Anatomy of cerebral circulation in MD anesthesia exam
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Reading File
Reading File
Finding Sources
Finding Sources
Loading Image
Reading File
I now have comprehensive content from Miller's Anesthesia (10e) and Neuroanatomy through Clinical Cases (3rd ed). Let me compile a thorough, exam-focused answer.
Anatomy of Cerebral Circulation — MD Anesthesia Exam
Sources: Miller's Anesthesia 10e (Ch. 10); Neuroanatomy through Clinical Cases 3rd ed (Ch. 10)
1. Overview
The brain receives blood from two pairs of arteries:
- Internal carotid arteries (ICA) → anterior circulation
- Vertebral arteries → posterior circulation (unite to form the basilar artery)
These connect at the circle of Willis, from which the three paired cerebral arteries arise.
2. Anterior Circulation — Internal Carotid Artery
Segments of the ICA (mnemonic: C P C S — Cervical, Petrous, Cavernous, Supraclinoid)
| Segment | Location | Key Relationships |
|---|---|---|
| Cervical | Neck | No branches |
| Petrous | Carotid canal of temporal bone | Passes through skull base |
| Cavernous | Within cavernous sinus | S-shaped "carotid siphon"; CN III, IV, V₁, VI nearby |
| Supraclinoid (intracranial) | Pierces dura, subarachnoid space | Main branches arise here |
Branches of supraclinoid ICA — mnemonic OPAAM
| Branch | Territory |
|---|---|
| Ophthalmic artery | Retina, orbit |
| Posterior communicating artery (PComm) | Connects ICA to PCA |
