Pathogenesis of ala
Asking for Preferences
Reading File
Finding Sources
Finding Sources
Searching PubMed
"amoebic liver abscess"[MeSH Terms] AND pathogenesis
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Loading Image
Reading File
I now have comprehensive material. Here is a detailed, well-structured answer on the pathogenesis of Amoebic Liver Abscess (ALA).
Pathogenesis of Amoebic Liver Abscess (ALA)
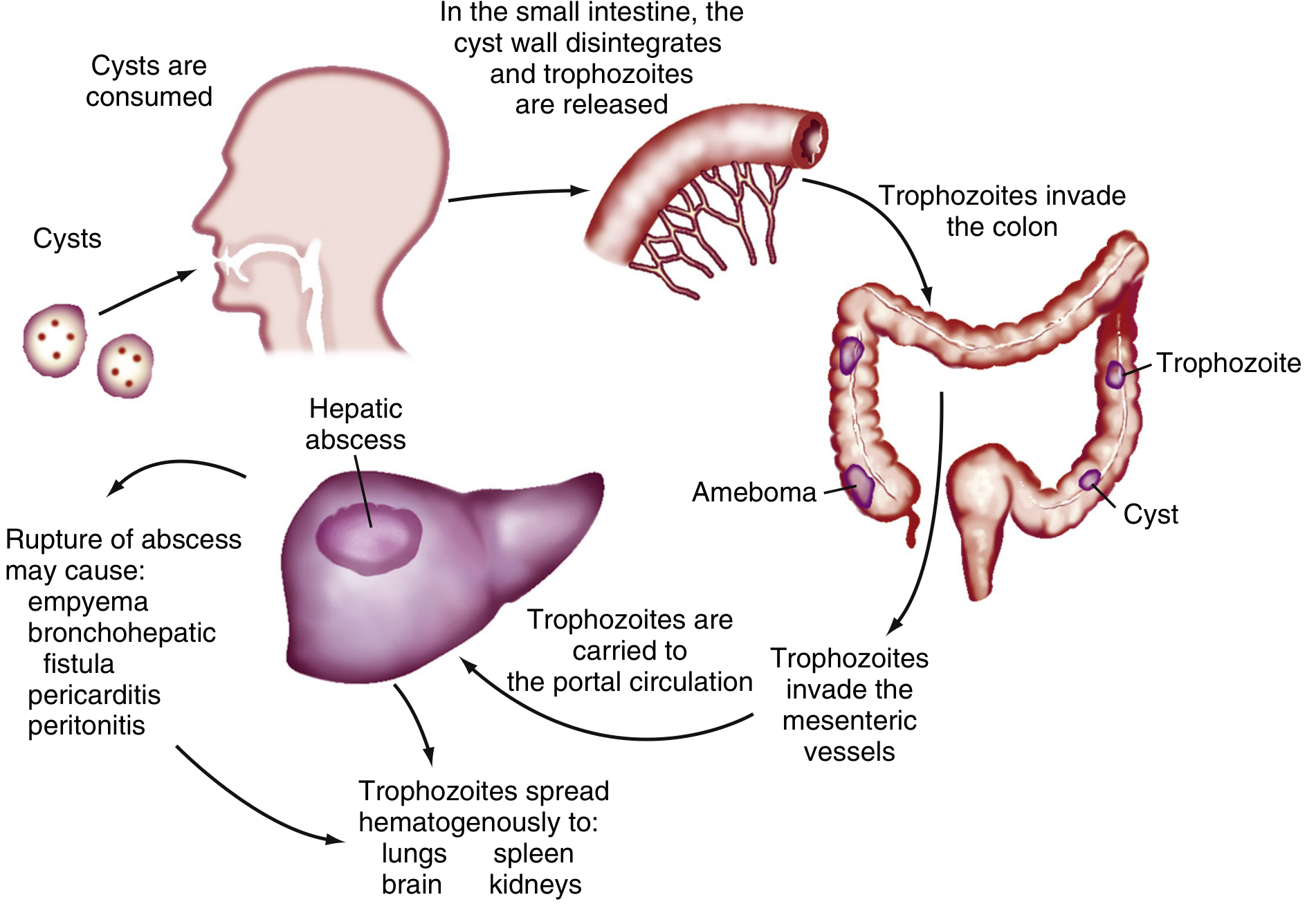
Life cycle and pathogenetic sequence of E. histolytica — Sleisenger & Fordtran's Gastrointestinal and Liver Disease
1. The Causative Organism
Entamoeba histolytica (named "histolytica" for its tissue-destructive properties) is the sole agent. It has a simple two-stage life cycle:
- Cyst — the infectious, environmentally stable form; low infectious dose (<100 organisms); resistant to chlorine
- Trophozoite — the motile, invasive stage responsible for disease
Certain genotypes are associated with colonization, while others are associated with tissue invasion. Only humans and some non-human primates are infected. — Goldman-Cecil Medicine
2. Step-by-Step Pathogenesis
Step 1 — Ingestion of Cysts
Cysts are ingested via fecal-oral route through contaminated food or water. An asymptomatic carrier can excrete up to 15 million cysts/day. — Harrison's Principles of Internal Medicine 22E
Step 2 — Excystation and Colonic Invasion
Cyst walls disintegrate in the small intestine; trophozoites are released and travel to the colon, where they invade the mucosa producing characteristic flask-shaped ulcers. — Sleisenger & Fordtran's
The molecular sequence of host cell destruction involves:
| Step | Mechanism |
|---|---|
| Adherence | Parasite's Gal/GalNAc-specific lectin binds carbohydrate determinants on intestinal epithelium |
| Trogytosis | A nibbling-like process where the ameba ingests pieces of host cells |
| Contact-dependent cytotoxicity | Host cells die by apoptosis — requires Gal/GalNAc lectin attachment + amebic pore-forming protein delivered by acid intracellular vesicles; caspase-3 is activated within minutes of adherence |
| Phagocytosis | The ameba ingests the host cell corpse; this step is required for continued mucosal invasion |
| Extracellular matrix degradation | ≥44 cysteine proteinase genes degrade colonic mucin glycoproteins, extracellular matrix, hemoglobin, villin, and inactivate IL-18 |
— Goldman-Cecil Medicine
Step 3 — Invasion of Mesenteric Vessels and Portal Transport
Trophozoites penetrate through mucosa and submucosal tissues, enter the mesenteric vessels, and reach the portal circulation. Crucially, overt amebic colitis is not required for liver abscess formation — portal seeding can occur without clinical intestinal disease (>90% of ALA patients have no bowel symptoms). — Goldman-Cecil Medicine, Harrison's
Step 4 — Hepatic Trapping and Abscess Formation
