explain me about metabolism of carbohydrates
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I now have comprehensive content from multiple authoritative textbooks. Here is a thorough, well-organized explanation of carbohydrate metabolism.
Carbohydrate Metabolism
Carbohydrate metabolism encompasses the entire set of biochemical processes by which the body digests, absorbs, stores, and oxidizes carbohydrates to produce energy and biosynthetic precursors. Carbohydrates provide ~4 kcal/g of energy and account for 50% or more of calories in most human diets.
1. Digestion and Absorption
Digestion begins in the mouth, where salivary amylase starts breaking down starch. Pancreatic amylase continues this in the small intestine, reducing complex polysaccharides to disaccharides. Brush-border enzymes (sucrase, lactase, maltase) then cleave disaccharides into monosaccharides:
- Sucrose → glucose + fructose
- Lactose → glucose + galactose
- Maltose → glucose + glucose
Absorption differs by monosaccharide:
- Glucose and galactose are absorbed by energy-dependent active transport coupled to the sodium pump (SGLT-1).
- Fructose is absorbed by concentration-dependent facilitated diffusion.
All absorbed sugars travel to the liver via the portal vein, where galactose and fructose are converted to glucose.
2. Central Molecule: Glucose-6-Phosphate (G6P)
Once inside cells, glucose is immediately phosphorylated to glucose-6-phosphate (G6P) — the metabolic hub of carbohydrate metabolism. G6P cannot leave the cell and is the entry point for:
- Glycolysis