| Anterior choroidal artery | Globus pallidus, putamen, posterior limb of internal capsule, thalamus, lateral geniculate |
| Anterior cerebral artery (ACA) | Medial frontal/parietal cortex, corpus callosum |
| Middle cerebral artery (MCA) | Lateral convexity |
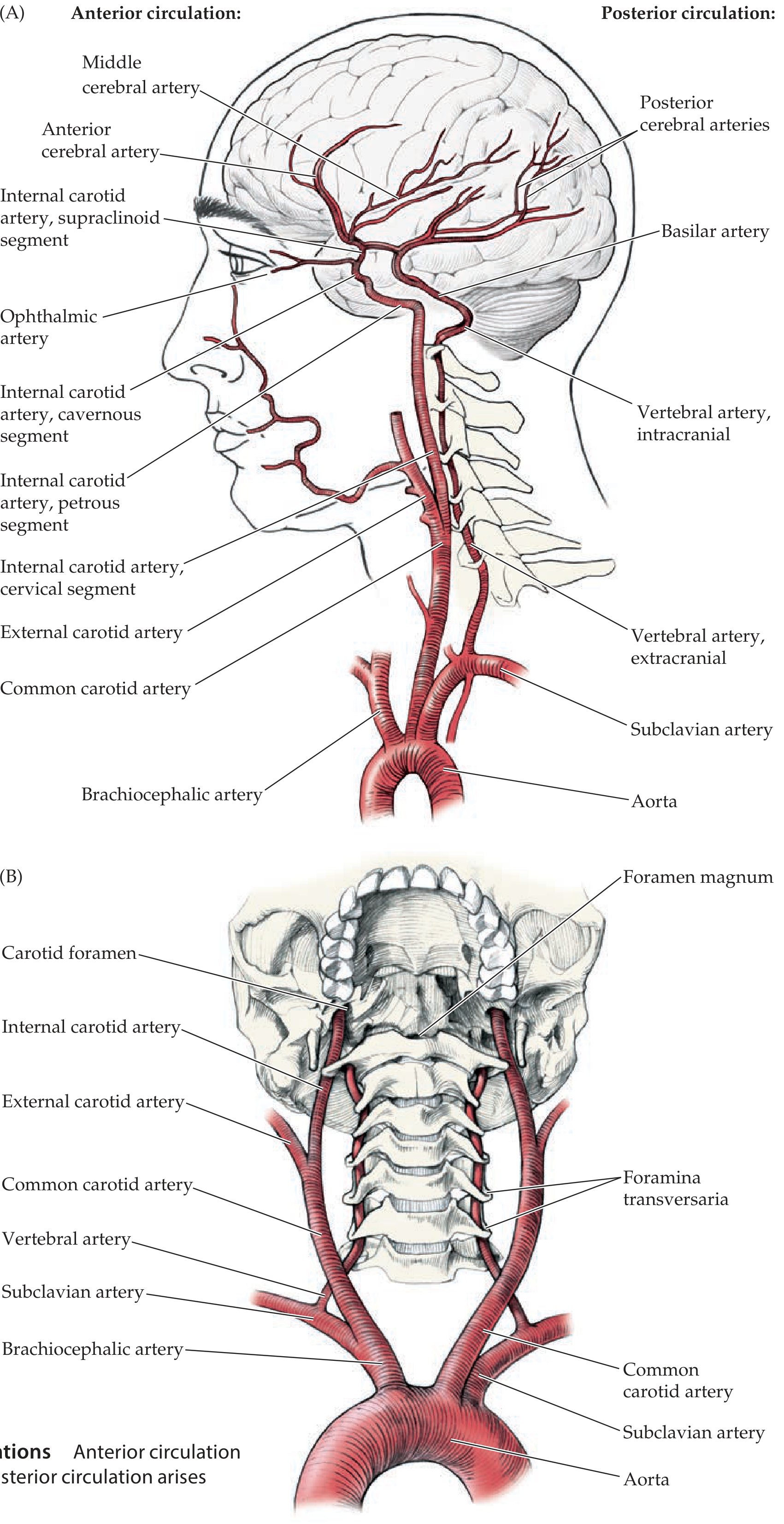
3. Posterior Circulation — Vertebrobasilar System
- Vertebral arteries arise from subclavian arteries → ascend through foramina transversaria of C1–C6 → enter foramen magnum → unite to form basilar artery
Key branches of vertebral arteries:
- PICA (posterior inferior cerebellar artery) — lateral medulla + inferior cerebellum (lesion = Wallenberg syndrome)
- Anterior spinal artery
Key branches of basilar artery:
- AICA (anterior inferior cerebellar artery) — CN VII/VIII territory
- SCA (superior cerebellar artery) — superior cerebellum
- PCA (posterior cerebral artery) — terminal branches → occipital and inferomedial temporal lobes
4. Circle of Willis
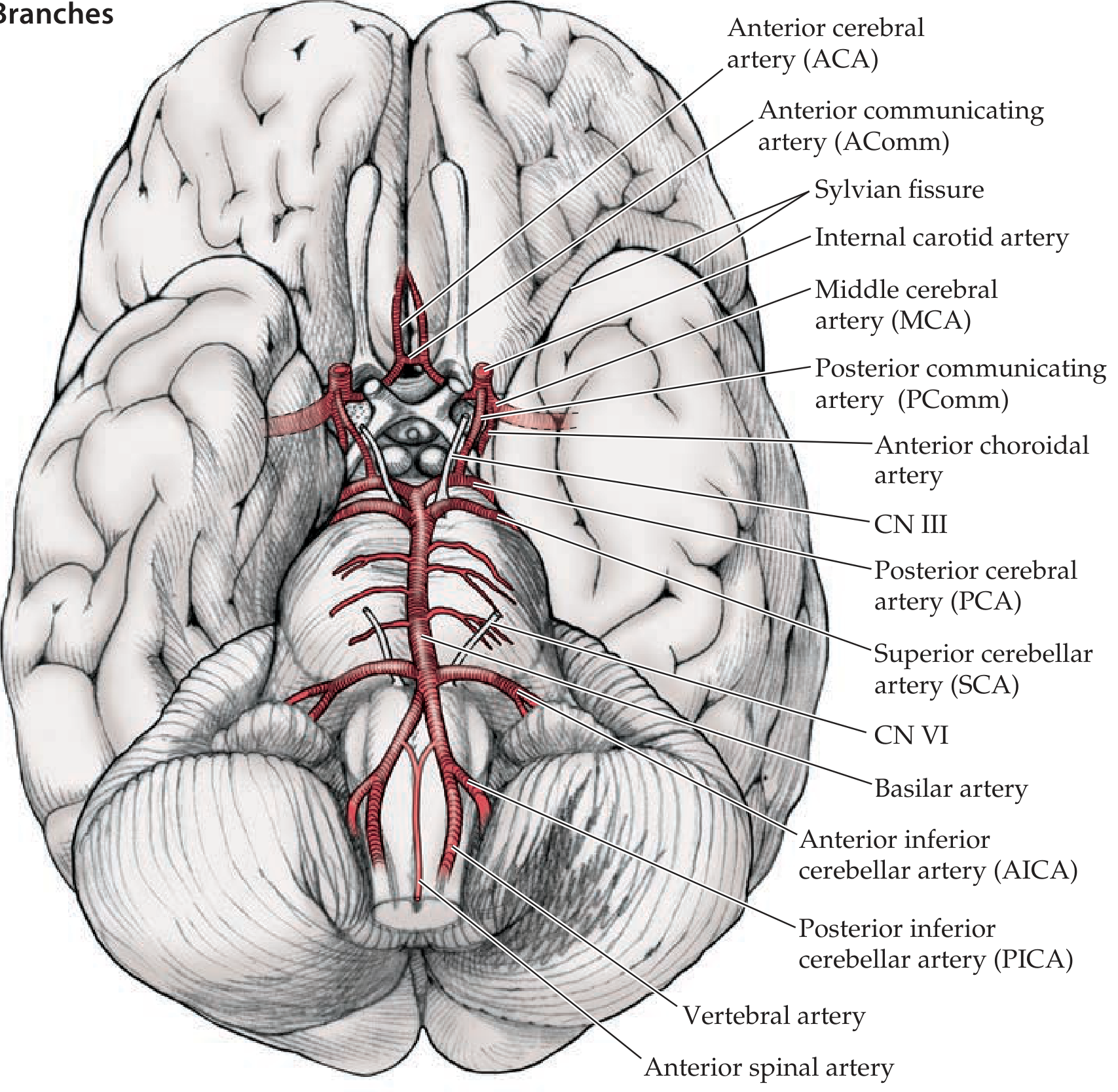
Components (anterior → posterior):
ACA ←—— AComm ——→ ACA
| |
ICA ICA
| |
PComm PComm
| |
PCA ←—— Basilar ——→ PCA
| Vessel | Role |
|---|---|
| ACA | Terminal branch of ICA |
| AComm (anterior communicating) | Connects the two ACAs |
| MCA | Terminal branch of ICA |
| PComm (posterior communicating) | Links ICA to PCA |
| PCA | Terminal branch of basilar |
Key exam point: A complete circle is present in only ~34% of individuals. Incomplete variants are common and have implications for collateral flow during carotid endarterectomy and cross-clamping.
