Explain every topic/aspect of CA cervix in breif
Reading File
Finding Sources
Finding Sources
Searching PubMed
cervical cancer management guidelines
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
I now have comprehensive content from the textbooks. Let me compile the complete answer on CA cervix covering all aspects.
Carcinoma of the Cervix (CA Cervix) — Comprehensive Overview
1. Epidemiology
- Global burden: Second most frequent cause of cancer death in women in developing countries; ~275,000 deaths/year worldwide.
- USA: ~12,578 new cases/year; mean age at diagnosis is 47 years; bimodal peaks at 35–39 years and 60–64 years.
- Lifetime risk (USA): 1 in 128.
- Ranks 3rd most common gynecologic malignancy in the US (behind endometrial and ovarian), mainly because of effective screening.
- 30% of cases in the US occur in women who have never had a Pap smear; in developing nations this rises to ~60%.
2. Etiology & Risk Factors
HPV is the causal agent — detected in up to 99% of squamous cervical carcinomas.
| Risk Factor | Details |
|---|---|
| HPV infection | High-risk types (esp. 16 & 18 → 70% of cervical carcinomas) |
| Early age at first intercourse | <16 years |
| Multiple sexual partners | — |
| Cigarette smoking | Cofactor |
| High parity | — |
| Low socioeconomic status | — |
| Chronic immune suppression | HIV (AIDS-defining illness per CDC) |
| Herpes simplex virus | Cofactor |
| Chlamydia trachomatis | Cofactor |
| Oral contraceptive use | Possibly increases glandular abnormalities (debated) |
HPV Molecular Mechanism:
- >100 HPV types; >30 affect the lower genital tract; 15 high-risk subtypes
- E6 protein → binds and degrades p53 (tumor suppressor)
- E7 protein → binds and inactivates Rb (retinoblastoma protein)
- This disrupts cell cycle control → dysplasia → carcinoma
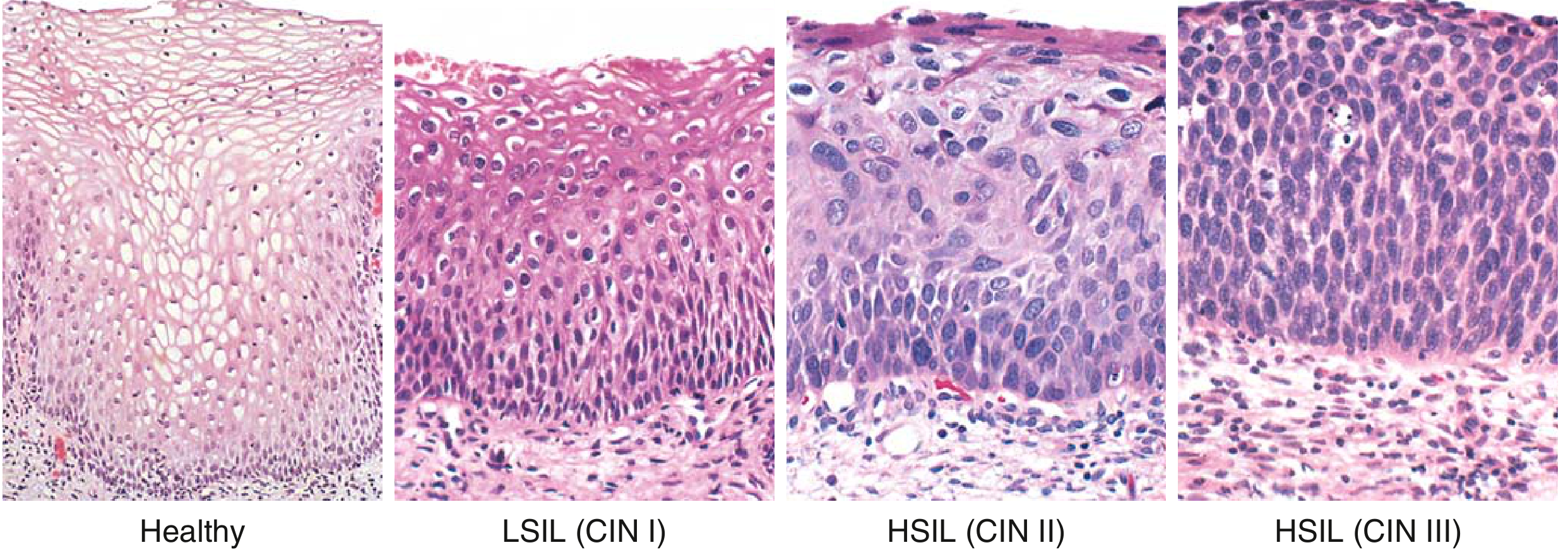
3. Premalignant Disease (CIN / SIL)
The cervix goes through a recognizable preinvasive phase before invasion.
| Terminology | Description |
|---|---|
| LSIL / CIN I | Dysplastic changes in lower ⅓ of epithelium + koilocytic change in superficial layers |
| HSIL / CIN II | Immature cells in lower ⅔ of epithelium |
| HSIL / CIN III (CIS) | Full-thickness involvement; no koilocytes; atypical mitoses |
Key site: Transformation zone (squamocolumnar junction) — where squamous metaplasia occurs and HPV preferentially infects.
