Neuromuscular monitoring in anaesthesia
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Searching PubMed
neuromuscular monitoring anaesthesia residual block
Loading Image
Loading Image
I have comprehensive content from both major anaesthesia textbooks. Here is a thorough, structured answer.
Neuromuscular Monitoring in Anaesthesia
Why Monitor?
Neuromuscular blocking drugs (NMBDs) do not alter consciousness or cause amnesia, yet they render patients unable to breathe or protect their airway. The most clinically important consequence of inadequate monitoring is residual neuromuscular blockade (RNMB) — partial paralysis persisting into the postoperative period. Depending on the drug and reversal strategy used, RNMB occurs in 30–40% of patients reversed with neostigmine, and may still occur in up to 5% reversed with sugammadex. With 230 million surgical procedures performed annually worldwide, the burden is enormous. — Barash's Clinical Anesthesia, 9e, p. 1626
Neither time elapsed since the last NMBD dose nor clinical tests (head lift, grip strength, tidal volume, eye opening, tongue protrusion) can reliably detect residual paralysis. A 5-second head lift was sustained by 11/12 volunteers despite a TOF ratio ≤0.5. — Barash, p. 1628
The Neuromuscular Junction: Physiology of Monitoring
Nondepolarizing NMBDs competitively occupy the α-subunits of postsynaptic nicotinic acetylcholine receptors. Crucially, only 25–30% of receptors need to be available (i.e., 70–75% occupied) for a normal train-of-four ratio — this is the "margin of safety" of the neuromuscular junction and explains why overt weakness appears only with high receptor occupancy, while subtle impairment persists at apparently "normal" clinical values. — Barash, p. 1647
Stimulation Site
The standard monitoring site is the ulnar nerve at the wrist → assessing contraction of the adductor pollicis muscle (thumb adduction). The negative electrode (black/white) is placed distally; the positive electrode (red) is proximal — "red toward the head."