Under normal conditions, blood from anterior and posterior circulations does not mix (equal pressures). In pathological occlusion, the circle acts as a shunt to supply ischemic territories. — Miller's Anesthesia 10e, p. 924
5. Vascular Territories of the Three Main Cerebral Arteries
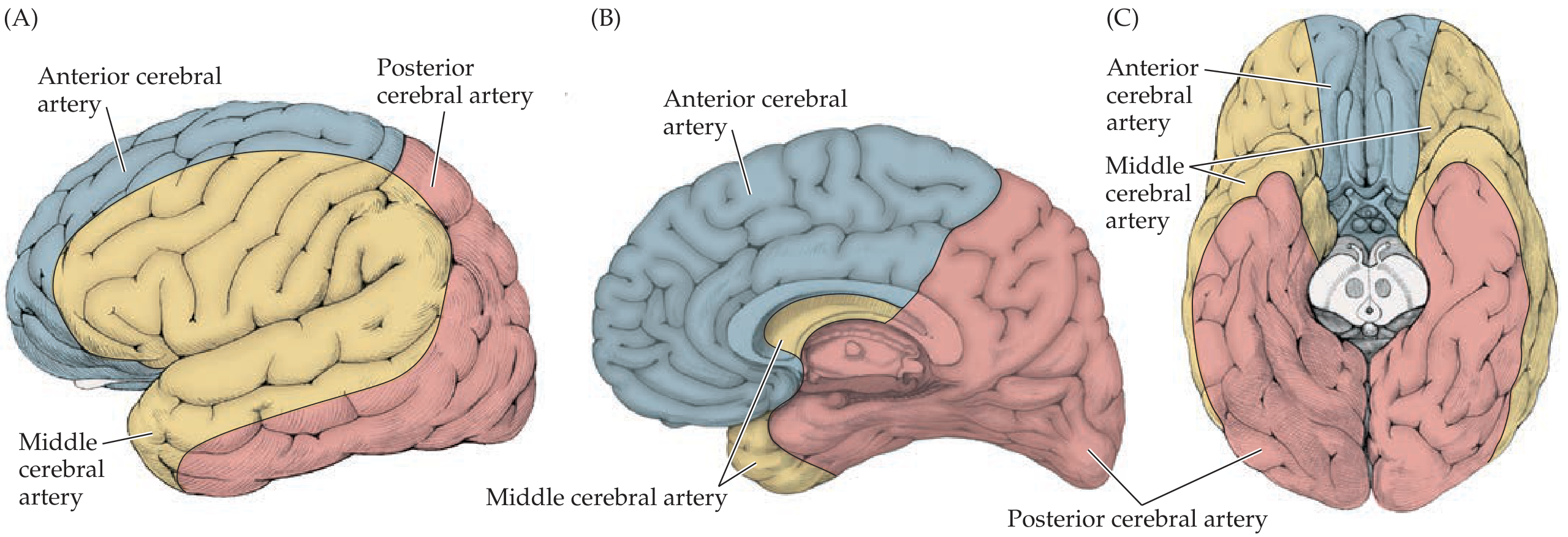
| Artery | Cortical Territory | Key Function |
|---|---|---|
| ACA | Medial frontal + anterior parietal (interhemispheric fissure) | Leg sensorimotor cortex; incontinence if bilateral |
| MCA | Entire lateral convexity (largest territory) | Arm/face sensorimotor, speech (dominant), neglect (non-dominant) |
| PCA | Inferior and medial temporal; occipital | Vision, memory |
ACA branches:
- Pericallosal artery, callosomarginal artery
- Recurrent artery of Heubner (off A1) → head of caudate, anterior putamen, anterior internal capsule
MCA segments:
- M1 (sphenoid) → gives off lenticulostriate arteries
- M2 (insular) → superior and inferior divisions within Sylvian fissure
- Superior division → frontal/parietal cortex above Sylvian fissure
- Inferior division → temporal lobe below Sylvian fissure
6. Deep Perforating Arteries — High-Yield for Anesthesia
These are end arteries — no collateral anastomosis → prone to lacunar infarction, especially in hypertension.
| Artery | Origin | Supplies |
|---|---|---|
| Lenticulostriate arteries | Proximal MCA (M1) | Putamen, caudate, globus pallidus, internal capsule (posterior limb) |
| Anterior choroidal artery | Supraclinoid ICA | Posterior limb IC, globus pallidus, thalamus, lateral geniculate |
| Recurrent artery of Heubner | Proximal ACA (A1) | Head of caudate, anterior putamen, anterior IC |
| Thalamoperforators | Proximal PCA (P1)/top of basilar | Thalamus, midbrain |
Anesthesia relevance: Lacunar strokes of the posterior limb of the internal capsule (lenticulostriate or anterior choroidal territory) produce pure motor hemiplegia — a key postoperative neurological complication.
