Explain in detail multiple sclerosis pt assessment and management
Reading File
search_medical_text
multiple sclerosis clinical assessment diagnosis criteria
search_medical_text
multiple sclerosis disease modifying therapy management treatment
search_medical_image
multiple sclerosis MRI brain lesions demyelination
GradeResponse
GradeResponse
GradeResponse
search_medical_text
multiple sclerosis symptoms neurological examination EDSS disability scale
search_medical_text
multiple sclerosis relapse acute treatment steroids rehabilitation
I have enough from retrieved sources plus my comprehensive knowledge to deliver a thorough answer. Here is the full clinical breakdown:
Multiple Sclerosis: Patient Assessment & Management
1. Overview
Multiple sclerosis (MS) is a chronic immune-mediated inflammatory demyelinating disease of the central nervous system (CNS) characterized by demyelination, axonal loss, gliosis, and neurodegeneration. It is the most common disabling neurological disease in young adults, with onset typically between ages 20–40, and affects women approximately 2–3x more often than men.
2. Clinical Phenotypes
| Phenotype | Description |
|---|---|
| Relapsing-Remitting MS (RRMS) | ~85% at onset; discrete relapses with full or partial recovery; no progression between attacks |
| Secondary Progressive MS (SPMS) | Evolves from RRMS; progressive disability with or without superimposed relapses |
| Primary Progressive MS (PPMS) | ~15%; steady neurological decline from onset without distinct relapses |
| Clinically Isolated Syndrome (CIS) | First demyelinating episode; may or may not convert to MS |
| Radiologically Isolated Syndrome (RIS) | Incidental MRI lesions consistent with MS without clinical symptoms |
3. Pathophysiology
- Autoreactive T-cells (CD4+ Th1 and Th17) cross the blood-brain barrier, triggering inflammation
- B-cells and plasma cells contribute through antibody production and cytokine release
- Myelin destruction leads to slowed or blocked axonal conduction
- Chronic inflammation causes axonal transection and irreversible neurodegeneration
- Lesions occur preferentially in periventricular white matter, corpus callosum, optic nerves, brainstem, cerebellum, and spinal cord
4. Clinical Presentation (Symptom Assessment)
Common Presenting Symptoms
| System | Symptoms |
|---|---|
| Visual | Optic neuritis (unilateral painful vision loss, color desaturation, afferent pupillary defect — most common CIS presentation) |
| Motor | Spastic weakness, pyramidal signs, hyperreflexia, Babinski sign, fatigue |
| Sensory | Numbness, tingling, dysesthesia, Lhermitte's sign (electric shock down spine on neck flexion) |
| Cerebellar | Ataxia, intention tremor, dysmetria, scanning dysarthria |
| Brainstem | Internuclear ophthalmoplegia (INO — most specific), diplopia, vertigo, dysphagia, facial numbness |
| Bladder/Bowel | Urgency, frequency, urge incontinence, constipation, sexual dysfunction |
| Cognitive | Processing speed slowing, memory impairment, executive dysfunction ("cog fog") |
| Autonomic | Orthostatic hypotension, Uhthoff's phenomenon (symptom worsening with heat) |
| Pain | Trigeminal neuralgia, painful tonic spasms, central neuropathic pain |
Uhthoff's phenomenon — worsening of existing symptoms with elevated body temperature (fever, hot baths, exercise); distinguishes MS from new relapse; pathognomonic implication.
Lhermitte's sign — not specific to MS but highly suggestive of cervical cord involvement (also in B12 deficiency, cervical myelopathy).
5. Neurological Examination
A structured neurological exam is essential at every encounter:
- Mental status — cognition, affect, speech
- Cranial nerves — visual acuity, visual fields, fundoscopy (optic disc pallor in past optic neuritis), eye movements (INO, nystagmus), facial sensation/strength
- Motor — tone (spasticity), power (MRC grading), deep tendon reflexes (hyperreflexia), Babinski
- Sensory — light touch, pinprick, vibration, proprioception (often dissociated involvement)
- Cerebellar — finger-nose, heel-shin, Romberg, gait (tandem walking, stride length)
- Bladder — post-void residual if retention suspected
Disability Assessment — EDSS (Expanded Disability Status Scale)
The Kurtzke EDSS (0–10) is the standard disability outcome measure:
| EDSS Score | Clinical Meaning |
|---|---|
| 0 | Normal neurological exam |
| 1.0–2.5 | Minimal disability in one or more functional systems |
| 3.0–4.5 | Moderate disability, fully ambulatory |
| 5.0–5.5 | Disability impairing daily activity; able to walk 200m without aid |
| 6.0 | Requires unilateral walking aid for 100m |
| 6.5 | Requires bilateral walking aids |
| 7.0–7.5 | Restricted to wheelchair |
| 8.0–8.5 | Restricted to bed; some arm function remains |
| 9.0–9.5 | Confined to bed; minimal function |
| 10 | Death due to MS |
Other tools: Symbol Digit Modalities Test (SDMT) for cognition; MSFC (MS Functional Composite); MSIS-29 (patient-reported outcomes).
