Medullary Carcinoma of Thyroid explained & Management essay with diagrams & flowcharts under 1000 words
Reading File
Finding Sources
Finding Sources
Searching Images
medullary carcinoma thyroid histology amyloid
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Now I have everything needed. Here is the comprehensive essay:
Medullary Carcinoma of the Thyroid (MTC)
1. Definition & Cell of Origin
Medullary thyroid carcinoma (MTC) is a neuroendocrine malignancy arising from the parafollicular C cells of the thyroid — not from follicular cells. C cells produce calcitonin, and this hormonal signature is the cornerstone of both diagnosis and follow-up. MTC accounts for approximately 5–10% of all thyroid cancers but carries a disproportionate prognostic burden. — Robbins & Kumar Basic Pathology
2. Classification: Sporadic vs. Familial
┌────────────────────────────────────────────────────────┐
│ MEDULLARY THYROID CARCINOMA │
│ │
│ ┌─────────────────────┐ ┌──────────────────────┐ │
│ │ SPORADIC (~70%) │ │ FAMILIAL (~30%) │ │
│ │ • Adults, 5th–6th │ │ • Germline RET │ │
│ │ decade │ │ mutation │ │
│ │ • Usually solitary │ │ • Bilateral & │ │
│ │ • Somatic RET mut. │ │ multicentric │ │
│ └─────────────────────┘ └──────────┬───────────┘ │
│ │ │
│ ┌────────────────────────┤ │
│ ▼ ▼ ▼ │
│ MEN 2A MEN 2B Familial MTC │
│ (MTC+Pheo+HPT) (MTC+Pheo+ (MTC alone, │
│ Marfanoid + no MEN features) │
│ Neuromas) │
└────────────────────────────────────────────────────────┘
- MEN 2A (Sipple syndrome): MTC + pheochromocytoma (~50%) + hyperparathyroidism (20–30%)
- MEN 2B: MTC + pheochromocytoma + marfanoid habitus + mucosal neuromas — most aggressive form; tumors arise earlier and grow faster
