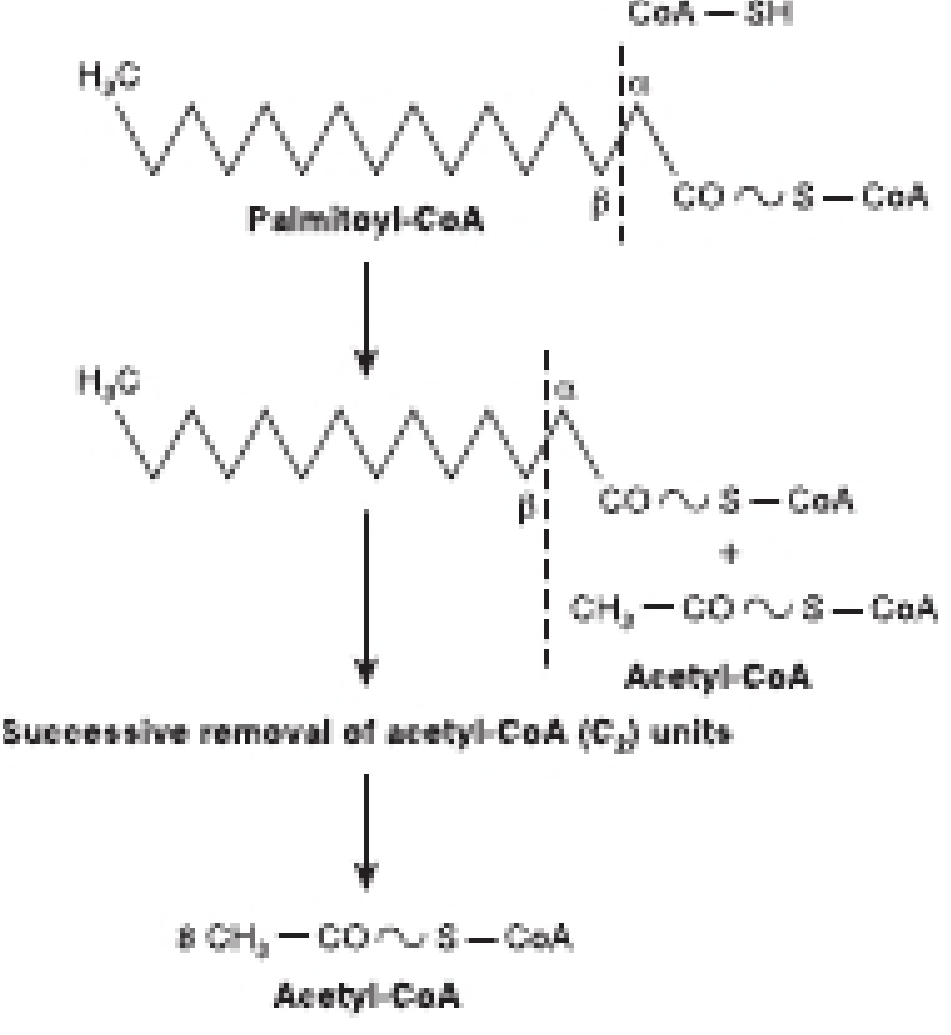
5. The Four Reactions of the Beta Oxidation Cycle
Once acyl-CoA is inside the mitochondrial matrix, it undergoes 4 repeating reactions (a "spiral"). Each turn removes 2 carbons as acetyl-CoA and yields 1 FADH₂ + 1 NADH.