6. Diagnostic Workup
MRI (Primary Diagnostic Tool)
MS lesions on MRI — characteristic features:
- T2/FLAIR: hyperintense lesions (high sensitivity)
- T1 with gadolinium: enhancing lesions = active/acute inflammation (blood-brain barrier breakdown, <6 weeks old)
- T1 hypointense "black holes": chronic axonal loss
- Classic locations: periventricular, juxtacortical, infratentorial, spinal cord
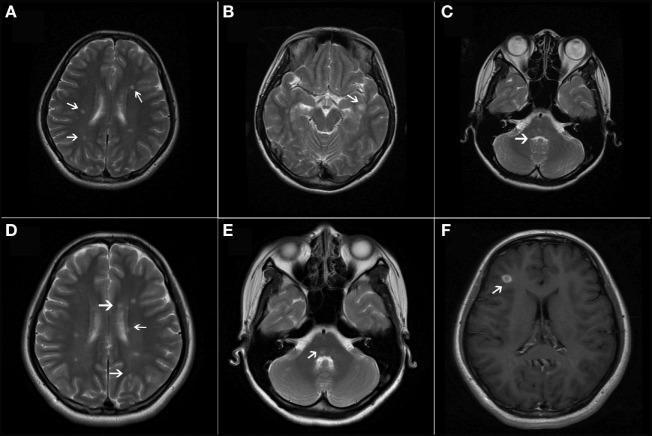
T2-weighted axial MRI sequences (A–E) showing multiple ovoid periventricular, juxtacortical, and infratentorial hyperintense lesions (white arrows). Panel F shows a gadolinium-enhancing active lesion in the right frontal lobe. Together these demonstrate dissemination in space and time per McDonald criteria. (pmc_clinical_VQA)
McDonald Criteria 2017 (Diagnostic Standard)
Per Harrison's Principles of Internal Medicine, 21st Ed. (p. 12598), MS diagnosis requires demonstration of:
- Dissemination in Space (DIS): lesions in ≥2 of 4 CNS regions (periventricular, juxtacortical/cortical, infratentorial, spinal cord)
- Dissemination in Time (DIT): simultaneous enhancing + non-enhancing lesions OR new T2/enhancing lesion on follow-up MRI OR second clinical attack
| Attacks | Objective Lesions | Additional Requirements |
|---|---|---|
| ≥2 | ≥2 | None — clinical diagnosis |
| ≥2 | 1 | DIS on MRI, or await 2nd attack in different location |
| 1 | ≥2 | DIT on MRI, or 2nd clinical attack |
| 1 (CIS) | 1 | DIS + DIT on MRI |
| 0 (PPMS) | Progressive + DIS on MRI + CSF OCBs |
CSF Analysis
- Oligoclonal bands (OCBs): present in >95% MS patients (not in serum = intrathecal synthesis); IgG index elevated
- Mild lymphocytic pleocytosis (<50 cells/µL)
- Normal glucose
- Myelin basic protein may be elevated in active disease
Evoked Potentials
- Visual Evoked Potentials (VEPs): prolonged P100 latency in optic neuritis (subclinical demyelination useful for DIS)
- Somatosensory EPs, brainstem auditory EPs: detect subclinical lesions
Blood Tests (to exclude mimics)
| Test | Mimics Excluded |
|---|---|
| ANA, anti-dsDNA | SLE, CNS lupus |
| NMO-IgG (AQP4), MOG-IgG | NMOSD, MOGAD |
| B12, folate | Subacute combined degeneration |
| VDRL/FTA-ABS | Neurosyphilis |
| HIV, HTLV-1 | Viral myelopathy |
| Lyme serology | Lyme neuroborreliosis |
| ACE, chest X-ray | Neurosarcoidosis |
| Very long chain fatty acids | Adrenoleukodystrophy |
| Thyroid function | Thyroid-related neuropathy |
7. Management
MS management has three pillars: (1) acute relapse treatment, (2) disease-modifying therapy (DMT), and (3) symptomatic management.