- Familial MTC without MEN: intermediate aggressiveness
- In familial forms, MTC is usually the first tumor to develop, typically in the second or third decade of life — Mulholland & Greenfield's Surgery
3. Molecular Pathogenesis
The RET proto-oncogene (chromosome 10q11.2) is central to MTC:
RET proto-oncogene mutation
│
Gain-of-function
│
Constitutive RET kinase
activation (tyrosine
kinase receptor)
│
┌──────┴──────┐
│ │
Germline Somatic
(Familial) (Sporadic ~50%)
│
▼
C-cell hyperplasia → Micromedullary CA → Invasive MTC
(precursor lesion)
All patients with MTC must be tested for RET mutations to enable genetic counselling and prophylactic management of family members. — Harrison's Principles of Internal Medicine 22E
4. Pathology
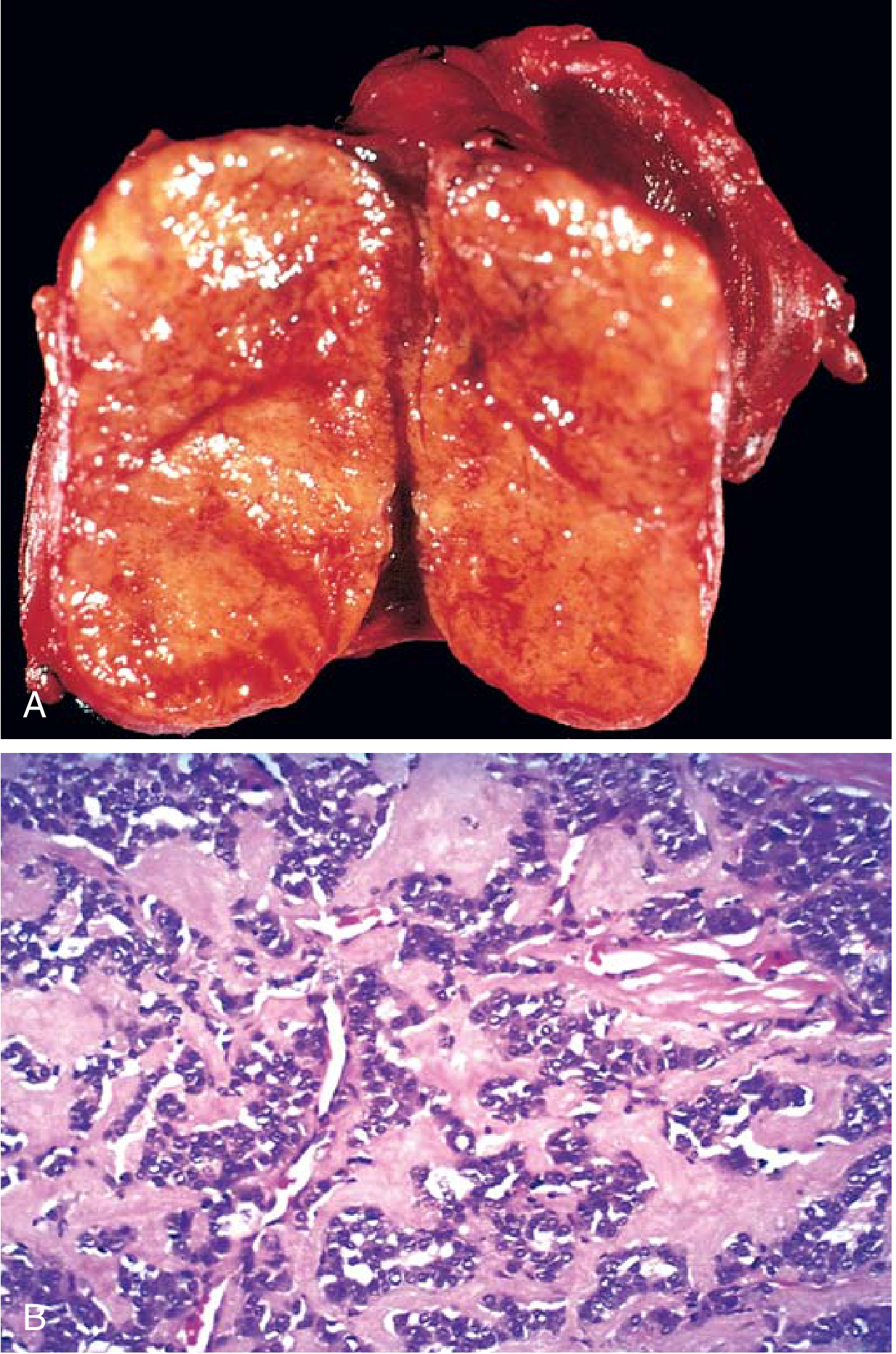
Gross: Solid, firm, tan-brown to grey mass, often without a capsule; larger tumours show haemorrhage and necrosis with possible capsular extension.
Microscopy: Polygonal to spindle-shaped cells arranged in nests, trabeculae, or gland-like structures. The hallmark is amyloid deposition in the stroma — derived from altered calcitonin molecules — identifiable as homogeneous pink extracellular material on H&E, and Congo red positive with apple-green birefringence under polarised light.
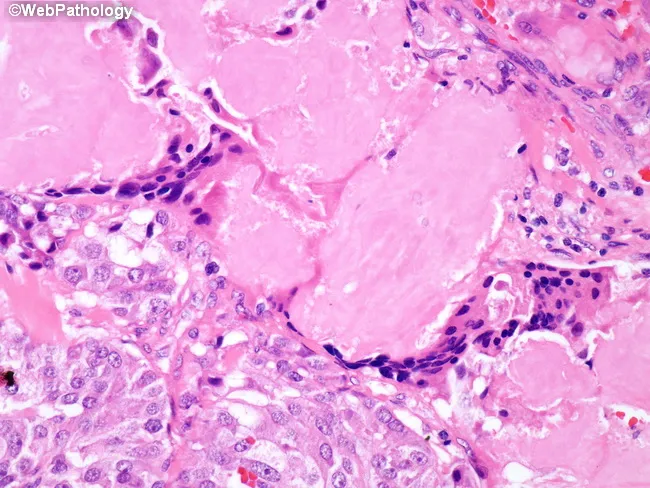
Immunohistochemistry: Positive for calcitonin, CEA, chromogranin A, synaptophysin, TTF-1; negative for thyroglobulin (distinguishing MTC from follicular-derived carcinomas).
Familial hallmark: C-cell hyperplasia in surrounding parenchyma — the precursor lesion — virtually absent in sporadic MTC.
