Tumors of the ear. In detailed information
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
I now have comprehensive information to provide a detailed, well-organized answer. Let me compile it.
Tumors of the Ear
Tumors of the ear are classified by anatomic location (external, middle, or inner ear/temporal bone/cerebellopontine angle), tissue of origin, and biological behavior (benign vs. malignant). The following is a systematic overview.
Classification Overview
Primary Benign Tumors
- Paraganglioma (glomus tympanicum, glomus jugulare)
- Schwannoma / Neurofibroma
- Meningioma
- Adenoma / Ceruminous adenoma
- Hemangioma
- Osteoma / Exostosis
- Choristoma / Lipoma
- Middle ear adenomatous neuroendocrine tumor
Primary Malignant Tumors
- Squamous cell carcinoma (SCC) — most common
- Basal cell carcinoma
- Adenocarcinoma / Ceruminous adenocarcinoma
- Adenoid cystic carcinoma
- Mucoepidermoid carcinoma
- Rhabdomyosarcoma
- Chondrosarcoma / Chordoma
- Melanoma
- Angiosarcoma, fibrosarcoma, others
Metastatic Tumors (prostate, breast, renal cell, lung, lymphoma, multiple myeloma)
— Cummings Otolaryngology Head and Neck Surgery, Box 178.1
I. BENIGN TUMORS
1. Paraganglioma (Glomus Tumors)
Paragangliomas of the temporal bone are the most frequently encountered temporal bone neoplasms after acoustic neuromas. They arise from neural crest elements associated with the jugular bulb adventitia and Jacobson's and Arnold's nerves.
Two main subtypes:
| Feature | Glomus Tympanicum | Glomus Jugulare |
|---|---|---|
| Origin | Glomus bodies of the middle ear | Jugular bulb adventitia |
| Presentation | Pulsatile tinnitus + conductive hearing loss (early, small) | Late presentation after bony destruction; cranial nerve IX–XII dysfunction |
| MRI hallmark | Enhancing mass behind TM | "Salt and pepper" T1/T2 appearance; arborizing flow voids |
| CT | Limited to middle ear; tympanic/hypotympanic location | Irregular destruction of jugular foramen; absent bony septum between ICA and IJV |
- 1–3% of glomus jugulare tumors secrete catecholamines → preoperative 24-hour urine metanephrines required before surgery
- MRI complements CT; angiography needed if embolization planned pre-operatively
- Exam: if all borders visible through the TM → glomus tympanicum; if borders incomplete → may be glomus jugulare extending into hypotympanum
Classification systems used: Fisch classification and Glasscock-Jackson classification
Treatment:
- Fisch class A/B (limited to middle ear): surgical resection preferred
- Fisch class C/D (extensive): stereotactic radiosurgery (SRT) is favored — lower complication rates and similar/better local control vs. surgery
- SRT achieves 90–95% tumor control; preserves lower cranial nerve function better than surgery
— Cummings Otolaryngology, pp. 3404–3436; K.J. Lee's Essential Otolaryngology, p. 488
2. Vestibular Schwannoma (Acoustic Neuroma)
- Most common tumor of the internal auditory canal (IAC) and cerebellopontine angle (CPA)
- 8–10% of all intracranial tumors; incidence ~1 per 100,000
- Arises most commonly from the vestibular nerve (though any cranial nerve may be involved)
- Benign, slow-growing: average growth 1–2 mm/year; some never grow
Histology: Antoni A fibers (fibrillary, polar, elongated tissue) + Antoni B fibers (loosely reticulated tissue)
Clinical Features:
- Asymmetric sensorineural hearing loss (SNHL)
- Tinnitus and imbalance
- Audiogram: poor speech discrimination score out of proportion to pure-tone loss; rollover (decline in discrimination at higher presentation levels)
- ABR historically used; MRI with gadolinium is now the gold standard
Treatment (three options):
| Approach | Indications | Notes |
|---|---|---|
| Observation | Small tumors, serviceable hearing, older age | Serial MRI; many tumors never grow |
| Radiosurgery | Tumors <2.5 cm, enlarging; 12–13 Gy | 90–95% control; does NOT preserve hearing. Facial nerve dysfunction <1%; rare malignant transformation (1:1000 over 30 years) |
| Microsurgery | All sizes depending on hearing goals | Three approaches (below) |
Surgical Approaches:
- Translabyrinthine: causes profound SNHL; no brain retraction; removes any size tumor; facial nerve identified at fundus before tumor removal
- Retrosigmoid: hearing preservation possible for tumors <1.5 cm in medial IAC; requires cerebellar retraction; higher rate of chronic headaches
- Middle fossa: hearing preservation 60–70%; tumors in IAC <1.5 cm; temporal lobe retraction required
— K.J. Lee's Essential Otolaryngology, p. 488
3. Facial Nerve Schwannoma
- Most common facial nerve tumor (followed by hemangioma)
- Geniculate ganglion most commonly affected
- MRI: enhancement with gadolinium
- CT: smooth bony remodeling (vs. ground-glass appearance + erosion of hemangioma)
Treatment:
- Normal/near-normal facial function → observe
- House-Brackmann Grade 3 or worse → surgical removal + nerve grafting
- Early/mild paresis → tumor decompression may be considered
4. Meningioma
- Second most common CPA tumor after vestibular schwannoma
- Arises from arachnoid cap cells
Imaging features distinguishing from schwannoma:
- Dural tail
- Eccentric location relative to the IAC
- Calcifications, hyperostosis of adjacent bone
- More homogenous enhancement with gadolinium
Symptoms: SNHL, tinnitus, imbalance, facial paresthesia, diplopia, ataxia
WHO Classification:
- Grade 1 (Benign): meningiothelial, fibrous, transitional, psammomatous, angiomatous, secretory
- Grade 2 (Atypical): chordoid, clear cell, atypical
- Grade 3 (Anaplastic/malignant): papillary, rhabdoid, anaplastic
Histology: Psammoma bodies
Hearing preservation (or even improvement) more common than with vestibular schwannomas.
Hearing preservation (or even improvement) more common than with vestibular schwannomas.