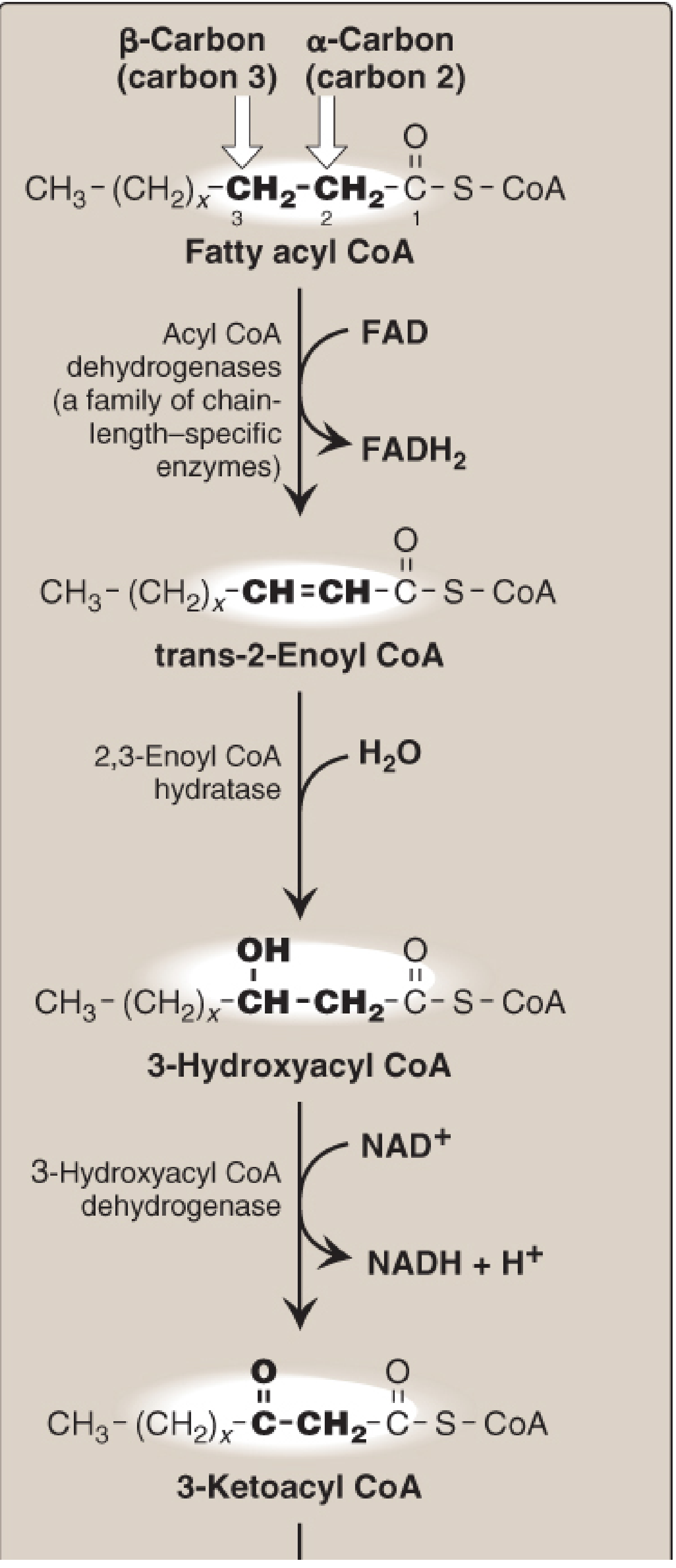
🔵 Step 1 — Oxidation (FAD-linked)
Acyl-CoA → trans-Δ²-Enoyl-CoA
- Enzyme: Acyl-CoA dehydrogenase (chain-length specific: VLCAD, LCAD, MCAD, SCAD)
- Coenzyme: FAD → FADH₂
- A double bond is introduced between C2 (α) and C3 (β), in the trans configuration
🔵 Step 2 — Hydration
trans-Δ²-Enoyl-CoA → L-3-Hydroxyacyl-CoA
- Enzyme: Enoyl-CoA hydratase (2,3-enoyl-CoA hydratase)
- Water is added across the double bond
- The hydroxyl group is added to the β-carbon (C3) → forms the L-stereoisomer
🔵 Step 3 — Second Oxidation (NAD⁺-linked)
L-3-Hydroxyacyl-CoA → 3-Ketoacyl-CoA
- Enzyme: L-3-Hydroxyacyl-CoA dehydrogenase
- Coenzyme: NAD⁺ → NADH + H⁺
- The hydroxyl on C3 is oxidized to a ketone
🔵 Step 4 — Thiolytic Cleavage
3-Ketoacyl-CoA → Acyl-CoA (shortened by 2C) + Acetyl-CoA
- Enzyme: Thiolase (β-ketoacyl-CoA thiolase)
- CoA-SH is used to cleave the bond between C2 and C3
- Products: 1 acetyl-CoA + a new acyl-CoA that is 2 carbons shorter (re-enters the spiral)
6. Energy Yield — Worked Example (Palmitic Acid, C16)
Palmitic acid (16 carbons, fully saturated) is the textbook example.