5. Clinical Features
| Feature | Detail |
|---|---|
| Presentation | Neck mass; hoarseness/dysphagia if compressing |
| Diarrhoea | Secretory; due to VIP/calcitonin (severe in advanced disease) |
| Cervical nodes | Palpable in many at presentation |
| Ectopic hormones | ACTH (Cushing's), serotonin, prostaglandins |
Basal plasma calcitonin is markedly elevated. After calcium/pentagastrin stimulation, levels rise >1,000 pg/mL (normal stimulated peak <200 pg/mL) — a sensitive test for occult or recurrent disease. — Mulholland & Greenfield's Surgery
6. Diagnosis & Workup
Suspected Thyroid Nodule / ↑ Calcitonin / Family History of MTC
│
┌─────────▼──────────┐
│ Serum Calcitonin │
│ & CEA levels │
└─────────┬──────────┘
│ Elevated
┌─────────▼──────────┐
│ FNA Cytology │ ← spindle/polygonal cells,
│ + IHC (calcitonin) │ amyloid
└─────────┬──────────┘
│
┌─────────▼──────────┐
│ RET Mutation │
│ Testing (germline)│
└─────────┬──────────┘
│
┌──────────────┴───────────────┐
▼ ▼
RET Positive RET Negative
→ Screen family → Sporadic MTC
→ Screen for Pheo → Local staging
→ Screen for HPT
│
┌────▼────────────────────────────────┐
│ If RET+ → EXCLUDE PHEO FIRST │
│ (urine/plasma metanephrines, CT/MRI)│
└─────────────────────────────────────┘
│
┌─────────▼──────────┐
│ Staging: │
│ Neck U/S, CT Chest │
│ Abdomen, FDG-PET │
│ (hepatic/skeletal │
│ metastasis) │
└────────────────────┘
7. Management
Surgery — the only curative modality
┌──────────────────────────────────────────────────────────────┐
│ SURGICAL MANAGEMENT │
├──────────────────────────────────────────────────────────────┤
│ │
│ CLINICALLY EVIDENT MTC: │
│ Total thyroidectomy + │
│ Bilateral central neck dissection │
│ + Ipsilateral lateral neck dissection │
│ (if tumour >1 cm or central node metastasis) │
│ │
│ PROPHYLACTIC (RET mutation, no clinical MTC): │
│ Total thyroidectomy + central neck dissection │
│ Timing by RET mutation risk category: │
│ Highest risk (MEN 2B, C634F/Y) → age <1 year │
│ High risk → age <5 years │
│ Moderate risk → age 5–10 years │
│ │
│ Post-op: lifelong levothyroxine replacement │
│ ~90% of young prophylactic patients: no recurrence at 7 yrs │
└──────────────────────────────────────────────────────────────┘
Important: Pheochromocytoma must be excluded before surgery in all RET-mutation carriers, as undetected phaeochromocytoma causes intraoperative hypertensive crises. — Harrison's Principles of Internal Medicine 22E
Postoperative Monitoring
- Serum calcitonin and CEA at 3 months post-op and then periodically
- Rising calcitonin → provocative testing → re-operative neck dissection (including mediastinal exploration) can normalise calcitonin and achieve biochemical cure
Advanced / Metastatic Disease
Unlike differentiated thyroid cancer, MTC does not take up radioiodine — ¹³¹I has no role.
| Treatment | Role |
|---|---|
| Vandetanib / Cabozantinib (TKIs) | First-line for unresectable/metastatic MTC; improve progression-free survival |
| Selpercatinib | Selective RET kinase inhibitor; most effective agent; preferred for RET-mutated MTC |
| ¹⁷⁷Lu-DOTATATE (PRRT) | For somatostatin receptor-positive MTC on SRS |
| External beam radiotherapy | Palliative; local disease control |
| Chemotherapy | Limited efficacy; not standard |
— Harrison's Principles of Internal Medicine 22E
8. Prognosis
- 10-year survival in metastatic MTC: ~20%
- MEN 2A: often indolent; survival prolonged even with metastases
- MEN 2B: aggressive; early death possible
- Familial MTC (non-MEN): intermediate
- Biochemical cure (normalised calcitonin) = best surrogate for survival
Summary Flowchart
MTC DIAGNOSIS
│
├─ Sporadic (70%) ──→ Total thyroidectomy + central dissection
│ → Monitor calcitonin/CEA post-op
│
└─ Familial (30%) ──→ Germline RET testing
│
├─ MEN 2A / 2B ──→ Exclude Pheo first
│ → Total thyroidectomy (timing by risk)
│ → Screen for HPT (MEN 2A)
│
└─ Recurrent/Metastatic ──→ Selpercatinib (RET+)
Vandetanib / Cabozantinib
PRRT if SSTR+
Radiotherapy (palliation)
Sources: Robbins & Kumar Basic Pathology; Harrison's Principles of Internal Medicine 22E; Mulholland & Greenfield's Surgery 7e; Robbins, Cotran & Kumar Pathologic Basis of Disease
Iatrogenic Facial Nerve Palsy - essay for ENT exam under 1000 words with flowcharts & diagrams.