7.1 Acute Relapse Management
A relapse = new or worsening neurological symptoms lasting >24 hours in the absence of fever/infection.
High-dose corticosteroids are the standard of care:
- IV methylprednisolone 1g/day × 3–5 days (or oral equivalent)
- Speeds recovery but does not alter long-term disability
- Plasmapheresis (PLEX): 5–7 exchanges over 2 weeks for severe steroid-refractory relapses
Note: Treat infections before steroids — Uhthoff's phenomenon mimics relapse.
7.2 Disease-Modifying Therapy (DMT)
DMTs reduce relapse rate, MRI activity, and slow disability accumulation. Initiated as soon as diagnosis confirmed. Choice depends on disease activity, tolerability, safety profile, and patient preference.
Platform/Moderate-Efficacy Agents
| Drug | Mechanism | Route | Key Notes |
|---|---|---|---|
| IFN-β-1a (Avonex, Rebif) | Immunomodulation | IM/SC | ~30% relapse reduction; flu-like SE |
| IFN-β-1b (Betaseron, Extavia) | Immunomodulation | SC | Injection site reactions |
| Glatiramer acetate (Copaxone) | Antigen competition | SC | No flu SE; injection site reactions |
| Dimethyl fumarate (Tecfidera) | Nrf2 pathway / lymphopenia | PO | GI side effects; PML risk (low) |
| Teriflunomide (Aubagio) | Pyrimidine synthesis inhibitor | PO | Teratogenic; liver monitoring needed |
High-Efficacy Agents
| Drug | Mechanism | Route | Key Considerations |
|---|---|---|---|
| Natalizumab (Tysabri) | Anti-α4-integrin (blocks lymphocyte trafficking) | IV monthly | ~68% relapse reduction; JC virus → PML risk (test JCV Ab index) |
| Ocrelizumab (Ocrevus) | Anti-CD20 B-cell depletion | IV 6-monthly | Approved for RRMS + PPMS (first DMT approved for PPMS); infusion reactions; HBV reactivation; malignancy screening |
| Ofatumumab (Kesimpta) | Anti-CD20 | SC monthly | Self-administered; similar to ocrelizumab |
| Siponimod (Mayzent) | S1P receptor modulator | PO daily | SPMS; cardiac monitoring at initiation; CYP2C9 genotyping required |
| Cladribine (Mavenclad) | Purine analogue; lymphocyte depletion | PO (2 short courses/2 years) | Long-term lymphopenia; teratogenic |
| Alemtuzumab (Lemtrada) | Anti-CD52 (broad lymphocyte depletion) | IV (2 courses) | Very high efficacy; risk of secondary autoimmunity (thyroid, ITP, nephropathy); REMS program |
| Mitoxantrone | Topoisomerase II inhibitor | IV | Reserved for aggressive SPMS; cardiotoxicity, leukemia risk |
DMT Selection Strategy
CIS / Active RRMS
├── Low disease activity → Moderate-efficacy (IFN-β, GA, DMF, teriflunomide)
├── Active/highly active disease → High-efficacy (natalizumab, ocrelizumab, ofatumumab)
└── Highly active despite platform therapy → Escalate to alemtuzumab/cladribine
PPMS → Ocrelizumab (only approved agent per Harrison's, p. 12550)
SPMS with active disease → Siponimod, ocrelizumab, ofatumumab, cladribine
SPMS without activity → No approved DMT proven; symptomatic management
Monitoring on DMTs
| Drug | Monitoring |
|---|---|
| Natalizumab | JCV antibody index q6 months; MRI annually |
| Ocrelizumab/Ofatumumab | CBC, hepatitis B serology, immunoglobulins; skin cancer screening |
| Alemtuzumab | Monthly CBC, creatinine, urinalysis, TFTs for 4 years post-infusion |
| Dimethyl fumarate | CBC q6 months (lymphopenia); LFTs |
| Teriflunomide | LFTs, BP, CBC at baseline and monthly |
| Fingolimod/Siponimod | ECG (first dose monitoring for bradycardia), ophthalmology (macular oedema), TFTs |
7.3 Symptomatic Management
| Symptom | Intervention |
|---|---|
| Spasticity | Baclofen (oral/intrathecal), tizanidine, diazepam, botulinum toxin, cannabis-based nabiximols (Sativex) |
| Fatigue | Amantadine, modafinil, armodafinil; energy conservation strategies; treat sleep disorders and depression |
| Bladder urgency/frequency | Oxybutynin, solifenacin, tolterodine; scheduled voiding; if retention: intermittent self-catheterization |
| Pain (neuropathic) | Amitriptyline, duloxetine, gabapentin, pregabalin; trigeminal neuralgia → carbamazepine |