5. Middle Ear Adenomatous Neuroendocrine Tumor
- Both neuroendocrine and glandular features
- Symptoms: hearing loss, aural fullness, tinnitus, otalgia
- CT similar to tympanic paraganglioma; may appear in atypical locations
- Male predominance (2:1 ratio)
- IHC: positive for pan-cytokeratins, chromogranin, synaptophysin, insulinoma-associated protein 1
- Rare metastatic potential
- Treatment: surgical resection + long-term follow-up (risk of recurrence)
6. Choristoma (Lipochoristoma / Middle Ear Lipoma)
- Rare middle ear or IAC mass
- Defined as histologically normal tissue in an abnormal location
- Most common type: salivary gland tissue; less common: glial choristoma
- Middle ear: conductive hearing loss + effusion
- IAC: SNHL, tinnitus, imbalance
- MRI: hyperintense on T1 without contrast; no enhancement with fat-saturation gadolinium sequences (unlike schwannoma)
- Treatment: observation with serial imaging — removal from facial nerve usually not feasible
II. MALIGNANT TUMORS
1. Squamous Cell Carcinoma (SCC) of the Temporal Bone
Most common primary malignancy of the temporal bone, followed by basal cell carcinoma, adenocarcinoma, adenoid cystic carcinoma, mucoepidermoid carcinoma, ceruminoma, melanoma, and sarcoma.
Risk factors: Chronic irritation or inflammation (e.g., chronic otitis media)
Symptoms (in decreasing frequency):
- Otalgia
- Otorrhea
- Hearing loss
- Facial palsy
- Parotid mass
Preoperative Evaluation:
- Audiogram
- CT temporal bone and neck (with IV contrast) — lymph node assessment
- CT chest / PET scan — distant metastases
Staging — Modified University of Pittsburgh System:
| T Stage | Extent of Disease |
|---|---|
| T1 | Tumor limited to EAC; no bony erosion or soft tissue involvement |
| T2 | Limited bony erosion (not full thickness) or <0.5 cm soft tissue involvement |
| T3 | Erosion through bony EAC with limited (<0.5 cm) soft tissue; or middle ear/mastoid involvement |
| T4 | Medial wall erosion (cochlea, labyrinth, petrous apex, jugular foramen, carotid canal, dura); >0.5 cm soft tissue; facial paresis |
Treatment:
- Low-grade EAC (no bone involvement): sleeve resection
- Bone involvement: lateral temporal bone resection (LTBR) minimum
- Middle ear extension: subtotal temporal bone resection (STBR)
- Total temporal bone resection: no proven survival benefit; rarely performed
- T3/T4 disease: parotidectomy ± neck dissection considered
— K.J. Lee's Essential Otolaryngology, pp. 491–492
2. Rhabdomyosarcoma
- Most common soft-tissue sarcoma in children
- Temporal bone is a parameningeal site → classified Stage II/III based on nodal status
- Types: embryonal (most common in children), alveolar, pleomorphic
Treatment:
- Wide surgical resection with clear margins
- Most pediatric high-grade cases: surgery + 50–70 Gy external beam radiation + multidrug chemotherapy
- SRS indicated for residual/recurrent disease at jugular foramen or perineurial invasion
Prognosis (5-year survival by risk group):
- Low risk: ~90%
- Intermediate risk: 60–80%
- High risk: 20–40%
Caution in children: External beam radiation to skull base can cause Eustachian tube dysfunction, osteoradionecrosis, SNHL, cataract development, impaired cranial bone/tooth development, pituitary dysfunction.
3. Chondrosarcoma
- Rare; 1% occur at the skull base — typically at spheno-occipital or petroclival synchondroses
- WHO Grades 1 and 2: low grade, more common
- WHO Grade 3 / mesenchymal subtype: high grade
Imaging: Similar to chordoma; eccentric location; no hallmark differentiating low from high grade
Pathology: Non-reactive for keratin and EMA (helps distinguish from chordoma)
Treatment: Surgery followed by radiation (proton beam / carbon ion preferred)
Prognosis (SEER): 5-year and 10-year survival: 87% and 68% (better in younger patients, females, smaller tumors)
4. Chordoma
- Derived from notochord remnants; 33% of all chordomas occur at the skull base
- Median age at presentation: 60 years (also occurs in children)
- Metastases rare at presentation; morbidity from local growth
Imaging:
- Hyperintense T2-MRI, hypointense T1-MRI (classic)
Treatment: Surgery followed by radiation; proton beam and carbon ion therapy preferred (5-year local control >70%)
Prognosis:
- Median overall survival: 7.7 years (SEER)
- Under 40 years: 68% 10-year survival
- Over 40 years: 43% 10-year survival
- Gross/near-total resection: 6× higher 5-year survival vs. subtotal resection
5. Adenoid Cystic Carcinoma and Other Ceruminous Gland Malignancies
Ceruminous gland tumors are a spectrum arising from the specialized apocrine glands of the external auditory canal:
- Ceruminous adenoma (benign)
- Ceruminous adenocarcinoma — low and high grade
- Adenoid cystic carcinoma — perineural invasion characteristic
- Mucoepidermoid carcinoma
All require surgical resection; adenoid cystic carcinoma has propensity for perineural spread and late recurrence.
III. Key Imaging Summary
| Tumor | CT | MRI |
|---|---|---|
| Glomus jugulare | Irregular jugular foramen destruction | "Salt and pepper"; flow voids |
| Vestibular schwannoma | IAC widening (smooth) | Enhances with gadolinium; centered on IAC |
| Meningioma | Hyperostosis, calcification | Dural tail; homogenous enhancement |
| Chordoma | Bone destruction at skull base | Hyperintense T2 |
| Chondrosarcoma | Eccentric, calcified; petroclival | Similar to chordoma |
| Choristoma/lipoma | — | Hyperintense T1; no enhancement with fat sat |
| Facial nerve schwannoma | Smooth bony remodeling | Gadolinium enhancement |
IV. Metastatic Tumors to the Temporal Bone
Common primary sites: prostate, breast, gastrointestinal, renal cell carcinoma, lung, also multiple myeloma, lymphoma, and leukemia (chloroma). Always consider metastatic disease in patients with known malignancy presenting with new unilateral hearing loss or facial palsy.
Sources:
- Cummings Otolaryngology Head and Neck Surgery, Box 178.1, pp. 3404–3436
- K.J. Lee's Essential Otolaryngology, pp. 488–493
In detailed information
Asking for Preferences
Checking Date & Time
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Searching Images
glomus tympanicum paraganglioma ear middle ear red mass
Searching Images
vestibular schwannoma acoustic neuroma MRI internal auditory canal
Finding Sources
Reading File
Reading File
Finding Sources
Searching Images
squamous cell carcinoma temporal bone external auditory canal CT scan
Finding Sources
Reading File
Reading File
Reading File
Here is the fully expanded, comprehensive reference on tumors of the ear, integrating all available textbook data with clinical imaging.
