Advances in design of modern ultrasound transducers in detail
Reading File
Finding Sources
Finding Sources
Searching PubMed
ultrasound transducer design advances
Searching the Web
modern ultrasound transducer technology advances 2024 2025 CMUT PMUT matrix array
Finding Sources
Finding Sources
Reading Article
Reading Article
Reading Article
Reading File
Reading Article
Reading Article
Reading Web Page
advances ultrasound transducer CMUT PMUT design fabrication wearable
Loading Image
Finding Sources
Finding Sources
Searching PubMed
matrix array transducer 3D ultrasound beamforming
Loading Image
Reading File
Reading File
Reading File
I now have comprehensive information from textbooks, PubMed reviews, and web sources. Let me synthesize a detailed, well-structured response.
Advances in the Design of Modern Ultrasound Transducers
Ultrasound transducers have evolved from simple single-crystal oscillators into sophisticated multi-element, multi-modal systems integrating microfabrication, AI, and flexible electronics. Below is a comprehensive account of the key design domains and recent advances.
1. Fundamental Architecture (Classical Baseline)
A conventional transducer consists of a piezoelectric crystal sandwiched between damping material (rear) and an acoustic lens/impedance matching layer (front), connected to the system via cable.
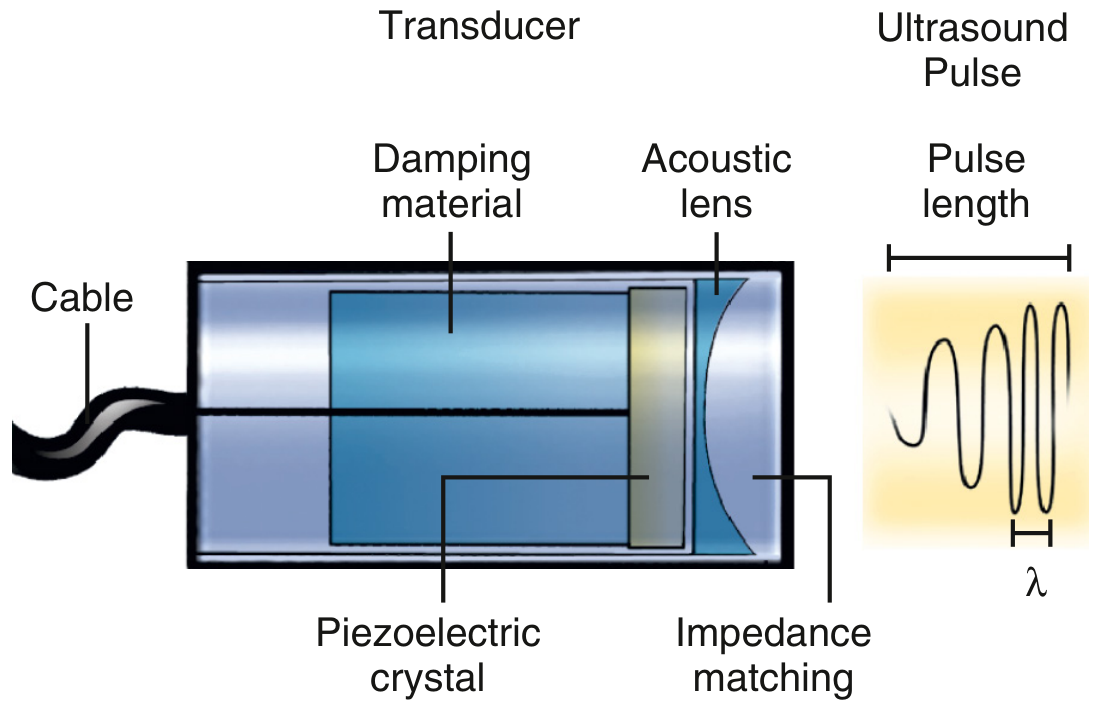
Schematic of a conventional transducer: piezoelectric crystal, damping material, acoustic lens, and impedance matching layer. — Textbook of Clinical Echocardiography
- Piezoelectric crystal: converts electrical current → mechanical compression (transmit) and pressure wave → electrical signal (receive). Common materials: lead zirconate titanate (PZT), barium titanate, quartz.