7. Venous Drainage
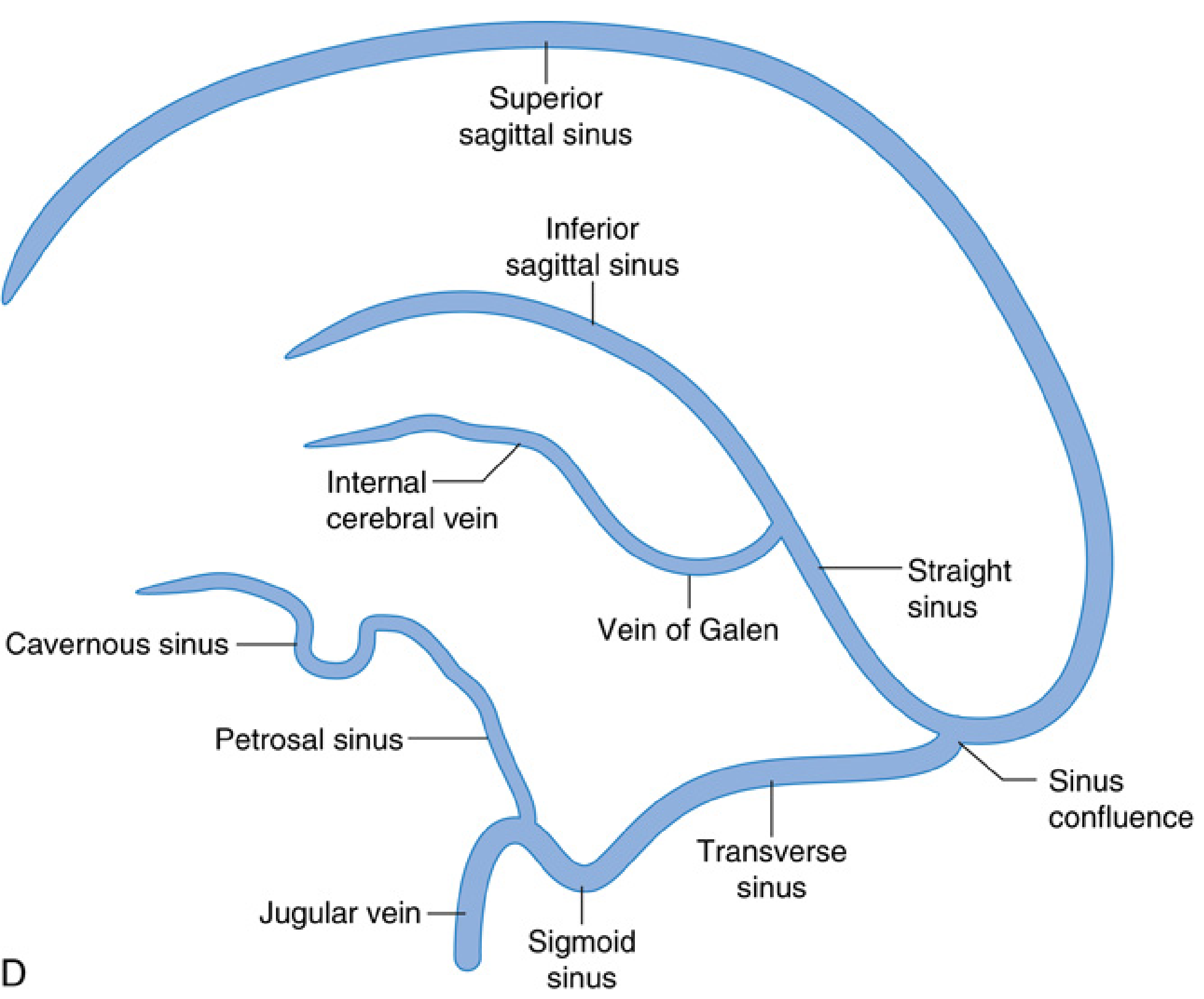
Three systems:
A. Superficial cortical veins (within pia mater on brain surface) → drain into dural sinuses
B. Deep cortical veins (drain deeper white matter/subcortical structures) → internal cerebral veins → Vein of Galen → straight sinus
C. Dural venous sinuses → ultimately drain into internal jugular veins
| Sinus | Location / Route |
|---|---|
| Superior sagittal sinus | Superior border of falx cerebri |
| Inferior sagittal sinus | Inferior border of falx |
| Straight sinus | Tentorium cerebelli; receives Vein of Galen |
| Transverse sinus | Lateral tentorium |
| Sigmoid sinus | Continues transverse → jugular foramen |
| Cavernous sinus | Around sella turcica; contains ICA, CN III, IV, V₁, V₂, VI |
| Petrosal sinus | Connects cavernous to transverse/sigmoid |
| Sinus confluence (torcular herophili) | Junction of superior sagittal, straight, transverse |
Anesthesia relevance: Air embolism via open dural sinuses (e.g., posterior fossa surgery in sitting position) is a life-threatening complication. The cavernous sinus surrounds the intracavernous ICA — relevant to carotid-cavernous fistulas and pituitary surgery.
8. Cerebral Blood Flow — Quantitative Data
| Parameter | Value |
|---|---|
| Mean CBF | 50 mL/100 g/min (at PaCO₂ 40 mmHg) |
| Gray matter CBF | ~80 mL/100 g/min |
| White matter CBF | ~20 mL/100 g/min |
| Total CBF (adult) | 750 mL/min (15–20% of cardiac output) |
| CBF causing EEG slowing | < 20–25 mL/100 g/min |
| CBF causing flat (isoelectric) EEG | < 20 mL/100 g/min |
| CBF causing irreversible damage | < 10 mL/100 g/min |
| Normal MCA velocity (TCD) | ~55 cm/s |
| Vasospasm threshold (TCD) | > 120 cm/s |
— Morgan & Mikhail's Clinical Anesthesiology 7e, p. 1075–1076
9. Exam Mnemonics Summary
| Mnemonic | Expands to |
|---|---|
| OPAAM | Branches of supraclinoid ICA: Ophthalmic, Posterior communicating, Anterior choroidal, Anterior cerebral, Middle cerebral |
| AComm + PComm | The two communicating arteries that complete the circle |
| PICA/AICA/SCA | Cerebellar arteries (inferior → superior, vertebrobasilar) |
| "Lenticulostriates = MCA perforators" | Deep supply to BG and IC from M1 |
10. Anesthesia-Specific High-Yield Points
- Carotid endarterectomy: Patchy or incomplete circle of Willis means cross-clamping the ICA may not allow adequate collateral — monitor with TCD or EEG; use shunt if needed.
