Give me full A2 style detailed answer with all introduction all etiology all pathology all pathophysiology all classification all pathogenesis all clinical features all diagnosis all treatment all hrct findings and drug with doses newer therapy from fishman and ers guidelines of bronchiectasis
Checking Date & Time
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Searching Images
bronchiectasis HRCT CT scan cylindrical varicose cystic
Reading File
Finding Sources
Searching PubMed
bronchiectasis management ERS guidelines treatment
Reading Article
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
I now have comprehensive data from all sources. Let me compose the full A2-style answer.
BRONCHIECTASIS — Comprehensive A2 Answer
Sources: Fishman's Pulmonary Diseases and Disorders (2-Volume Set), Murray & Nadel's Textbook of Respiratory Medicine, Harrison's Principles of Internal Medicine 22E (2025), Robbins & Kumar Pathologic Basis of Disease, Grainger & Allison's Diagnostic Radiology; ERS Clinical Practice Guideline 2025 (Chalmers et al., PMID 41016738)
1. INTRODUCTION & DEFINITION
Bronchiectasis is a chronic, irreversible structural disorder of the lung characterized by permanent, abnormal dilatation of the bronchi due to destruction of the bronchial wall's muscular and elastic components — resulting from persistent inflammation, infection, and impaired mucociliary clearance.
The condition was first formally described by René Laennec in the early 19th century. It is defined clinically by the triad of:
- Chronic productive cough
- Daily sputum production
- Recurrent respiratory tract infections/exacerbations
Epidemiology:
- Prevalence: 52–139 cases/100,000 in the USA (increasing by ~8.7%/year)
- Total US cases estimated >110,000
- More common in women (1.3–1.6× higher) and Asians (2.5–3.9× higher vs. Caucasians/African Americans)
- Prevalence increases with age
- European data show incidence and point prevalence rising yearly from 2004–2013
- Significant economic burden: increased hospitalization vs. age-matched controls
- Associated with impaired QoL and mortality (influenced by sputum volume, airflow obstruction severity, chronic gram-negative infection — especially P. aeruginosa)
— Fishman's Pulmonary Diseases and Disorders, Chapter 51
2. ETIOLOGY
Bronchiectasis is a heterogeneous syndrome with many contributing etiologies. Etiology determines the pattern of lung involvement:
A. Focal Bronchiectasis (localized)
- Bronchial obstruction — extrinsic (lymphadenopathy, tumor compression) or intrinsic (endobronchial tumor, aspirated foreign body, scarring/stenosis, bronchial atresia)
B. Diffuse Bronchiectasis (bilateral/widespread)
| Category | Examples |
|---|---|
| Post-infectious | Bacterial pneumonia (Staphylococcus, Klebsiella, Mycobacterium tuberculosis), severe viral pneumonia, whooping cough, measles, NTM |
| Genetic/Congenital | Cystic fibrosis (CF), Primary Ciliary Dyskinesia (PCD), Kartagener syndrome, α1-antitrypsin deficiency, Williams-Campbell syndrome (cartilage deficiency), Mounier-Kuhn syndrome (tracheobronchomegaly), intralobar sequestration |
| Immunodeficiency | Hypogammaglobulinemia (IgA, IgG subclass, common variable immunodeficiency), HIV, post-transplant bronchiolitis obliterans |
| Autoimmune/Rheumatologic | Rheumatoid arthritis, Sjögren's syndrome, SLE, inflammatory bowel disease |
| Allergic/Immune-mediated | Allergic bronchopulmonary aspergillosis (ABPA) — central/upper lobe predilection |
| Recurrent aspiration | Neuromuscular disease, GERD |
| Toxic inhalation | Ammonia, chlorine |
| Traction bronchiectasis | Idiopathic pulmonary fibrosis, post-radiation fibrosis (parenchymal traction on airways) |
| Miscellaneous | Yellow nail syndrome, Young's syndrome |
| Idiopathic | Up to 50% of cases — dysfunctional host immunity to infectious agents |
Distribution Clues to Etiology:
- Upper lobe: CF, ABPA, post-TB
- Lower lobe: Post-infectious, aspiration, immune deficiency
- Middle lobe/lingula (Lady Windermere pattern): NTM infection (MAC)
- Central: ABPA
— Harrison's 22E, Table 30-1; Robbins Pathologic Basis of Disease, p. 642
3. PATHOGENESIS
Cole's "Vicious Cycle" Hypothesis (Key Framework)
Peter Cole described the foundational conceptual model:
Initial insult → Neutrophilic airway inflammation → Airway destruction in susceptible individuals → Anatomic disruption → Abnormal mucus clearance → Chronic bacterial infection → Further airway inflammation → Perpetuating cycle
Molecular Steps:
- Initial insult (infection, obstruction, inflammation) damages ciliated airway epithelium
- Bacteria form biofilms; glycoproteins and IL-8 drive neutrophil recruitment
- Human Neutrophil Elastase (HNE) — a key proteolytic enzyme — cleaves bronchial wall structural proteins (elastin, collagen, smooth muscle)
- Reactive oxygen species and proinflammatory cytokines (TNF-α, IL-1β) amplify destruction
- Goblet cell hyperplasia → excess mucus production
- Impaired mucociliary clearance → mucus stasis → bacterial colonization
- Loss of elastin and smooth muscle → permanent airway dilation
- Smaller bronchioles become obliterated by fibrosis (bronchiolitis obliterans)
- Antiproteases (α1-antitrypsin) are overwhelmed — hence α1-AT deficiency worsens the process
In Cystic Fibrosis: CFTR mutation → defective ion transport → thick viscous secretions → mucostasis → chronic Pseudomonas and Staphylococcus infections → widespread airway wall destruction
In PCD: Dynein arm mutations → ciliary dysfunction → recurrent infections → bronchiectasis; ~50% have situs inversus (Kartagener syndrome)
In ABPA: Th2-driven immune response against Aspergillus antigens → proximal bronchial wall damage → central bronchiectasis with mucus plugging
Traction bronchiectasis: No infection required — peripheral fibrosis mechanically distorts and dilates airways
— Fishman's Ch. 51; Robbins Pathologic Basis of Disease p. 642, 668–674
4. PATHOLOGY
Gross Pathology
- Dilated, thick-walled bronchi extending to the lung periphery (normally bronchi do not extend within 1 cm of pleura)
- Airways may be filled with mucopurulent secretions
- Surrounding lung parenchyma: fibrosis, consolidation, and collapse (atelectasis) — especially lower lobes
- In severe disease: lung abscess formation
Microscopic Pathology
- Transmural inflammation — chronic inflammatory cell infiltrate in all layers of the bronchial wall
- Destruction of smooth muscle and elastic tissue — hallmark of irreversibility
- Goblet cell hyperplasia and mucous gland hypertrophy
- Squamous metaplasia of bronchial epithelium
- Fibrosis of bronchial wall and peribronchial tissue
- Bronchiolitis obliterans in small airways
- Neovascularization — bronchial arteries hypertrophy (source of hemoptysis)
- Mucous plugging in smaller airways
- Surrounding alveoli: pneumonia, fibrosis, and emphysema
The three-mechanism pathological model (Grainger & Allison):
- Bronchial obstruction → mucus plugging + infection → airway damage
- Bronchial wall damage → direct infection/inflammation
- Parenchymal fibrosis → traction bronchiectasis
— Robbins Pathologic Basis of Disease; Grainger & Allison's Diagnostic Radiology
5. CLASSIFICATION
A. Morphological Classification (Reid, 1950; most widely used on HRCT)
| Type | Description | Severity |
|---|---|---|
| Cylindrical (Tubular) | Uniform, parallel bronchial dilation without tapering; "tram-track" appearance | Mild — most common |
| Varicose | Irregular, beaded, serpiginous dilation ("string of pearls"); alternating areas of constriction and dilation | Moderate |
| Cystic (Saccular) | Marked sac-like dilation forming "cluster of grapes"; often with air-fluid levels | Severe |
B. Anatomical Distribution
- Focal — single lobe/segment (usually obstructive cause)
- Multifocal — multiple lobes
- Diffuse — bilateral widespread (systemic cause)
C. Etiological Classification
- CF-related bronchiectasis
- Non-CF bronchiectasis (the broader clinical entity, focus of ERS 2025 guidelines)
D. By Bronchial Distribution (Anatomy)
- Upper lobe (ABPA, CF, post-TB)
- Lower lobe (post-infectious, aspiration)
- Middle lobe / lingula (NTM)
- Right middle lobe syndrome (specific entity)
— Harrison's 22E; Grainger & Allison's Diagnostic Radiology; Fishman's Ch. 51
6. PATHOPHYSIOLOGY
Pulmonary Function
- Obstructive ventilatory defect predominates (reduced FEV1/FVC)
- Increased RV and TLC (air trapping)
- Reduced DLCO in advanced disease
- The obstructive defect is primarily due to obliterative bronchiolitis (small airway disease), NOT large airway collapse or mucus plugging