- Damping (backing) material: absorbs rearward energy; shortens pulse length → improves axial resolution.
- Acoustic lens: converges the beam to a focal zone.
- Impedance matching layer: λ/4-thick layer that reduces acoustic mismatch between the crystal (~30 MRayl) and soft tissue (~1.5 MRayl), maximizing energy transfer.
- Frequency: determined by crystal thickness — thinner crystal = higher frequency. Higher frequency = better resolution, less penetration. Clinical range: 2.5 MHz (cardiac TTE) to ≥20 MHz (intravascular).
- Bandwidth: range of frequencies in the pulse; wider bandwidth = better axial resolution and better reception of harmonic frequencies. — Textbook of Clinical Echocardiography
2. Array Architectures
Linear & Phased Arrays
The transition from single-crystal to multi-element phased arrays (64–256 elements) was the dominant advance of the 1980s–2000s. Firing elements in precise time sequences (beam-steering) allows electronic sweeping of the ultrasound beam without mechanical movement — enabling real-time 2D imaging. A phased array produces a sector-shaped image from a small footprint. Each array element is ~λ/2 wide, and the final beam shape depends on aperture size, element spacing, and electronic focusing. — Textbook of Clinical Echocardiography; Harrison's 22E
1.5D Arrays
Rows of elements are grouped in the elevation direction to provide limited beam-steering in that plane, improving slice-thickness uniformity compared to 1D arrays.
Full 2D Matrix Arrays (for 3D imaging)
The key hardware enabling volumetric (3D/4D) imaging. Matrix array transducers contain thousands of independently addressable piezoelectric elements arranged in a rectangular grid (e.g., 64×64 = 4,096 elements), producing a pyramid-shaped ultrasound volume from a single transducer position. The tradeoff is between temporal resolution, spatial resolution, and sector size. — Miller's Anesthesia 10e; Fuster & Hurst's The Heart 15e
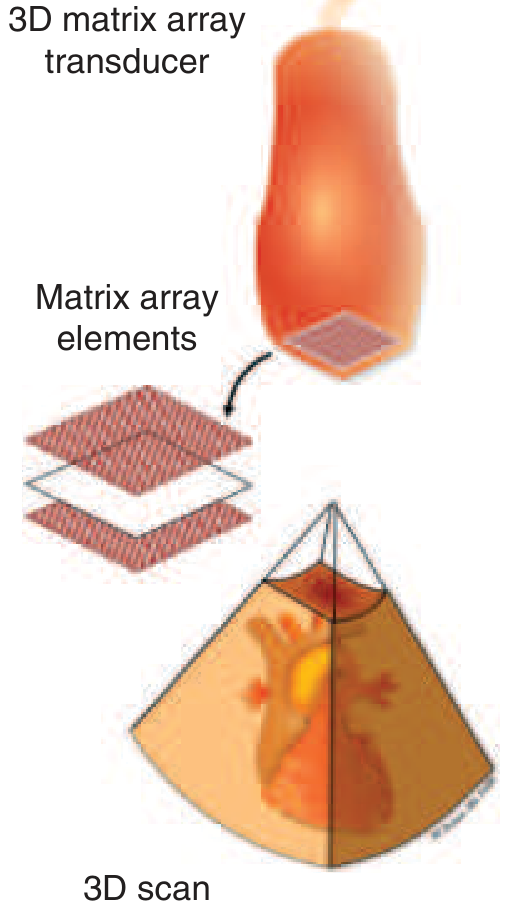
3D matrix array transducer (layered square element base) and the resulting pyramidal 3D cardiac scan. — Harrison's Principles of Internal Medicine 22E
Matrix array probes also offer multiplane imaging — simultaneously displaying two or more rotatable live 2D planes from a single acquisition, especially useful in intraoperative TEE. — Miller's Anesthesia 10e
3. Micromachined Ultrasound Transducers (MUTs)
The most transformative platform shift in the last two decades is the move from bulk piezoelectric crystals to MEMS-based micromachined transducers, fabricated using semiconductor lithography on silicon wafers. MEMS-based devices enable:
- Batch (wafer-scale) fabrication → dramatically lower cost
- Integration with CMOS electronics on the same chip
- High element density — 2D arrays with pitches as small as 20 µm
- Miniaturization for catheter-based and wearable applications
- High-frequency operation (tens of MHz for IVUS)
Two main MUT types exist:
a. Capacitive Micromachined Ultrasound Transducers (CMUTs)