- Sitting/beach-chair position: Open dural sinuses → venous air embolism risk.
- Posterior fossa surgery: PICA/AICA/SCA territory; cranial nerve monitoring essential.
- Pituitary surgery (transsphenoidal): Cavernous sinus with ICA immediately lateral; risk of ICA injury.
- Cerebral perfusion pressure = MAP − ICP; normal ICP < 15 mmHg.
- Autoregulation: Maintained between MAP 50–150 mmHg; abolished by hypoxia, hypercarbia, volatile agents.
Make a presentation on cerebral autoregulation and anesthetic effects on CBF
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Running Command
Running Command
Running Command
Writing File
Running Command
Running Command
Your presentation is ready:
📥
Cerebral Autoregulation Anesthesia
Presentation · PPTX
What's Inside (11 Slides)
| Slide | Content |
|---|---|
| 1 | Title slide — topic overview with sub-topic tags |
| 2 | Normal CBF values — data cards + threshold color-coded blocks (EEG slowing, isoelectric, irreversible damage, TCD) |
| 3 | Classic autoregulation concept — Lassen curve description + integrated regulation image from Miller's |
| 4 | 4 mechanisms of autoregulation — Myogenic, Chemical/CO₂-O₂, Neurogenic, Neurovascular Coupling in grid cards |
| 5 | CO₂ and O₂ reactivity — side-by-side panels with clinical cutoffs |
| 6 | Contemporary integrated regulation view — textbook diagram from Miller's 10e with key takeaways |
| 7 | Volatile agents — CBF vs MAC graph (halothane/isoflurane/sevoflurane/desflurane) + comparison table |
| 8 | IV agents — 6-agent grid: Propofol, Thiopental, Ketamine, Etomidate, Dexmedetomidine, Midazolam |
| 9 | ICP, CPP & Monro-Kellie — doctrine + avoid/use management strategy boxes |
| 10 | 6 clinical scenarios — CEA, craniotomy, TBI, sitting position, SAH, controlled hypotension |
| 11 | Summary comparison table — all agents: CBF, CMRO₂, autoregulation, CO₂ reactivity, ICP, best use |
Design: navy/teal/gold palette, textbook images embedded directly from Miller's Anesthesia 10e and Barash 9e.
How to manage the case of cleft lip and palate in Anaesthesia case
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Loading Image
Reading File
Finding Sources
Loading Image
Searching the Web
anesthesia management cleft lip palate repair intubation airway preformed RAE tube 2024 guidelines
Reading Web Page
anesthesia management cleft lip palate airway intubation induction analgesia postoperative complications
I now have comprehensive content from Miller's Anesthesia 10e, Mulholland & Greenfield's Surgery, and the peer-reviewed PMC article on anaesthesia for cleft lip/palate surgery. Here is the complete guide:
Anaesthesia Management of Cleft Lip and Palate
Sources: Miller's Anesthesia 10e (Ch. 79); Mulholland & Greenfield's Surgery 7e (Ch. 107); Anaesthesia for cleft lip and palate surgery, BJA Education 2021
1. Overview and Surgical Timing
Cleft lip and palate (CLP) is the most common congenital craniofacial anomaly — incidence ~1 in 650 live births. Approximately one-third are associated with a syndrome (e.g., Pierre Robin sequence, Treacher Collins, Stickler, Van der Woude).
Staged Surgical Repair
| Age | Procedure |
|---|---|