- Glycogen synthesis (glycogenesis)
- The pentose phosphate pathway (PPP)
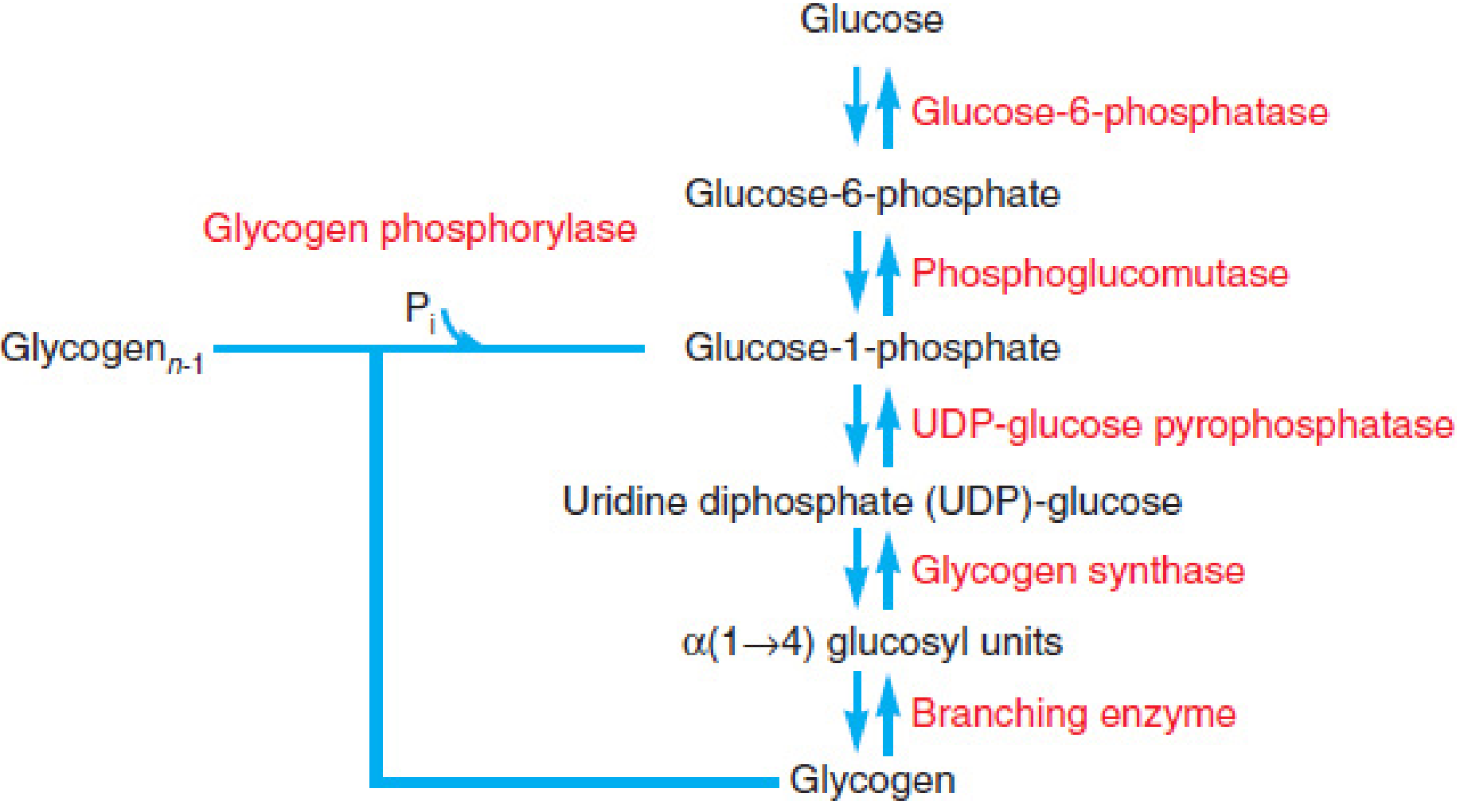
Glycogenesis/glycogenolysis reactions, with glucose-6-phosphate at the center — Mulholland & Greenfield's Surgery, 7e
3. Glycogenesis and Glycogenolysis
Glycogenesis (Storage)
When glucose is abundant (postprandial, insulin-dominant state):
- Glucose → Glucose-6-phosphate (hexokinase/glucokinase, uses 1 ATP)
- G6P → Glucose-1-phosphate (phosphoglucomutase)
- Glucose-1-P + UTP → UDP-glucose (UDP-glucose pyrophosphorylase)
- UDP-glucose → glycogen chain (glycogen synthase, α-1→4 linkages; branching enzyme creates α-1→6 branch points)
The liver can store ~100 g of glycogen; muscles store the rest. Because complete oxidation of one molecule of G6P generates 37 ATP and its creation uses only 1 ATP, the efficiency of glucose storage is ~97%.
Glycogenolysis (Mobilization)
Glycogen breakdown is not a reversal of synthesis. It requires:
- Glycogen phosphorylase → releases glucose-1-phosphate from the chain
- Phosphoglucomutase → converts G1P → G6P
- Glucose-6-phosphatase → converts G6P → free glucose (only in liver, kidney, intestinal epithelial cells)
This explains why muscle glycogen cannot directly raise blood glucose — muscle cells lack glucose-6-phosphatase.
Hormonal regulation:
- Insulin (fed state) → activates glycogen synthase (dephosphorylation) → favors storage
- Glucagon / Epinephrine (fasted/stress) → activates glycogen phosphorylase (via cAMP cascade) → favors release
4. Glycolysis
Glycolysis is the 10-step cytoplasmic pathway that converts 1 molecule of glucose into 2 molecules of pyruvate.