CMUTs operate as miniaturized parallel-plate capacitors. A thin, metallized membrane (typically silicon nitride) is suspended over a vacuum-sealed cavity above a fixed bottom electrode. An applied DC bias deflects the membrane; an AC voltage drives oscillation, generating ultrasound. Received pressure waves deflect the membrane, changing capacitance and generating current.
Key advantages:
- Exceptional bandwidth (up to 175%), enabling broadband imaging and multi-frequency operation
- High electromechanical coupling (kT² ~0.85 at optimal bias)
- Wide dynamic range
- CMOS-compatible → on-chip signal processing and beamforming
Key challenges:
- Require large DC bias near "collapse voltage" (risk of membrane failure)
- Separate cavity heights may be needed for transmit vs. receive
- CMUT fabrication uses either sacrificial layer release (etch away a sacrificial material to form the air gap) or wafer bonding (bond a pre-thinned silicon wafer over a patterned cavity)
CMUT technology has since been developed by Philips, Hitachi, and imec. — Herickhoff & van Schaijk, Z Med Phys 2023 [PMID: 37316428]
b. Piezoelectric Micromachined Ultrasound Transducers (PMUTs)
PMUTs use a thin-film piezoelectric layer (PZT, AlN, ZnO, or ScAlN) deposited on a thin suspended membrane. Applied voltage causes flexural bending, generating pressure waves. Conversely, incident ultrasound bends the membrane, generating a voltage.
Key advantages:
- No DC bias required (simpler drive electronics, safer)
- Operate on standard CMOS-compatible low voltages
- Flexible substrate compatibility for wearable/conformable devices
- Mass-producible via standard photolithography
Key materials:
- PZT (Lead zirconate titanate): highest coupling coefficients, but lead-based (environmental concern)
- AlN (Aluminum nitride): CMOS-compatible, lead-free, moderate coupling
- ScAlN (Scandium-doped AlN): significantly higher piezoelectric response than AlN, emerging standard
- ZnO: biocompatible, used in flexible devices
PMUT adoption was catalyzed by PMUT-based fingerprint sensors (Qualcomm Snapdragon Sense ID), which demonstrated reliable mass production. This spurred medical imaging integration — 256–512 element PMUT arrays have been demonstrated at 5 MHz for 3D intracardiac echocardiography. — He et al., Biosensors 2022 [PMID: 36671890]
4. Bandwidth and Broadband Design
Modern transducers are designed for broadband operation (fractional bandwidth >80%) rather than narrow resonance. This is achieved by:
- Matching layer optimization: multiple λ/4 matching layers between crystal and tissue, reducing acoustic impedance mismatch
- Composite piezoelectrics (1-3 composites): PZT pillars embedded in polymer matrices — reduces lateral coupling, lowers acoustic impedance, broadens bandwidth
- Heavy damping backing: sacrifices sensitivity but greatly shortens pulse and broadens bandwidth
- Apodization: applying non-uniform voltage weighting across array elements to reduce side-lobe artifacts