Tumors of the Ear — Comprehensive Reference
Anatomic Classification Table
| Location | Benign | Malignant |
|---|---|---|
| External Auditory Canal (EAC) | Osteoma, Exostosis, Adenoma (ceruminous) | SCC, BCC, Ceruminous adenocarcinoma, Melanoma, Direct extension |
| Middle Ear / Mastoid | Glomus tympanicum, Hemangioma, Lipoma/Choristoma, Endolymphatic sac tumor, Adenomatous neuroendocrine tumor | SCC, Adenoid cystic carcinoma, Acinic cell carcinoma, Rhabdomyosarcoma |
| IAC / CPA / Skull Base | Vestibular schwannoma, Facial nerve schwannoma, Meningioma, Choristoma/Lipoma | Chondrosarcoma, Chordoma, Metastases |
— K.J. Lee's Essential Otolaryngology, Table 24-1; Cummings Otolaryngology, Box 178.1
PART I — BENIGN TUMORS
1. Paraganglioma (Glomus Tumor / Chemodectoma)
Epidemiology & Biology
- Most common neoplasm of the middle ear and second most common temporal bone tumor overall
- Also called chemodectoma; three types: Glomus tympanicum (GT), Glomus jugulare (GJ), Glomus vagale (GV)
- Sex ratio: M:F = 1:5 (predominantly female); Caucasians more frequently affected
- 10% are multicentric
- Majority sporadic; up to 30% familial
- Rarely malignant: 2–4% (highest risk in vagal paragangliomas); diagnosis of malignancy requires metastasis to non-neuroendocrine tissue; most common sites: lymph nodes, bone, lung, liver, spleen
- Rarely functional: 5% or less secrete neuroactive peptides → palpitations, sweating, flushing, syncope, hypertension, headaches; may cause catastrophic hypertension at anesthesia induction if unrecognized
- If secretory: treat with phentolamine (nonselective reversible α-adrenergic blocker)
- Arise from chemoreceptor (Type I chief) cells and Type II sustentacular cells of parasympathetic paraganglia associated with:
- Jugular dome adventitia
- Tympanic promontory
- Along Jacobson's nerve (branch of CN IX) and Arnold's nerve (branch of CN X)
Genetics — Familial Paraganglioma Syndromes
| Gene | Syndrome | Locus | Inheritance | Head & Neck | Adrenal | Malignancy Risk | Notes |
|---|---|---|---|---|---|---|---|
| SDHD | PGL1 | 11q23 | AD (imprinted) | 91–98% | 16–21% | 8% | Multiple tumors |
| SDHAF2 | PGL2 | 11q13.1 | AD (imprinted) | 70–91% | 0% | 0% | Imprinting |
| SDHC | PGL3 | 1q21 | AD | 88% | <10% | <10% | Single tumors |
| SDHB | PGL4 | 1p35-36 | AD | 27–31% | 18–28% | 13–23% | Malignancy risk |
| SDHA | PGL5 | 5p15.33 | AD | <3% | <10% | <10% | — |
Mutations in mitochondrial complex II (succinate dehydrogenase subunits); involved in the electron transport chain. SDHB carries the highest malignancy risk.
Tumor Classification
Fisch Classification:
| Type | Extent |
|---|---|
| A | Limited to the middle ear |
| B | Limited to the tympanomastoid area; no infralabyrinthine involvement |
| C1 | Infralabyrinthine/petrous apex; limited carotid canal involvement |
| C2 | Invasion of vertical carotid canal |
| C3 | Invasion of horizontal carotid canal (not foramen lacerum) |
| C4 | Entire intrapetrous carotid |
| De1 | Extradural intracranial extension <2 cm |
| De2 | Extradural extension >2 cm |
| Di1 | Intradural extension <2 cm |
| Di2 | Intradural extension >2 cm |
| Di3 | Intradural, unresectable |
Glasscock-Jackson — Glomus Tympanicum:
- Type I: Limited to promontory
- Type II: Completely filling the middle ear
- Type III: Fills middle ear, extends into mastoid
- Type IV: Extends into EAC; may extend anterior to ICA
Clinical Presentation
- GT: Pulsatile tinnitus + conductive hearing loss (symptoms when tumor is still small due to proximity to TM and ossicles)
- GJ: Late presentation — often sizeable when first identified; bony destruction of jugular foramen; cranial nerve IX–XII dysfunction; facial paresis (mastoid extension); SNHL (labyrinthine erosion); pulsatile tinnitus
- Either type may erode TM and appear as a bleeding mass in the EAC
Diagnosis
- Otoscopy: Reddish-blue/vascular pulsatile retrotympanic mass ("rising sun" sign inferiorly); if all borders visible through TM → GT; if borders not fully visible → GJ must be excluded
- CT (bone windows): GT shows soft tissue on promontory; GJ shows irregular destruction of jugular foramen; absent bony septum between ICA and IJV
- MRI: "Salt and pepper" appearance on T1/T2 (arborizing flow voids from prominent vascularity); low signal flow voids on T1/T2 reflecting large vessels
- Angiography: Hypervascular pattern; essential before preoperative embolization
- Preoperative 24-hour urine metanephrines: Mandatory — 1–3% secrete catecholamines (GJ > GT)
- If arterial invasion suspected: preoperative balloon occlusion testing (99mTc-HMPAO SPECT or Xenon CT) of ipsilateral carotid
Endoscopic Images — Paraganglioma
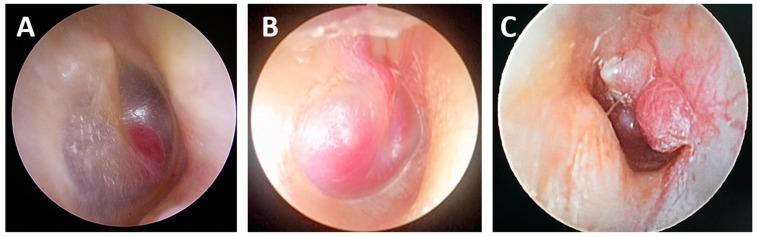
Endoscopic views of paraganglioma at different Fisch stages: A1 (small localized deep-red mass), B1 (larger pinkish-red mass filling most of middle ear), B2 (multi-lobulated mass with increased vascularity).