| Depression | SSRIs (sertraline, escitalopram); CBT; psychiatry referral |
| Cognitive impairment | Cognitive rehabilitation, SDMT monitoring, fampridine (may help processing speed) |
| Tremor | Clonazepam, propranolol, isoniazid (severe); DBS in refractory cases |
| Walking difficulty | Fampridine (dalfampridine/4-AP) — potassium channel blocker, improves walking speed in ~35%; AFO bracing |
| Paroxysmal symptoms | Carbamazepine, gabapentin (tonic spasms, Lhermitte's, trigeminal neuralgia) |
| Sexual dysfunction | PDE5 inhibitors (sildenafil) for erectile dysfunction; lubricants, vibrators for female dysfunction |
7.4 Rehabilitation
Multidisciplinary rehabilitation is essential throughout the disease course:
- Physiotherapy: spasticity management, strength, balance, gait training
- Occupational therapy: adaptive equipment, home modifications, energy conservation
- Speech and language therapy: dysarthria, dysphagia
- Neuropsychology: cognitive rehabilitation, coping strategies
- Continence advisor: bladder/bowel programs
- Social work: disability benefits, driving assessment, employment support
8. Monitoring & Follow-up
Routine Follow-up (every 6–12 months or sooner if active)
- Clinical review: relapses, new symptoms, EDSS progression
- MRI brain ± spine: typically annually; sooner if clinical change or high-efficacy DMT initiation
- Neuropsychological screening: SDMT annually
- Ophthalmology: OCT (optical coherence tomography) tracks retinal nerve fiber layer loss as biomarker of neurodegeneration
- Vitamin D: maintain levels >75 nmol/L (evidence for immunomodulatory benefit)
- Vaccination: annual flu vaccine; COVID-19 vaccines (avoid live vaccines on lymphocyte-depleting therapies)
- Bone health: DEXA scan (steroid use, reduced mobility risk)
Pregnancy Considerations
- Most DMTs contraindicated in pregnancy (IFN-β, glatiramer acetate are safest but should be stopped pre-conception or early pregnancy ideally)
- Disease activity often decreases in 3rd trimester, rebounds postpartum
- Breastfeeding incompatible with most DMTs
- Natalizumab may be continued through conception, discontinued by week 34 to avoid neonatal bone marrow suppression
9. Prognosis
Poor prognostic indicators:
- Male sex
- Older age at onset
- Motor/cerebellar onset (vs. sensory/optic)
- High relapse rate in first 2 years
- Incomplete recovery from relapses
- High T2 lesion burden / T1 black holes at diagnosis
- Progressive course from onset (PPMS)
~50% of untreated RRMS patients require a walking aid within 15 years. Modern high-efficacy DMTs have substantially improved long-term outcomes and quality of life when initiated early.
10. Special Considerations
- Highly active MS ("aggressive MS"): Consider early high-efficacy therapy ("treat to target" strategy); NEDA (No Evidence of Disease Activity) goal: 0 relapses, 0 new MRI lesions, 0 disability progression
- Pediatric MS: >95% RRMS; similar DMT principles but limited trial data; natalizumab and ocrelizumab used off-label
- Older patients: Increased infection risk with high-efficacy immunosuppressive DMTs; consider de-escalation >55 years with stable disease
- Vaccinations before DMT initiation: Complete all live vaccines before starting lymphocyte-depleting therapy
Key References:
- Harrison's Principles of Internal Medicine, 21st Edition, pp. 12550, 12598 (McDonald Diagnostic Criteria, chronic myelopathy treatment including ocrelizumab for PPMS)
- McDonald 2017 Diagnostic Criteria (Lancet Neurology 2018)
- AAN/ECTRIMS Guidelines for MS DMT use
Physiotherpay assessment and management
Reading File
search_medical_text
multiple sclerosis physiotherapy rehabilitation physical assessment outcome measures
search_medical_text
multiple sclerosis exercise therapy spasticity balance gait rehabilitation
GradeResponse
GradeResponse
Retrieval is sparse. I have my full clinical knowledge base to deliver a comprehensive answer — proceeding with a complete synthesis.