| 0–3 months | Presurgical orthodontics (optional) |
| 3–6 months | Cleft lip repair (cheiloplasty) |
| 9–12 months | Cleft palate repair (palatoplasty) |
| 3–6 years | Pharyngoplasty / palatal lengthening |
| 7–9 years | Alveolar bone grafting |
| 10–12 years | Rhinoplasty + lip revision |
| 16–18 years | Orthognathic surgery |
— Mulholland & Greenfield's Surgery 7e, Table 107.5
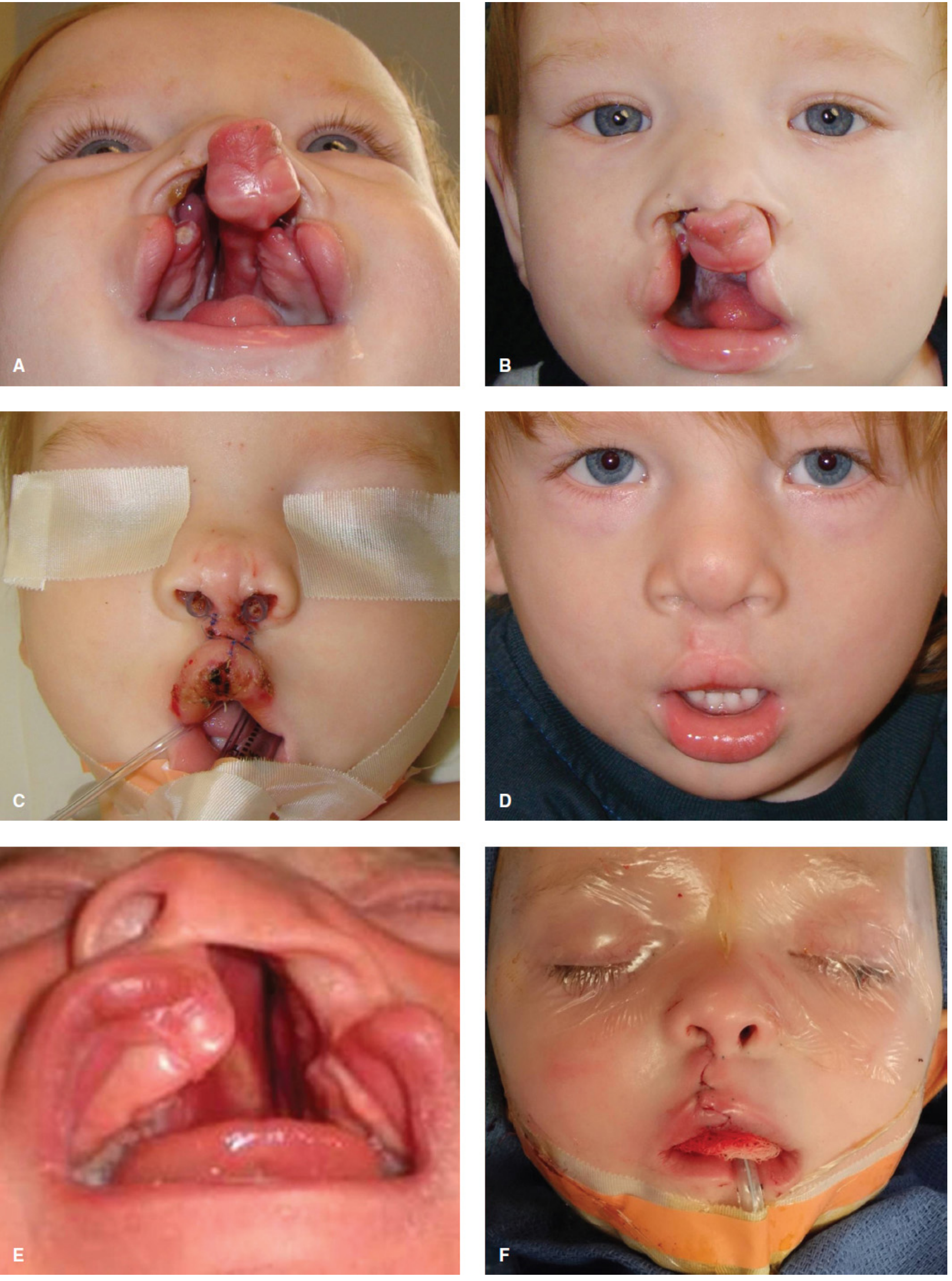
2. Preoperative Assessment
A. Airway Evaluation (Most Critical Step)
- Retrognathia (common in Pierre Robin, Treacher Collins) → anticipated difficult airway
- Large or bilateral cleft: tongue may prolapse into cleft, obstructing the airway
- Laryngoscope blade may lodge in the cleft during direct laryngoscopy
- Assess mouth opening, neck mobility, Mallampati score (as applicable for age)
- Review any prior intubation records
- Syndromic associations must be identified — the syndrome often defines the airway difficulty more than the cleft itself
B. Systemic Assessment
| System | What to Check |
|---|---|
| Cardiac | ~10–15% have associated CHD (especially in syndromic CLP); ECHO if indicated |
| Respiratory | Upper respiratory tract infection (URTI) — see below |
| Nutritional/Weight | Feeding difficulties → poor weight gain; aim for weight ≥5 kg for cleft lip repair |
| Hearing | Chronic otitis media / hearing loss (Eustachian tube dysfunction) |
| Genetic | Syndromes: Pierre Robin, Treacher Collins, CHARGE, Down syndrome |
C. URTI Policy
- Active URTI with fever, purulent secretions, wheeze → postpone 4–6 weeks
- Mild clear rhinorrhea alone: anaesthetist's discretion (often acceptable to proceed)
- Post-URTI airway hyperreactivity persists for up to 6 weeks
D. Premedication
- Oral midazolam 0.5 mg/kg (max 15 mg) — 20–30 minutes before induction; reduces separation anxiety
- Atropine 20 mcg/kg IV at induction (some centres give IM pre-op) — reduces secretions, prevents bradycardia during intubation
- Routine fasting: 6h solid food, 4h breast milk, 2h clear fluids (234 rule)
3. Induction of Anaesthesia
Standard Approach (Uncomplicated Airway)
- Inhalational induction with sevoflurane in O₂/air — preferred in infants; avoids IV needle in awake child
- Establish IV access once adequately deep
- Muscle relaxant only after confirming facemask ventilation is possible
- Options: Suxamethonium 2 mg/kg or atracurium/rocuronium (non-depolarising)
Anticipated Difficult Airway (Retrognathia, Syndromic)
| Priority | Strategy |
|---|---|
| 1st | Inhalational induction maintaining spontaneous ventilation |
| 2nd | Video laryngoscopy (Airtraq, C-MAC paediatric) — reduces failed laryngoscopy |
| 3rd | Hybrid technique: video laryngoscopy + flexible bronchoscopy via ETT — most effective for anticipated difficult airways |
| Rescue | Nasopharyngeal airway (does NOT damage palate repair); LMA for oxygenation as bridge |
| Last resort | Tracheostomy (rarely needed in primary cleft repair) |
Never administer a muscle relaxant until you have confirmed the ability to ventilate by mask.
4. Airway Device — The RAE Tube
The oral preformed south-facing RAE (Ring-Adair-Elwyn) tube is the device of choice.