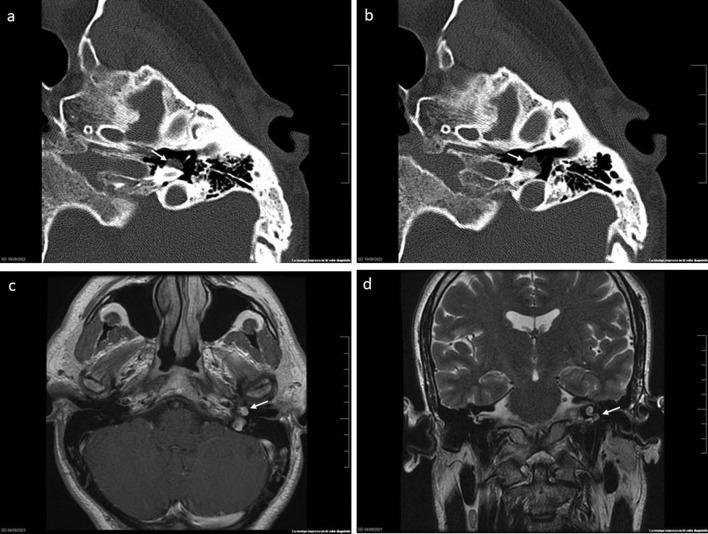
CT (axial bone windows) + MRI (T1+contrast, T2 coronal) of glomus tympanicum on the cochlear promontory with no bony erosion.
Treatment
Surgical:
- Fisch A/B (GT and small GJ): Transcanal or anterior tympanotomy for small GT limited to promontory; mastoidectomy + extended facial recess for larger GT
- Fisch A infratemporal fossa (Fisch type A) approach is standard for GJ — requires proximal control of great vessels and sigmoid sinus
- Large tumors: may need facial nerve transposition for anterior exposure
- Blood supply: ascending pharyngeal and stylomastoid arteries (branches of external carotid); contributions from ICA or vertebral possible
- Preoperative embolization significantly reduces intraoperative blood loss (well-documented benefit)
- Intraoperative vessel ligation: ascending pharyngeal, stylomastoid, caroticotympanic, superior tympanic arteries
- Lower cranial nerve preservation paramount: morbidity from CN IX–XII resection may require tracheostomy and gastrostomy
- Very large tumors: may need staged surgery (if blood loss >3 L during neck/temporal bone phase, intracranial resection proceeds at a later date)
Radiation:
- Both IMRT and stereotactic radiotherapy achieve 90–100% tumor control
- Mechanism: obliterative endarteritis
- Doses: single fraction SRS 15 Gy; external beam 40–45 Gy
- SRS preferred for Fisch C/D and GJ — fewer complications, better cranial nerve preservation vs. surgery
- Risks of SRS: radiation-induced malignancy, osteoradionecrosis of skull base, temporal lobe necrosis, cranial nerve injury
2. Vestibular Schwannoma (Acoustic Neuroma)
Epidemiology
- Most common tumor of the IAC and CPA
- 8–10% of all intracranial tumors; incidence ~1/100,000
- Arises predominantly from the superior or inferior vestibular nerve; less commonly from cochlear, facial, or other cranial nerves
- Bilateral schwannomas = Neurofibromatosis Type 2 (NF2)
Pathology
- Benign; average growth 1–2 mm/year; up to 50% show no growth on serial imaging
- Histology: Two cell populations:
- Antoni A: Dense, fibrillary, bipolar spindle cells in palisading arrangement (Verocay bodies)
- Antoni B: Loosely arranged, hypocellular, myxoid, reticulated stroma
Clinical Features
- Asymmetric sensorineural hearing loss (SNHL) — most common presentation
- Tinnitus (usually unilateral, high-pitched)
- Imbalance / unsteady gait
- Rollover on audiogram: speech discrimination score paradoxically worsens at higher presentation levels
- Larger tumors: facial paresthesia (CN V compression), facial weakness (CN VII compression), ataxia (brainstem/cerebellar compression), hydrocephalus
Diagnosis
- MRI with gadolinium — gold standard; shows strongly enhancing mass centered on IAC/CPA
- CT: smooth, funnel-shaped widening of the IAC
- ABR: prolonged Wave V latency or absent Wave V — historically used as screening test, now replaced by MRI
Koos Grading:
- Grade 1: Intracanalicular
- Grade 2: Small CPA component (<2 cm)
- Grade 3: Large CPA component without brainstem contact
- Grade 4: Large CPA component with brainstem displacement
Vestibular Schwannoma — MRI Images
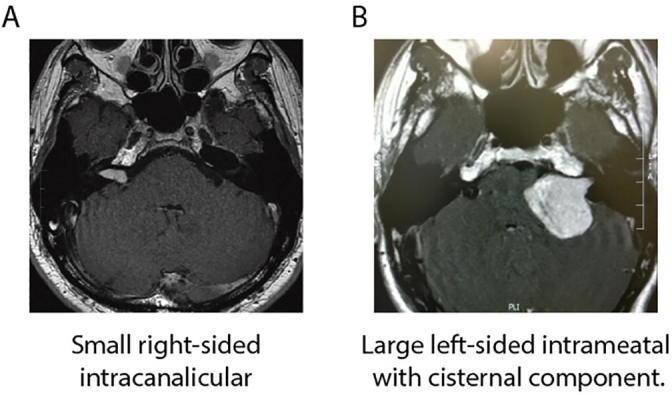
Left: Small intracanalicular (Koos 1) vestibular schwannoma. Right: Large CPA mass with brainstem displacement (Koos 3–4).
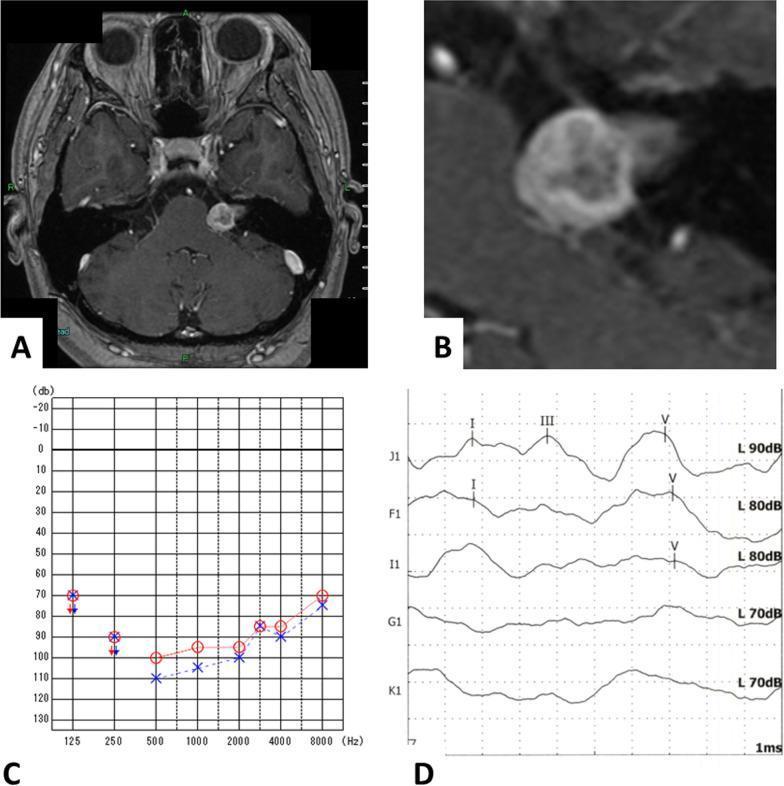
Heterogeneously enhancing IAC mass with associated profound SNHL and abnormal ABR waveforms.