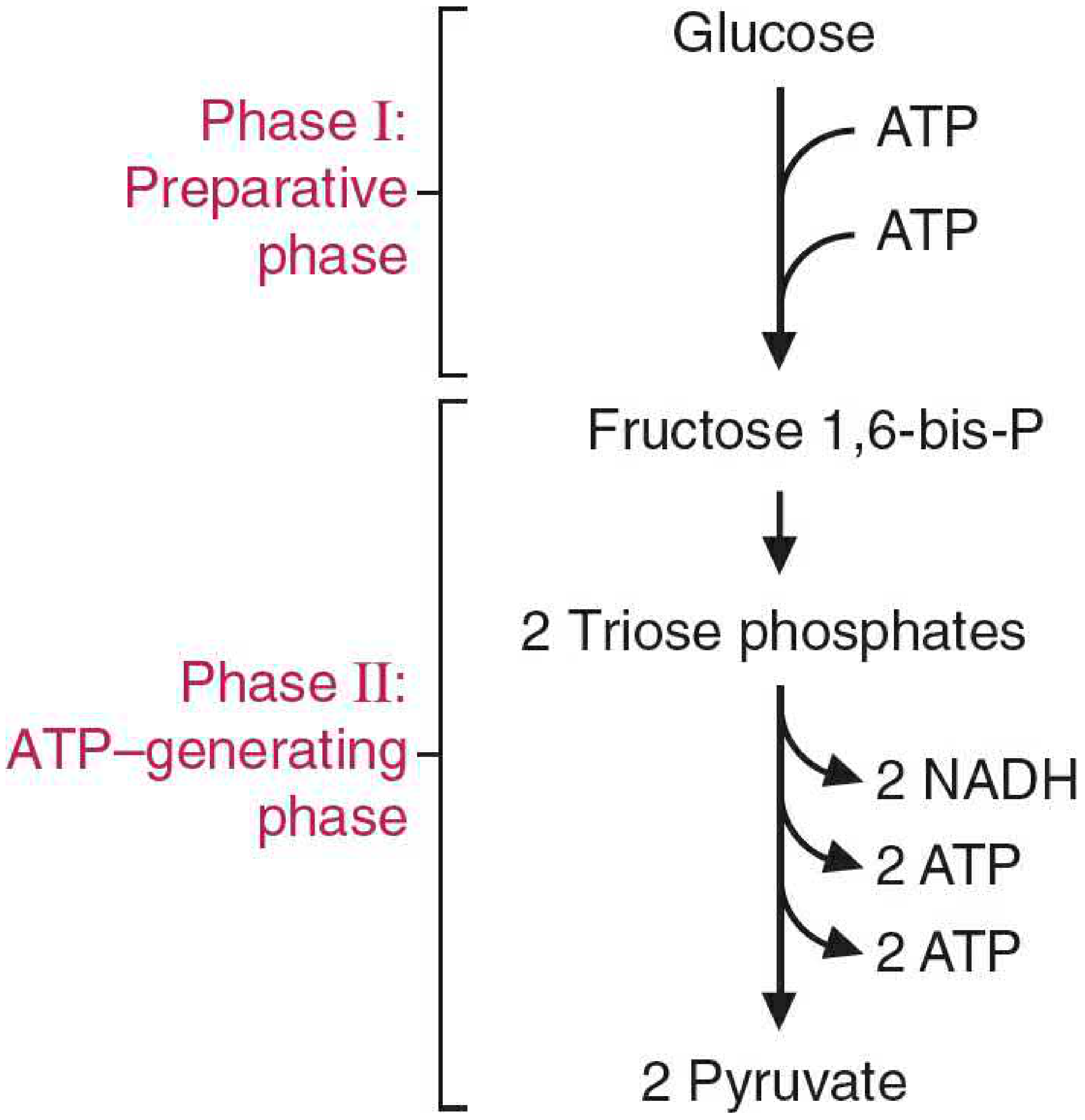
Phases of glycolysis — Basic Medical Biochemistry, 6e (Lippincott)
Two Phases:
Phase I — Preparative (energy investment):
- Glucose is phosphorylated twice using 2 ATP → Fructose-1,6-bisphosphate
- Then split into 2 triose phosphates (glyceraldehyde-3-phosphate)
Phase II — ATP-generating:
- Each triose phosphate → pyruvate, yielding 2 NADH + 4 ATP (substrate-level phosphorylation)
Net yield per glucose: 2 pyruvate + 2 NADH + 2 net ATP
Three Rate-Limiting Enzymes (irreversible steps):
| Enzyme | Step | Regulation |
|---|---|---|
| Hexokinase (Glucokinase in liver) | Glucose → G6P | Inhibited by G6P; glucokinase induced by insulin |
| Phosphofructokinase-1 (PFK-1) | F6P → F1,6-BP | Activated by AMP, F2,6-BP; inhibited by ATP, citrate |
| Pyruvate kinase | PEP → Pyruvate | Activated by F1,6-BP; inhibited by ATP, alanine |
Overall equation:
Glucose + 2 NAD⁺ + 2 ADP + 2Pᵢ → 2 Pyruvate + 2 NADH + 2H⁺ + 2 ATP
5. Fate of Pyruvate
Pyruvate is at a critical junction:
A. Aerobic: Entry into the TCA Cycle
In the presence of oxygen, pyruvate dehydrogenase (mitochondria) converts:
Pyruvate + CoA + NAD⁺ → Acetyl-CoA + CO₂ + NADH
Acetyl-CoA then enters the Krebs (TCA/Citric Acid) cycle.
B. Anaerobic: Lactate Production
Without oxygen (or when mitochondrial capacity is exceeded), lactate dehydrogenase converts:
Pyruvate + NADH → Lactate + NAD⁺
This regenerates NAD⁺ so glycolysis can continue. Lactate can be measured as a marker of tissue hypoperfusion in clinical settings.