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Searching Images
facial nerve anatomy parotid branches surgical
Loading Image
Reading File
I now have comprehensive textbook content. Let me compile the essay.
Iatrogenic Facial Nerve Palsy
An ENT Exam Essay
1. Introduction
Iatrogenic facial nerve palsy is one of the most feared complications in head and neck surgery. The facial nerve (CN VII) has a long, tortuous course through the temporal bone and parotid gland, rendering it vulnerable at multiple points during surgical procedures. Consequences range from temporary weakness to permanent paralysis, with profound functional (eye closure, oral competence) and psychological impact on the patient. — Cummings Otolaryngology
2. Anatomy at Risk
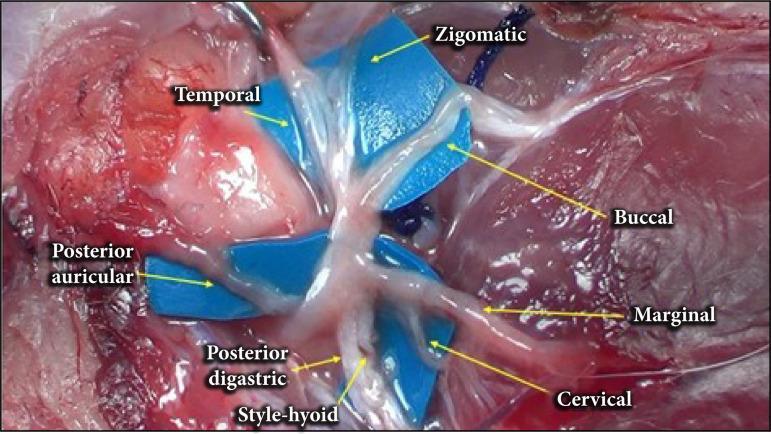
COURSE OF THE FACIAL NERVE — SITES OF SURGICAL VULNERABILITY
═══════════════════════════════════════════════════════════════
Brainstem (pons)
│
Internal Auditory Canal (IAC) ←─── Acoustic neuroma surgery
│
Labyrinthine segment
(geniculate ganglion) ←──────────── Temporal bone fracture /
│ middle fossa approach
Tympanic (horizontal) segment ←─── Cholesteatoma / tympanoplasty /
│ ossiculoplasty
Mastoid (vertical) segment ←─────── Mastoidectomy (most common
│ iatrogenic site)
Stylomastoid foramen
│
Extratemporal (parotid) ←────────── Parotidectomy / parotid surgery
│
┌────┴────────────────────────────┐
Temporal | Zygomatic | Buccal |
Marginal Mandibular | Cervical ←── Facelift, neck dissection,
submandibular gland excision
The mastoid (vertical) segment is the most commonly injured intratemporal site; the marginal mandibular branch is the most commonly injured extratemporal branch. — Schwartz's Principles of Surgery
3. Causes & Operations at Risk
| Operation | Mechanism of Injury |
|---|---|
| Mastoidectomy | Direct trauma to vertical segment; most common cause overall |
| Parotidectomy | Traction, thermal, or transection injury to main trunk / branches |
| Acoustic neuroma / CPA surgery | Stretch/traction on nerve at IAC or brainstem |
| Middle ear / tympanoplasty | Exposed or dehiscent tympanic segment |
| Stapedectomy | Abnormal nerve overhanging oval window |
| Neck dissection | Marginal mandibular branch at submandibular triangle |
| Parotid / submandibular excision | Peripheral branch injury |
| Facelift (rhytidectomy) | Temporal or zygomatic branches |
| Temporal bone fracture surgery | Compression, haematoma, entrapment at labyrinthine segment / geniculate ganglion |
4. Mechanisms of Nerve Injury (Sunderland Classification)
┌──────────────────────────────────────────────────────────────┐
│ MECHANISMS OF IATROGENIC NERVE INJURY │
├────────────────┬─────────────────────────────────────────────┤
│ Neuropraxia │ Conduction block only; axon intact │
│ (Grade I) │ → Full spontaneous recovery expected │
├────────────────┼─────────────────────────────────────────────┤
│ Axonotmesis │ Axon disrupted; endoneurium intact │
│ (Grade II-IV) │ → Partial/full recovery with time │
├────────────────┼─────────────────────────────────────────────┤
│ Neurotmesis │ Complete transection │
│ (Grade V) │ → No spontaneous recovery; repair required │
└────────────────┴─────────────────────────────────────────────┘
Intraoperative causes:
• Traction / stretching
• Thermal (diathermy, drill heat)
• Compression (packing, retractors)
• Transection / laceration
• Devascularisation
• Oedema / haematoma (delayed onset)
5. Recognition & Intraoperative Management
INTRAOPERATIVE FACIAL NERVE CONCERN
│
┌────────▼─────────┐
│ Positively ID │
│ nerve using │
│ distant landmarks │
└────────┬──────────┘
│
┌────────▼──────────────────────────────────┐
│ Trace nerve to site of suspected injury │
└────────┬──────────────────────────────────┘
│
┌────────▼─────────────────────────────────────────┐
│ Assess extent of injury │
│ < 50% diameter → local decompression, observe │
│ > 50% diameter → primary reanastomosis │
│ Gap / loss of tissue → interposition nerve graft │
│ (great auricular or sural nerve) │
└────────┬─────────────────────────────────────────┘
│
If landmarks NOT discernible:
STOP dissection. Seek senior / specialist consultation.