- LSIL: Managed conservatively with observation (high spontaneous regression rate).
- HSIL / persistent LSIL: Treated with surgical excision (cone biopsy/LEEP).
4. Pathology / Histological Types
| Type | Frequency | Notes |
|---|---|---|
| Squamous cell carcinoma | 80% | Most common; arising from ectocervix |
| Adenocarcinoma | ~15% | Arising from endocervical glands; increasing in relative incidence |
| Adenosquamous carcinoma | Mixed | Combined features |
| Small cell / neuroendocrine carcinoma | <5% | Very aggressive; worst prognosis |
| Sarcoma | Rare | — |
| Malignant melanoma | Very rare | — |
- The proportion of adenocarcinoma is rising because the Pap smear has limited ability to detect precancerous glandular lesions.
- All common types are caused by high-risk HPV.
5. Screening
- Papanicolaou (Pap) smear: Mainstay; detects morphologic changes (koilocytes, nuclear atypia, abnormal N:C ratio)
- HPV DNA testing: Co-testing with Pap smear (age ≥30); primary HPV testing also now used
- Colposcopy: Performed for abnormal Pap/HPV results; acetic acid turns dysplastic epithelium white (acetowhite)
- Cervicography / VIA: Used in low-resource settings
Screening recommendations (general):
- Begin at age 21
- Age 21–29: Pap every 3 years
- Age 30–65: Co-testing (Pap + HPV) every 5 years OR Pap alone every 3 years
6. Diagnosis & Workup
Symptoms:
- Early: Often asymptomatic (detected on screening)
- Classic: Postcoital bleeding, intermenstrual bleeding, vaginal discharge (watery/bloody)
- Late: Pelvic pain, urinary symptoms, hematuria, rectal bleeding, lower limb edema (lymphatic obstruction), ureteral obstruction (uremia)
Diagnostic steps:
- Pap smear / colposcopy → directed biopsy
- Cervical punch biopsy / cone biopsy → histologic confirmation
- Endocervical curettage (ECC)
- Examination under anesthesia (EUA) for clinical staging
- Imaging: CT, MRI (best for parametrial invasion and local extent), PET-CT (for LN staging and distant mets)
Laboratory: CBC, renal function (ureteral obstruction), liver function
7. Staging — FIGO 2018
The 2018 revision incorporated imaging and pathologic findings (lymph node status now included).
| Stage | Description |
|---|---|
| IA | Microscopy only; depth of invasion <5 mm |
| IA1 | Stromal invasion <3 mm |
| IA2 | Stromal invasion ≥3 mm and <5 mm |
| IB | Deepest invasion ≥5 mm; confined to cervix |
| IB1 | <2 cm in greatest dimension |
| IB2 | ≥2 cm and <4 cm |
| IB3 | ≥4 cm |
| IIA | Beyond uterus; upper ⅔ vagina; NO parametrium |
| IIA1 | <4 cm |
| IIA2 | ≥4 cm |
| IIB | Parametrial involvement (not to pelvic wall) |
| IIIA | Lower ⅓ vagina; no pelvic wall |
| IIIB | Pelvic wall / hydronephrosis / non-functioning kidney |
| IIIC1 | Pelvic lymph node metastasis (r = radiologic, p = pathologic) |
| IIIC2 | Para-aortic lymph node metastasis |
| IVA | Bladder or rectal mucosa invasion |
| IVB | Distant metastasis |
Key 2018 changes:
- Horizontal spread no longer used in IA; only depth of invasion matters
- IB split into 3 substages (IB1/IB2/IB3)
- Stage III now includes lymph node metastasis (IIIC1/IIIC2)
Stage at diagnosis distribution: Stage I ~38%, Stage II ~32%, Stage III ~26%, Stage IV ~4%
8. Patterns of Spread
- Direct extension: Parametrium, vagina, bladder (IVA), rectum (IVA), pelvic side wall
- Lymphatic spread (most important): Parametrial → paracervical → obturator → external iliac → common iliac → para-aortic nodes → supraclavicular nodes
- Hematogenous: Lungs, liver, bones (less common; usually late)
9. Treatment by Stage
| Stage | Primary Treatment |
|---|---|
| IA1 (no LVSI) | Cone biopsy (if fertility desired) or simple hysterectomy |
| IA1 (with LVSI), IA2 | Radical hysterectomy + pelvic lymph node dissection (PLND), or radiation |
| IB1, IB2, IIA1 | Radical hysterectomy (type III) + PLND OR Concurrent chemoradiation (CRT) |