Setup:
- Activation: costs 2 ATP equivalents
- Number of cycles: 7 (each cycle removes 2C; 7 cycles reduce C16 → 8 acetyl-CoA)
- Acetyl-CoA produced: 8
- FADH₂ produced: 7
- NADH produced: 7
ATP calculation (using P:O ratios):
| Source | Quantity | ATP per unit | Total ATP |
|---|---|---|---|
| FADH₂ → ETC | 7 | 1.5 | 10.5 |
| NADH → ETC | 7 | 2.5 | 17.5 |
| Acetyl-CoA → TCA cycle | 8 | 10 | 80 |
| Subtotal | 108 | ||
| Minus activation cost | −2 | ||
| Net ATP | ~106 ATP |
This is why fats yield more energy per gram than carbohydrates — they are more reduced (more C–H bonds = more electrons to donate).
7. Odd-Chain Fatty Acids
Most human fatty acids have even numbers of carbons. Odd-chain fatty acids (found in ruminant fat and some fish) generate propionyl-CoA in the final cycle instead of acetyl-CoA.
Propionyl-CoA is converted to succinyl-CoA (a TCA intermediate) via:
- Propionyl-CoA → D-methylmalonyl-CoA (enzyme: propionyl-CoA carboxylase, requires biotin + CO₂)
- D-methylmalonyl-CoA → L-methylmalonyl-CoA (racemase)
- L-methylmalonyl-CoA → Succinyl-CoA (enzyme: methylmalonyl-CoA mutase, requires Vitamin B12)
Clinical: Vitamin B12 deficiency → methylmalonic acid accumulates in blood/urine → methylmalonic acidemia/aciduria (a metabolic marker of B12 deficiency)
8. Unsaturated Fatty Acids
Unsaturated fatty acids require extra enzymes to handle their double bonds:
| Double bond position | Extra enzyme needed |
|---|---|
| Monounsaturated (e.g., oleic acid, Δ9) | Enoyl-CoA isomerase converts cis-Δ³ → trans-Δ² (so hydratase can act) |
| Polyunsaturated (e.g., linoleic acid, Δ9,12) | 2,4-dienoyl-CoA reductase (requires NADPH) + isomerase |
Consequence: unsaturated fatty acids yield slightly less ATP than saturated ones (some FADH₂ is not produced because the cis-double bond already introduces partial oxidation).
9. Peroxisomal Beta Oxidation
For very-long-chain fatty acids (VLCFA, >C22), the initial cycles occur in peroxisomes.
Key differences from mitochondrial beta oxidation:
| Feature | Mitochondria | Peroxisomes |
|---|---|---|
| First enzyme | Acyl-CoA dehydrogenase (uses FAD) | Acyl-CoA oxidase (uses O₂ → H₂O₂) |
| FADH₂ fate | Enters ETC → ATP | H₂O₂ formed instead → less ATP |
| Acetyl-CoA fate | TCA cycle | Exported (as acetylcarnitine) to mitochondria |
| Chain length specificity | C4–C20 | C22 and above |
Clinical: Zellweger syndrome (peroxisome biogenesis disorder) → failure to oxidize VLCFA → VLCFA accumulate in brain/adrenals → neurodevelopmental failure, death in infancy. X-linked adrenoleukodystrophy (X-ALD) — defective VLCFA transporter (ABCD1) into peroxisomes — same accumulation, treated with Lorenzo's Oil.