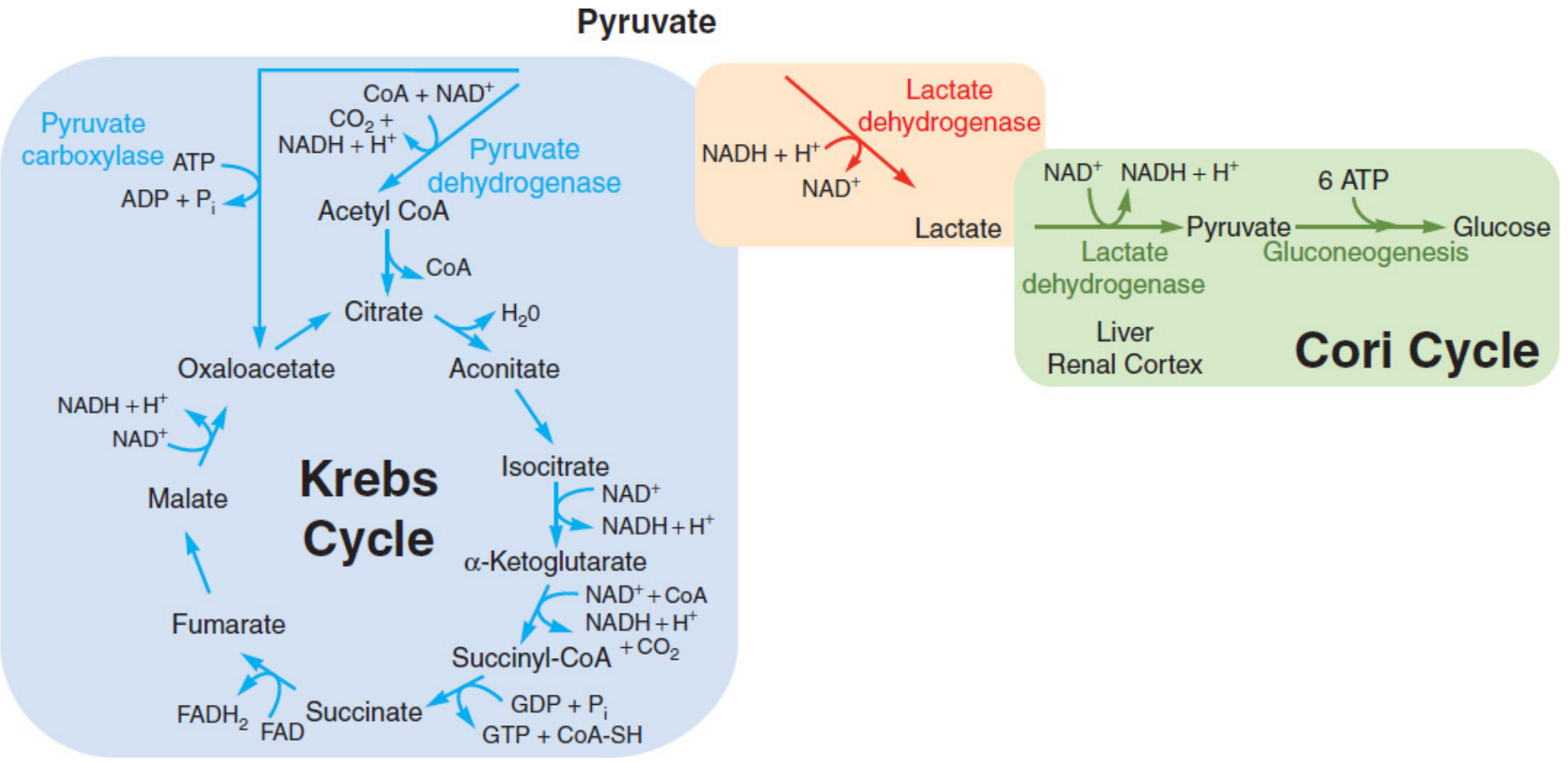
Pyruvate: gateway to the Krebs cycle (aerobic) and lactic acid fermentation (anaerobic), plus the Cori cycle — Mulholland & Greenfield's Surgery, 7e
6. The TCA Cycle (Krebs / Citric Acid Cycle)
The TCA cycle occurs in the mitochondrial matrix and fully oxidizes acetyl-CoA to CO₂, capturing energy as NADH, FADH₂, and GTP.
Key steps:
- Acetyl-CoA (2C) + Oxaloacetate (4C) → Citrate (6C) [citrate synthase]
- Citrate → Isocitrate → α-Ketoglutarate (→ CO₂ + NADH)
- α-Ketoglutarate → Succinyl-CoA (→ CO₂ + NADH)
- Succinyl-CoA → Succinate → GTP
- Succinate → Fumarate → Malate → Oxaloacetate (regenerated)
Yield per acetyl-CoA: 3 NADH + 1 FADH₂ + 1 GTP
These electron carriers (NADH, FADH₂) then feed the electron transport chain (ETC), where oxidative phosphorylation generates the bulk of ATP (~2.5 ATP per NADH; ~1.5 ATP per FADH₂).