- Mixed obstructive-restrictive pattern in fibrotic cases
Gas Exchange
- V/Q mismatch → hypoxemia
- Chronic hypoxemia → pulmonary vasoconstriction → pulmonary hypertension
- Cor pulmonale in advanced disease
Mucociliary Dysfunction
- Impaired cilia → mucus stasis → promotes colonization
- Daily mucus production >30 mL/day
Hemoptysis Mechanism
- Hypertrophied bronchial arteries (neovascularization at inflammation sites)
- Erosion of superficial mucosal vessels by recurrent infection
- Massive hemoptysis: life-threatening (>200–300 mL/24h)
Systemic Effects
- Chronic inflammatory state → cachexia, weight loss
- Amyloidosis (rare, secondary AA)
- Metastatic abscess (brain) — rare
7. CLINICAL FEATURES
Symptoms
| Symptom | Details |
|---|---|
| Chronic productive cough | Most common; daily; often worse in mornings |
| Sputum production | >30 mL/day; mucoid, mucopurulent, or purulent; "three-layer" sputum (foam/mucus/pus) on standing |
| Hemoptysis | Common; may be mild streaking to massive; life-threatening in severe cases |
| Dyspnea | Proportional to extent of disease |
| Pleuritic chest pain | During exacerbations |
| Fever, malaise, fatigue | During exacerbations |
| Weight loss | Chronic disease burden |
| Rhinosinusitis | Common association (upper airway disease) |
| Wheezing | Bronchospasm; associated asthma |
Physical Examination
| Finding | Description |
|---|---|
| Crackles (coarse) | Most characteristic; bilateral in diffuse disease; basal predominance |
| Wheeze | Reversible airflow obstruction |
| Digital clubbing | ~3–7%; correlates with disease severity |
| Cyanosis | Advanced disease |
| Barrel chest | Air trapping/hyperinflation |
| Signs of cor pulmonale | JVD, peripheral edema, loud P2 — advanced disease |
| Nasal polyps | CF association |
Exacerbation Features
- Increased sputum volume and purulence
- Worsening dyspnea
- New or increased hemoptysis
- Fever ± new infiltrate
- May NOT have fever/infiltrate (unlike pneumonia)
— Harrison's 22E; Fishman's Ch. 51; Murray & Nadel's Respiratory Medicine
8. DIAGNOSIS
Clinical Diagnostic Criteria (International Expert Consensus)
Radiological criteria (at least ONE of the following on chest CT):
- Inner or outer airway-artery diameter ratio ≥1.5
- Lack of airway tapering
- Visibility of airways in the periphery (within 1 cm of pleura)
Clinical syndrome (at least TWO of the following):
- Cough most days of the week
- Sputum production most days of the week
- History of exacerbations
Chest X-Ray Findings
- Increased bronchial wall thickening — "tram-tracks" (dilated airways en face)
- Ring shadows (dilated airways seen end-on)
- "Tubular shadows" or "gloved finger" opacities (mucus-filled bronchi)
- Multiple thin-walled ring shadows with air-fluid levels (cystic)
- Volume loss/atelectasis in affected lobes
- Overinflation (generalized disease/CF)
- Poor sensitivity; may be normal in mild disease
Sputum Microbiology
- Haemophilus influenzae (most common colonizer)
- Pseudomonas aeruginosa (key prognostic organism; associated with accelerated decline)
- Staphylococcus aureus (CF especially)
- Moraxella catarrhalis
- Streptococcus pneumoniae
- Nontuberculous mycobacteria (NTM) — MAC most common
- Culture for AFB and fungi in appropriate clinical contexts
Pulmonary Function Tests
- FEV1/FVC: obstructive pattern
- ± Mixed pattern
- Baseline PFTs and serial monitoring recommended
Additional Investigations for Underlying Cause
| Investigation | Disorder Sought |
|---|---|
| Sweat chloride; CFTR mutation testing | Cystic fibrosis |
| Nasal brush/biopsy; genetic testing (dynein genes) | Primary ciliary dyskinesia |
| Serum immunoglobulins (IgG, IgA, IgM, IgE) | Hypogammaglobulinemia, CVID |
| IgE; IgE anti-Aspergillus; Aspergillus skin test; eosinophils | ABPA |
| RF, ANA, anti-CCP, anti-Ro/La | Rheumatoid arthritis, Sjögren's |
| HIV serology | HIV-related immunodeficiency |
| α1-antitrypsin level | α1-AT deficiency |
| CBC + differential | Neutrophil disorders |
| Ciliary electron microscopy (TEM) | PCD ultrastructural defects |
— Harrison's 22E; Fishman's Ch. 51
9. HRCT FINDINGS
HRCT (thin-section, ≤1 mm collimation) is the gold standard for diagnosis and morphological characterization of bronchiectasis.
