Tell me about diabetes
Reading File
Finding Sources
Searching PubMed
"diabetes mellitus"[MeSH Terms] AND "review"[pt]
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have substantial information from Harrison's Principles of Internal Medicine (2025) and other sources. Let me compile a comprehensive answer.
Diabetes Mellitus
Definition & Overview
Diabetes mellitus (DM) is a group of metabolic diseases characterized by hyperglycemia resulting from defects in insulin secretion, insulin action, or both. Chronic hyperglycemia damages the vasculature and peripheral nerves, leading to the microvascular and macrovascular complications that define the long-term burden of the disease.
Globally, DM affects hundreds of millions of people, with prevalence rising sharply in all world regions — particularly Sub-Saharan Africa, South/East Asia, and the Western Pacific — and type 2 DM accounts for the vast majority of cases.
Classification
The ADA recognizes four main clinical types:
| Type | Core Mechanism |
|---|---|
| Type 1 DM (T1DM) | Autoimmune destruction of pancreatic beta cells → absolute insulin deficiency |
| Type 2 DM (T2DM) | Progressive beta cell failure on a background of insulin resistance |
| Gestational DM (GDM) | Diabetes diagnosed in pregnancy, not clearly overt prior to gestation |
| Other specific types | Monogenic (MODY), exocrine pancreatic disease, drug-induced, endocrinopathies, etc. |
Other specific causes include:
- Genetic defects of beta cell function (MODY subtypes: HNF-4α, glucokinase, HNF-1α, etc.)
- Exocrine pancreatic disease — pancreatitis, cystic fibrosis, hemochromatosis, pancreatectomy
- Endocrinopathies — acromegaly, Cushing's syndrome, glucagonoma, pheochromocytoma
- Drug-induced — glucocorticoids, calcineurin inhibitors, antiretrovirals, second-generation antipsychotics, thiazides, PCSK9 inhibitors
- Rare immune-mediated forms — anti-insulin receptor antibodies, immune checkpoint inhibitor therapy
— Harrison's Principles of Internal Medicine, 22E, Table 415-1
Diagnosis
Diagnosis (in non-pregnant adults) is based on any one of:
- Fasting plasma glucose ≥ 7.0 mmol/L (126 mg/dL)
- 2-hour plasma glucose ≥ 11.1 mmol/L (200 mg/dL) on OGTT
- HbA1c ≥ 6.5%
- Random plasma glucose ≥ 11.1 mmol/L with classic symptoms
Prediabetes is defined by impaired fasting glucose (IFG) or impaired glucose tolerance (IGT) — values above normal but below the diagnostic threshold.
The ADA recommends screening all adults ≥35 years every 3 years, and earlier in overweight individuals with additional risk factors.
Pathophysiology
Type 1 DM
T1DM is a chronic autoimmune disease in which T-cell-mediated destruction of islet beta cells results in absolute insulin deficiency. It accounts for 5–10% of all DM. Key autoantibodies include anti-GAD65, anti-insulin, anti-islet cell, and anti-IA-2. Genetic susceptibility (>60 loci identified, most in HLA region) combined with environmental triggers (enteroviruses implicated) drives disease onset.