Treatment
| Modality | Indications | Key Points |
|---|---|---|
| Observation | Small tumors, serviceable hearing, older patients, initial diagnosis | Serial MRI; ~50% never grow |
| Stereotactic Radiosurgery (SRS) | Tumors <2.5 cm; enlarging tumor; patient preference | 12–13 Gy; 90–95% control rate; does NOT preserve hearing; facial nerve dysfunction <1%; trigeminal paresthesia possible; malignant transformation 1:1000 at 30 years |
| Microsurgery | All sizes depending on hearing goals and tumor extent | Three approaches (below) |
Surgical Approaches — Detailed Comparison:
| Approach | Hearing | Tumor Size | Brain Retraction | Special Notes |
|---|---|---|---|---|
| Translabyrinthine | Destroys hearing (profound SNHL) | Any size | None required | Facial nerve identified at IAC fundus BEFORE tumor removal; safest facial nerve approach |
| Retrosigmoid (suboccipital) | Preservation possible if <1.5 cm, medial IAC | All sizes | Cerebellar retraction required | Higher rate of chronic postoperative headaches; supplemental endoscopy may be needed for lateral IAC tumor |
| Middle Fossa | 60–70% hearing preservation | IAC tumors <1.5 cm | Temporal lobe retraction | Rare: memory changes, seizure; ideal for purely intracanalicular or small CPA with serviceable hearing |
Most common surgical risks: CSF leak, meningitis, facial paralysis
Cochlear schwannomas (rare, within cochlea): usually observed unless intractable dizziness or growth
3. Facial Nerve Schwannoma
- Most common facial nerve tumor (hemangioma is second)
- Geniculate ganglion most commonly affected
- May involve any segment: tympanic, mastoid, labyrinthine, IAC
- Present with progressive facial weakness (House-Brackmann grade increasing over months–years); rarely: sudden onset
Imaging:
- CT: Smooth bony remodeling of facial nerve canal
- MRI: Gadolinium enhancement; tubular shape following facial nerve course
Differential from hemangioma on CT:
- Schwannoma → smooth remodeling
- Hemangioma → infiltrative erosion + stippled/honeycomb calcifications at geniculate ganglion
Treatment algorithm based on facial function:
| Facial Function | Management |
|---|---|
| Normal or near-normal | Observation with serial imaging |
| Mild early paresis | Tumor decompression |
| House-Brackmann Grade 3+ | Tumor removal + nerve grafting (improved outcome with shorter preoperative weakness duration) |
| Surgical approaches based on hearing | Hearing preserved → middle fossa or transmastoid; no serviceable hearing → translabyrinthine |
4. Hemangioma
- 0.7% of all temporal bone neoplasms; peak in 5th–6th decade
- Almost exclusively associated with the facial nerve at the geniculate ganglion; rarely IAC or tympanic/mastoid segments
- Key clinical pearl: Produces facial weakness out of proportion to tumor size (unlike schwannoma)
- Rarely causes hearing loss or vestibular symptoms
- Locally aggressive — bony destruction via nerve compression, nerve infiltration, or vascular steal
- Cochlear erosion → SNHL; middle ear extension → CHL
Imaging:
- CT: Infiltrative erosive lesion at geniculate ganglion with intratumoral stippled (honeycomb) calcifications — characteristic
- MRI: Hypo- to isointense on T1; avid enhancement with contrast; may erode cochlea or labyrinth
Treatment:
- Small lesions: Meticulous dissection from nerve — full facial function preservation possible
- Early intervention maximizes chance of anatomic and functional nerve preservation
- IAC lesions (non-paralyzed): Decompression of IAC canal (resection requires grafting)
- Large lesions / complete paralysis: Resection + nerve grafting (outcome better with short duration of preoperative weakness)
- Subtotal resection acceptable if facial function preserved — mandatory postoperative surveillance imaging
5. Endolymphatic Sac Tumor (ELST)
- Locally aggressive, slow-growing neoplasm of the endolymphatic sac or duct
- Histologic description: destructive papillary cystic adenomatous tumor of the temporal bone
- IHC profile: Positive: cytokeratin, vimentin, S-100; Negative: chromogranin (↔ paraganglioma), thyroglobulin (↔ metastatic thyroid), transthyretin/prealbumin (↔ choroid plexus tumor)
Association with Von Hippel-Lindau (VHL):
- 11% of VHL patients develop ELST; 30% of those bilateral
- VHL: loss-of-function mutation at chromosome 3p25.5 (ubiquitin ligase → fails to degrade HIF-1α → uncontrolled angiogenesis)
- VHL-associated: 3rd–4th decade, more common in females
- Sporadic: 5th–6th decade, no gender predilection
- Screening: cranial MRI every 1–3 years in VHL patients
Clinical Presentation:
- Dysfunction of the otic capsule — most common
- Symptoms mimic endolymphatic hydrops: SNHL (most common), tinnitus, vertigo, aural fullness
- Sudden SNHL — from intralabyrinthine hemorrhage
- Middle ear extension: mimics Eustachian tube dysfunction or otitis media
- Late: facial paralysis, brainstem compression, lower cranial neuropathies
- No distant metastases reported; rare drop metastasis into thecal sac
Imaging:
- CT: Bony destruction of posterior fossa plate at the operculum; central calcifications; may extend into mastoid
- MRI T1: Heterogeneous; hyperintense areas (intralesional hemorrhage — methemoglobin, hemosiderin, cholesterol); hypointense areas (calcifications, residual bone)
- MRI T2: Heterogeneous (highly vascular)
- MRI T1+Gad: Heterogeneous enhancement
- Angiography: Hypervascular; blood supply from inferior tympanic artery + dural stylomastoid branch (ascending pharyngeal and postauricular arteries); may also receive ICA or vertebral contributions
Differential: Paraganglioma, choroid plexus tumor, metastasis, eosinophilic granuloma, meningioma, arachnoid granulation, aneurysmal bone cyst
Treatment:
- Surgery — method of choice; must remove both surfaces of the dura for complete resection
- Hearing-sparing approaches (retrolabyrinthine–transdural) for small tumors
- Translabyrinthine for nonserviceable hearing
- Preoperative embolization for large tumors to minimize blood loss
6. Meningioma (CPA/Temporal Bone)
- Second most common CPA tumor (after vestibular schwannoma)
- Arises from arachnoid cap cells
- WHO Grade classification:
- Grade 1 (Benign): Meningothelial, fibrous, transitional, psammomatous, angiomatous, secretory, lymphoplasmacyte-rich, metaplastic
- Grade 2 (Atypical): Chordoid, clear cell, atypical (mitoses, brain invasion)
- Grade 3 (Anaplastic/Malignant): Papillary, rhabdoid, anaplastic
- Histology: Psammoma bodies (concentric calcified lamellae) — characteristic but not pathognomonic
Imaging features distinguishing meningioma from vestibular schwannoma:
- Dural tail (enhancing dural thickening extending from tumor)
- Eccentric location relative to IAC (does not funnel into it)
- Calcifications
- Hyperostosis of adjacent bone
- More homogeneous enhancement
- Bone algorithm CT essential
Symptoms: SNHL, tinnitus, imbalance, facial paresthesia/numbness, diplopia, ataxia — depending on location and size
Clinical note: Hearing preservation (or even improvement) more likely with meningioma than with vestibular schwannoma.