Primary HRCT Signs
| Sign | Description |
|---|---|
| Signet-Ring Sign ⭐ | Cross-section of dilated bronchus with adjacent smaller pulmonary artery; bronchus:artery ratio >1:1 (normally <1:1, threshold ≥1.5:1 diagnostic); the ring (bronchus) with the "gemstone" (artery) = signet ring |
| Tram-Track Sign | Two parallel lines (thickened bronchial walls) when bronchus is imaged along its long axis |
| Lack of Tapering | Cardinal sign — bronchial lumen does not narrow toward periphery |
| Peripheral Bronchi Visibility | Airways visible within 1 cm of costal pleura or abutting mediastinal pleura (normal airways not visible here) |
| Bronchial Wall Thickening | Thickened walls of dilated bronchi |
CT Morphological Types
| HRCT Type | Appearance |
|---|---|
| Cylindrical | Parallel "tram-track" lines; uniform dilation; failure to taper |
| Varicose | Beaded/irregular appearance; "string of pearls"; alternating dilation and constriction |
| Cystic | Cluster of cysts/"bunch of grapes"; most severe; may contain air-fluid levels; extends to subpleural surface |
Additional/Associated HRCT Findings
| Finding | Significance |
|---|---|
| Tree-in-bud pattern | Centrilobular nodules + branching lines; bronchiolitis/mucus plugging in small airways |
| Mucoid impaction | Glove-finger, V- or Y-shaped tubular densities (branching opacities) |
| Mosaic attenuation/perfusion | Obliterative bronchiolitis causing regional air trapping |
| Expiratory air trapping | Confirms obliterative bronchiolitis |
| Volume loss/atelectasis | Lobar/segmental collapse from chronic obstruction |
| Peribronchial consolidation | Active infection superimposed |
| Pleural thickening | Chronic disease |
| Lung cysts | In severe/chronic cases |
CT Distribution Clues to Etiology
- Bilateral upper lobe → CF, ABPA, post-TB
- Bilateral lower lobe → Post-infectious, aspiration, immunodeficiency
- Central/proximal bronchi → ABPA (pathognomonic central bronchiectasis with mucoid impaction)
- Right middle lobe + lingula ("Lady Windermere") → NTM (MAC)
CT-Based Scoring Systems
- Bhalla score — severity scoring based on number of bronchopulmonary segments involved
- Reiff score — number of segments affected + type (cylindrical = 1, varicose = 2, cystic = 3)
— Grainger & Allison's Diagnostic Radiology p. 4299–4314; Harrison's 22E; Fishman's Ch. 51
HRCT Images:
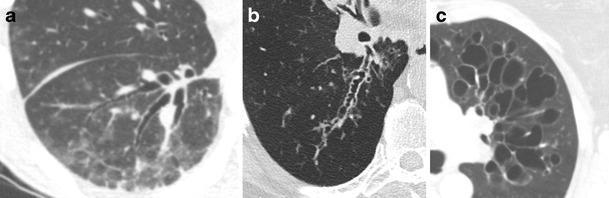
HRCT classification: (a) Cylindrical bronchiectasis — uniform dilation, tram-track pattern; (b) Varicose bronchiectasis — beaded/irregular appearance; (c) Cystic bronchiectasis — cluster-of-grapes configuration
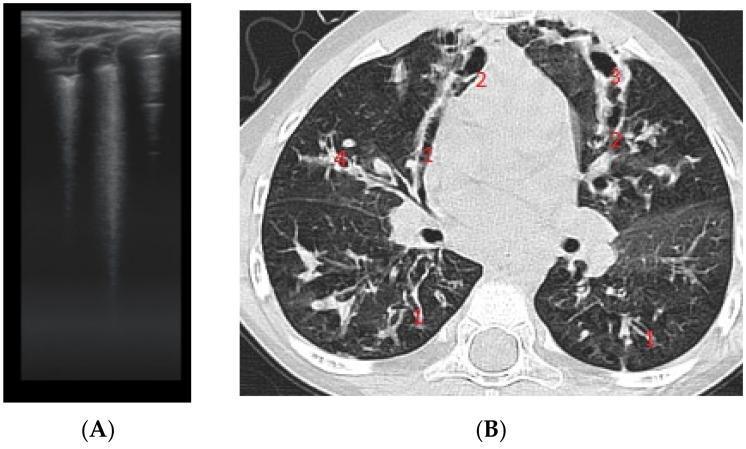
Multi-morphic bronchiectasis: (1) cylindrical, (2) varicose, (3) saccular/cystic, (4) mucus plugging. Signet-ring sign present.