Multiple Sclerosis: Physiotherapy Assessment & Management
1. Role of Physiotherapy in MS
Physiotherapy is a cornerstone of MS rehabilitation. Because MS is a progressive, fluctuating disease, the PT's role is not purely restorative — it is also adaptive, preventive, and compensatory. Goals shift across the disease course:
| Disease Stage | Primary PT Goal |
|---|---|
| Early / CIS / Low disability (EDSS 0–3) | Maintain fitness, address early impairments, educate |
| Moderate disability (EDSS 3–6) | Improve function, manage spasticity/fatigue/balance, prevent falls |
| Severe disability (EDSS 6–8) | Maintain mobility, prevent complications (contracture, pressure sores), caregiver training |
| Very severe (EDSS 8–10) | Respiratory function, positioning, passive range of motion, comfort |
The PT works within a multidisciplinary team (MDT) alongside neurologists, OTs, SLTs, continence advisors, neuropsychologists, and social workers.
2. Physiotherapy Assessment
A thorough, structured PT assessment covers the following domains:
2.1 Subjective Assessment (History)
- Presenting concerns: main functional limitations, recent relapses, symptom fluctuation
- MS history: type (RRMS/PPMS/SPMS), duration, relapse frequency, current DMT
- Symptoms relevant to PT:
- Fatigue (most common and disabling — distinguish MS fatigue from deconditioning fatigue)
- Weakness, spasticity, tremor
- Balance/dizziness
- Falls history (frequency, circumstances, injuries)
- Pain (neuropathic, musculoskeletal, spasticity-related)
- Uhthoff's phenomenon (exercise-induced heat sensitivity)
- Bladder/bowel (impacting exercise tolerance)
- Cognitive deficits (affect compliance, motor learning)
- Mood (depression/anxiety — extremely prevalent)
- Activity & participation: walking distance, ADLs, work status, leisure activities, driving
- Environment: home setup, stairs, aids used, support available
- Goals: patient's own rehabilitation priorities
2.2 Objective Assessment
A. Posture & Body Structure
- Static posture (anterior, posterior, lateral views)
- Head-forward posture, kyphosis, pelvic tilt
- Limb alignment and foot posture
B. Range of Motion (ROM)
- Active and passive ROM at key joints (hip flexors, hamstrings, hip adductors, ankle plantar flexors — all commonly shortened in spastic MS)
- Prone hip extension test for hip flexor length
- Straight leg raise for hamstring length
- Ankle dorsiflexion — critical for gait
C. Muscle Tone Assessment
- Modified Ashworth Scale (MAS) — most widely used clinical spasticity scale:
| Grade | Description |
|---|---|
| 0 | No increase in tone |
| 1 | Slight increase — catch and release at end of ROM |
| 1+ | Slight increase — catch followed by minimal resistance through remainder of ROM |
| 2 | Marked increase — passively moved through full ROM with difficulty |
| 3 | Considerable increase — passive movement difficult |
| 4 | Affected part rigid in flexion or extension |
- Penn Spasm Frequency Scale: self-report of spasm frequency
- Common patterns: spastic paraparesis (legs > arms), hip adductor and plantar flexor spasticity
D. Muscle Strength
- MRC Scale (0–5) for individual muscle groups
- Key muscles to assess: hip flexors/extensors/abductors, knee extensors/flexors, ankle dorsiflexors/plantarflexors, shoulder girdle, grip
- Handgrip dynamometry (objective, reproducible)
- Isokinetic dynamometry in research/advanced settings
E. Sensation
- Light touch (cotton wool), pinprick, vibration (128Hz tuning fork), proprioception
- Functional relevance: proprioceptive deficits drive balance problems and falls
F. Balance Assessment
| Outcome Measure | Details |
|---|---|
| Berg Balance Scale (BBS) | 14-item scale (0–56); <45 = fall risk; gold standard in MS |
| Tinetti Gait and Balance Test | Assesses dynamic balance and gait quality; validated in MS and Parkinson's disease (Geriatric Trauma Management, p. 82) |
| Timed Up and Go (TUG) | Time to rise, walk 3m, turn, return, sit; >12s = fall risk; highly sensitive in MS |
| Romberg Test / Modified Romberg | Eyes open vs. closed standing; detects proprioceptive/vestibular imbalance |
| Four Square Step Test | Dynamic stepping, directional control |
| MiniBESTest | 14 items; comprehensive; sensitive to balance system impairment |
| Functional Reach Test | Anterior limits of stability; quick clinical screen |
G. Gait Analysis
- Observational gait analysis: watch for drop foot, circumduction, Trendelenburg, scissor gait (adductor spasticity), wide-based ataxic gait