Type 2 DM
T2DM involves a dual defect:
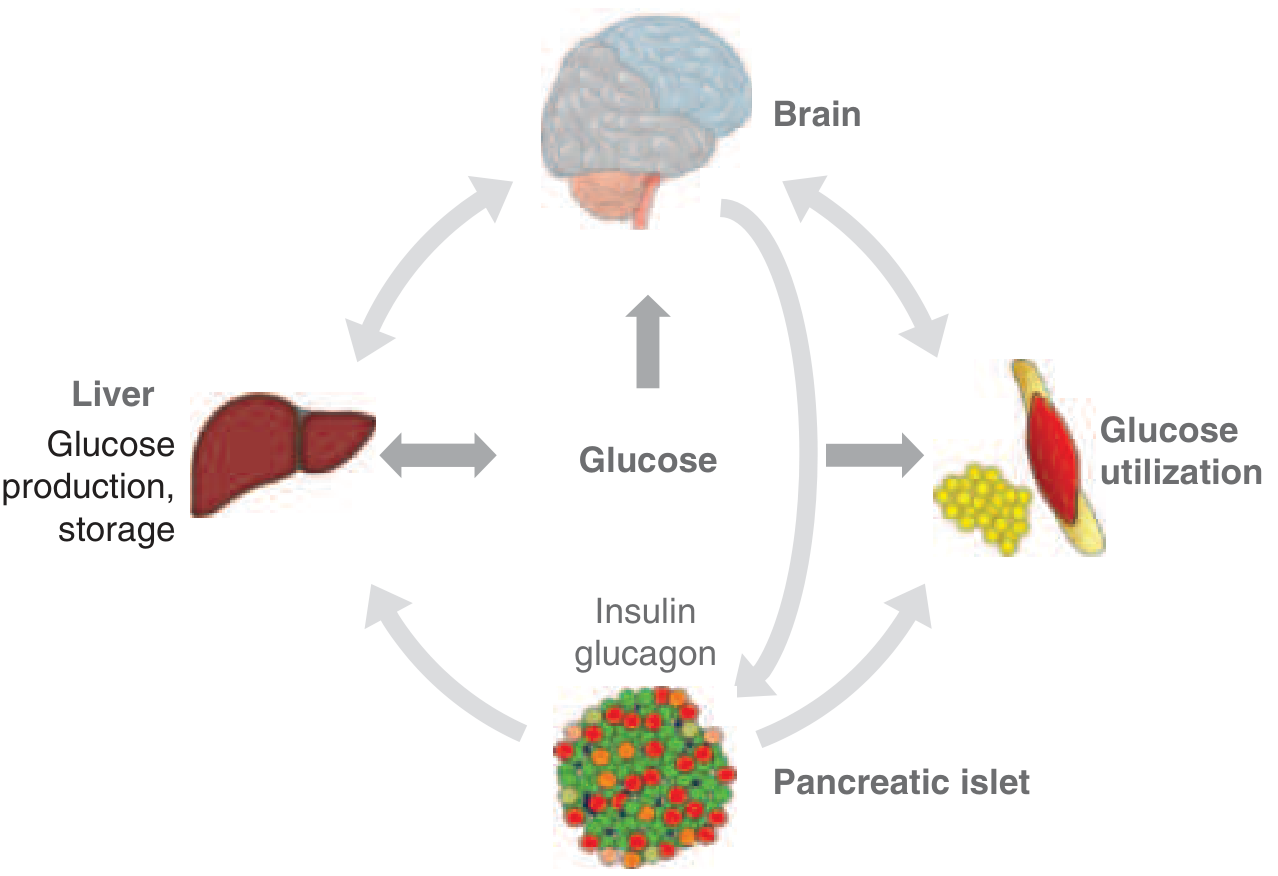
- Insulin resistance — reduced insulin action in skeletal muscle, liver, and adipose tissue. In T2DM, glucose utilization is 30–60% lower than in non-diabetic individuals. The molecular mechanism involves post-receptor defects in insulin signalling, accumulation of lipid intermediates in skeletal myocytes impairing mitochondrial oxidative phosphorylation, and low-grade metabolic inflammation driven by reactive oxygen species.
- Beta cell failure — compensatory hyperinsulinemia initially maintains glucose homeostasis. As beta cells fail to compensate, postprandial hyperglycemia (IGT) develops first, followed by fasting hyperglycemia, and ultimately frank T2DM.
Visceral obesity is a major driver: enlarged adipocytes secrete free fatty acids, TNF-α, resistin, IL-6, and reduce adiponectin — all worsening insulin resistance. Elevated free fatty acids drain into the portal circulation, directly impairing hepatic insulin signalling and driving excess hepatic glucose output.