Intraoperative adjuncts:
- Facial nerve integrity monitor (FNIM / EMG): Provides real-time feedback via auditory alarm on nerve stimulation; mandatory in acoustic neuroma, parotid, and revision ear surgery
- Nerve stimulator probe: Localises the nerve in difficult fields
6. Postoperative Management Flowchart
POST-OP FACIAL WEAKNESS NOTED
│
┌────────▼──────────────────────────────┐
│ Is this EXPECTED (warned) or SURPRISE? │
└────────┬──────────────────────────────┘
│
┌───────┴──────────┐
▼ ▼
Expected Surprise (unexpected)
(e.g. planned ─────────────────────
nerve sacrifice) 1. Release tight dressings & packing
│ 2. Allow local anaesthetic to dissipate
▼ (wait 30–60 min)
Conservative 3. If no recovery → URGENT re-exploration
management and decompression
+ rehab 4. Identify & address site of injury
5. Intraop repair if needed
SEVERITY ASSESSMENT (House-Brackmann Grade)
I — Normal
II — Mild dysfunction; complete eye closure with effort
III — Moderate; obvious asymmetry; complete eye closure
IV — Moderately severe; incomplete eye closure
V — Severe; barely perceptible movement
VI — Total paralysis
Electrophysiological testing (ENoG / EMG):
- ENoG showing >90% degeneration within 6 days → consider surgical decompression
- EMG: detects fibrillation potentials (denervation) vs. polyphasic units (reinnervation)
- Long-standing paralysis: if no clinical or EMG recovery at 12 months → surgical exploration indicated — Cummings Otolaryngology
7. Treatment of Established Palsy
| Timing | Intervention |
|---|---|
| Immediate (intraop) | Primary neurorrhaphy / interposition nerve graft |
| Early (<3 weeks) | Re-explore; nerve graft if needed |
| Intermediate (weeks–months) | Eye care (lubricants, taping, moisture chamber); physiotherapy; gold weight upper eyelid implant for lagophthalmos |
| Late / permanent | Facial reanimation: hypoglossal-facial anastomosis; microneurovascular free muscle transfer (temporalis/latissimus dorsi); static slings; brow lift |
Eye care is paramount — incomplete lid closure causes corneal exposure, dryness, and potential blindness.
8. Prevention
KEY PREVENTIVE PRINCIPLES
─────────────────────────
✓ Thorough anatomical knowledge of the facial nerve course
✓ Pre-op imaging (MRI/CT) to identify aberrant anatomy
✓ Routine intraoperative nerve monitoring
✓ Meticulous surgical technique; avoid excessive traction
✓ Minimal use of monopolar diathermy near nerve
✓ Adequate decompression; avoid tight postoperative packing
✓ Informed consent — discuss risk explicitly before surgery
✓ Identify high-risk patients (revision surgery, parotid tumours,
cholesteatoma, temporal bone fractures)
9. Summary
Iatrogenic facial nerve palsy results from surgical injury during mastoidectomy, parotidectomy, CPA surgery, or neck dissection. The vertical segment of the mastoid is the most common intratemporal site; the marginal mandibular branch is most vulnerable extracranially. Intraoperative recognition demands immediate assessment and repair; unexpected postoperative palsy requires urgent re-exploration. Outcomes depend on extent of injury — neuropraxia recovers fully, while neurotmesis requires surgical reconstruction. Prevention through nerve monitoring, anatomical expertise, and careful dissection remains the gold standard.
Sources: Cummings Otolaryngology Head and Neck Surgery; Schwartz's Principles of Surgery 11e; Localization in Clinical Neurology 8e
This is a shared conversation. Sign in to Orris to start your own chat.