The first dorsal interosseous muscle is the next best alternative and most closely mimics the sensitivity of the adductor pollicis.
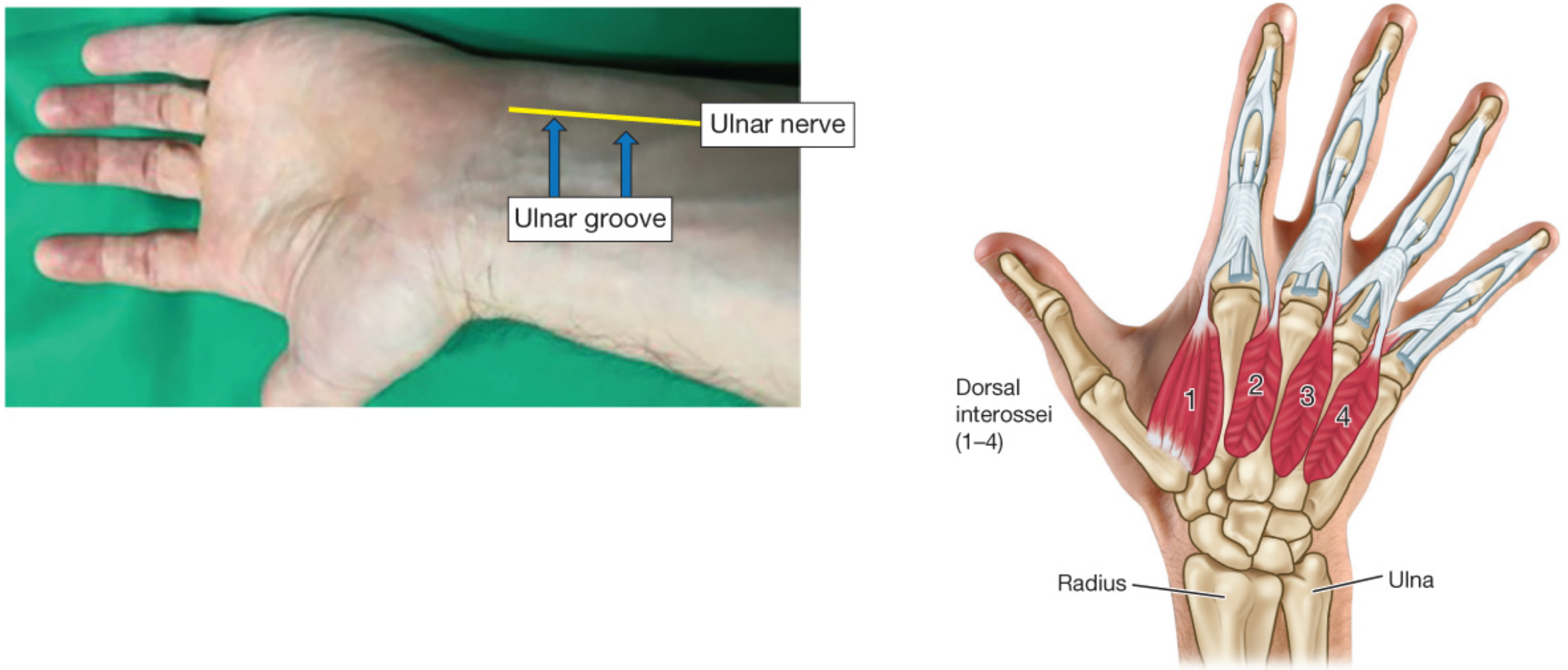
Critical caveat on facial muscles: The orbicularis oculi and corrugator supercilii are among the most resistant muscles to blockade. Monitoring at eye muscles may falsely suggest recovery when the adductor pollicis (and diaphragm) are still significantly blocked. FDA-approved sugammadex dosing recommendations do not apply to corrugator supercilii measurements. If facial monitoring is used intraoperatively, the site must be switched to the adductor pollicis before reversal. — Barash, p. 1651–1652
Peripheral Nerve Stimulator (PNS) — Qualitative (Subjective) Monitoring
A PNS delivers a monophasic square-wave current of 10–80 mA for 100–300 µs (pulse widths >300 µs risk exceeding the nerve refractory period). Skin resistance should be minimised with alcohol cleansing and abrasion (from ~100,000 Ω to <5,000 Ω).
Stimulation Patterns:
1. Single Twitch (ST)
- Frequency 0.1–1 Hz; only one stimulus per train
- Primarily used during onset of block to observe loss of twitch
- Compares to a pre-drug baseline; cannot detect fade
- Limited clinical value for recovery assessment
2. Train-of-Four (TOF) — the clinical workhorse
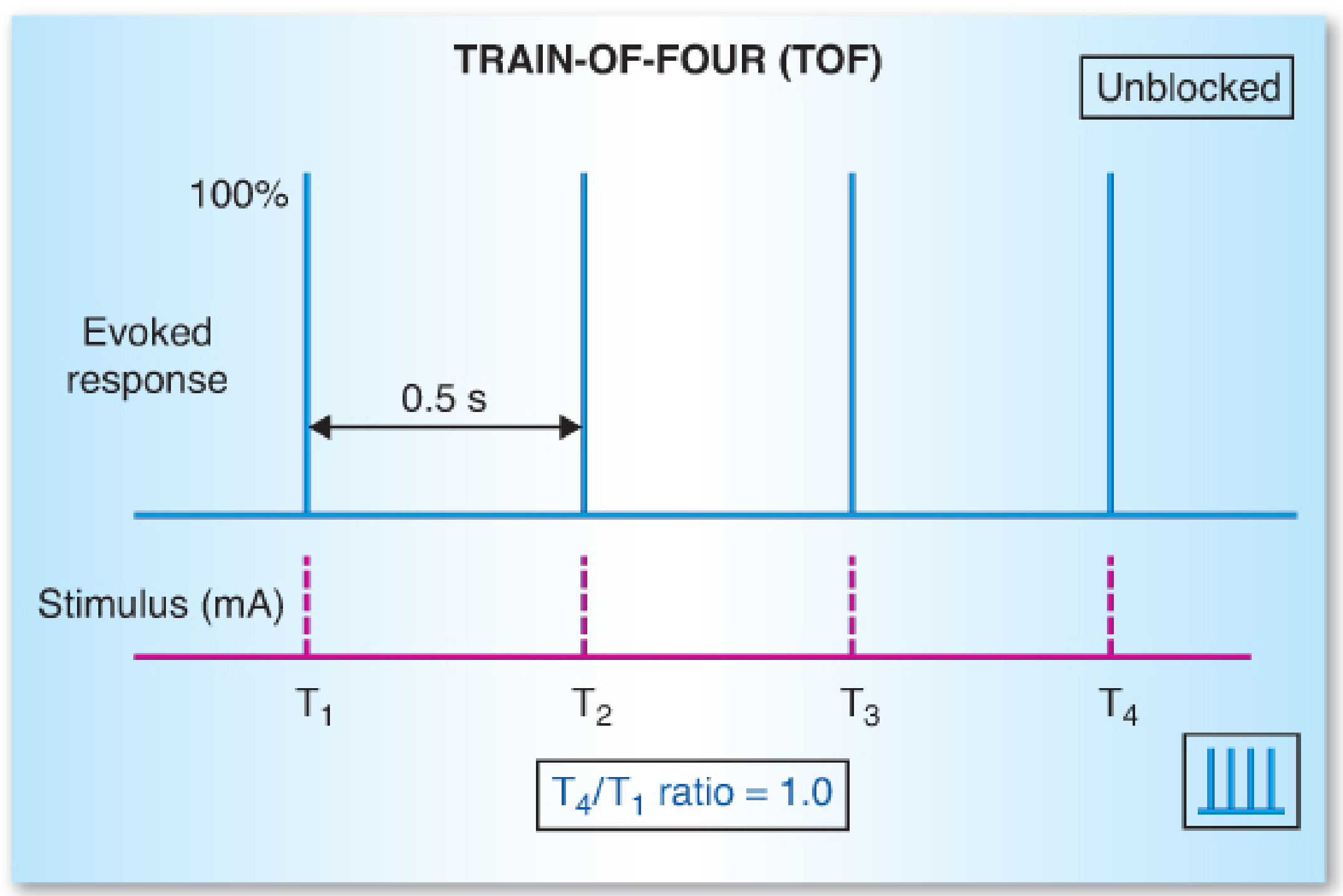
- Four stimuli at 2 Hz (0.5 s apart); can be repeated every 10–12 s
- The TOF ratio = T4/T1 (4th to 1st twitch amplitude)
- With a nondepolarising block, successive twitches fade (T4 < T3 < T2 < T1); the TOF ratio falls below 1.0
- Does not require a pre-drug baseline
- TOF count (number of visible/palpable twitches, 0–4) is used during profound block when the ratio cannot be measured
| TOF Count | Approximate Receptor Occupancy | Depth of Block |
|---|---|---|
| 0 | >95% | Intense/complete block |
| 1–2 | ~90–95% | Deep block |