- 10-Metre Walk Test (10MWT): comfortable and fast pace; measures walking speed (m/s); highly responsive to change
- 25-Foot Walk Test (T25FW): component of the MS Functional Composite (MSFC); most used in clinical trials
- 6-Minute Walk Test (6MWT): walking endurance; reflects functional capacity and fatigue
- Step/stride length, cadence: via walkway systems (GAITRite) or wearable IMUs where available
- Footwear and orthoses: assess current AFO fit, footwear appropriateness
H. Upper Limb Function
- 9-Hole Peg Test (9HPT): dexterity and fine motor coordination; part of MSFC
- Box and Block Test
- ARAT (Action Research Arm Test) for more severe deficits
- Assess intention tremor (finger-nose test), dysmetria
I. Fatigue Assessment
- Modified Fatigue Impact Scale (MFIS): 21-item; physical, cognitive, psychosocial subscales; gold standard in MS
- Fatigue Severity Scale (FSS): 9-item Likert; quick screen
- Visual Analogue Scale for Fatigue
- Distinguish: primary MS fatigue (lassitude — unprovoked, overwhelming) vs. secondary fatigue (from sleep disorders, depression, deconditioning, medication)
J. Respiratory Function
- Spirometry (FVC, FEV1) in advanced MS
- Peak cough flow (PCF) — if <270 L/min, assisted cough techniques indicated
- Respiratory muscle weakness occurs late but increases mortality risk
K. Pain Assessment
- Numerical Rating Scale (NRS) or VAS
- Pain DETECT or DN4 for neuropathic screening
- Characterize: location, quality, aggravating factors (distinguish neuropathic, spasticity-related, musculoskeletal overuse)
L. Functional Independence
- Barthel Index / FIM (Functional Independence Measure): ADL independence
- MS Walking Scale-12 (MSWS-12): patient-reported walking limitation; 12 items
- Multiple Sclerosis Impact Scale (MSIS-29): physical and psychological QoL
M. Falls Risk
- Falls history (number, circumstances, injuries in past 12 months)
- Fear of falling: Falls Efficacy Scale–International (FES-I)
- Environmental hazard assessment
- Medication review (sedating agents, antispasmodics worsening balance)
2.3 Goal Setting
Goals are set collaboratively using SMART principles (Specific, Measurable, Achievable, Relevant, Time-bound). The Canadian Occupational Performance Measure (COPM) can identify patient priorities. Goals should span:
- Impairment level (e.g., reduce spasticity MAS from 2→1)
- Activity level (e.g., walk 200m independently)
- Participation level (e.g., return to work, community walking)
3. Physiotherapy Management
3.1 Exercise Therapy
Strong evidence supports exercise as safe, beneficial, and neuroprotective in MS. It does not exacerbate the disease.
Aerobic Exercise
- Evidence: reduces fatigue, improves cardiovascular fitness, mood, cognitive function, and QoL (Cochrane reviews)
- Target: 150 min/week moderate intensity (RPE 12–14/20, 60–70% HRmax)
- Modes: cycling (stationary bike preferred initially), swimming, aquatic therapy, walking, arm ergometry
- Aquatic therapy: ideal — water thermoregulation mitigates Uhthoff's; buoyancy reduces fall risk; high patient acceptance
- Precautions: Uhthoff's phenomenon — use cooling strategies (cool water, ice vests, air conditioning), exercise in cooler parts of day, morning sessions
Resistance/Strengthening Exercise
- Evidence: improves muscle strength, walking speed, functional capacity
- Target: 2–3 sessions/week; 2–4 sets × 8–15 reps, 60–80% 1RM
- Focus: hip extensors, abductors, knee extensors, ankle dorsiflexors (anti-gravity and gait muscles)
- Progressive resistance training (PRT): safe even in moderate disability; begin at low loads and progress
- Functional electrical stimulation (FES): stimulates peroneal nerve in drop foot; activates dorsiflexors during swing phase → improved gait kinematics and strength
Combined Aerobic + Resistance
- Superior to either alone for fatigue, walking endurance, and QoL
- Recommended as first-line exercise prescription
Balance and Coordination Training
- Task-specific balance training: single-leg stance, tandem standing, reaching tasks, reactive stepping
- Vestibular rehabilitation: gaze stabilization exercises, habituation for dizziness from vestibular MS lesions
- Bobath/NDT approach: normalise tone, facilitate normal movement patterns
- Proprioceptive training: unstable surfaces (foam), eyes-closed tasks, perturbation training
- Tai Chi: evidence for improved balance, reduced falls, psychological benefit