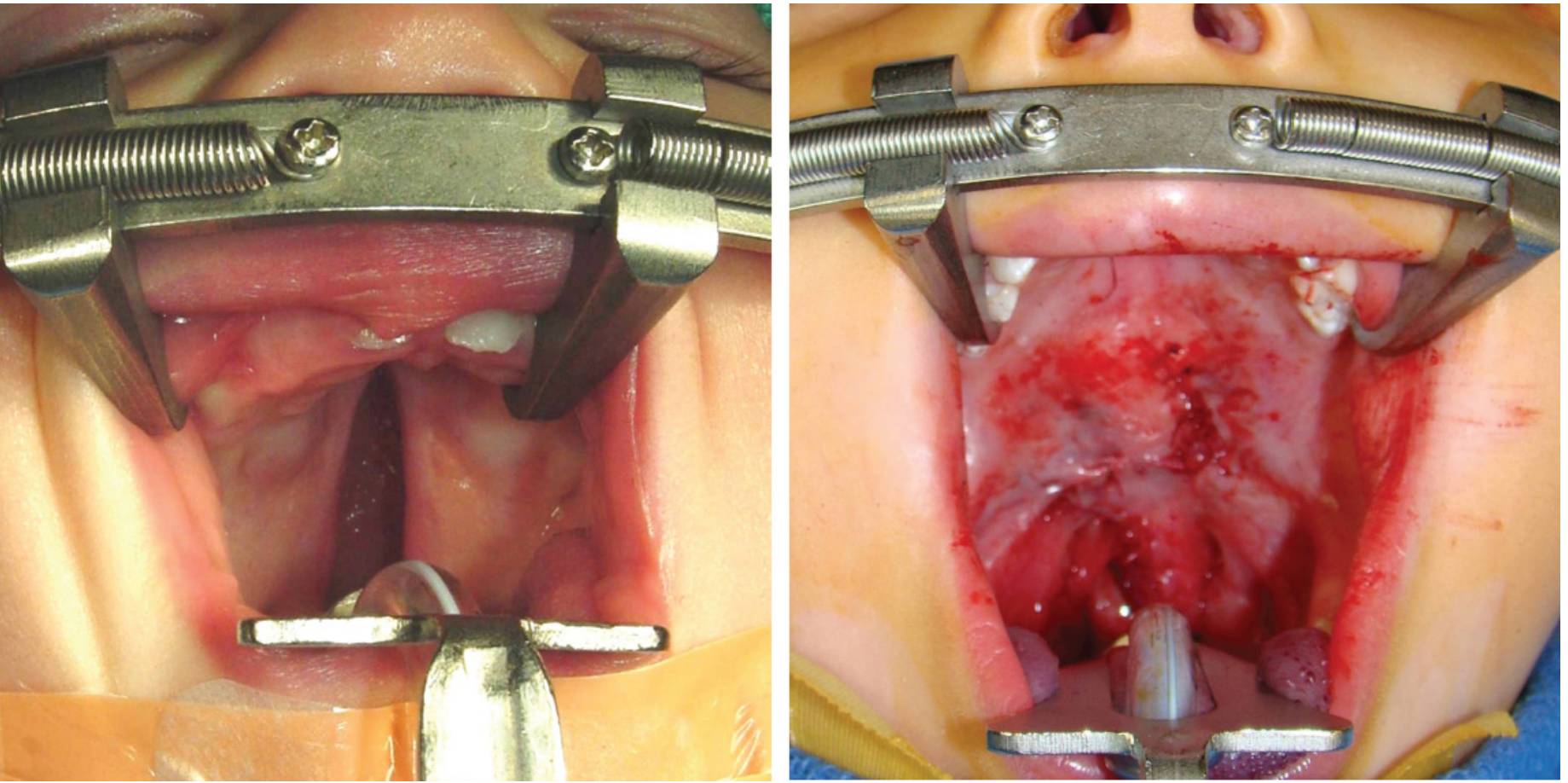
Why RAE?