| 3 | ~85–90% | Deep-moderate |
| 4 + fade | ~70–85% | Moderate block |
| 4, no fade (subjective) | Cannot exclude <0.9 ratio | Inadequate to confirm recovery |
| TOF ratio ≥0.9 (quantitative) | ~25–30% available | Adequate recovery |
3. Double-Burst Stimulation (DBS)
- Two short tetanic bursts (3 stimuli at 50 Hz) separated by 750 ms
- Designed to make fade more perceptible tactilely/visually than TOF
- Still a subjective technique; cannot reliably detect ratios >0.60
4. Tetanic Stimulation
- 50 Hz or 100 Hz for 5 s
- Sustained contraction = no fade = deep block unlikely; fade indicates residual block
- Painful in awake patients; causes post-tetanic potentiation (confounds subsequent twitches for ~3–5 min)
5. Post-Tetanic Count (PTC)
- Used when TOF count = 0 (intense block) to gauge how deeply blocked a patient is
- A 50 Hz tetanus for 5 s is given, then after 3 s a single twitch at 1 Hz is repeated
- Number of post-tetanic twitches correlates inversely with depth: PTC 1–2 = very deep; PTC >15 = TOF count about to return
The Core Problem with Subjective Assessment
Clinicians are consistently unable to subjectively detect fade when TOF ratios exceed 0.30–0.40. At TOF ratios of 0.70–0.90, subtle but clinically important impairment persists:
- Impaired pharyngeal function and airway obstruction
- Increased aspiration risk
- Impaired hypoxic ventilatory drive
- Unpleasant symptoms (diplopia, facial weakness, generalised weakness)
- Prolonged PACU stay
This means a patient who feels "fine" and can lift their head may still have dangerous residual block undetectable by touch or sight. — Miller's Anesthesia, 10e, p. 3349–3350; Barash, p. 1628
Quantitative (Objective) Monitoring
Quantitative monitors objectively measure the amplitude of the evoked muscle response and calculate the TOF ratio precisely. Guidelines from Canada, France, Spain, Australia/New Zealand, Czech Republic, Portugal, Japan, and the USA now advocate for their routine use.