| IB3, IIA2 | Concurrent CRT (preferred) ± brachytherapy |
| IIB–IVA | Concurrent CRT (external beam + brachytherapy) + cisplatin |
| IVB / recurrent | Palliative chemotherapy (cisplatin + paclitaxel ± bevacizumab); immunotherapy (pembrolizumab) |
Surgery
- Cone biopsy / LEEP: IA1 without LVSI (fertility-sparing)
- Radical trachelectomy: IA2–IB1 (<2 cm); fertility-sparing; removes cervix but preserves uterus
- Radical hysterectomy (Wertheim's): Removes uterus, parametria, upper vagina + pelvic LN dissection
- Reserved for stages I–IIA; not for lesions >4 cm (will need post-op RT)
- Urinary fistula rate: <2%; operative mortality: <1%
- Sentinel lymph node evaluation: Increasingly used
Radiation
- External beam radiation therapy (EBRT): Pelvic field
- Brachytherapy: Intracavitary (tandem & ovoid/ring) — essential for local control
- Intensity-modulated radiation therapy (IMRT): Reduces bowel/bladder toxicity
- Complications: Bladder fibrosis, bowel dysfunction, vaginal stenosis (in up to 8% long-term)
Chemotherapy
- Concurrent cisplatin (40 mg/m²/week) with radiation — standard of care for ≥IB3
- Recurrent/metastatic: Cisplatin + paclitaxel ± topotecan; bevacizumab improves survival in recurrent disease
- Pembrolizumab (PD-L1 inhibitor): Approved for recurrent/metastatic PD-L1+ cervical cancer
10. Special Situations
| Situation | Management |
|---|---|
| Cervical cancer in pregnancy | Stage-dependent; delay treatment if near viability (>28 weeks) |
| Barrel-shaped cervix | Large endophytic tumors; often need CRT + adjuvant hysterectomy |
| Cancer of cervical stump | After subtotal hysterectomy; radiation preferred |
| Ureteral obstruction | Stenting; may indicate stage IIIB |
| Acute hemorrhage | Packing, embolization, palliative radiation |
11. Recurrent Cervical Cancer
- Occurs in ~35% of patients (more common with advanced stage)
- Central recurrence (vault) → potentially curable by pelvic exenteration
- Lateral/pelvic wall recurrence → poor prognosis; chemotherapy ± palliative radiation
- Pelvic exenteration: Anterior (bladder), posterior (rectum), or total; requires urinary/bowel diversion
- Laterally extended endopelvic resection (LEER): For selected cases involving the lateral pelvic wall
12. Prognosis / Survival
| Stage | 5-Year Survival |
|---|---|
| Stage I | ~85% |
| Stage II | ~60–65% |
| Stage III | ~30–40% |
| Stage IV | <15% |
Prognostic factors (for early-stage post-radical hysterectomy):
- Positive lymph nodes (strongest adverse factor)
- Parametrial involvement
- Positive surgical margins
- Lymphovascular space invasion (LVSI)
- Tumor size >4 cm
- Deep stromal invasion
13. Prevention & Vaccination
| Vaccine | HPV types covered |
|---|---|
| Cervarix (bivalent) | 16, 18 |
| Gardasil 4 (quadrivalent) | 6, 11, 16, 18 |
| Gardasil 9 (nonavalent) | 6, 11, 16, 18, 31, 33, 45, 52, 58 |
- Recommended for girls and boys aged 9–14 (ideally before sexual debut); catch-up to age 26 (up to 45 in select cases)
- Gardasil 9 covers ~90% of HPV strains causing cervical cancer
- A 2025 Cochrane network meta-analysis (PMID: 41276263) confirms HPV vaccination is highly effective for preventing cervical cancer and related HPV diseases.
14. Follow-Up After Treatment
- Every 3 months for first 2 years; every 6 months for years 3–5; annually thereafter
- Pap smear of vaginal vault
- Physical + pelvic exam at each visit
- CT/PET-CT for symptomatic patients or high-risk cases
- Watch for: recurrence, radiation-induced bowel/bladder injury, lymphedema, vaginal stenosis, psychosexual morbidity
Sources: Berek & Novak's Gynecology; Robbins & Kumar Basic Pathology; Schwartz's Principles of Surgery 11e; Grainger & Allison's Diagnostic Radiology.
This is a shared conversation. Sign in to Orris to start your own chat.