10. Regulation of Beta Oxidation
| Regulator | Mechanism | Effect |
|---|---|---|
| Malonyl-CoA | Inhibits CPT-I | ↓ Fatty acid entry into mitochondria (fed state) |
| Insulin | ↑ Malonyl-CoA synthesis | Inhibits β-oxidation (fed state) |
| Glucagon/Epinephrine | ↓ Malonyl-CoA | Activates β-oxidation (fasted/exercise) |
| Acetyl-CoA/CoA ratio | High ratio inhibits thiolase | Slows the cycle when energy is abundant |
| NADH/NAD⁺ ratio | High ratio inhibits Step 3 | Slows cycle when reducing equivalents are excess |
11. Clinical Relevance — Nigeria Exam Favorites 🇳🇬
| Condition | Defect | Key Feature |
|---|---|---|
| MCAD deficiency | Medium-chain acyl-CoA dehydrogenase deficiency | Most common FAO defect; hypoketotic hypoglycemia during fasting; "sweaty feet" smell; can cause sudden death in infants; treat with avoiding fasting |
| Carnitine deficiency | Reduced carnitine transport/synthesis | Muscle weakness, cardiomyopathy, hypoglycemia |
| CPT-I deficiency | Cannot import LCFA into mitochondria | Liver: severe fasting hypoglycemia |
| CPT-II deficiency | Cannot reform acyl-CoA in matrix | Muscle: rhabdomyolysis with exercise |
| Vitamin B12 deficiency | Methylmalonyl-CoA mutase impaired | Methylmalonic aciduria + macrocytic anemia + subacute combined degeneration |
| Diabetic ketoacidosis | Excess FFA → excess acetyl-CoA → ketone bodies | Ketonemia, metabolic acidosis, Kussmaul breathing |
12. Summary Mnemonics
The 4 steps (OHOT):
Oxidation (FAD) → Hydration → Oxidation (NAD⁺) → Thiolysis
Carnitine shuttle: "CPT-I Invites fatty acids IN, CPT-II IIdentifies them inside"
Malonyl-CoA = the traffic policeman: when fatty acid synthesis is ON (fed state), it blocks CPT-I so no fat goes to mitochondria.
Sources: Harper's Illustrated Biochemistry 32e, pp. 229–234 | Lippincott's Illustrated Reviews: Biochemistry 8e, pp. 541–550 | Basic Medical Biochemistry: A Clinical Approach 6e
In the bt
It looks like your message got cut off — "In the bt..."
What were you trying to ask? Feel free to complete your question!
In beta oxidation proper you didn't. Talk about nadh and fadh2 formed at each step and how
Great catch! Let me go deeper on exactly how and why FADH₂ and NADH are formed at each specific step.
NADH and FADH₂ in Beta Oxidation — Step by Step
Why FAD in Step 1, not NAD⁺?
This is a classic exam question. The answer is about thermodynamics and bond type.
In Step 1, you are removing two hydrogens from a fully saturated C–C single bond (between C2 and C3). This bond has very little difference in energy between the substrate and product — the oxidation is not energetically favourable enough to reduce NAD⁺ (which has a more negative reduction potential, −0.32 V).
FAD has a higher (less negative) reduction potential (−0.06 V), so it can accept electrons from this relatively weak oxidation. FAD is physically bound to the enzyme (acyl-CoA dehydrogenase) and passes its electrons directly to the electron transport chain via Coenzyme Q (ubiquinone) through the electron transfer flavoprotein (ETF).
In Step 3, you are oxidising a hydroxyl group (–OH) to a ketone (C=O). This is a much more energetically favourable oxidation — enough to reduce NAD⁺ (more demanding electron acceptor). This is the same reason NAD⁺ is used in the TCA cycle at alcohol/hydroxyl oxidations (e.g., isocitrate dehydrogenase, malate dehydrogenase).
The 4 Steps — Detailed Electron Accounting
🔵 Step 1 — Acyl-CoA Dehydrogenase → FADH₂ formed
Fatty Acyl-CoA + FAD → trans-2-Enoyl-CoA + FADH₂
What happens chemically:
- Two hydrogen atoms are removed — one from C2 (α-carbon) and one from C3 (β-carbon)
- A trans double bond is introduced between C2 and C3
- Both hydrogens (as H⁺ + 2e⁻) go to FAD → FADH₂
- The enzyme is acyl-CoA dehydrogenase (chain-length specific — VLCAD, LCAD, MCAD, SCAD)
Fate of FADH₂:
- FAD is covalently bound to the enzyme
- FADH₂ is reoxidised by electron transfer flavoprotein (ETF)
- ETF passes electrons to ETF-ubiquinone oxidoreductase → Coenzyme Q (ubiquinol)
- From CoQ → Complex III → cytochrome c → Complex IV → O₂
- Yields ~1.5 ATP (P:O ratio ≈ 1.5)
Note: FADH₂ does NOT go directly to the ETC itself — it takes a shuttle route via ETF. This is unique to fatty acid oxidation.