Technologies
| Technology | Mechanism | Notes |
|---|---|---|
| Mechanomyography (MMG) | Measures isometric force of thumb adduction | Reference standard; not commercially available |
| Electromyography (EMG) | Measures compound muscle action potential (area under biphasic curve or peak amplitude) | FDA-approved; values similar to or slightly lower than MMG — most accurate commercially available option |
| Acceleromyography (AMG) | Accelerometer on thumb; F = ma, force inferred from acceleration | Most widely used; values slightly higher than MMG → prone to overestimating recovery. Normalisation (dividing by pre-drug baseline) is recommended |
| Kinemography (KMG) | Isoelastic polymer strip between thumb and index finger measures range of motion | Values slightly higher than MMG |
| TOFcuff | Electrodes embedded in BP cuff; measures pressure change from muscular contraction | Volumetric measurement |
Key principle: All technologies are more accurate than subjective assessment. Because AMG (and KMG) tend to overestimate recovery vs. MMG, the safe threshold differs by device:
- TOF ratio ≥0.9 by EMG/MMG
- TOF ratio ≥1.0 by AMG (to compensate for overestimation)
— Miller's Anesthesia, 10e, p. 3349; Barash, p. 1556–1561
Depths of Neuromuscular Block — Clinical Definitions
| Depth | Monitoring Criterion | Clinical Implication |
|---|---|---|
| Intense/complete | PTC = 0 | No reversal possible; continue block if needed |
| Deep | PTC ≥1, TOF count = 0 | Sugammadex 16 mg/kg can reverse rocuronium/vecuronium |
| Moderate | TOF count 1–3 | Inadequate for extubation; sugammadex preferred |
| Shallow/minimal | TOF count 4 + fade | Neostigmine most effective here |
| Full recovery | TOF ratio ≥0.9 (EMG/MMG) or ≥1.0 (AMG) | Safe for extubation |
Reversal and the Role of Monitoring
Monitoring directly guides reversal drug choice and dosing:
- Neostigmine (anticholinesterase): most effective when residual block is minimal (TOF count 4 with subjective no-fade, or TOF ratio measurable). Ineffective and potentially harmful if given during deep block. Paradoxically, high doses of neostigmine can cause neostigmine-induced weakness by desensitising ACh receptors.
- Sugammadex (selective relaxant binding agent): can reverse any depth of rocuronium/vecuronium block; dose depends on quantitative monitoring:
- TOF count ≥2: 2 mg/kg
- TOF count 0–1 (deep): 4 mg/kg
- Intense (PTC 0): 16 mg/kg
Without quantitative monitoring, neostigmine dosing is guesswork, and even sugammadex dosing may be subtherapeutic if the depth of block is unknown.
Confounders and Practical Considerations
- Respiratory acidosis limits anticholinesterase efficacy and potentiates residual block; volatile anaesthetics, opioids, and benzodiazepines may compound this via ventilatory depression
- Temperature: hypothermia prolongs block duration and reduces recovery rates
- Cholinesterase deficiency: succinylcholine and mivacurium rely on plasma cholinesterase for hydrolysis — homozygous atypical cholinesterase can extend block to 4–8 hours
- Electrode placement: skin preparation is essential; the current intensity should be supramaximal (confirm by increasing current until no further twitch increase)
- AMG normalisation: always obtain a baseline TOF measurement before NMBD administration; divide all subsequent readings by this baseline
Summary: Current Recommendations
- Quantitative neuromuscular monitoring should be used routinely for all patients receiving NMBDs — it is the only way to confirm TOF ratio ≥0.9 and exclude residual block.
- The adductor pollicis (ulnar nerve at wrist) is the preferred monitoring site.
- Avoid extubation based on time elapsed, clinical tests, or subjective TOF assessment alone.
- TOF ratio thresholds: ≥0.9 by EMG/MMG, ≥1.0 by AMG before extubation.
- Reversal drug and dose must be guided by the monitored depth of block.
- Facial muscle monitoring (especially corrugator supercilii) should not be the basis for reversal decisions.
Sources:
- Miller's Anesthesia, 10e — Chapters 24 & 39
- Barash, Cullen, and Stoelting's Clinical Anesthesia, 9e — Chapter 21
Recent evidence note: A 2025 systematic review (Bijkerk et al., PMID 39443187, Br J Anaesth) confirms that RNMB in the PACU remains a significant problem with high real-world incidence, consistent with the textbook figures above.
This is a shared conversation. Sign in to Orris to start your own chat.