7. Osteoma and Exostosis of the EAC
Both are benign bony outgrowths of the external auditory canal and are histopathologically identical but clinically distinct:
| Feature | Osteoma | Exostosis |
|---|---|---|
| Cause | Spontaneous / idiopathic | Cold water exposure ("surfer's ear") |
| Laterality | Unilateral | Bilateral |
| Shape | Pedunculated (on a stalk) | Broad-based |
| Location | Lateral to bony isthmus | Multiple; closer to TM |
| Multiplicity | Usually single | Multiple outgrowths |
Clinical significance: May cause sufficient narrowing of the EAC to require surgery (conductive hearing loss, recurrent otitis externa from trapped keratin)
Radiology questions to answer on CT (HRCT/CBCT):
- Maximum depth and transverse diameter of the exostosis
- Exact site of origin of the osteoma
- Distance between the medial aspect of exostosis and TM and between the deep aspect and facial nerve canal
- Obstructed secretions medial to the exostosis?
- Is the middle ear cleft normally pneumatized?
Treatment: Surgical canaloplasty (meatoplasty) when symptomatic
8. Middle Ear Adenomatous Neuroendocrine Tumor
- Dual neuroendocrine + glandular differentiation
- Sex ratio: M:F = 2:1
- Symptoms: hearing loss, aural fullness, tinnitus, otalgia
- CT: similar to tympanic paraganglioma; may occur in atypical positions
- IHC: Positive: pan-cytokeratins, chromogranin, synaptophysin, insulinoma-associated protein 1 (INSM1)
- Rare regional and distant metastatic potential
- Treatment: surgical resection + long-term follow-up (recurrence risk)
9. Choristoma (Middle Ear / IAC Lipoma)
- Histologically normal tissue in abnormal location (definition of choristoma)
- Most common type: salivary gland tissue; less common: glial choristoma
- Middle ear location → conductive hearing loss + effusion
- IAC location → SNHL, tinnitus, imbalance
- MRI: Hyperintense on T1 without contrast; no enhancement on fat-saturation gadolinium sequences (key distinction from schwannoma)
- Unlike ELST or schwannoma: no surgical removal possible (inseparable from facial nerve in most cases)
- Treatment: serial imaging and observation
PART II — MALIGNANT TUMORS
1. Squamous Cell Carcinoma (SCC) of the Temporal Bone
Epidemiology
- Most common primary malignancy of the temporal bone
- Frequency order: SCC > BCC > Adenocarcinoma > Adenoid cystic carcinoma > Mucoepidermoid carcinoma > Ceruminoma > Melanoma > Sarcoma