Broadband design enables tissue harmonic imaging — the transducer transmits at a fundamental frequency f₀ and receives at the second harmonic 2f₀. Nonlinear propagation of the ultrasound wave through tissue generates harmonic frequencies. Since harmonics are generated progressively as the beam travels (narrowing the effective beam in the near-field), harmonic images have reduced side-lobe noise, better contrast resolution, improved signal-to-noise ratio, and fewer artifacts. — Miller's Anesthesia 10e
5. High-Frequency and Intravascular Transducers (IVUS)
Intravascular ultrasound (IVUS) requires transducers at 20–60 MHz to image coronary arterial walls from within the vessel lumen. Design requirements are extreme:
- Transducer diameter: ≤1 mm (to fit within 3–3.5 Fr catheters)
- Very high frequency: 40–60 MHz for atherosclerotic plaque characterization
- Single-element rotating design (mechanical) or solid-state phased array
Recent advances include:
- PVDF (polyvinylidene fluoride) and P(VDF-TrFE) polymer transducers — better acoustic impedance match to tissue, high-frequency response, flexible
- PMN-PT and PIN-PMN-PT single crystal piezoelectrics — higher coupling coefficients than PZT for superior sensitivity at small apertures
- CMUT-IVUS catheters — forward-looking (not just side-viewing) imaging of vessel bifurcations; improved bandwidth for multimodality integration
- IVUS + OCT hybrid catheters — co-registered optical coherence tomography and ultrasound on the same catheter — Peng et al., Sensors 2021 [PMID: 34069613]
6. Conformable and Wearable Transducers
One of the most active frontiers is conformable ultrasound electronics (cUSE) — transducers built on flexible/stretchable substrates that conform to curved body surfaces for continuous monitoring.
Design elements:
- Substrate: polyimide, PDMS, or other polymer films replace rigid PCBs
- Active layer: PZT thin films, PVDF, or flexible PMUT membranes on polymer substrates
- Interconnects: serpentine copper traces allow stretching without fracture
- Fabrication: spin-coating piezoelectric films on flexible substrates; laser lift-off for substrate release; ICP-CVD silicon nitride structural layers
A landmark 2024 Nature Biotechnology paper (PMID: 37217752) demonstrated a fully integrated wearable ultrasonic-system-on-patch (USoP) — a miniaturized flexible control circuit interfaced with a transducer array for signal conditioning and wireless communication. Using machine learning for real-time tissue target tracking, the device monitored central blood pressure, heart rate, and cardiac output from tissues as deep as 164 mm, continuously for 12 hours in mobile subjects. — Lin et al., Nat Biotechnol 2024
imec has demonstrated 64×64 polymer-based PMUT arrays over a 4×4 cm² area, integrating thin-film transistor (TFT) backplanes as driving electronics, fabricated on display-manufacturing-compatible process lines — opening the possibility of body-surface-scale ultrasound arrays.