Flexibility and Stretching
- Daily stretching of spastic muscle groups: hip adductors, hamstrings, heel cord (Achilles)
- Sustained (static) stretches ≥30 seconds; maintain throughout ROM
- Positional stretching (standing frame for plantar flexors/hip extensors)
- Serial casting: for fixed or dynamic ankle equinus
3.2 Spasticity Management
PT plays the primary non-pharmacological role:
| Intervention | Details |
|---|---|
| Stretching | Daily passive/active-assisted stretching; sustained holds preferred to ballistic |
| Standing programs | Tilt table or standing frame; weight-bearing inhibits spasticity via proprioceptive input; also benefits bone density and bowel function |
| Cryotherapy | Cold packs/cool baths transiently reduce spasticity; facilitates stretching and exercise |
| Electrical stimulation (TENS/NMES) | Reduces spasticity post-application; NMES to antagonists (dorsiflexors) inhibits plantar flexor spasticity |
| Hydrotherapy | Warm water (≤35°C) — caution re Uhthoff's; reduces spasm frequency |
| Positioning | Correct seating, lying postures prevent abnormal patterns; anti-spasm positions |
| Splinting/Orthoses | Resting hand splints, ankle foot orthoses (AFO) to maintain muscle/tendon length |
| Intrathecal Baclofen (ITB) | Not PT-delivered but PT essential for post-pump rehab, goal setting, monitoring |
3.3 Gait Rehabilitation
| Problem | PT Intervention |
|---|---|
| Drop foot | Ankle-foot orthosis (AFO) — rigid or dynamic; FES (WalkAide, NESS L300); peroneal strengthening |
| Scissor gait (adductor spasticity) | Hip abductor strengthening; botulinum toxin to adductors; gait retraining |
| Ataxic/wide-based gait | Weighted vest/ankle weights (attenuates ataxia); walking poles; task-specific gait training; treadmill training |
| Reduced walking speed | Fampridine (PT monitors walking response); treadmill training at faster speeds; body-weight-supported treadmill training (BWSTT) |
| Festination/short steps | Visual/auditory cueing; rhythmic auditory stimulation (RAS) using metronome |
| Reduced endurance | 6MWT-guided aerobic conditioning; pacing strategies; energy conservation |
Treadmill Training: evidence supports improvement in walking speed and endurance; body-weight-supported treadmill training (BWSTT) allows earlier/safer gait training in higher disability levels.
Robot-Assisted Gait Training (RAGT): Lokomat and similar systems; evidence in moderate-to-severe MS; augments stepping pattern, provides repetition; more beneficial in earlier stages.
3.4 Fatigue Management
Fatigue is the most prevalent and disabling MS symptom. PT interventions:
| Strategy | Details |
|---|---|
| Aerobic conditioning | Improves central and peripheral fatigue mechanisms; 30–60 min moderate exercise, 3–5×/week |
| Energy conservation | Pacing (activity vs. rest ratio), activity planning, prioritisation |
| Cooling strategies | Pre-cooling (cold shower/ice vest before exercise), cool exercise environments |
| Sleep hygiene education | Address sleep disorders contributing to fatigue |
| Graded activity programs | Graduated increase in activity avoiding boom-bust cycle |
| Mindfulness-based exercise | Yoga, Tai Chi — reduces fatigue severity and improves QoL |
3.5 Falls Prevention
- Multifactorial falls assessment and individualized program
- Exercise: balance training + strengthening most effective intervention
- Environmental modification: liaise with OT; remove trip hazards, install grab rails, stair rails
- Footwear assessment: supportive, flat, closed heel
- Orthoses: AFO improves foot clearance and reduces trip risk
- Walking aids: prescribe appropriate aid (stick, Lofstrand crutches, rollator/wheeled walker, wheelchair) at the right time
- Assistive technology: FES, wearable fall detectors
- Dual-task training: practice walking while performing cognitive tasks (fall risk often higher during dual-task in MS)
3.6 Upper Limb and Tremor Rehabilitation
- Strengthening: proximal stabilizer strengthening (shoulder girdle, core) improves distal control
- Coordination retraining: repetitive task training, reaching/grasping tasks, fine motor exercises
- Weighted cuffs/utensils: dampen intention tremor during ADLs (evidence modest but patient-reported benefit)
- Constraint-induced movement therapy (CIMT): in unilateral weakness, forced use of weaker limb
- Mirror therapy: cortical remapping in unilateral impairment