- Risk factors: chronic otitis media, chronic irritation/inflammation, prior radiation, cholesteatoma
Clinical Presentation (in decreasing frequency):
- Otalgia
- Otorrhea (often blood-stained)
- Hearing loss
- Facial palsy
- Parotid mass
Preoperative Evaluation
- Audiogram
- CT temporal bone + neck with IV contrast (assess bony erosion and lymph nodes)
- CT chest with IV contrast / PET-CT (distant metastases)
Staging — Modified University of Pittsburgh System
| T Stage | Extent of Disease |
|---|---|
| T1 | Tumor limited to EAC; no bony erosion or soft tissue involvement |
| T2 | Limited bony erosion (not full-thickness) OR <0.5 cm soft tissue involvement |
| T3 | Erosion through bony EAC with <0.5 cm soft tissue; OR middle ear/mastoid involvement |
| T4 | Medial wall erosion (cochlea, labyrinth, petrous apex, jugular foramen, carotid canal, dura); >0.5 cm soft tissue (TMJ, styloid, parotid); facial paresis/paralysis |
Surgical Resection Types
| Surgery | Tissues Removed | Medial Limit |
|---|---|---|
| Sleeve resection | EAC skin, cartilaginous EAC portion, ± TM | Leaves bony EAC intact |
| LTBR (Lateral Temporal Bone Resection) | En bloc: cartilaginous + bony EAC, TM, malleus, incus ± parotidectomy, neck dissection, mandibular condyle | Medial: stapes |
| STBR (Subtotal TBR) | LTBR + middle ear/mastoid contents, otic capsule, medial wall ± facial nerve, dura, sigmoid | IAC, petrous apex with neurovascular structures |
| TTBR (Total TBR) | STBR + petrous apex + neurovascular bundle ± ICA, clivus | No proven survival benefit; rarely performed |
Adjuvant Treatment
- Postoperative radiation (50–60 Gy) offered to all T2, T3, T4 disease
- Chemotherapy considered for T4 tumors, residual disease, or metastatic disease
- Parotidectomy + neck dissection performed for locoregional spread; prophylactic parotidectomy ± neck dissection for T3/T4
Survival by T Stage
- T1: ~90% 5-year
- T2: ~70–80% 5-year
- T3: ~50–60% 5-year
- T4: <30% 5-year
SCC Temporal Bone — Imaging
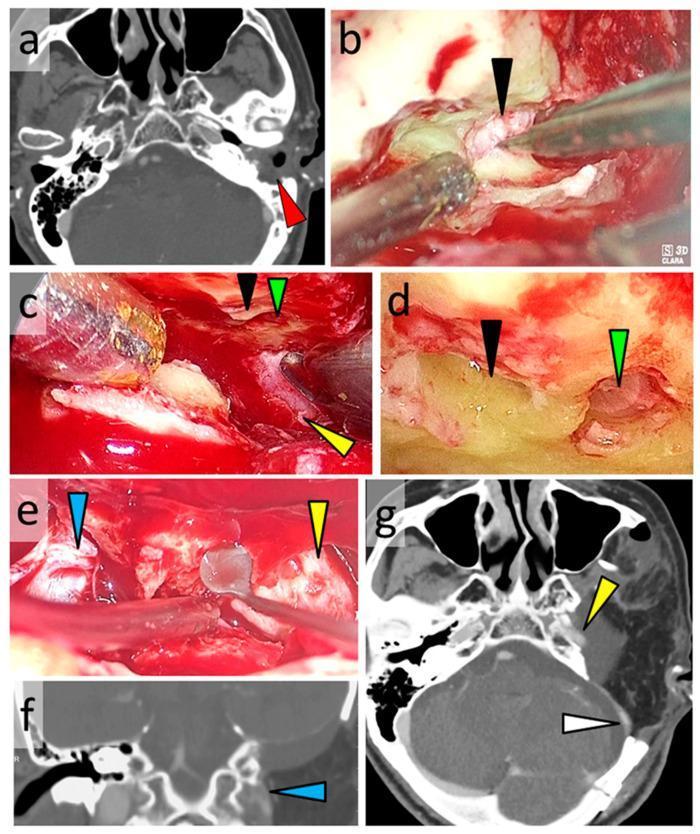
CT showing EAC SCC with mastoid involvement (left). Intraoperative exoscopic views showing IAC, cochlea, ICA, and jugular foramen during subtotal temporal bone resection.
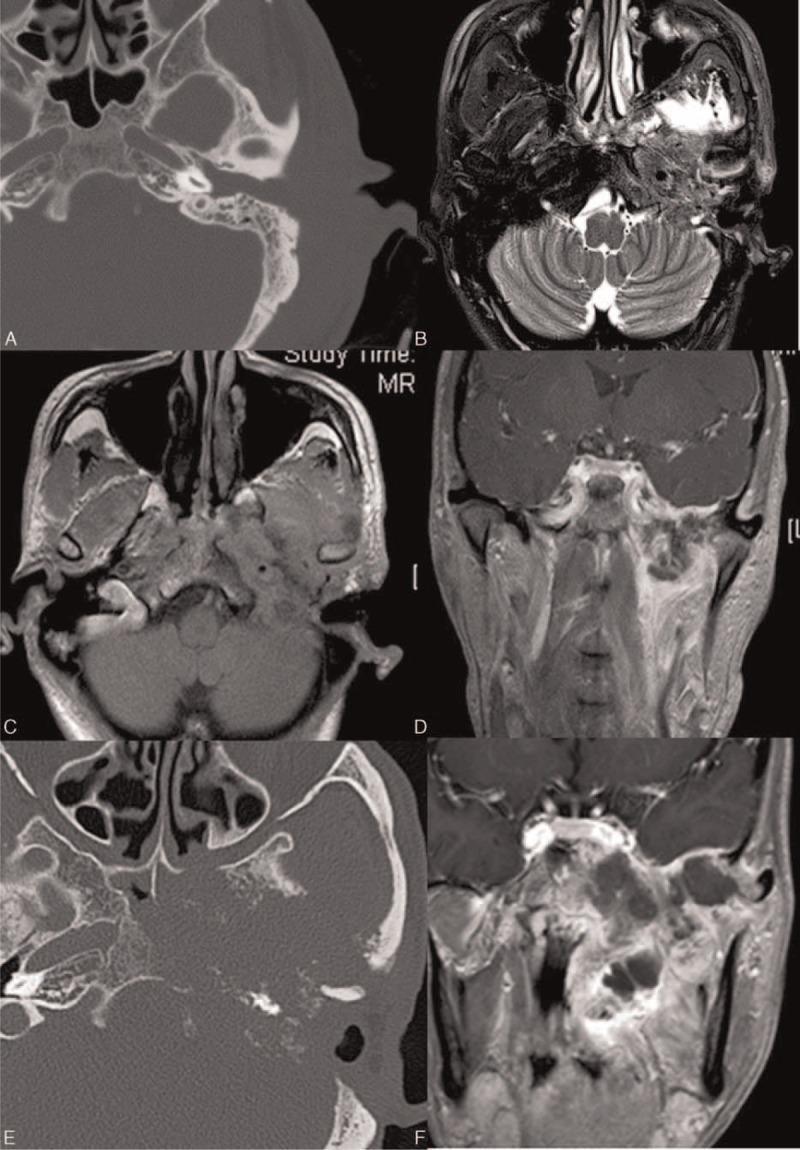
Advanced T4 SCC: HRCT demonstrates 'moth-eaten' petrous apex erosion; MRI shows carotid encasement, sigmoid sinus involvement, and dural invasion.