10. TREATMENT
Management of bronchiectasis targets five core goals: (1) treat/identify the underlying cause; (2) enhance secretion clearance; (3) control infection; (4) reduce inflammation; (5) manage complications.
A. AIRWAY CLEARANCE TECHNIQUES (ACT) — ERS 2025: Strong Recommendation
- Chest physiotherapy (CPT) — postural drainage, manual percussion — cornerstone
- Active Cycle of Breathing Technique (ACBT)
- Oscillatory Positive Expiratory Pressure (OPEP) devices: Flutter valve, Acapella, RC-Cornet
- High-Frequency Chest Wall Oscillation (HFCWO) vest
- Minimum: 2×/day; individualized to disease severity
- Goal: clear secretions, reduce bacterial load, break vicious cycle
B. MUCOACTIVE AGENTS
| Agent | Dose | Notes |
|---|---|---|
| Inhaled Hypertonic Saline (7%) | 4 mL nebulized BD | Increases mucus hydration, mucociliary clearance; ERS 2025: conditional recommendation |
| Mannitol (inhaled dry powder) | 400 mg BD | Osmotic; improves clearance; conditionally recommended ERS 2025 |
| Carbocisteine | 750 mg TDS oral | Mucoregulator; reduces exacerbations |
| N-Acetylcysteine | 600 mg BD oral | Mucolytic + antioxidant |
| Dornase alfa (DNase) | 2.5 mg once or twice daily inhaled | Recommended in CF-related bronchiectasis ONLY; NOT recommended in non-CF (lack of efficacy + potential harm) |
| Erdosteine | 300 mg BD | Emerging evidence |
| Bromhexine | 8–16 mg TDS oral | Mucolytic |
C. BRONCHODILATORS
| Drug | Class | Dose |
|---|---|---|
| Salbutamol (Albuterol) | SABA | 100–200 μg inhaled PRN/BD (preferably before ACT) |
| Ipratropium bromide | SAMA | 20–40 μg QID inhaled |
| Formoterol | LABA | 12 μg BD inhaled |
| Tiotropium | LAMA | 18 μg once daily inhaled |
| Salmeterol | LABA | 50 μg BD inhaled |
| Salbutamol + ipratropium nebulized | Combination | Particularly in acute exacerbations |
- Inhaled beta-2 agonists improve mucociliary clearance and reverse bronchoconstriction
- No strong RCT evidence in non-CF bronchiectasis; used by analogy and clinical benefit
D. ANTIBIOTIC THERAPY
1. Acute Exacerbations
-
Duration: 14 days (minimum 7–10 days)
-
Oral therapy (mild-moderate exacerbations):
- Amoxicillin-clavulanate 625 mg TDS (H. influenzae, S. pneumoniae, M. catarrhalis)
- Ciprofloxacin 500–750 mg BD (Pseudomonas aeruginosa or gram-negatives)
- Azithromycin 500 mg OD × 3 days (alternative)
- Levofloxacin 500–750 mg OD
- Doxycycline 100 mg BD
-
IV therapy (severe exacerbations / Pseudomonas):
- Piperacillin-tazobactam 4.5 g TID IV
- Ceftazidime 2 g TID IV
- Meropenem 1 g TID IV
- Ciprofloxacin 400 mg BD IV ± aminoglycoside (gentamicin 5–7 mg/kg OD IV)
- Tobramycin 5–7 mg/kg OD IV (with level monitoring)
- Colistin (polymyxin E) 2–3 MU TID IV (MDR Pseudomonas)
2. Long-Term/Chronic Oral Antibiotic Therapy (LTAT)
| Drug | Dose | Indication / Notes |
|---|---|---|
| Azithromycin ⭐ | 250 mg OD or 500 mg 3×/week | Most evidence; reduces exacerbations (EMBRACE, BAT, BLESS trials); ERS 2025: Strong Recommendation for high-risk patients |
| Erythromycin | 250–500 mg BD oral | Alternative macrolide; reduces exacerbations (BLESS trial) |
| Roxithromycin | 150 mg BD | Used in Asian populations (BAT trial) |
Macrolides work both by antimicrobial and anti-inflammatory mechanisms (reduce IL-8, TNF-α, downregulate NF-κB, inhibit biofilm formation, reduce neutrophil recruitment)
Important caveats for macrolides:
- Screen for NTM before starting (NTM growth → macrolide resistance)
- Baseline audiology and ECG (QTc prolongation risk)
- ERS 2025: Against routine use in patients with NTM due to risk of macrolide-resistant NTM
| Drug | Dose | Indication / Notes |
|---|---|---|
| Long-term oral doxycycline | 100 mg OD | Non-macrolide alternative; ERS 2025 suggests NOT using routinely |
| Co-trimoxazole | 960 mg BD | Certain pathogens; evidence limited |
ERS 2025 Strong Recommendation: Do NOT use long-term oral non-macrolide antibiotics routinely
3. Long-Term Inhaled Antibiotic Therapy — ERS 2025: Strong Recommendation (chronic P. aeruginosa, high-risk)
| Drug | Formulation | Dose | Notes |
|---|---|---|---|
| Tobramycin (TOBI/TOBI Podhaler) | Solution for nebulization / DPI | 300 mg BD nebulized 28 days on/28 days off (TOBI); 112 mg BD DPI (TOBI Podhaler) | FDA-approved for CF; used in non-CF bronchiectasis with chronic PA |
| Aztreonam lysine (Cayston) | Nebulized | 75 mg TID 28 days on/28 days off | Gram-negative activity; less Pseudomonas activity alone |
| Colistimethate (Colobreathe) | DPI | 125 mg (1,662,500 IU) BD | Active against MDR Pseudomonas |
| Ciprofloxacin (Ciprofloxacin DPI/liposomal) | DPI/liposomal nebulized | 32.5 mg BD (DPI) / 150 mg BD liposomal | ORBIT-3/4 trials (DPI); RESPIRE trials (liposomal); emerging ERS guidance |
| Levofloxacin liposomal (Quinsair) | Nebulized solution | 240 mg BD | Approved in EU; ORBIT data |
4. Pseudomonas Eradication Treatment — ERS 2025: Conditional Recommendation
- Goal: eradicate new P. aeruginosa culture positivity before chronic colonization established
- Ciprofloxacin 500–750 mg BD oral × 3 weeks, PLUS
- Tobramycin 300 mg BD nebulized × 28 days
- OR Colistimethate nebulized
- Systematic review (PMID 38296344): benefit shown in CF; limited non-CF RCT data
5. NTM Treatment (MAC)
- Confirmed MAC lung disease (ATS criteria): ≥2 positive sputum cultures or 1 BAL positive
- Macrolide-based triple therapy: Azithromycin 500–600 mg 3×/week (or clarithromycin 1000 mg/day) + Rifampicin 600 mg/day + Ethambutol 15 mg/kg/day
- Duration: 12 months culture-negative (typically 18–24 months total)
E. INHALED CORTICOSTEROIDS — ERS 2025: Suggest Against Routine Use
- No significant benefit on exacerbation rate or lung function in non-CF bronchiectasis
- Risk: increased risk of NTM infection, local adverse effects
- Exception: Use if concurrent asthma or COPD indication
F. ORAL/SYSTEMIC CORTICOSTEROIDS
- Not recommended routinely in infectious bronchiectasis
- Indicated in ABPA: Prednisolone 0.5–1 mg/kg/day → taper over months
- ABPA + itraconazole: Itraconazole 200 mg BD × 16 weeks (reduces steroid requirement, reduces Aspergillus load)
- Active autoimmune cause (RA, Sjögren's): systemic immunosuppression per underlying disease
G. PULMONARY REHABILITATION — ERS 2025: Strong Recommendation (impaired exercise capacity)
- Supervised exercise training + education
- Duration: minimum 8 weeks (ideally 12–20 weeks)
- Improves exercise capacity (6MWD), QoL, dyspnea scores
- Combines aerobic, resistance, and breathing training
H. OXYGEN THERAPY
- Long-term oxygen therapy (LTOT) if resting SpO2 ≤88% on 2+ assessments
- Target SpO2 88–92% in those with CO2 retention risk
I. MANAGING COMPLICATIONS
Massive Hemoptysis (>200–300 mL/24h):