Total ATP yield per glucose (aerobic): ~30–32 ATP
7. Gluconeogenesis
Gluconeogenesis is the synthesis of new glucose from non-carbohydrate precursors. It occurs primarily in the liver (and kidney during prolonged fasting, critical illness, diabetes).
Precursors (in order of contribution in humans):
- Lactate (~most significant) — via the Cori cycle
- Alanine and glutamine — from muscle protein breakdown
- Glycerol — from triglyceride lipolysis
- Odd-chain fatty acids, glucogenic amino acids
Key principle: Gluconeogenesis is NOT a simple reversal of glycolysis. The three irreversible glycolytic steps must be bypassed:
| Glycolytic step | Gluconeogenic bypass enzyme |
|---|---|
| Pyruvate → PEP | Pyruvate carboxylase (pyruvate → OAA, mitochondria) + PEPCK (OAA → PEP) |
| F1,6-BP → F6P | Fructose-1,6-bisphosphatase |
| G6P → Glucose | Glucose-6-phosphatase (liver/kidney only) |
Regulation: Stimulated by glucagon and high acetyl-CoA; inhibited by ADP, AMP, and F2,6-bisphosphate.
The Cori Cycle
Lactate from anaerobic muscle metabolism is transported to the liver → converted back to pyruvate → gluconeogenesis → new glucose released into blood → taken up again by muscle. This is an elegant inter-organ glucose recycling loop.
8. Pentose Phosphate Pathway (PPP / Hexose Monophosphate Shunt)
The PPP is an anabolic, cytosolic pathway that branches from glycolysis at G6P. It serves two main purposes:
Phase 1 — Oxidative (irreversible):
- G6P → Ribulose-5-phosphate (by G6P dehydrogenase, rate-limiting)
- Generates 2 NADPH per G6P
- NADPH is used for: fatty acid synthesis, cholesterol synthesis, steroid synthesis, and reducing oxidative stress via glutathione reductase
Phase 2 — Nonoxidative (reversible):
- Ribulose-5-P → Ribose-5-phosphate (for DNA, RNA, ATP, NAD, FAD, CoA synthesis)
- Intermediates can re-enter glycolysis or gluconeogenesis
Clinical relevance — G6PD Deficiency:
RBCs lack mitochondria and depend entirely on the PPP for NADPH to fight oxidative stress. G6PD deficiency → inadequate NADPH → hemolysis when exposed to oxidative triggers (fava beans, infections, primaquine, aspirin).
9. Hormonal Regulation — Summary
| State | Dominant Hormone | Major Effects on Carbohydrate Metabolism |
|---|---|---|
| Fed (absorptive) | Insulin ↑ | ↑ Glycogenesis, ↑ Glycolysis, ↑ PPP, ↑ Lipogenesis; ↓ Gluconeogenesis |
| Fasted | Glucagon ↑ | ↑ Glycogenolysis, ↑ Gluconeogenesis; ↓ Glycogen synthesis |
| Stress/exercise | Epinephrine ↑ | ↑ Glycogenolysis (in liver and muscle), ↑ Gluconeogenesis |
10. Integration Across Organs
| Organ | Key Role |
|---|---|
| Liver | Glucose buffer — stores glycogen, performs gluconeogenesis; contains glucose-6-phosphatase (can release free glucose) |
| Muscle | Major consumer; stores glycogen for its own use (no G6Pase); produces lactate under anaerobic conditions |
| Brain | Obligate glucose consumer under normal conditions; no significant glycogen stores |
| Adipose tissue | Uses glucose for glycerol-3-phosphate (TAG synthesis); GLUT-4 transporter (insulin-sensitive) |
| RBCs | No mitochondria → entirely dependent on anaerobic glycolysis |
| Kidney | Secondary site of gluconeogenesis (especially during prolonged fasting and diabetes) |
Sources:
- Basic Medical Biochemistry: A Clinical Approach, 6e (Lippincott), blocks 4–5
- Mulholland & Greenfield's Surgery: Scientific Principles and Practice, 7e, block 1
- Guyton and Hall Textbook of Medical Physiology, block 8
- Schwartz's Principles of Surgery, 11e, block 1
This is a shared conversation. Sign in to Orris to start your own chat.