Trophozoites block intrahepatic portal venules, then invade liver parenchyma. The resulting abscess is characterized by:
- Well-circumscribed regions of dead hepatocytes, liquefied cells, and cellular debris
- Surrounded by a rim of connective tissue, a few inflammatory cells, and amebic trophozoites
- Adjacent liver parenchyma is unaffected
- Given the small numbers of amebae relative to the large abscess size, E. histolytica can cause hepatocyte death without direct contact — suggesting a remote toxin/immune mechanism
— Goldman-Cecil Medicine
Step 5 — Potential Spread Beyond the Liver
Via hematogenous spread or direct extension, trophozoites can reach:
- Lungs (pleuropulmonary amebiasis — most common extraintestinal site after liver)
- Brain (rare brain abscess)
- Pericardium, pleura, peritoneum — from direct rupture of a liver abscess (→ empyema, bronchohepatic fistula, pericarditis, peritonitis)
3. Immune Response
Innate Immunity
- Granulocytes (neutrophils, eosinophils) are the earliest responders (within 1–2 days) — depletion worsens hepatic and intestinal disease in murine models
- Epithelial cytokines (TNF-α, IL-1α, IL-6, IL-8, GRO-α, GM-CSF) coordinate the initial response
- Macrophages and T lymphocytes recruited by day 3; macrophages acquire amebicidal activity when stimulated with IFN-γ, TNF-α, or CSF-1
- The IL-33–ILC2 pathway is an important host defense mechanism
Acquired Immunity
- Protective immunity requires both systemic IFN-γ and mucosal IgA directed at the Gal/GalNAc lectin
- Children with mucosal IgA against Gal/GalNAc lectin have 86% fewer new infections the following year
- IL-4 and IFN-γ have opposing roles: a TH2/IL-4 response promotes persistence, whereas an IFN-γ response promotes clearance
- Immune evasion: ameba's cysteine proteinases inactivate IL-18, undermining the host's inflammatory signaling
— Goldman-Cecil Medicine
4. Host Risk Factors for Severe Disease
- Male sex (10:1 male:female for ALA), age 18–50
- HLA-DR3 genotype
- Alcohol intake, malnutrition, pregnancy, glucocorticoid use, malignancy
- Immunosuppression (particularly HIV/AIDS)
- Post-menopausal women (suggesting hormonal protection in premenopausal females)
— Goldman-Cecil Medicine, Sleisenger & Fordtran's
5. Abscess Characteristics
- Typically solitary, right lobe (due to greater portal blood flow from the right colon)
- Acute presentation → more often multifocal; chronic/indolent → more often solitary
- "Anchovy paste" content (brown, odorless fluid = lysed hepatocytes + cellular debris, not frank pus)
- True pus suggests bacterial superinfection
Sources: Goldman-Cecil Medicine International Edition; Sleisenger & Fordtran's Gastrointestinal and Liver Disease; Harrison's Principles of Internal Medicine 22E
This is a shared conversation. Sign in to Orris to start your own chat.