- Preformed bend exits over the lower lip and chin, pointing downward
- Taped centrally in the midline
- Keeps the tube out of the surgical field while allowing the surgeon full access to the lips and palate
- Fixed intra-oral length is both advantage (airway security) and disadvantage (risk of endobronchial intubation)
RAE Tube Practical Points
| Issue | Solution |
|---|---|
| Tube too long → endobronchial intubation | Place gauze/dental roll padding between tube and chin as a platform; secure to it |
| Tube not fitting well | Use a reinforced (armoured) tube — flexible enough to follow chin contour |
| Surgeon inserts mouth gag (Dingman/Boyle-Davis) | Check tube position — gag can kink or displace the tube → most common cause of intraoperative extubation |
| Throat pack insertion | Document, write on patient's forehead, remove before extubation |
Throat Pack
- A moist throat pack may be placed to absorb blood and debris from the palate
- Must be documented and counted — retained throat pack is a serious never event
- "Throat pack in — throat pack out" checklist is mandatory
5. Maintenance of Anaesthesia
| Parameter | Target |
|---|---|
| Agent | Volatile (sevoflurane/isoflurane) or TIVA (propofol) |
| Ventilation | IPPV; normocapnia |
| Temperature | Active warming — neonates and infants lose heat rapidly; use warming mattress, warm IV fluids |
| Fluids | 4:2:1 rule maintenance + deficit; glucose-containing maintenance in neonates |
| Blood loss | Usually minimal in cleft lip; palate repair can bleed more; have blood grouped and held |
| Local anaesthetic infiltration | Surgeon typically infiltrates adrenaline 1:200,000 — watch for tachyarrhythmia; max adrenaline dose 10 mcg/kg |
Patient Positioning
- Supine, shoulder roll to extend neck → improves surgical access
- Head ring to stabilise position
- Recheck tube position after every positional change — neck extension can advance the tube tip
6. Analgesia (Multimodal)
Cleft Lip Repair
| Drug | Dose | Notes |
|---|---|---|
| Paracetamol (acetaminophen) | 15–20 mg/kg IV/PR | Regular dosing, backbone of analgesia |
| Infraorbital nerve block | 0.1 mL/kg of 0.25% bupivacaine | Bilateral; provides excellent postop analgesia for lip repair; landmark or US-guided |
| NSAIDs (ketorolac/ibuprofen) | Age-appropriate dosing | If no bleeding concern; ≥3 months |
| Opioids | Fentanyl 1–2 mcg/kg intraop | Judicious use — risk of postop respiratory depression |
Cleft Palate Repair
- Paracetamol + NSAIDs (regular)
- Greater palatine nerve block (by surgeon intraoperatively)
- Opioids used cautiously — palate repair patients at higher risk of postoperative airway obstruction
- Dexamethasone 0.15 mg/kg IV — reduces swelling and has antiemetic effect
Infraorbital nerve block is the most effective regional technique for cleft lip and is strongly recommended. — Miller's Anesthesia 10e, p. 10644
7. Extubation
Timing: Awake Extubation is Standard for Palate Repair
- Miller's Anesthesia explicitly states: "The child should be extubated awake" after palate repair
- Full reversal of neuromuscular block (check TOF ratio ≥0.9)
- Suction oropharynx under direct visualisation — careful not to disrupt suture lines
- Never blindly suction after palatoplasty
- Remove throat pack (confirm count)
- Extubate in left lateral or semi-prone position to allow drainage of secretions
- Have airway adjuncts ready: nasopharyngeal airway, facemask, LMA, reintubation equipment
Deep vs Awake Extubation
| Cleft Lip | Cleft Palate |
|---|---|
| Either approach acceptable | Awake extubation preferred |
| Deep extubation: ↓coughing/bucking (protects repair) | Awake: ensures airway protective reflexes present |
| Risk: post-extubation obstruction if deep | Higher risk of obstruction — avoid deep extubation |
8. Postoperative Care
Immediate (Recovery Room)
- Lateral/semi-prone position until fully awake
- Supplemental oxygen until stable SpO₂
- Monitor closely for 12–24 hours after palate repair (risk period for obstruction and bleeding)
- Nasopharyngeal airway may be left in situ for up to 6 hours if obstruction occurs post-palatoplasty (does NOT damage the repair)
Complications to Watch For
| Complication | Timing | Action |
|---|---|---|
| Airway obstruction | Immediate (0–48h) | Jaw thrust, nasopharyngeal airway, nebulised adrenaline (0.5 mg/kg, max 5 mg); reintubation if needed |
| Postextubation stridor | Immediate | Nebulised epinephrine; IV dexamethasone; may prevent reintubation in syndromic patients |
| Bleeding | Up to 24h | Return to theatre; correct coagulopathy |
| Disrupted suture line | Intraop/postop | Avoid oral instrumentation; tongue stitch if Pierre Robin |
| Inadvertent extubation | Intraop | Most common after mouth gag insertion — prevention is key |
| Retained throat pack | Post-extubation | Strict count protocol |
Admission Policy
- Cleft lip: overnight stay, usually discharge next day
- Cleft palate: 1–3 days admission (higher risk of bleeding and obstruction)
- Syndromic patients with comorbidities → PICU level care
9. Associated Syndromes — Anaesthetic Relevance
| Syndrome | Key Anaesthetic Concern |
|---|---|
| Pierre Robin sequence | Micrognathia + glossoptosis → very difficult airway; use video laryngoscopy; prone positioning may open airway; consider tongue stitch |
| Treacher Collins (TCS) | Bilateral mandibular hypoplasia, choanal atresia, absent zygomas → one of the most difficult airways; fibreoptic intubation often needed |
| Stickler syndrome | Myopia, retinal detachment, hearing loss; airway usually manageable |
| CHARGE syndrome | Coloboma, Heart defects, Atresia choanae, Restricted growth, Genital abnormalities, Ear abnormalities → choanal atresia makes nasal route impossible |
| Down syndrome (T21) | Atlantoaxial instability — avoid excessive neck flexion; large tongue; small subglottis |
| Crouzon/Apert | Craniosynostosis + midface hypoplasia → ↑ICP, difficult mask ventilation and intubation |
10. High-Yield Exam Summary
| Topic | Key Point |
|---|---|
| Cleft lip repair age | 3–6 months |
| Cleft palate repair age | 9–12 months |
| Airway device of choice | Oral south-facing RAE tube, taped midline below lower lip |
| Most dangerous intraop event | Inadvertent extubation when mouth gag inserted |
| Extubation after palatoplasty | Awake extubation mandatory |
| Best regional block (lip) | Bilateral infraorbital nerve block |
| Postop obstruction rescue | Nasopharyngeal airway (does not damage palate repair) |
| Throat pack | Must be documented and counted out — never event if retained |
| Highest risk group | Syndromic patients (Pierre Robin, Treacher Collins) → PICU |
| ~⅓ of CLPs | Associated with other syndromes — thorough pre-op assessment mandatory |
This is a shared conversation. Sign in to Orris to start your own chat.