- Stabilize — airway protection; selective intubation of non-bleeding lung if needed
- Identify source — rigid bronchoscopy
- Bronchial Artery Embolization (BAE) — first-line interventional treatment (hypertrophied bronchial arteries); ~80% success
- Surgical resection — if BAE fails or focal disease
- Tranexamic acid 500–1000 mg IV/oral (adjunct)
Recurrent Infections / Antibiotic Resistance:
- Combination antibiotics may be necessary
- Antibiotic cycling strategy (rotating inhaled antibiotics)
Chronic Respiratory Failure / Advanced Disease:
- Lung transplantation — consideration for CF and non-CF bronchiectasis with FEV1 <30%, rapid decline, increasing hospitalizations
J. SURGICAL TREATMENT
- Indications: Focal, drug-resistant disease; massive hemoptysis not controlled by BAE; resectable suppurative locus
- Procedure: Segmentectomy or lobectomy
- Rarely curative in diffuse disease
K. VACCINATION
- Annual influenza vaccine
- Pneumococcal vaccine (PCV13 + PPSV23 schedule)
- COVID-19 booster vaccination
11. NEWER / EMERGING THERAPIES
| Agent | Mechanism | Status |
|---|---|---|
| Brensocatib (Insmed) | Oral DPP1 inhibitor — inhibits neutrophil serine protease (NSP) activation (most novel); reduces neutrophil elastase, cathepsin G, proteinase-3 activity | ASPEN Phase 3 trial ongoing; Phase 2 showed reduced exacerbation rate and improved FEV1; referenced in Harrison's 22E as "improved bronchiectasis outcomes with an oral inhibitor of neutrophil serine protease activity" |
| Liposomal ciprofloxacin (ARD-3150) | Inhaled; sustained-release biofilm penetration | ORBIT-3/4 trials; approved in Europe |
| Inhaled colistimethate sodium (iCS) | Inhaled polymyxin; Pseudomonas and MDR pathogens | PROMIS-II trial data |
| Dupilumab | Anti-IL-4Rα (IL-4/IL-13 blockade) | Study in eosinophilic/allergic endotype bronchiectasis |
| Mepolizumab | Anti-IL-5 (anti-eosinophil) | Eosinophilic bronchiectasis phenotype investigation |
| Inhaled Mannitol | Osmotic airway hydration | ERS 2025 conditional recommendation; Bronchitol approved EU/Australia |
| Statins | Anti-inflammatory | Early phase investigation |
| Hypertonic saline 6–7% | Mucus hydration | ERS 2025 conditionally recommended |
| Airway microbiome modulation | Bacteriotherapy concepts | Pre-clinical/early clinical |
| CFTR modulators (Elexacaftor/tezacaftor/ivacaftor — Trikafta) | Corrects/potentiates CFTR protein in CF | Game-changing therapy for CF-related bronchiectasis; reduces pulmonary exacerbations by >80%, improves FEV1; approved ≥2 years with eligible mutations |
12. SEVERITY SCORING & PROGNOSTIC TOOLS
| Score | Components |
|---|---|
| BSI (Bronchiectasis Severity Index) | Age, BMI, FEV1%, prior hospital admissions, exacerbations/year, dyspnea, colonization (H. influenzae/Pseudomonas), radiologic extent; predicts mortality and hospitalization |
| FACED | FEV1, Age, Chronic colonization (Pseudomonas), Extension (radiologic), Dyspnea; 0–7 score; 5-year mortality prediction |
| E-FACED | Extended FACED incorporating prior exacerbations |
13. ERS 2025 GUIDELINE SUMMARY (Chalmers et al., PMID 41016738)
| Recommendation | Strength |
|---|---|
| Airway clearance techniques for most patients | Strong ✔ |
| Pulmonary rehabilitation for impaired exercise capacity | Strong ✔ |
| Long-term macrolide therapy for patients at high risk of exacerbations | Strong ✔ |