2. Rhabdomyosarcoma (RMS)
- Most common pediatric temporal bone malignancy; most common pediatric soft-tissue sarcoma overall
- 35–40% of all RMS are in the head and neck; only 5% in the temporal bone; temporal bone is a parameningeal site → unfavorable, minimum Stage II
- Origin: embryologic skeletal muscle cells
- Can arise from EAC, middle ear, or mastoid cavity
Symptoms
Otalgia, otorrhea, hearing loss, headache, aural polyp, facial nerve weakness, regional lymphadenopathy
Histological Classification
| Type | Notes |
|---|---|
| Embryonal (most common in children) | Includes conventional, spindle cell, and botryoid subtypes |
| Alveolar | Worse prognosis than embryonal |
| Pleomorphic | Most common in adults |
- Spindle cell and botryoid: more favorable outlook
- Alveolar > embryonal in aggressiveness
Staging — Children's Oncology Group (COG) System
| Stage | Site | T | N | M |
|---|---|---|---|---|
| I | Orbit, HN (non-parameningeal), GU non-bladder | T1 or T2a/b | N0 or N1 | M0 |
| II | Bladder/prostate, extremity, cranial/parameningeal | T1 or T2a/b | N0 | M0 |
| III | Same | T1 or T2a/b | N1 | M0 |
| IV | All sites | Any T | N0/N1 | M1 |
T2 = tumor invades structures outside EAC/tympanomastoid space; "a" = <5 cm, "b" = >5 cm
Surgical group assigned after resection (Groups 1–4: complete resection to distant metastasis)
Treatment
- Debate: Upfront resection vs. biopsy → primary chemoradiation
- Many advocate wide surgical resection — extent of resection determines prognosis
- Most pediatric high-grade: Surgery + 50–70 Gy EBRT + multidrug chemotherapy
- SRS for residual/recurrent disease at jugular foramen or perineural invasion into IAC/Meckel cave/cavernous sinus
Radiation risks in children: Eustachian tube dysfunction, osteoradionecrosis, SNHL, cataract, impaired cranial bone/tooth development, pituitary dysfunction, chronic headache
Prognosis
- Low risk: ~90% 5-year survival
- Intermediate risk: 60–80%
- High risk: 20–40%
3. Chondrosarcoma
- Rare cartilaginous malignancy; 1% occur at skull base, typically at spheno-occipital or petroclival synchondroses
- Rarely metastasizes; morbidity from local growth
- WHO Grades 1–2 (low grade): significantly more frequent; better prognosis
- WHO Grade 3 / mesenchymal subtype (high grade): worse prognosis
- IHC: Non-reactive for keratin and EMA (contrast with chordoma which is keratin+)
Imaging: Similar to chordoma; tends to be eccentrically located; calcifications common; no hallmark to distinguish low from high grade
Treatment: Surgery + radiation (proton beam / carbon ion preferred)
Prognosis (SEER):
- 5-year survival: 87%; 10-year: 68% (substantially higher for low-grade alone)
- Better prognosis: younger age, female sex, smaller tumor
4. Chordoma
- Derived from notochord remnants; 33% of all chordomas occur at the skull base
- Median age of presentation: 60 years (also in children)
- Metastases rare at presentation; morbidity/mortality from local progressive growth
- Frequent presenting symptom: headache
Imaging (classic):
- T1-MRI: Hypointense
- T2-MRI: Hyperintense (key characteristic)
- Bone destruction at skull base on CT
Treatment:
- Surgery + radiation; proton beam / carbon ion therapy most commonly recommended
- 5-year local control rates: >70%
Prognosis (SEER):
- Median overall survival: 7.7 years
- Age <40 years: 68% 10-year survival
- Age >40 years: 43% 10-year survival
- Gross or near-total resection: 6× higher 5-year survival vs. subtotal
5. Ceruminous Gland Tumors (EAC)
Arising from the specialized apocrine ceruminous glands of the outer EAC:
| Tumor | Behavior | Notes |
|---|---|---|
| Ceruminous adenoma | Benign | Well-circumscribed; local excision curative |
| Ceruminous adenocarcinoma | Malignant | Low or high grade; wide excision required |
| Adenoid cystic carcinoma | Malignant | Perineural invasion characteristic; late recurrence common; may occur decades later |
| Mucoepidermoid carcinoma | Malignant | Intermediate behavior |
| Sebaceous adenocarcinoma, papillary cystadenocarcinoma | Malignant | Rare |
All ceruminous gland malignancies require wide surgical resection; adenoid cystic carcinoma specifically requires long-term follow-up given propensity for late, perineural recurrence.
6. Skull Base Malignancies Extending to the Ear
Jugular Foramen Schwannomas (Lower CN)
- Originate from CN IX, X, or XI at jugular foramen
- May be intracranial, infraforaminal, cervical, or combined
- CT: Smooth symmetric enlargement of jugular foramen (vs. paraganglioma's irregular destruction)
- Treatment: suboccipital approach for intracranial component; Fisch infratemporal fossa approaches for infraforaminal
Metastatic Tumors to the Temporal Bone
Common primaries:
- Prostate (most common)
- Breast
- Renal cell carcinoma
- Lung, GI, multiple myeloma, lymphoma, leukemia (chloroma)
- Always suspect in patients with known malignancy presenting with new unilateral hearing loss or facial palsy
Key Imaging Differentiation Summary
| Tumor | CT Appearance | MRI Appearance |
|---|---|---|
| Glomus tympanicum | Soft tissue on cochlear promontory; no bony erosion | Enhancing vascular mass |
| Glomus jugulare | Irregular jugular foramen destruction; absent ICA-IJV septum | "Salt and pepper" T1/T2; flow voids |
| Vestibular schwannoma | Smooth funnel-shaped IAC widening | Enhancing mass centered on IAC/CPA; isointense T1, hyperintense T2 |
| Meningioma | Hyperostosis; calcification; eccentric to IAC | Dural tail; homogeneous enhancement |
| Hemangioma | Infiltrative erosion at geniculate; honeycomb calcifications | Avid enhancement; T1 hypo- to isointense |
| Endolymphatic sac tumor | Posterior fossa plate destruction; central calcifications | Heterogeneous T1 (hyperintense hemorrhage); heterogeneous T2; heterogeneous enhancement |
| Facial nerve schwannoma | Smooth remodeling of facial nerve canal | Tubular enhancement along CN VII |
| Cholesteatoma | Bony erosion of ossicular chain/tegmen/EAC | Restricted diffusion on DWI (non-EPI) |
| SCC temporal bone | Soft tissue mass ± bony erosion ± mastoid involvement | Enhancing soft tissue; perineural spread along CN VII/V |
| Chordoma | Midline/skull base bone destruction | Hyperintense T2 — characteristic |
| Chondrosarcoma | Eccentric; calcifications; petroclival | Similar to chordoma but off-midline |
| Choristoma/Lipoma IAC | Normal bony canal | Hyperintense T1; no fat-sat enhancement |
Primary Sources:
- K.J. Lee's Essential Otolaryngology, Ch. 24 "Tumors of the Temporal Bone", pp. 481–493
- Cummings Otolaryngology Head and Neck Surgery, Ch. 178 (Temporal Bone Neoplasms), pp. 3404–3436; Ch. 179 (Glomus Tumors), pp. 3436–3489
- Grainger & Allison's Diagnostic Radiology, Ch. 61 (Temporal Bone), pp. 1589–1590
This is a shared conversation. Sign in to Orris to start your own chat.