🔵 Step 2 — Enoyl-CoA Hydratase → No coenzyme reduced
trans-2-Enoyl-CoA + H₂O → L-3-Hydroxyacyl-CoA
What happens chemically:
- Water is added across the double bond (hydration)
- The –OH goes to C3 (β-carbon), and –H goes to C2 (α-carbon)
- Strictly the L (S) stereoisomer is formed — the hydratase is stereospecific
- No oxidation/reduction occurs here — no NADH, no FADH₂
- This step simply prepares the β-carbon for the next oxidation
🔵 Step 3 — L-3-Hydroxyacyl-CoA Dehydrogenase → NADH formed
L-3-Hydroxyacyl-CoA + NAD⁺ → 3-Ketoacyl-CoA + NADH + H⁺
What happens chemically:
- The hydroxyl group (–OH) on C3 is oxidised to a ketone (C=O)
- Two hydrogen atoms are removed: the H from the –OH and a hydride (H⁻) from C3
- The hydride is transferred directly to NAD⁺ → NADH
- The H⁺ is released into solution
Fate of NADH:
- NADH passes its electrons to Complex I (NADH dehydrogenase) of the ETC
- Electrons flow: Complex I → CoQ → Complex III → Cyt c → Complex IV → O₂
- Yields ~2.5 ATP (P:O ratio ≈ 2.5)
🔵 Step 4 — Thiolase → No coenzyme reduced
3-Ketoacyl-CoA + CoA-SH → Acetyl-CoA + Acyl-CoA (shortened by 2C)
What happens chemically:
- The bond between C2 and C3 is cleaved by CoA (thiolytic cleavage)
- The C1–C2 fragment leaves as acetyl-CoA
- The remaining chain, now 2 carbons shorter, is a new acyl-CoA that re-enters the spiral
- No oxidation occurs — no NADH, no FADH₂
- The ketone on C3 (from Step 3) activates the bond for cleavage — it withdraws electrons, making the C2–C3 bond weak and susceptible to thiolysis
Summary Table — Coenzymes Per Cycle
| Step | Enzyme | Reaction Type | Coenzyme | Product |
|---|---|---|---|---|
| 1 | Acyl-CoA dehydrogenase | Oxidation (C–C → C=C) | FAD | FADH₂ |
| 2 | Enoyl-CoA hydratase | Hydration | None | — |
| 3 | L-3-Hydroxyacyl-CoA dehydrogenase | Oxidation (C–OH → C=O) | NAD⁺ | NADH |
| 4 | Thiolase | Thiolytic cleavage | CoA-SH | Acetyl-CoA |
Per cycle: 1 FADH₂ + 1 NADH + 1 Acetyl-CoA
Why FAD Before NAD⁺? — The Simple Rule
| Feature | FAD (Step 1) | NAD⁺ (Step 3) |
|---|---|---|
| What is oxidised | C–C (alkane, very reduced) | C–OH (alcohol, partially oxidised) |
| Reduction potential | −0.06 V (weaker oxidiser) | −0.32 V (stronger oxidiser) |
| Why it works | Weak oxidation → only FAD can accept | Stronger oxidation → NAD⁺ can accept |
| ETC entry point | CoQ (via ETF) | Complex I |
| ATP yield | ~1.5 ATP | ~2.5 ATP |
Think of it this way: NAD⁺ is a more "demanding" electron acceptor — it only accepts electrons from reactions that release enough energy. Oxidising an alcohol to a ketone (Step 3) releases more energy than oxidising an alkane to an alkene (Step 1), hence the different coenzymes.
For Palmitate (C16) — Full Coenzyme Tally
| Cycle | FADH₂ | NADH | Acetyl-CoA |
|---|---|---|---|
| 1 | 1 | 1 | 1 |
| 2 | 1 | 1 | 1 |
| 3 | 1 | 1 | 1 |
| 4 | 1 | 1 | 1 |
| 5 | 1 | 1 | 1 |
| 6 | 1 | 1 | 1 |
| 7 | 1 | 1 | 2* |
| Total | 7 | 7 | 8 |
*The last cycle produces 2 acetyl-CoA directly (the final 4-carbon acetoacetyl-CoA is cleaved by thiolase into 2 acetyl-CoA)
Bottom line to remember: Each complete turn of the beta oxidation spiral costs nothing but produces 1 FADH₂ (Step 1), 1 NADH (Step 3), and 1 Acetyl-CoA (Step 4) — and the reason FAD comes before NAD⁺ is purely thermodynamic: the first oxidation is too weak for NAD⁺ to handle.
This is a shared conversation. Sign in to Orris to start your own chat.