- Splinting: wrist/hand supports to stabilize proximal joints during functional tasks
3.7 Respiratory Physiotherapy
In advanced MS (EDSS >7):
- Breathing exercises: diaphragmatic breathing, segmental expansion
- Assisted cough techniques: manual chest compression, mechanical insufflation-exsufflation (MIE/CoughAssist) when PCF <270 L/min
- Postural drainage / airway clearance: if aspiration/recurrent chest infections
- Inspiratory muscle training (IMT): threshold IMT devices; evidence for improved respiratory muscle strength and perception of breathlessness in MS
3.8 Pain Management (PT Contribution)
| Pain Type | PT Approach |
|---|---|
| Spasticity-related pain | Stretching, positioning, TENS, hydrotherapy |
| Musculoskeletal overuse pain (compensatory gait) | Biomechanical correction, orthoses, targeted strengthening |
| Central neuropathic pain | TENS (gate control), TENS, hydrotherapy, graded exercise; PT adjunct to pharmacological management |
| Back pain (from poor posture/sitting) | Core stability, postural correction, ergonomic seating assessment |
3.9 Wheelchair and Assistive Technology Prescription
When walking becomes unsafe or too effortful:
- Walking aids: hierarchical progression — stick → Lofstrand crutches → rollator → wheelchair
- Manual wheelchair: lightweight, individually prescribed; upper limb assessment critical (shoulder overuse risk)
- Power wheelchair/scooter: for upper limb involvement or severe fatigue limiting self-propulsion
- Environmental control systems: voice-activated devices, ramps, hoists
- Liaise closely with OT and seating specialists
3.10 Neuroplasticity-Based Approaches
PT leverages the CNS's capacity for neuroplastic adaptation:
- Task-specific repetitive practice: high repetitions of meaningful tasks drive cortical remapping
- Motor learning principles: blocked → random practice; external focus of attention; augmented feedback
- Constraint-induced movement therapy (CIMT)
- Virtual reality (VR) rehabilitation: evidence for balance, gait, and motivation in MS; Wii Fit balance games studied
- Transcranial magnetic stimulation (TMS) / tDCS: research stage; combined with PT may enhance motor learning
4. Outcome Monitoring & Reassessment
| Timepoint | Action |
|---|---|
| Baseline | Full assessment as above; establish goals and baseline measures |
| 4–6 weeks | Reassess primary outcome measures; progress or modify program |
| 3 months | Full reassessment; liaise with MDT; adjust goals |
| Following relapse | Acute inpatient/outpatient PT; reassess and rehabilitate new deficits |
| Annually | Comprehensive review; update equipment, home setup, exercise program |
Key Outcome Measures Summary
| Domain | Measure |
|---|---|
| Disability | EDSS, FIM, Barthel Index |
| Walking | 25FWT, 10MWT, 6MWT, MSWS-12 |
| Balance | BBS, TUG, Tinetti, MiniBESTest |
| Spasticity | Modified Ashworth Scale, Penn Spasm Scale |
| Fatigue | MFIS, FSS |
| Upper limb | 9-Hole Peg Test |
| QoL | MSIS-29, SF-36 |
| Falls | FES-I, Falls diary |
| Cognition | SDMT (screen) |
5. Principles Guiding MS Physiotherapy
- Treat to function, not diagnosis — individual goals, not disease labels
- Exercise is medicine — prescribe it like a drug (type, dose, frequency, progression)
- Fatigue-aware practice — avoid over-fatiguing; schedule PT at optimal times (not post-DMT infusion, not peak heat)
- Promote self-management — home exercise programs, activity monitoring, education
- Pacing and graded activity — boom-bust cycle is common; teach consistent moderate activity
- Relapse awareness — suspend vigorous PT during active relapse; gentle ROM and positioning maintained
- Psychological integration — screen for depression/anxiety; motivational interviewing; psychologically informed practice
- Longitudinal relationship — MS is lifelong; PT involvement at key transitions (diagnosis, relapse, EDSS progression, loss of ambulation, pregnancy)
Key References:
- Geriatric Trauma Management (p. 82) — Tinetti Gait and Balance Test validation in MS and neurodegenerative disease
- NICE Guideline NG220: Multiple Sclerosis in Adults (2022)
- Cochrane Reviews: Exercise therapy for MS (Rietberg et al.; Learmonth et al.)
- MS Society UK / National MS Society Clinical Practice Guidelines
- Harrison's Principles of Internal Medicine, 21st Ed. (p. 12550, 12598) — MS overview informing PT context
This is a shared conversation. Sign in to Orris to start your own chat.