| Long-term inhaled antibiotics in chronic P. aeruginosa infection with high exacerbation risk | Strong ✔ |
| Eradication treatment for new P. aeruginosa | Conditional ✔ |
| Mucoactive drugs (hypertonic saline, mannitol) in specific circumstances | Conditional ✔ |
| Long-term oral non-macrolide antibiotics — do NOT use routinely | Strong Against ✗ |
| Inhaled corticosteroids — do NOT use routinely | Against ✗ |
14. KEY ORGANISMS, BUGS, AND DRUG DOSES (Quick Reference Table)
| Pathogen | First-Line | Dose | Duration |
|---|---|---|---|
| H. influenzae | Amoxicillin-clavulanate | 625 mg TDS oral | 14 days |
| S. pneumoniae | Amoxicillin | 500 mg–1g TDS oral | 14 days |
| P. aeruginosa (oral) | Ciprofloxacin | 750 mg BD oral | 14 days |
| P. aeruginosa (severe/IV) | Piperacillin-tazobactam | 4.5 g TID IV | 14 days |
| P. aeruginosa (severe/IV) | Meropenem | 1–2 g TID IV | 14 days |
| Staphylococcus aureus MSSA | Flucloxacillin | 500 mg–1 g QID oral | 14 days |
| MRSA | Co-trimoxazole or linezolid | 960 mg BD / 600 mg BD | 14 days |
| MAC (NTM) | Azithromycin + Rifampicin + Ethambutol | 500 mg 3×/wk + 600 mg OD + 15 mg/kg OD | 12 months culture-negative |
| Long-term macrolide | Azithromycin | 250 mg OD or 500 mg 3×/wk | Long-term (≥12 months) |
| Inhaled anti-Pseudomonas | Tobramycin (TOBI) | 300 mg BD nebulized 28 days on/off | Ongoing cycles |
| Inhaled anti-Pseudomonas | Colistimethate DPI | 125 mg BD | Ongoing |
| ABPA | Prednisolone + Itraconazole | 0.5 mg/kg/day → taper + 200 mg BD | 16+ weeks |
Summary Schematic
BRONCHIECTASIS
│
├── ETIOLOGY: Post-infectious | CF/PCD/genetic | Immune deficiency |
│ Autoimmune | ABPA | Obstruction | Idiopathic (50%)
│
├── PATHOGENESIS: Vicious cycle (Cole)
│ Initial insult → Neutrophil elastase + HNE + cytokines
│ → Wall destruction → Mucostasis → Chronic infection → ↑ inflammation
│
├── PATHOLOGY: Dilated thick-walled bronchi, smooth muscle/elastic loss,
│ goblet cell hyperplasia, fibrosis, neovascularization
│
├── HRCT: Signet-ring sign, tram-tracks, lack of tapering,
│ peripheral airways visible, tree-in-bud, mosaic attenuation
│
├── CLASSIFICATION: Cylindrical > Varicose > Cystic
│
├── CLINICAL: Productive cough, purulent sputum, hemoptysis,
│ crackles, clubbing, recurrent infections
│
└── TREATMENT:
├── ACT (Strong) + Pulmonary Rehab (Strong)
├── Antibiotics: Acute (14d) → Exacerbation-directed
├── Long-term macrolide (Strong) → Azithromycin 250mg OD
├── Inhaled antibiotics if chronic PA (Strong) → Tobramycin/Colistin
├── Mucoactives: Hypertonic saline, Mannitol (Conditional)
├── No ICS routinely (ERS 2025)
├── Newer: Brensocatib (DPP1 inhibitor) – most promising
└── CF: CFTR modulators (Trikafta) – transformative
References:
- Fishman's Pulmonary Diseases and Disorders, 2-Volume Set — Chapter 51 (Gregory Tino)
- Harrison's Principles of Internal Medicine 22E (2025) — Chapter 301 (Baron et al.)
- Murray & Nadel's Textbook of Respiratory Medicine, 2-Volume Set
- Robbins, Cotran & Kumar: Pathologic Basis of Disease — Chapter 15
- Grainger & Allison's Diagnostic Radiology — Chapter 6
- ERS Clinical Practice Guideline for Adult Bronchiectasis 2025 — Chalmers JD et al. Eur Respir J 2025 (PMID 41016738)
- Pseudomonas Eradication Meta-Analysis — Conceição M et al. Eur Respir Rev 2024 (PMID 38296344)
This is a shared conversation. Sign in to Orris to start your own chat.