Clinical Features
Symptoms of hyperglycemia:
- Polyuria, polydipsia, polyphagia
- Weight loss (especially T1DM)
- Blurred vision, fatigue
Acute complications:
- Diabetic ketoacidosis (DKA) — predominantly T1DM; ketone production from unrestrained lipolysis; life-threatening
- Hyperosmolar hyperglycaemic state (HHS) — predominantly T2DM; severe hyperglycaemia without significant ketosis
- Hypoglycaemia — major risk with insulin and sulfonylurea therapy
Chronic complications:
| Category | Manifestations |
|---|---|
| Microvascular | Diabetic nephropathy, retinopathy, neuropathy |
| Macrovascular | Coronary artery disease, stroke, peripheral arterial disease |
| Other | Diabetic foot ulcers, infections, gastroparesis, sexual dysfunction |
Management
The three overarching goals are: (1) eliminate hyperglycaemia symptoms, (2) prevent or reduce microvascular and macrovascular complications, (3) maintain quality of life.
Lifestyle
- Nutrition: Reduce caloric intake; limit refined carbohydrates and sugar-sweetened beverages; low-carbohydrate diets can produce rapid glucose lowering in T2DM. Sodium <2,300 mg/day.
- Physical activity: ADA recommends ≥150 min/week of moderate aerobic activity spread over ≥3 days; resistance training also advised. Exercise independently improves insulin sensitivity.
- Weight loss: 5–10% body weight reduction significantly improves glycaemic control in T2DM.
Pharmacotherapy
Type 1 DM: Insulin replacement is mandatory (basal-bolus regimens, insulin pumps). Carbohydrate counting guides mealtime dosing.
Type 2 DM (stepwise approach):
- Metformin remains first-line (unless contraindicated)
- Add agents based on comorbidities:
- GLP-1 receptor agonists (e.g., semaglutide, liraglutide) — weight loss, CV benefit
- SGLT-2 inhibitors (e.g., empagliflozin, dapagliflozin) — renal and CV protection
- DPP-4 inhibitors, sulfonylureas, TZDs, basal insulin as needed
- Insulin therapy when oral/injectable agents are insufficient
Recent evidence (2025): A large meta-analysis of 99,599 patients (PMID 40892610) confirmed GLP-1 receptor agonists have significant cardiovascular benefits and favourable tolerability in T2DM. Daily step counts also show a dose-response relationship with reduced diabetes-related mortality (PMID 40713949).
Bariatric surgery
In severe obesity with T2DM, bariatric surgery (Roux-en-Y gastric bypass, sleeve gastrectomy) produces 68% complete remission of T2DM at 5 years and reduces cardiovascular and all-cause mortality. Earlier-stage T2DM (no insulin requirement, shorter duration, lower HbA1c) responds best.
Glycaemic targets
- HbA1c target: generally <7% for most adults
- Tighter targets (<6.5%) may suit younger, newly diagnosed patients without hypoglycaemia risk
- Less stringent targets (7.5–8%) appropriate for elderly or those with hypoglycaemia unawareness
Multidisciplinary team
Optimal care requires collaboration among primary care, endocrinology, nursing/diabetes educators, dietitians, ophthalmology, nephrology, podiatry, cardiology, and behavioural health.
Epidemiology
Prevalence is rising globally. T2DM has a strong association with obesity, physical inactivity, and ageing populations. Type 2 DM may be present for up to a decade before diagnosis, and some patients already have complications at first presentation — underscoring the importance of screening.
Sources:
- Harrison's Principles of Internal Medicine, 22E (2025) — Chapters 415 & 416
- Creasy & Resnik's Maternal-Fetal Medicine
- Tietz Textbook of Laboratory Medicine, 7th Ed.
- Recent PubMed evidence: GLP-1 agonist meta-analysis [PMID 40892610], daily steps meta-analysis [PMID 40713949]
This is a shared conversation. Sign in to Orris to start your own chat.