7. ASIC Integration and On-Chip Beamforming
Conventional probes require a cable wire per element — completely impractical for matrix arrays with thousands of elements. Modern solutions:
- Micro-beamforming ASICs: custom integrated circuits placed immediately behind the transducer array (in the probe head) perform partial beamforming on subgroups of elements, reducing the cable count from thousands to tens
- CMUT/PMUT + CMOS monolithic integration: transducer fabricated directly on top of CMOS read-out circuitry, minimizing parasitic capacitance and maximizing sensitivity
- Deep learning beamforming: neural networks replace delay-and-sum algorithms to reconstruct images from sparse receive data, enabling high-frame-rate 3D imaging at reduced channel count (PMID: 36253231)
- Row-column addressed (RCA) arrays: instead of N×N individual element addressing, only N+N connections needed — drastically simplifying wiring while retaining volumetric imaging capability
8. Advanced Acoustic Design Features
| Feature | Mechanism | Clinical Benefit |
|---|---|---|
| Acoustic lens | Converges near-field beam | Fixed focal zone |
| Electronic multi-focus | Sequential transmissions at different focal depths | Improved lateral resolution across depth |
| Dynamic receive focusing | Continuously adjusts receive focus as echoes return | Near-optimal lateral resolution at all depths |
| Apodization | Tapered element weighting | Reduced side lobes, fewer artifacts |
| Coded excitation | Chirp/Golay sequences instead of single pulses | Higher SNR, deeper penetration, or lower output power |
| Diverging wave / plane wave transmit | Full aperture unfocused transmission | Ultrafast imaging (>10,000 frames/sec) enabling shear wave elastography and ultrasound localization microscopy |
9. Specialized Transducer Types
| Transducer | Frequency Range | Design Specifics |
|---|---|---|
| Adult TTE (transthoracic echo) | 2–5 MHz | Phased array, small footprint for intercostal access |
| TEE (transesophageal echo) | 5–7 MHz | Miniaturized array on flexible endoscope tip |
| Intracardiac echo (ICE) | 5–10 MHz | Array on 8–10 Fr catheter; rotational or phased array |
| IVUS | 20–60 MHz | Single element rotational or solid-state array on coronary catheter |
| Vascular / linear array | 5–15 MHz | Flat array for near-surface structures |
| Endocavitary (transvaginal, transrectal) | 5–10 MHz | Curved array, small probe diameter |
| High-frequency small-parts | 12–20 MHz | Linear array, thyroid, skin, superficial masses |
| Intraoperative 3D TEE | 5–7 MHz | Full matrix array in miniaturized TEE scope |
— Textbook of Clinical Echocardiography; Miller's Anesthesia 10e; Fuster & Hurst's The Heart 15e
10. Emerging and Future Directions
- Single-crystal piezoelectrics (PMN-PT, PIN-PMN-PT): electromechanical coupling kT ≥ 0.9, enabling high-sensitivity transducers with bandwidth exceeding 100%
- Lead-free piezoelectrics: KNN (potassium sodium niobate), BaTiO₃-based — driven by RoHS environmental regulations
- Large-area PMUT arrays on display-compatible processes: imec's 4×4 cm² arrays point toward ultrasound imaging patches worn like a band-aid
- Volumetric ultrasound localization microscopy (vULM): matrix array transducers performing super-resolution vascular imaging by tracking individual microbubbles in 3D — resolution below the diffraction limit (PMID: 39453807)
- Internet-of-Medical-Things (IoMT) integration: wearable USoP patches transmitting continuous hemodynamic data wirelessly
- Photoacoustic ultrasound hybrids: same transducer receives both ultrasound echoes and laser-excited photoacoustic signals for simultaneous anatomical + molecular imaging
- AI-assisted adaptive beamforming: neural networks optimizing transmit sequences in real time based on tissue characteristics
Key References
- Textbook of Clinical Echocardiography — Piezoelectric Crystal, Types of Transducers, Beam Shape and Focusing
- Harrison's Principles of Internal Medicine 22E — Echocardiography, 3D matrix array transducer
- Miller's Anesthesia 10e — Harmonic Imaging, 3D Image Acquisition, Matrix Array
- Fuster & Hurst's The Heart 15e — Three-Dimensional Echocardiography
- He Y et al. Piezoelectric Micromachined Ultrasound Transducer Technology. Biosensors 2022 [PMID: 36671890]
- Herickhoff CD & van Schaijk R. cMUT technology developments. Z Med Phys 2023 [PMID: 37316428]
- Zhang L et al. Conformable Ultrasound Electronics. Adv Mater 2024 [PMID: 37792426]
- Lin M et al. Wearable ultrasound system-on-patch. Nat Biotechnol 2024 [PMID: 37217752]
- Peng C et al. Advances in IVUS Transducers. Sensors 2021 [PMID: 34069613]
- Denis L et al. Volumetric Ultrasound Localization Microscopy. IEEE TUFFC 2024 [PMID: 39453807]
This is a shared conversation. Sign in to Orris to start your own chat.